Abstract
The terpene synthase encoded by the SCO5222 (SC7E4.19) gene ofStreptomyces coelicolor was cloned by PCR and expressed inEscherichia coli as an N-terminal-His6-tag protein. Incubation of the recombinant protein, SCO5222p, with farnesyl diphosphate (1, FPP) in the presence of Mg(II) gave a new sesquiterpene, (+)-epi-isozizaene (2), whose structure and stereochemistry were determined by a combination of1H,13C, COSY, HMQC, HMBC, and NOESY NMR. The steady state kinetic parameters werekcat 0.049 ± 0.001 s−1 and aKm (FPP) of 147 ± 14 nM. Individual incubations of recombinant epiisozizaene synthase with {1,1-2H2}FPP (1a), (1R)-{1-2H}-FPP (1b), and (1 S)-{1-2H}-FPP (1c) and NMR analysis of the resulting deuterated epi-isozizaenes supported an isomerization-cyclization-rearrangement mechanism involving the intermediacy of (3R)-nerolidyl diphosphate (3).
Streptomyces are Gram-positive, filamentous, soil-dwelling saprophytic bacteria that are the source of a vast array of bioactive secondary metabolites, including antibiotics, immunosuppressants, and anticancer agents.1 Among the thousands of such natural products are only a relative handful of cyclic terpenoids. For example the antibiotic pentalenolactone, which is produced by a large number of Streptomyces species, is derived from the sesquiterpene hydrocarbon, pentalenene.2 Pentalenene itself is formed by the cyclization of farnesyl diphosphate (1, FPP), the universal acyclic precursor of more than 300 distinct sesquiterpenes. Pentalenene synthase from Streptomyces sp. UC5319 has been cloned and expressed, and the structure determined by X-ray crystallography.3a We have also recently cloned and expressed a closely related pentalenene synthase from S. avermitilis.3 b
Streptomyces coelicolor A3(2) is genetically the most thoroughly studied member of the genus.4 The 8.7 Mb linear chromosome harbors 7,825 open-reading frames (ORFs), among which at least three show significant levels of similarity to pentalenene synthase.5 The 2-kb SCO6073 (SC9B1.20) encodes a protein of 726 amino acids that catalyzes the cyclization of FPP to a mixture of germacradienol and germacrene D, implicated in the biosynthesis of geosmin, a degraded sesquiterpene that is responsible for the characteristic odor of soil.6 We now report the molecular cloning and biochemical characterization of a second S. coelicolor terpene synthase, SCO5222 (SC7E4.19) that is shown to catalyze the Mg2+-dependent cyclization of FPP (1) to a novel sesquiterpene, epi-isozizaene (2) (Scheme 1).
Scheme 1.
Cyclization of farnesyl diphosphate (1) to epi-isozizaene (2)
The 361 amino acid (aa) SCO5222 protein has 23.8% amino acid sequence identity and 51.1% similarity over 311 aa with the 336-aa pentalenene synthase of Streptomyces UC5319. Importantly, the protein encoded by SCO5222 includes the two universally conserved Mg2+-binding domains, the asparate-rich motif DDRHD, beginning at Asp99, and the characteristic triad NDLCSLPKE, at Asn240. We used PCR to amplify the 1,086-bp ORF of SCO5222 S. coelicolor cosmid SC7E4 DNA as template, while introducing NdeI and XhoI restrictions sites at the 5′- and 3′-ends, respectively. The doubly-digested DNA amplicon was ligated into the corresponding sites of the expression vector pET28a(+), and the ligation mixture was used to transform competent cells of Escherichia coli XL1-Blue. The resultant plasmid, pET28a/SCO5222 was introduced into the T7-RNA polymerase-based expression host, E. coli BL21(DE3), which was grown to an OD600 of 0.5, then induced with 0.1 mM IPTG. After further incubation at 20 °C for 18 h, the resultant soluble N-terminal His6-tag protein was harvested and purified to homogeneity by Ni2+-affinity chromatography to yield 13 mg protein/L culture with M D (MALDI-TOF) 43665±22 (calc. His6-tag-SCO5222 43650). Incubation of the purified SCO5222 protein with FPP (1) and analysis by GC-MS revealed the formation of a single sesquiterpene hydrocarbon product, C15H24, m/z 204.
