Abstract
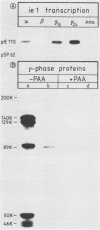
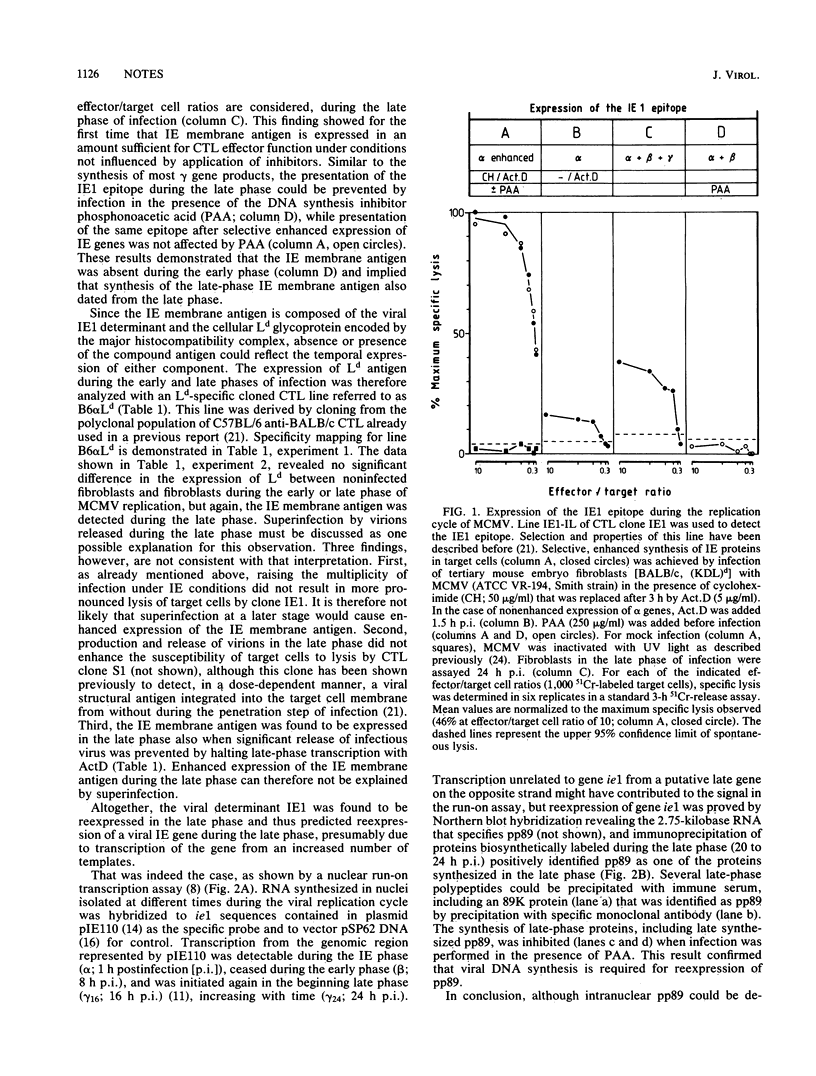
The cloned murine cytolytic T-lymphocyte line IE1-IL and several sublines detect a murine cytomegalovirus immediate-early (IE) membrane determinant in conjunction with Ld class I major histocompatibility glycoprotein. The lines retained cytolytic activity, strict antigen specificity, and self-restriction even when adapted to long-term, antigen-independent growth in the presence of interleukin-2 only (M. J. Reddehase, H.-J. Bühring, and U. H. Koszinowski, J. Virol. 57:408-412). These attributes allowed us to use IE1-IL as a stable, monospecific probe for tracing the expression of the IE membrane antigen throughout the viral replication cycle. Presentation of the antigen at the cell membrane proved to be most effective when expression of IE genes in infected mouse embryo fibroblasts was selectively enhanced by consecutive cycloheximide-actinomycin D treatment, whereas without enhancement high numbers of IE1-IL cytolytic T lymphocytes were required to demonstrate the antigen in the IE phase. In the early phase of infection when IE genes were no longer transcribed, cytolysis was not observed, although IE proteins were detectable in the nuclei of the infected cells. Without application of inhibitors IE membrane antigen expression was most prominent during the late phase of infection. Reinitiation of transcription from the genomic region encoding the major IE protein (pp89) and de novo synthesis of pp89 correlated with this reexpression of the IE membrane antigen.
