Abstract
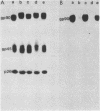
Previous results from our laboratory have demonstrated that equine infectious anemia virus displays structural variations in its surface glycoproteins and RNA genome during passage and chronic infections in experimentally infected Shetland ponies (Montelaro et al., J. Biol. Chem. 259:10539-10544, 1984; Payne et al., J. Gen. Virol. 65:1395-1399, 1984). The present study was undertaken to obtain an antigenic and biochemical characterization of equine infectious anemia virus isolates recovered from an experimentally infected pony during sequential disease episodes, each separated by intervals of only 4 to 8 weeks. The virus isolates could be distinguished antigenically by neutralization assays with serum from the infected pony and by Western blot analysis with a monoclonal antibody against the major surface glycoprotein gp90, thus demonstrating that novel antigenic variants of equine infectious anemia virus predominate during each clinical episode. The respective virion glycoproteins displayed different electrophoretic mobilities on sodium dodecyl sulfate-polyacrylamide gels, indicating structural variation. Tryptic peptide and glycopeptide maps of the viral proteins of each virus isolate revealed biochemical alterations involving amino acid sequence and glycosylation patterns in the virion surface glycoproteins gp90 and gp45. In contrast, no structural variation was observed in the internal viral proteins pp15, p26, and p9 from any of the four virus isolates. Oligonucleotide mapping experiments revealed similar but unique RNase T1-resistant oligonucleotide fingerprints of the RNA genomes of each of the virus isolates. Localization of altered oligonucleotides for one virus isolate placed two of three unique oligonucleotides within the predicted env gene region of the genome, perhaps correlating with the structural variation observed in the envelope glycoproteins. Thus these results support the concept that equine infectious anemia virus is indeed capable of relatively rapid genomic variations during replication, some of which result in altered glycoprotein structures and antigenic variants which are responsible for the unique periodic disease nature observed in persistently infected animals. The findings of envelope specific differences in isolates of visna virus and of human T-cell lymphotropic virus III (acquired immune deficiency syndrome-related virus) suggest that this variation may be a common characteristic of the subfamily Lentivirinae.
Full text
PDF







Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Alexander S., Elder J. H. Carbohydrate dramatically influences immune reactivity of antisera to viral glycoprotein antigens. Science. 1984 Dec 14;226(4680):1328–1330. doi: 10.1126/science.6505693. [DOI] [PubMed] [Google Scholar]
- Aviv H., Leder P. Purification of biologically active globin messenger RNA by chromatography on oligothymidylic acid-cellulose. Proc Natl Acad Sci U S A. 1972 Jun;69(6):1408–1412. doi: 10.1073/pnas.69.6.1408. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bister K., Löliger H. C., Duesberg P. H. Oligoribonucleotide map and protein of CMII: detection of conserved and nonconserved genetic elements in avian acute leukemia viruses CMII, MC29, and MH2. J Virol. 1979 Oct;32(1):208–219. doi: 10.1128/jvi.32.1.208-219.1979. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bruck C., Portetelle D., Glineur C., Bollen A. One-step purification of mouse monoclonal antibodies from ascitic fluid by DEAE Affi-Gel blue chromatography. J Immunol Methods. 1982 Sep 30;53(3):313–319. doi: 10.1016/0022-1759(82)90178-8. [DOI] [PubMed] [Google Scholar]
- Bruck C., Rensonnet N., Portetelle D., Cleuter Y., Mammerickx M., Burny A., Mamoun R., Guillemain B., van der Maaten M. J., Ghysdael J. Biologically active epitopes of bovine leukemia virus glycoprotein gp51: their dependence on protein glycosylation and genetic variability. Virology. 1984 Jul 15;136(1):20–31. doi: 10.1016/0042-6822(84)90244-7. [DOI] [PubMed] [Google Scholar]
