Abstract
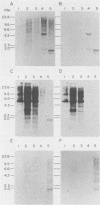
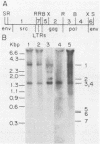
We detected sequences related to the avian retrovirus Rous sarcoma virus within the genome of the Japanese quail, a species previously considered to be free of endogenous avian leukosis virus elements. Using low-stringency conditions of hybridization, we screened a quail genomic library for clones containing retrovirus-related information. Of five clones so selected, one, lambda Q48, contained sequence information related to the gag, pol, and env genes of Rous sarcoma virus arranged in a contiguous fashion and spanning a distance of approximately 5.8 kilobases. This organization is consistent with the presence of an endogenous retroviral element within the Japanese quail genome. Use of this element as a high-stringency probe on Southern blots of genomic digests of several quail DNA demonstrated hybridization to a series of high-molecular-weight bands. By slot hybridization to quail DNA with a cloned probe, it was deduced that there were approximately 300 copies per diploid cell. In addition, the quail element also hybridized at low stringency to the DNA of the White Leghorn chicken and at high stringency to the DNAs of several species of jungle fowl and both true and ruffed pheasants. Limited nucleotide sequencing analysis of lambda Q48 revealed homologies of 65, 52, and 46% compared with the sequence of Rous sarcoma virus strain Prague C for the endonuclease domain of pol, the pol-env junction, and the 3'-terminal region of env, respectively. Comparisons at the amino acid level were also significant, thus confirming the retrovirus relatedness of the cloned quail element.
Full text
PDF








Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Astrin S. M., Buss E. G., Haywards W. S. Endogenous viral genes are non-essential in the chicken. Nature. 1979 Nov 15;282(5736):339–341. doi: 10.1038/282339a0. [DOI] [PubMed] [Google Scholar]
- Astrin S. M., Crittenden L. B., Buss E. G. Ev 2, a genetic locus containing structural genes for endogenous virus, codes for Rous-associated virus type 0 produced by line 72 chickens. J Virol. 1980 Jan;33(1):250–255. doi: 10.1128/jvi.33.1.250-255.1980. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Astrin S. M. Endogenous viral genes of the White Leghorn chicken: common site of residence and sites associated with specific phenotypes of viral gene expression. Proc Natl Acad Sci U S A. 1978 Dec;75(12):5941–5945. doi: 10.1073/pnas.75.12.5941. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Astrin S. M., Robinson H. L., Crittenden L. B., Buss E. G., Wyban J., Hayward W. S. Ten genetic loci in the chicken that contain structural genes for endogenous avian leukosis viruses. Cold Spring Harb Symp Quant Biol. 1980;44(Pt 2):1105–1109. doi: 10.1101/sqb.1980.044.01.119. [DOI] [PubMed] [Google Scholar]
- Astrin S. M., Robinson H. L. Gs, an allele of chickens for endogenous avian leukosis viral antigens, segregates with ev 3, a genetic locus that contains structural genes for virus. J Virol. 1979 Aug;31(2):420–425. doi: 10.1128/jvi.31.2.420-425.1979. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bauer G., Friis R. R., Mattersberger H., Hofschneider P. H. Controlled release of particle-associated RNA-dependent DNA polymerase by primary chick embryo cell cultures. Exp Cell Res. 1978 Dec;117(2):383–392. doi: 10.1016/0014-4827(78)90151-9. [DOI] [PubMed] [Google Scholar]
- Bauer G., Hofschneider P. H. An RNA-dependent DNA polymerase, different from the known viral reverse transcriptases, in the chicken system. Proc Natl Acad Sci U S A. 1976 Sep;73(9):3025–3029. doi: 10.1073/pnas.73.9.3025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bauer G., Jilek G., Hofschneider P. H. Purification and further characterization of an RNA-dependent DNA polymerase from the allantoic fluid of leukosis-virus-free chicken eggs. Eur J Biochem. 1977 Oct 3;79(2):345–354. doi: 10.1111/j.1432-1033.1977.tb11815.x. [DOI] [PubMed] [Google Scholar]