Full text
PDF



Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Borysiewicz L. K., Morris S., Page J. D., Sissons J. G. Human cytomegalovirus-specific cytotoxic T lymphocytes: requirements for in vitro generation and specificity. Eur J Immunol. 1983 Oct;13(10):804–809. doi: 10.1002/eji.1830131005. [DOI] [PubMed] [Google Scholar]
- Brautigam A. R., Dutko F. J., Olding L. B., Oldstone M. B. Pathogenesis of murine cytomegalovirus infection: the macrophage as a permissive cell for cytomegalovirus infection, replication and latency. J Gen Virol. 1979 Aug;44(2):349–359. doi: 10.1099/0022-1317-44-2-349. [DOI] [PubMed] [Google Scholar]
- Brody A. R., Craighead J. E. Pathogenesis of pulmonary cytomegalovirus infection in immunosuppressed mice. J Infect Dis. 1974 Jun;129(6):677–689. doi: 10.1093/infdis/129.6.677. [DOI] [PubMed] [Google Scholar]
- Bukowski J. F., Warner J. F., Dennert G., Welsh R. M. Adoptive transfer studies demonstrating the antiviral effect of natural killer cells in vivo. J Exp Med. 1985 Jan 1;161(1):40–52. doi: 10.1084/jem.161.1.40. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dorsch-Häsler K., Keil G. M., Weber F., Jasin M., Schaffner W., Koszinowski U. H. A long and complex enhancer activates transcription of the gene coding for the highly abundant immediate early mRNA in murine cytomegalovirus. Proc Natl Acad Sci U S A. 1985 Dec;82(24):8325–8329. doi: 10.1073/pnas.82.24.8325. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dutko F. J., Oldstone M. B. Cytomegalovirus causes a latent infection in undifferentiated cells and is activated by induction of cell differentiation. J Exp Med. 1981 Nov 1;154(5):1636–1651. doi: 10.1084/jem.154.5.1636. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ebeling A., Keil G. M., Knust E., Koszinowski U. H. Molecular cloning and physical mapping of murine cytomegalovirus DNA. J Virol. 1983 Sep;47(3):421–433. doi: 10.1128/jvi.47.3.421-433.1983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Greenberg M. E., Ziff E. B. Stimulation of 3T3 cells induces transcription of the c-fos proto-oncogene. Nature. 1984 Oct 4;311(5985):433–438. doi: 10.1038/311433a0. [DOI] [PubMed] [Google Scholar]
- Honess R. W., Roizman B. Regulation of herpesvirus macromolecular synthesis. I. Cascade regulation of the synthesis of three groups of viral proteins. J Virol. 1974 Jul;14(1):8–19. doi: 10.1128/jvi.14.1.8-19.1974. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jordan M. C., Takagi J. L., Stevens J. G. Activation of latent murine cytomegalovirus in vivo and in vitro: a pathogenetic role for acute infection. J Infect Dis. 1982 May;145(5):699–705. doi: 10.1093/infdis/145.2.699. [DOI] [PubMed] [Google Scholar]
- Keil G. M., Ebeling-Keil A., Koszinowski U. H. Temporal regulation of murine cytomegalovirus transcription and mapping of viral RNA synthesized at immediate early times after infection. J Virol. 1984 Jun;50(3):784–795. doi: 10.1128/jvi.50.3.784-795.1984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Keil G. M., Fibi M. R., Koszinowski U. H. Characterization of the major immediate-early polypeptides encoded by murine cytomegalovirus. J Virol. 1985 May;54(2):422–428. doi: 10.1128/jvi.54.2.422-428.1985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kim K. S., Sapienza V. J., Carp R. I., Moon H. M. Analysis of structural proteins of purified murine cytomegalovirus. J Virol. 1976 Mar;17(3):906–915. doi: 10.1128/jvi.17.3.906-915.1976. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Koszinowski U. H., Keil G. M., Volkmer H., Fibi M. R., Ebeling-Keil A., Münch K. The 89,000-Mr murine cytomegalovirus immediate-early protein activates gene transcription. J Virol. 1986 Apr;58(1):59–66. doi: 10.1128/jvi.58.1.59-66.1986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mayo D. R., Armstrong J. A., Ho M. Reactivation of murine cytomegalovirus by cyclophosphamide. Nature. 1977 Jun 23;267(5613):721–723. doi: 10.1038/267721a0. [DOI] [PubMed] [Google Scholar]
- Melton D. A., Krieg P. A., Rebagliati M. R., Maniatis T., Zinn K., Green M. R. Efficient in vitro synthesis of biologically active RNA and RNA hybridization probes from plasmids containing a bacteriophage SP6 promoter. Nucleic Acids Res. 1984 Sep 25;12(18):7035–7056. doi: 10.1093/nar/12.18.7035. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Meyers J. D., Flournoy N., Thomas E. D. Nonbacterial pneumonia after allogeneic marrow transplantation: a review of ten years' experience. Rev Infect Dis. 1982 Nov-Dec;4(6):1119–1132. doi: 10.1093/clinids/4.6.1119. [DOI] [PubMed] [Google Scholar]