- Burnette W. N. "Western blotting": electrophoretic transfer of proteins from sodium dodecyl sulfate--polyacrylamide gels to unmodified nitrocellulose and radiographic detection with antibody and radioiodinated protein A. Anal Biochem. 1981 Apr;112(2):195–203. doi: 10.1016/0003-2697(81)90281-5. [DOI] [PubMed] [Google Scholar]
- Charman H. P., Bladen S., Gilden R. V., Coggins L. Equine infectious anemia virus: evidence favoring classification as a retravirus. J Virol. 1976 Sep;19(3):1073–1079. doi: 10.1128/jvi.19.3.1073-1079.1976. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Clements J. E., Pedersen F. S., Narayan O., Haseltine W. A. Genomic changes associated with antigenic variation of visna virus durig persistent infection. Proc Natl Acad Sci U S A. 1980 Aug;77(8):4454–4458. doi: 10.1073/pnas.77.8.4454. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Coffin J. M., Champion M., Chabot F. Nucleotide sequence relationships between the genomes of an endogenous and an exogenous avian tumor virus. J Virol. 1978 Dec;28(3):972–991. doi: 10.1128/jvi.28.3.972-991.1978. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Darlix J. L., Spahr P. F. High spontaneous mutation rate of Rous sarcoma virus demonstrated by direct sequencing of the RNA genome. Nucleic Acids Res. 1983 Sep 10;11(17):5953–5967. doi: 10.1093/nar/11.17.5953. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Elder J. H., Gautsch J. W., Jensen F. C., Lerner R. A., Hartley J. W., Rowe W. P. Biochemical evidence that MCF murine leukemia viruses are envelope (env) gene recombinants. Proc Natl Acad Sci U S A. 1977 Oct;74(10):4676–4680. doi: 10.1073/pnas.74.10.4676. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Elder J. H., Pickett R. A., 2nd, Hampton J., Lerner R. A. Radioiodination of proteins in single polyacrylamide gel slices. Tryptic peptide analysis of all the major members of complex multicomponent systems using microgram quantities of total protein. J Biol Chem. 1977 Sep 25;252(18):6510–6515. [PubMed] [Google Scholar]
- Gonda M. A., Wong-Staal F., Gallo R. C., Clements J. E., Narayan O., Gilden R. V. Sequence homology and morphologic similarity of HTLV-III and visna virus, a pathogenic lentivirus. Science. 1985 Jan 11;227(4683):173–177. doi: 10.1126/science.2981428. [DOI] [PubMed] [Google Scholar]
- Griffin D. E., Narayan O., Adams R. J. Early immune responses in visna, a slow viral disease of sheep. J Infect Dis. 1978 Sep;138(3):340–350. doi: 10.1093/infdis/138.3.340. [DOI] [PubMed] [Google Scholar]
- Gudnadóttir M. Visna-maedi in sheep. Prog Med Virol. 1974;18(0):336–349. [PubMed] [Google Scholar]
- Hahn B. H., Shaw G. M., Arya S. K., Popovic M., Gallo R. C., Wong-Staal F. Molecular cloning and characterization of the HTLV-III virus associated with AIDS. Nature. 1984 Nov 8;312(5990):166–169. doi: 10.1038/312166a0. [DOI] [PubMed] [Google Scholar]
- Holland J., Spindler K., Horodyski F., Grabau E., Nichol S., VandePol S. Rapid evolution of RNA genomes. Science. 1982 Mar 26;215(4540):1577–1585. doi: 10.1126/science.7041255. [DOI] [PubMed] [Google Scholar]
- Kawai S., Hanafusa H. Isolation of defective mutant of avian sarcoma virus. Proc Natl Acad Sci U S A. 1973 Dec;70(12):3493–3497. doi: 10.1073/pnas.70.12.3493. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kawaoka Y., Naeve C. W., Webster R. G. Is virulence of H5N2 influenza viruses in chickens associated with loss of carbohydrate from the hemagglutinin? Virology. 1984 Dec;139(2):303–316. doi: 10.1016/0042-6822(84)90376-3. [DOI] [PubMed] [Google Scholar]
- Kono Y., Kobayashi K., Fukunaga Y. Antigenic drift of equine infectious anemia virus in chronically infected horses. Arch Gesamte Virusforsch. 1973;41(1):1–10. doi: 10.1007/BF01249923. [DOI] [PubMed] [Google Scholar]