- Bauer G., Temin H. M. RNA-directed DNA polymerase from particles released by normal goose cells. J Virol. 1979 Mar;29(3):1006–1013. doi: 10.1128/jvi.29.3.1006-1013.1979. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bencini D. A., Houghton J. E., Hoover T. A., Foltermann K. F., Wild J. R., O'Donovan G. A. The DNA sequence of argI from Escherichia coli K12. Nucleic Acids Res. 1983 Dec 10;11(23):8509–8518. doi: 10.1093/nar/11.23.8509. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Benton W. D., Davis R. W. Screening lambdagt recombinant clones by hybridization to single plaques in situ. Science. 1977 Apr 8;196(4286):180–182. doi: 10.1126/science.322279. [DOI] [PubMed] [Google Scholar]
- Bonner T. I., O'Connell C., Cohen M. Cloned endogenous retroviral sequences from human DNA. Proc Natl Acad Sci U S A. 1982 Aug;79(15):4709–4713. doi: 10.1073/pnas.79.15.4709. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Callahan R., Drohan W., Gallahan D., D'Hoostelaere L., Potter M. Novel class of mouse mammary tumor virus-related DNA sequences found in all species of Mus, including mice lacking the virus proviral genome. Proc Natl Acad Sci U S A. 1982 Jul;79(13):4113–4117. doi: 10.1073/pnas.79.13.4113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Callahan R., Drohan W., Tronick S., Schlom J. Detection and cloning of human DNA sequences related to the mouse mammary tumor virus genome. Proc Natl Acad Sci U S A. 1982 Sep;79(18):5503–5507. doi: 10.1073/pnas.79.18.5503. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen P. J., Cywinski A., Taylor J. M. Reverse transcription of 7S L RNA by an avian retrovirus. J Virol. 1985 May;54(2):278–284. doi: 10.1128/jvi.54.2.278-284.1985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chiu I. M., Andersen P. R., Aaronson S. A., Tronick S. R. Molecular cloning of the unintegrated squirrel monkey retrovirus genome: organization and distribution of related sequences in primate DNAs. J Virol. 1983 Sep;47(3):434–441. doi: 10.1128/jvi.47.3.434-441.1983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Crittenden L. B., Fadly A. M., Smith E. J. Effect of endogenous leukosis virus genes on response to infection with avian leukosis and reticuloendotheliosis viruses. Avian Dis. 1982 Apr-Jun;26(2):279–294. [PubMed] [Google Scholar]
- DeLorbe W. J., Luciw P. A., Goodman H. M., Varmus H. E., Bishop J. M. Molecular cloning and characterization of avian sarcoma virus circular DNA molecules. J Virol. 1980 Oct;36(1):50–61. doi: 10.1128/jvi.36.1.50-61.1980. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dunwiddie C., Faras A. J. Presence of retrovirus reverse transcriptase-related gene sequences in avian cells lacking endogenous avian leukosis viruses. Proc Natl Acad Sci U S A. 1985 Aug;82(15):5097–5101. doi: 10.1073/pnas.82.15.5097. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Frisby D. P., Weiss R. A., Roussel M., Stehelin D. The distribution of endogenous chicken retrovirus sequences in the DNA of galliform birds does not coincide with avian phylogenetic relationships. Cell. 1979 Jul;17(3):623–634. doi: 10.1016/0092-8674(79)90270-8. [DOI] [PubMed] [Google Scholar]
- Grandgenett D. P., Vora A. C., Schiff R. D. A 32,000-dalton nucleic acid-binding protein from avian retravirus cores possesses DNA endonuclease activity. Virology. 1978 Aug;89(1):119–132. doi: 10.1016/0042-6822(78)90046-6. [DOI] [PubMed] [Google Scholar]
- Groudine M., Eisenman R., Weintraub H. Chromatin structure of endogenous retroviral genes and activation by an inhibitor of DNA methylation. Nature. 1981 Jul 23;292(5821):311–317. doi: 10.1038/292311a0. [DOI] [PubMed] [Google Scholar]
