Abstract
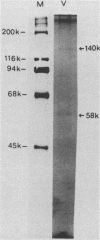
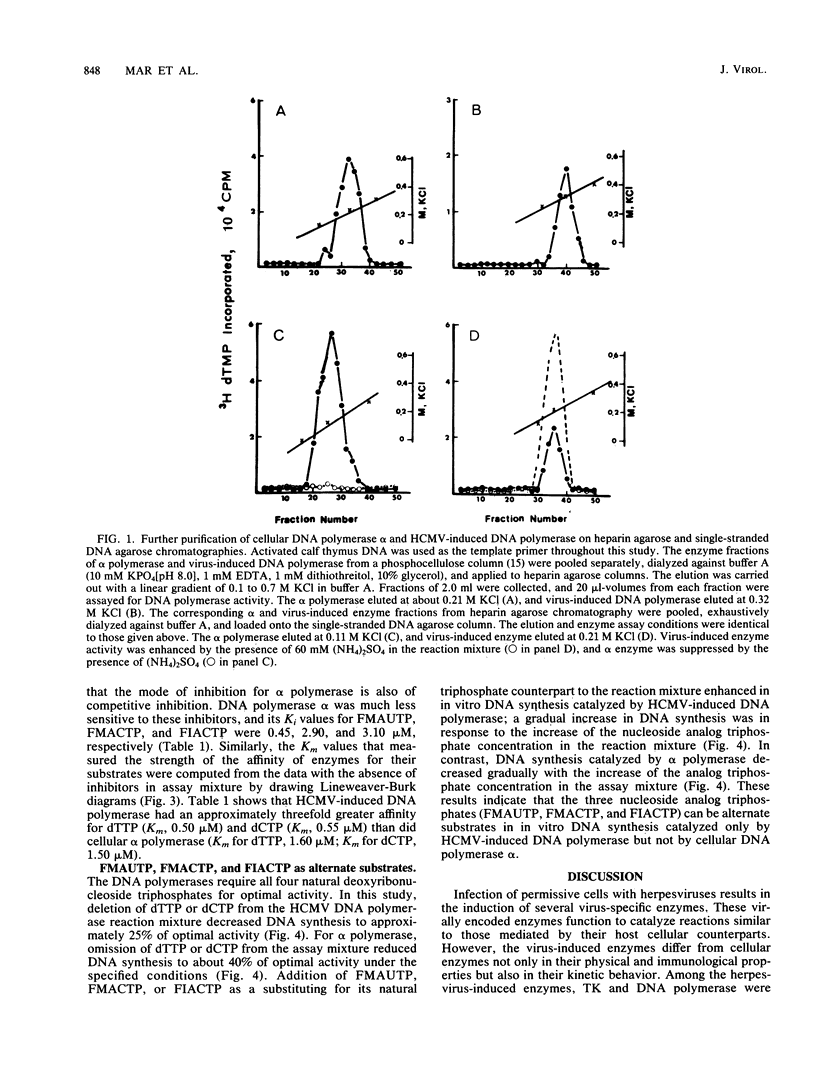
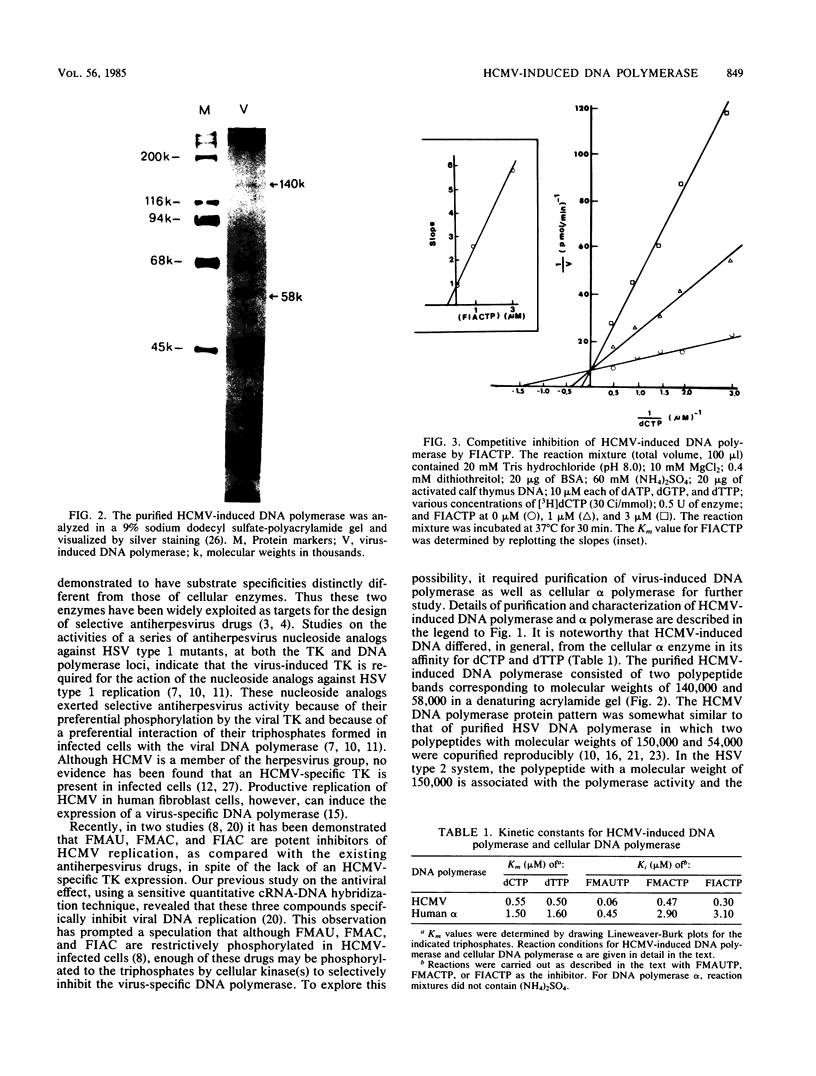
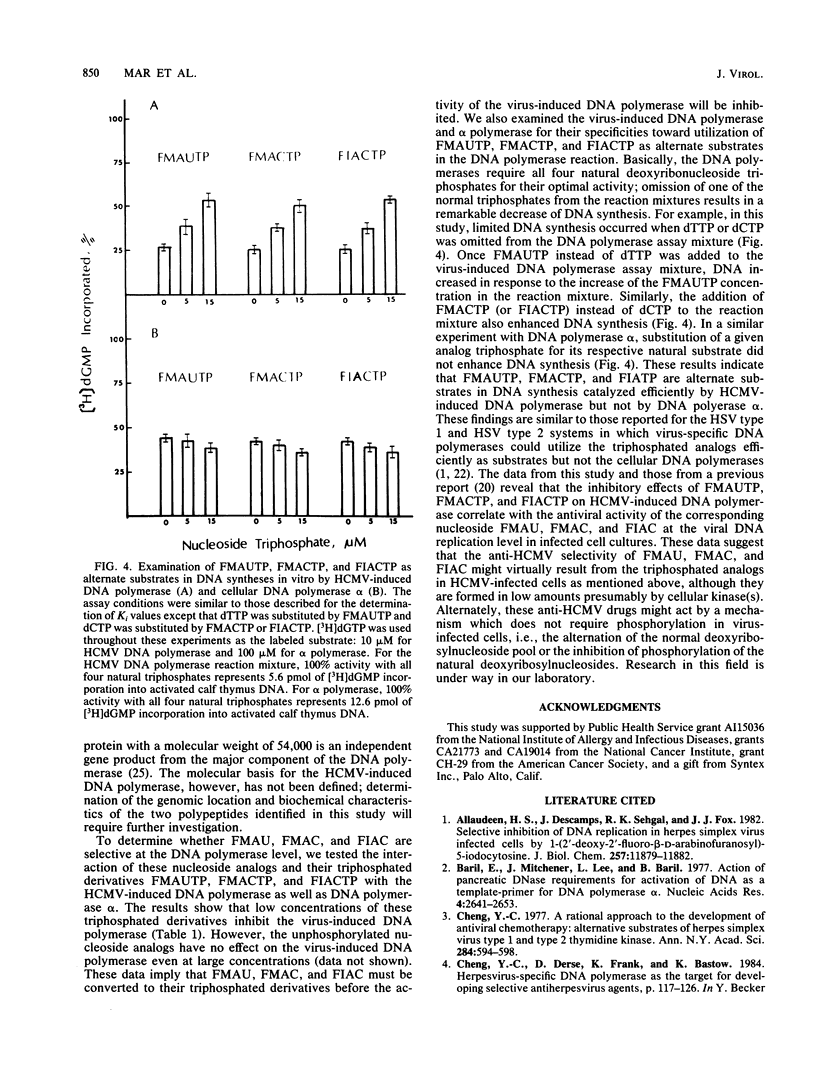
Human cytomegalovirus-induced DNA polymerase and cellular DNA polymerase alpha were purified by successive chromatography on DEAE-cellulose, phosphocellulose, heparin agarose, and single-stranded DNA agarose columns. The purified virus-induced DNA polymerase was resolved to consist of two polypeptides corresponding to molecular weights of 140,000 and 58,000, as analyzed by sodium dodecyl sulfate-polyacrylamide gel electrophoresis. Virus-induced DNA polymerase and cellular alpha polymerase were examined for their sensitivities to the triphosphates of 1-(2'-deoxy-2'-fluoro-beta-D-arabinofuranosyl)-5-methyluracil (FMAUTP), -5-iodocytosine (FIACTP), and -5-methylcytosine (FMACTP). The inhibitive effects of these triphosphates on the DNA polymerases were competitive with regard to the natural substrates; thus FMAUTP competes with dTTP, and FIACTP and FMACTP compete with dCTP. The inhibition constants (Ki) for FMAUTP, FIACTP, and FMACTP of virus-induced DNA polymerase are 0.06, 0.30, and 0.47 microM, respectively. Cellular DNA polymerase alpha is much less sensitive to these inhibitors, and its Ki values for FMAUTP, FIACTP, and FMACTP are 0.45, 3.10, and 2.90 microM, respectively. In addition, human cytomegalovirus-induced DNA polymerase, but not cellular DNA polymerase alpha, can utilize these analog triphosphates as alternate substrates for their corresponding natural deoxyribonucleoside triphosphates in in vitro DNA synthesis.
Full text
PDF


Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Allaudeen H. S., Descamps J., Sehgal R. K., Fox J. J. Selective inhibition of DNA replication in herpes simplex virus infected cells by 1-(2'-deoxy-2'-fluoro-beta-D-arabinofuranosyl)-5-iodocytosine. J Biol Chem. 1982 Oct 25;257(20):11879–11882. [PubMed] [Google Scholar]