- O'Connell K. A., Gooding L. R. Cloned cytotoxic T lymphocytes recognize cells expressing discrete fragments of the SV40 tumor antigen. J Immunol. 1984 Feb;132(2):953–958. [PubMed] [Google Scholar]
- Ponta H., Ball R., Steinmetz M., Groner B. Hormonal regulation of cell surface expression of the major histocompatibility antigen H-2Ld in transfected cells. EMBO J. 1985 Dec 16;4(13A):3447–3453. doi: 10.1002/j.1460-2075.1985.tb04103.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Quinnan G. V., Manischewitz J. E., Ennis F. A. Cytotoxic T lymphocyte response to murine cytomegalovirus infection. Nature. 1978 Jun 15;273(5663):541–543. doi: 10.1038/273541a0. [DOI] [PubMed] [Google Scholar]
- Reddehase M. J., Bühring H. J., Koszinowski U. H. Cloned long-term cytolytic T-lymphocyte line with specificity for an immediate-early membrane antigen of murine cytomegalovirus. J Virol. 1986 Jan;57(1):408–412. doi: 10.1128/jvi.57.1.408-412.1986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Reddehase M. J., Keil G. M., Koszinowski U. H. The cytolytic T lymphocyte response to the murine cytomegalovirus. I. Distinct maturation stages of cytolytic T lymphocytes constitute the cellular immune response during acute infection of mice with the murine cytomegalovirus. J Immunol. 1984 Jan;132(1):482–489. [PubMed] [Google Scholar]
- Reddehase M. J., Keil G. M., Koszinowski U. H. The cytolytic T lymphocyte response to the murine cytomegalovirus. II. Detection of virus replication stage-specific antigens by separate populations of in vivo active cytolytic T lymphocyte precursors. Eur J Immunol. 1984 Jan;14(1):56–61. doi: 10.1002/eji.1830140111. [DOI] [PubMed] [Google Scholar]
- Reddehase M. J., Koszinowski U. H. Significance of herpesvirus immediate early gene expression in cellular immunity to cytomegalovirus infection. Nature. 1984 Nov 22;312(5992):369–371. doi: 10.1038/312369a0. [DOI] [PubMed] [Google Scholar]
- Reddehase M. J., Weiland F., Münch K., Jonjic S., Lüske A., Koszinowski U. H. Interstitial murine cytomegalovirus pneumonia after irradiation: characterization of cells that limit viral replication during established infection of the lungs. J Virol. 1985 Aug;55(2):264–273. doi: 10.1128/jvi.55.2.264-273.1985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rice G. P., Schrier R. D., Oldstone M. B. Cytomegalovirus infects human lymphocytes and monocytes: virus expression is restricted to immediate-early gene products. Proc Natl Acad Sci U S A. 1984 Oct;81(19):6134–6138. doi: 10.1073/pnas.81.19.6134. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shanley J. D., Pesanti E. L., Nugent K. M. The pathogenesis of pneumonitis due to murine cytomegalovirus. J Infect Dis. 1982 Sep;146(3):388–396. doi: 10.1093/infdis/146.3.388. [DOI] [PubMed] [Google Scholar]
- Tevethia S. S., Lewis A. J., Campbell A. E., Tevethia M. J., Rigby P. W. Simian virus 40 specific cytotoxic lymphocyte clones localize two distinct TSTA sites on cells synthesizing a 48 kD SV40 T antigen. Virology. 1984 Mar;133(2):443–447. doi: 10.1016/0042-6822(84)90411-2. [DOI] [PubMed] [Google Scholar]
- Townsend A. R., Gotch F. M., Davey J. Cytotoxic T cells recognize fragments of the influenza nucleoprotein. Cell. 1985 Sep;42(2):457–467. doi: 10.1016/0092-8674(85)90103-5. [DOI] [PubMed] [Google Scholar]
- Townsend A. R., Rothbard J., Gotch F. M., Bahadur G., Wraith D., McMichael A. J. The epitopes of influenza nucleoprotein recognized by cytotoxic T lymphocytes can be defined with short synthetic peptides. Cell. 1986 Mar 28;44(6):959–968. doi: 10.1016/0092-8674(86)90019-x. [DOI] [PubMed] [Google Scholar]
- Wathen M. W., Stinski M. F. Temporal patterns of human cytomegalovirus transcription: mapping the viral RNAs synthesized at immediate early, early, and late times after infection. J Virol. 1982 Feb;41(2):462–477. doi: 10.1128/jvi.41.2.462-477.1982. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Watson R. J., Clements J. B. A herpes simplex virus type 1 function continuously required for early and late virus RNA synthesis. Nature. 1980 May 29;285(5763):329–330. doi: 10.1038/285329a0. [DOI] [PubMed] [Google Scholar]
- Yewdell J. W., Bennink J. R., Smith G. L., Moss B. Influenza A virus nucleoprotein is a major target antigen for cross-reactive anti-influenza A virus cytotoxic T lymphocytes. Proc Natl Acad Sci U S A. 1985 Mar;82(6):1785–1789. doi: 10.1073/pnas.82.6.1785. [DOI] [PMC free article] [PubMed] [Google Scholar]