- Guntaka R. V., Richards O. C., Shank P. R., Kung H. J., Davidson N. Covalently closed circular DNA of avian sarcoma virus: purification from nuclei of infected quail tumor cells and measurement by electron microscopy and gel electrophoresis. J Mol Biol. 1976 Sep 15;106(2):337–357. doi: 10.1016/0022-2836(76)90090-5. [DOI] [PubMed] [Google Scholar]
- Hanafusa H., Miyamoto T., Hanafusa T. A cell-associated factor essential for formation of an infectious form of Rous sarcoma virus. Proc Natl Acad Sci U S A. 1970 Jun;66(2):314–321. doi: 10.1073/pnas.66.2.314. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hayward W. S., Braverman S. B., Astrin S. M. Transcriptional products and DNA structure of endogenous avian proviruses. Cold Spring Harb Symp Quant Biol. 1980;44(Pt 2):1111–1121. doi: 10.1101/sqb.1980.044.01.120. [DOI] [PubMed] [Google Scholar]
- Hohn B., Murray K. Packaging recombinant DNA molecules into bacteriophage particles in vitro. Proc Natl Acad Sci U S A. 1977 Aug;74(8):3259–3263. doi: 10.1073/pnas.74.8.3259. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hsu T. W., Taylor J. M., Aldrich C., Townsend J. B., Seal G., Mason W. S. Tandem duplication of the proviral DNA in an avian sarcoma virus-transformed quail clone. J Virol. 1981 Apr;38(1):219–223. doi: 10.1128/jvi.38.1.219-223.1981. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jones T. R., Hyman R. W. Specious hybridization between herpes simplex virus DNA and human cellular DNA. Virology. 1983 Dec;131(2):555–560. doi: 10.1016/0042-6822(83)90521-4. [DOI] [PubMed] [Google Scholar]
- Martin M. A., Bryan T., McCutchan T. F., Chan H. W. Detection and cloning of murine leukemia virus-related sequences from African green monkey liver DNA. J Virol. 1981 Sep;39(3):835–844. doi: 10.1128/jvi.39.3.835-844.1981. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Martin M. A., Bryan T., Rasheed S., Khan A. S. Identification and cloning of endogenous retroviral sequences present in human DNA. Proc Natl Acad Sci U S A. 1981 Aug;78(8):4892–4896. doi: 10.1073/pnas.78.8.4892. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mason W. S., Aldrich C., Summers J., Taylor J. M. Asymmetric replication of duck hepatitis B virus DNA in liver cells: Free minus-strand DNA. Proc Natl Acad Sci U S A. 1982 Jul;79(13):3997–4001. doi: 10.1073/pnas.79.13.3997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Maxam A. M., Gilbert W. Sequencing end-labeled DNA with base-specific chemical cleavages. Methods Enzymol. 1980;65(1):499–560. doi: 10.1016/s0076-6879(80)65059-9. [DOI] [PubMed] [Google Scholar]
- May F. E., Westley B. R., Rochefort H., Buetti E., Diggelmann H. Mouse mammary tumour virus related sequences are present in human DNA. Nucleic Acids Res. 1983 Jun 25;11(12):4127–4139. doi: 10.1093/nar/11.12.4127. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mondal H., Hofschneider P. H. Demonstration of free reverse transcriptase in the nuclei of embryonic tissues of the Japanese quail. Biochem Biophys Res Commun. 1983 Oct 14;116(1):303–311. doi: 10.1016/0006-291x(83)90415-1. [DOI] [PubMed] [Google Scholar]
- Moroni C., Schumann G. Are endogenous C-type viruses involved in the immune system? Nature. 1977 Oct 13;269(5629):600–601. doi: 10.1038/269600a0. [DOI] [PubMed] [Google Scholar]
- Morse H. C., 3rd, Chused T. M., Boehm-Truitt M., Mathieson B. J., Sharrow S. O., Hartley J. W. XenCSA: cell surface antigens related to the major glycoproteins (gp70) of xenotropic murine leukemia viruses. J Immunol. 1979 Feb;122(2):443–454. [PubMed] [Google Scholar]
- Noda M., Kurihara M., Takano T. Retrovirus-related sequences in human DNA: detection and cloning of sequences which hybridize with the long terminal repeat of baboon endogenous virus. Nucleic Acids Res. 1982 May 11;10(9):2865–2878. doi: 10.1093/nar/10.9.2865. [DOI] [PMC free article] [PubMed] [Google Scholar]