- Baril E., Mitchener J., Lee L., Baril B. Action of pancreatic DNase: requirements for activation of DNA as a template-primer for DNA polymerase. Nucleic Acids Res. 1977 Aug;4(8):2641–2653. doi: 10.1093/nar/4.8.2641. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cheng Y. C. A rational approach to the development of antiviral chemotherapy: alternative substrates of herpes simplex virus Type 1 (HSV-1) and Type 2 (HSV-2) thymidine kinase (TK). Ann N Y Acad Sci. 1977 Mar 4;284:594–598. doi: 10.1111/j.1749-6632.1977.tb21992.x. [DOI] [PubMed] [Google Scholar]
- Cheng Y. C., Dutschman G., De Clercq E., Jones A. S., Rahim S. G., Verhelst G., Walker R. T. Differential affinities of 5-(2-halogenovinyl)-2'-deoxyuridines for deoxythymidine kinases of various origins. Mol Pharmacol. 1981 Jul;20(1):230–233. [PubMed] [Google Scholar]
- Cheng Y. C., Dutschman G., Fox J. J., Watanabe K. A., Machida H. Differential activity of potential antiviral nucleoside analogs on herpes simplex virus-induced and human cellular thymidine kinases. Antimicrob Agents Chemother. 1981 Sep;20(3):420–423. doi: 10.1128/aac.20.3.420. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cheng Y. C., Huang E. S., Lin J. C., Mar E. C., Pagano J. S., Dutschman G. E., Grill S. P. Unique spectrum of activity of 9-[(1,3-dihydroxy-2-propoxy)methyl]-guanine against herpesviruses in vitro and its mode of action against herpes simplex virus type 1. Proc Natl Acad Sci U S A. 1983 May;80(9):2767–2770. doi: 10.1073/pnas.80.9.2767. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Colacino J. M., Lopez C. Efficacy and selectivity of some nucleoside analogs as anti-human cytomegalovirus agents. Antimicrob Agents Chemother. 1983 Oct;24(4):505–508. doi: 10.1128/aac.24.4.505. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Derse D., Bastow K. F., Cheng Y. Characterization of the DNA polymerases induced by a group of herpes simplex virus type I variants selected for growth in the presence of phosphonoformic acid. J Biol Chem. 1982 Sep 10;257(17):10251–10260. [PubMed] [Google Scholar]