- Kronvall G., Grey H. M., Williams R. C., Jr Protein A reactivity with mouse immunoglobulins. Structural relationship between some mouse and human immunoglobulins. J Immunol. 1970 Nov;105(5):1116–1123. [PubMed] [Google Scholar]
- Kung H. J., Bailey J. M., Davidson N., Nicolson M. O., McAllister R. M. Structure, subunit composition, and molecular weight of RD-114 RNA. J Virol. 1975 Aug;16(2):397–411. doi: 10.1128/jvi.16.2.397-411.1975. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Laemmli U. K. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature. 1970 Aug 15;227(5259):680–685. doi: 10.1038/227680a0. [DOI] [PubMed] [Google Scholar]
- Lutley R., Pétursson G., Pálsson P. A., Georgsson G., Klein J., Nathanson N. Antigenic drift in visna: virus variation during long-term infection of Icelandic sheep. J Gen Virol. 1983 Jul;64(Pt 7):1433–1440. doi: 10.1099/0022-1317-64-7-1433. [DOI] [PubMed] [Google Scholar]
- Mackenzie M. R., Warner N. L., Mitchell G. F. The binding of murine immunoglobulins to staphylococcal protein A. J Immunol. 1978 May;120(5):1493–1496. [PubMed] [Google Scholar]
- Malmquist W. A., Barnett D., Becvar C. S. Production of equine infectious anemia antigen in a persistently infected cell line. Arch Gesamte Virusforsch. 1973;42(4):361–370. doi: 10.1007/BF01250717. [DOI] [PubMed] [Google Scholar]
- Massey R. J., Schochetman G. Topographical analysis of viral epitopes using monoclonal antibodies: mechanism of virus neutralization. Virology. 1981 Nov;115(1):20–32. doi: 10.1016/0042-6822(81)90085-4. [DOI] [PubMed] [Google Scholar]
- Montelaro R. C., Bolognesi D. P. Structure and morphogenesis of type-C retroviruses. Adv Cancer Res. 1978;28:63–89. doi: 10.1016/s0065-230x(08)60646-6. [DOI] [PubMed] [Google Scholar]
- Montelaro R. C., Lohrey N., Parekh B., Blakeney E. W., Issel C. J. Isolation and comparative biochemical properties of the major internal polypeptides of equine infectious anemia virus. J Virol. 1982 Jun;42(3):1029–1038. doi: 10.1128/jvi.42.3.1029-1038.1982. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Montelaro R. C., Parekh B., Orrego A., Issel C. J. Antigenic variation during persistent infection by equine infectious anemia virus, a retrovirus. J Biol Chem. 1984 Aug 25;259(16):10539–10544. [PubMed] [Google Scholar]
- Montelaro R. C., West M., Issel C. J. Antigenic reactivity of the major glycoprotein of equine infectious anemia virus, a retrovirus. Virology. 1984 Jul 30;136(2):368–374. doi: 10.1016/0042-6822(84)90173-9. [DOI] [PubMed] [Google Scholar]
- Montelaro R. C., West M., Issel C. J. Isolation of equine infectious anemia virus glycoproteins. Lectin affinity chromatography procedures for high avidity glycoproteins. J Virol Methods. 1983 Jun;6(6):337–346. doi: 10.1016/0166-0934(83)90056-3. [DOI] [PubMed] [Google Scholar]
- Mullins J. I., Casey J. W., Nicolson M. O., Davidson N. Sequence organization of feline leukemia virus DNA in infected cells. Nucleic Acids Res. 1980 Aug 11;8(15):3287–3305. doi: 10.1093/nar/8.15.3287. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Narayan O., Griffin D. E., Chase J. Antigenic shift of visna virus in persistently infected sheep. Science. 1977 Jul 22;197(4301):376–378. doi: 10.1126/science.195339. [DOI] [PubMed] [Google Scholar]
- Nilsson B., Nordén N. E., Svensson S. Structural studies on the carbohydrate portion of fetuin. J Biol Chem. 1979 Jun 10;254(11):4545–4553. [PubMed] [Google Scholar]
- Nottay B. K., Kew O. M., Hatch M. H., Heyward J. T., Obijeski J. F. Molecular variation of type 1 vaccine-related and wild polioviruses during replication in humans. Virology. 1981 Jan 30;108(2):405–423. doi: 10.1016/0042-6822(81)90448-7. [DOI] [PubMed] [Google Scholar]
- O'Rear J. J., Temin H. M. Spontaneous changes in nucleotide sequence in proviruses of spleen necrosis virus, an avian retrovirus. Proc Natl Acad Sci U S A. 1982 Feb;79(4):1230–1234. doi: 10.1073/pnas.79.4.1230. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Orrego A., Issel C. J., Montelaro R. C., Adams W. V., Jr Virulence and in vitro growth of a cell-adapted strain of equine infectious anemia virus after serial passage in ponies. Am J Vet Res. 1982 Sep;43(9):1556–1560. [PubMed] [Google Scholar]