- O'Brien S. J., Bonner T. I., Cohen M., O'Connell C., Nash W. G. Mapping of an endogenous retroviral sequence to human chromosome 18. Nature. 1983 May 5;303(5912):74–77. doi: 10.1038/303074a0. [DOI] [PubMed] [Google Scholar]
- O'Connell C., O'Brien S., Nash W. G., Cohen M. ERV3, a full-length human endogenous provirus: chromosomal localization and evolutionary relationships. Virology. 1984 Oct 30;138(2):225–235. doi: 10.1016/0042-6822(84)90347-7. [DOI] [PubMed] [Google Scholar]
- Rabson A. B., Steele P. E., Garon C. F., Martin M. A. mRNA transcripts related to full-length endogenous retroviral DNA in human cells. Nature. 1983 Dec 8;306(5943):604–607. doi: 10.1038/306604a0. [DOI] [PubMed] [Google Scholar]
- Repaske R., O'Neill R. R., Steele P. E., Martin M. A. Characterization and partial nucleotide sequence of endogenous type C retrovirus segments in human chromosomal DNA. Proc Natl Acad Sci U S A. 1983 Feb;80(3):678–682. doi: 10.1073/pnas.80.3.678. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Repaske R., Steele P. E., O'Neill R. R., Rabson A. B., Martin M. A. Nucleotide sequence of a full-length human endogenous retroviral segment. J Virol. 1985 Jun;54(3):764–772. doi: 10.1128/jvi.54.3.764-772.1985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rigby P. W., Dieckmann M., Rhodes C., Berg P. Labeling deoxyribonucleic acid to high specific activity in vitro by nick translation with DNA polymerase I. J Mol Biol. 1977 Jun 15;113(1):237–251. doi: 10.1016/0022-2836(77)90052-3. [DOI] [PubMed] [Google Scholar]
- Sanger F., Nicklen S., Coulson A. R. DNA sequencing with chain-terminating inhibitors. Proc Natl Acad Sci U S A. 1977 Dec;74(12):5463–5467. doi: 10.1073/pnas.74.12.5463. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schwartz D. E., Tizard R., Gilbert W. Nucleotide sequence of Rous sarcoma virus. Cell. 1983 Mar;32(3):853–869. doi: 10.1016/0092-8674(83)90071-5. [DOI] [PubMed] [Google Scholar]
- Simon M. C., Smith R. E., Hayward W. S. Mechanisms of oncogenesis by subgroup F avian leukosis viruses. J Virol. 1984 Oct;52(1):1–8. doi: 10.1128/jvi.52.1.1-8.1984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Smith H. O. Recovery of DNA from gels. Methods Enzymol. 1980;65(1):371–380. doi: 10.1016/s0076-6879(80)65048-4. [DOI] [PubMed] [Google Scholar]
- Southern E. M. Detection of specific sequences among DNA fragments separated by gel electrophoresis. J Mol Biol. 1975 Nov 5;98(3):503–517. doi: 10.1016/s0022-2836(75)80083-0. [DOI] [PubMed] [Google Scholar]
- Staden R. Further procedures for sequence analysis by computer. Nucleic Acids Res. 1978 Mar;5(3):1013–1016. doi: 10.1093/nar/5.3.1013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Suzuki A., Kitasato H., Kawakami M., Ono M. Molecular cloning of retrovirus-like genes present in multiple copies in the Syrian hamster genome. Nucleic Acids Res. 1982 Oct 11;10(19):5733–5746. doi: 10.1093/nar/10.19.5733. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Taylor J. M., Cywinski A., Smith J. K. Discontinuities in the DNA synthesized by an avian retrovirus. J Virol. 1983 Dec;48(3):654–659. doi: 10.1128/jvi.48.3.654-659.1983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thomas P. S. Hybridization of denatured RNA transferred or dotted nitrocellulose paper. Methods Enzymol. 1983;100:255–266. doi: 10.1016/0076-6879(83)00060-9. [DOI] [PubMed] [Google Scholar]
- Vogt P. K. A virus released by "nonproducing" Rous sarcoma cells. Proc Natl Acad Sci U S A. 1967 Sep;58(3):801–808. doi: 10.1073/pnas.58.3.801. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wilbur W. J., Lipman D. J. Rapid similarity searches of nucleic acid and protein data banks. Proc Natl Acad Sci U S A. 1983 Feb;80(3):726–730. doi: 10.1073/pnas.80.3.726. [DOI] [PMC free article] [PubMed] [Google Scholar]