- Elion G. B., Furman P. A., Fyfe J. A., de Miranda P., Beauchamp L., Schaeffer H. J. Selectivity of action of an antiherpetic agent, 9-(2-hydroxyethoxymethyl) guanine. Proc Natl Acad Sci U S A. 1977 Dec;74(12):5716–5720. doi: 10.1073/pnas.74.12.5716. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Estes J. E., Huang E. S. Stimulation of cellular thymidine kinases by human cytomegalovirus. J Virol. 1977 Oct;24(1):13–21. doi: 10.1128/jvi.24.1.13-21.1977. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Frank K. B., Chiou J. F., Cheng Y. C. Interaction of herpes simplex virus-induced DNA polymerase with 9-(1,3-dihydroxy-2-propoxymethyl)guanine triphosphate. J Biol Chem. 1984 Feb 10;259(3):1566–1569. [PubMed] [Google Scholar]
- Furman P. A., St Clair M. H., Spector T. Acyclovir triphosphate is a suicide inactivator of the herpes simplex virus DNA polymerase. J Biol Chem. 1984 Aug 10;259(15):9575–9579. [PubMed] [Google Scholar]
- Huang E. S. Human cytomegalovirus. III. Virus-induced DNA polymerase. J Virol. 1975 Aug;16(2):298–310. doi: 10.1128/jvi.16.2.298-310.1975. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Knopf K. W. Properties of herpes simplex virus DNA polymerase and characterization of its associated exonuclease activity. Eur J Biochem. 1979 Jul;98(1):231–244. doi: 10.1111/j.1432-1033.1979.tb13181.x. [DOI] [PubMed] [Google Scholar]
- Lopez C., Watanabe K. A., Fox J. J. 2'-fluoro-5-iodo-aracytosine, a potent and selective anti-herpesvirus agent. Antimicrob Agents Chemother. 1980 May;17(5):803–806. doi: 10.1128/aac.17.5.803. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mancini W. R., De Clercq E., Prusoff W. H. The relationship between incorporation of E-5-(2-Bromovinyl)-2'-deoxyuridine into herpes simplex virus type 1 DNA with virus infectivity and DNA integrity. J Biol Chem. 1983 Jan 25;258(2):792–795. [PubMed] [Google Scholar]