- Parekh B., Issel C. J., Montelaro R. C. Equine infectious anemia virus, a putative lentivirus, contains polypeptides analogous to prototype-C oncornaviruses. Virology. 1980 Dec;107(2):520–525. doi: 10.1016/0042-6822(80)90319-0. [DOI] [PubMed] [Google Scholar]
- Payne S., Parekh B., Montelaro R. C., Issel C. J. Genomic alterations associated with persistent infections by equine infectious anaemia virus, a retrovirus. J Gen Virol. 1984 Aug;65(Pt 8):1395–1399. doi: 10.1099/0022-1317-65-8-1395. [DOI] [PubMed] [Google Scholar]
- Pedersen F. S., Haseltine W. A. A micromethod for detailed characterization of high molecular weight RNA. Methods Enzymol. 1980;65(1):680–687. doi: 10.1016/s0076-6879(80)65066-6. [DOI] [PubMed] [Google Scholar]
- Pedersen F. S., Haseltine W. A. Analysis of the genome of an endogenous, ecotropic retrovirus of the AKR strain of mice: micromethod for detailed characterization of high-molecular-weight RNA. J Virol. 1980 Jan;33(1):349–365. doi: 10.1128/jvi.33.1.349-365.1980. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ratner L., Haseltine W., Patarca R., Livak K. J., Starcich B., Josephs S. F., Doran E. R., Rafalski J. A., Whitehorn E. A., Baumeister K. Complete nucleotide sequence of the AIDS virus, HTLV-III. Nature. 1985 Jan 24;313(6000):277–284. doi: 10.1038/313277a0. [DOI] [PubMed] [Google Scholar]
- Rice N. R., Simek S., Ryder O. A., Coggins L. Detection of proviral DNA in horse cells infected with equine infectious anemia virus. J Virol. 1978 Jun;26(3):577–583. doi: 10.1128/jvi.26.3.577-583.1978. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sanchez-Pescador R., Power M. D., Barr P. J., Steimer K. S., Stempien M. M., Brown-Shimer S. L., Gee W. W., Renard A., Randolph A., Levy J. A. Nucleotide sequence and expression of an AIDS-associated retrovirus (ARV-2). Science. 1985 Feb 1;227(4686):484–492. doi: 10.1126/science.2578227. [DOI] [PubMed] [Google Scholar]
- Schmid K., Nimerg R. B., Kimura A., Yamaguchi H., Binette J. P. The carbohydrate units of human plasma alpha1-acid glycoprotein. Biochim Biophys Acta. 1977 Jun 24;492(2):291–302. doi: 10.1016/0005-2795(77)90080-0. [DOI] [PubMed] [Google Scholar]
- Scott J. V., Stowring L., Haase A. T., Narayan O., Vigne R. Antigenic variation in visna virus. Cell. 1979 Oct;18(2):321–327. doi: 10.1016/0092-8674(79)90051-5. [DOI] [PubMed] [Google Scholar]
- Shane B. S., Issel C. J., Montelaro R. C. Enzyme-linked immunosorbent assay for detection of equine infectious anemia virus p26 antigen and antibody. J Clin Microbiol. 1984 Mar;19(3):351–355. doi: 10.1128/jcm.19.3.351-355.1984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shtivelman E., Zakut R., Canaani E. Frequent generation of nonrescuable reorganized Moloney murine sarcoma viral genomes. Proc Natl Acad Sci U S A. 1984 Jan;81(2):294–298. doi: 10.1073/pnas.81.2.294. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stowring L., Haase A. T., Charman H. P. Serological definition of the lentivirus group of retroviruses. J Virol. 1979 Feb;29(2):523–528. doi: 10.1128/jvi.29.2.523-528.1979. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thormar H., Barshatzky M. R., Arnesen K., Kozlowski P. B. The emergence of antigenic variants is a rare event in long-term visna virus infection in vivo. J Gen Virol. 1983 Jul;64(Pt 7):1427–1432. doi: 10.1099/0022-1317-64-7-1427. [DOI] [PubMed] [Google Scholar]
- Zarling D. A., Temin H. M. High spontaneous mutation rate of an avian sarcoma virus. J Virol. 1975 Jan;17(1):74–84. doi: 10.1128/jvi.17.1.74-84.1976. [DOI] [PMC free article] [PubMed] [Google Scholar]
- de StGroth S. F., Scheidegger D. Production of monoclonal antibodies: strategy and tactics. J Immunol Methods. 1980;35(1-2):1–21. doi: 10.1016/0022-1759(80)90146-5. [DOI] [PubMed] [Google Scholar]