- Mar E. C., Patel P. C., Cheng Y. C., Fox J. J., Watanabe K. A., Huang E. S. Effects of certain nucleoside analogues on human cytomegalovirus replication in vitro. J Gen Virol. 1984 Jan;65(Pt 1):47–53. doi: 10.1099/0022-1317-65-1-47. [DOI] [PubMed] [Google Scholar]
- Powell K. L., Purifoy D. J. Nonstructural proteins of herpes simplex virus. I. Purification of the induced DNA polymerase. J Virol. 1977 Nov;24(2):618–626. doi: 10.1128/jvi.24.2.618-626.1977. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ruth J. L., Cheng Y. C. Nucleoside analogues with clinical potential in antivirus chemotherapy. The effect of several thymidine and 2'-deoxycytidine analogue 5'-triphosphates on purified human (alpha, beta) and herpes simplex virus (types 1, 2) DNA polymerases. Mol Pharmacol. 1981 Sep;20(2):415–422. [PubMed] [Google Scholar]
- Ruyechan W. T., Weir A. C. Interaction with nucleic acids and stimulation of the viral DNA polymerase by the herpes simplex virus type 1 major DNA-binding protein. J Virol. 1984 Dec;52(3):727–733. doi: 10.1128/jvi.52.3.727-733.1984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Smith K. O., Galloway K. S., Kennell W. L., Ogilvie K. K., Radatus B. K. A new nucleoside analog, 9-[[2-hydroxy-1-(hydroxymethyl)ethoxyl]methyl]guanine, highly active in vitro against herpes simplex virus types 1 and 2. Antimicrob Agents Chemother. 1982 Jul;22(1):55–61. doi: 10.1128/aac.22.1.55. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vaughan P. J., Purifoy D. J., Powell K. L. DNA-binding protein associated with herpes simplex virus DNA polymerase. J Virol. 1985 Feb;53(2):501–508. doi: 10.1128/jvi.53.2.501-508.1985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wray W., Boulikas T., Wray V. P., Hancock R. Silver staining of proteins in polyacrylamide gels. Anal Biochem. 1981 Nov 15;118(1):197–203. doi: 10.1016/0003-2697(81)90179-2. [DOI] [PubMed] [Google Scholar]
- Závada V., Erban V., Rezácová D., Vonka V. Thymidine-kinase in cytomegalovirus infected cells. Arch Virol. 1976;52(4):333–339. doi: 10.1007/BF01315622. [DOI] [PubMed] [Google Scholar]