The steady-state kinetic parameters were determined by carrying out a series of 15-min incubations at 30 °C in pH 6.5 PIPES using 83 nM - 2.49 μM [1-3H]-FPP (164 mCi/mmol), giving a kcat of 0.049±0.001 s−1 and a Km (FPP) of 147±14 nM.7
Preparative-scale (500-mL) incubations were carried out for 18 h at 30 °C using 12.6 mg (30 μmol) of FPP and a total of 0.6 μmol of recombinant SCO5222 protein, added in equal batches at 0, 6, and 12 h. The hydrocarbon product was extracted into HPLC-grade pentane and purified by SiO2 chromatography, yielding 3–4 mg of 2 that was >98% pure by GC-MS. The structure and stereochemistry of the enzymatic cyclization product were determined by a combination of 1H, 1 3C, 1H-1H COSY, NOESY, HMQC, and HMBC NMR (Figure 1). The presence of a fully substituted double bond in 2 was indicated by the observation of two quaternary olefinic carbons at 127.4 (C-3) and 143.0 ppm (C-4) in the 75.48 MHz 1 3C NMR spectrum and the absence of any signals due to olefinic protons in the 300 MHz 1H NMR spectrum. Four methyl groups were readily identified: a geminal methyl pair (H-12, δ 0.99; C-12, 28.3 ppm and H-13, δ 0.96; C-13 25.0 ppm) attached to a quaternary carbon (C-2, 40.4 ppm); an allylic methyl (H-14, δ 1.42; C-14, 12.8 ppm) attached to the olefinic C-3 carbon at 127.4 ppm; and a secondary methyl (H-15, δ 0.90, d, J=6.3 Hz; C-15, 14.0 ppm) attached to a C–H (H-7, δ 1.77, m; C-7, 39.7 ppm). The 1H-1H COSY spectrum revealed long range couplings between the allylic H-14 methyl and each of the allylic H-5 methylene protons at δ 2.06 and 2.2, both of which were correlated with C-5 (27.3 ppm). The HMBC spectrum also revealed crosspeaks between each of the geminal methyl protons and the olefinic C-3 signal at 127.4 ppm as well as a second set of crosspeaks with the C-1 methine (47.1 ppm; H-1,δ 1.81). The secondary methyl (H-15) also showed a 3-bond HMBC crosspeak to C-8 (52.6 ppm). The remaining 4 methylene carbons each carried a pair of diastereotopic protons, which were identified by their characteristic sets of crosspeaks in the HMQC, COSY, and NOESY spectra and assigned based on the observed coupling constants and the NOESY spectrum.8 The configuration of the secondary methyl group in 2 was unambiguously assigned as anti to the C-11 methylene bridge, based on the strong 600 MHz NOESY crosspeak between H-15 (δ 0.90) and H-9b (δ 1.35). A weaker NOESY crosspeak was also observed between H-15 and H-11anti (δ 1.47), as well as a faint correlation to H-9a (δ 1.16). On the other hand, there was no detectable NOE interaction between H-15 and H-11syn (δ 1.39), which itself was unambiguously assigned based on its NOESY crosspeak with the H-12 exo-methyl singlet at δ 0.99.
Figure 1.
NOESY (A) and COSY (B) correlations for epi-isozizaene (2). Correlations with H-6b (δ 1.77) and with H–7 (δ 1.77) are not assigned due to signal overlap. s, strong; w, weak.
Although the enzymatic cyclization product 2 has not previously been isolated as a natural product, the structure and relative stereochemistry of 2 correspond to the known compound (−)-epi-isozizaene, originally synthesized by formic acid-catalyzed rearrangement of (+)-zizaene.9,10 Enzymatically generated (+)-(1S,7S,8R)-epi-isozizaene is enantiomeric to (−)-2 prepared from (+)-zizaene, based on the [α]D +21° (CHCl3, c=0.10) ((−)-2, [α]D −17° (CHCl3, c=0.10)9a).
The formation of epi-isozizaene by cyclization of FPP can be readily explained by the mechanism illustrated in Scheme 2. Ionization and isomerization of FPP (1) will give (3R)-nerolidyl diphosphate (NPP, 3). Rotation about the newly generated C-2–C-3 bond generates the corresponding cisoid NPP conformer which can undergo ionization and cyclization to form bisabolyl cation 4. Following 1,2-hydride shift and spirocyclization, the resultant acorenyl cation 5 can undergo further cyclization, ring contraction, methyl migration, and deprotonation to yield (+)-epi-isozizaene (2). The proposed mechanism was tested by incubation of individual samples of [1,1-2H2]-FPP (1a), (1R)-[1-2H]-FPP (1b), and (1S)-[1-2H]-FPP (1c),2 with recombinant epi-isozizaene synthase and NMR analysis of the resultant labeled 2. Epi-isozizaene (2a) derived from [1,1-2H2]-FPP (1a) lacked the characteristic H-11 signals at δ 1.39 and 1.47, as well as both the corresponding pair of crosspeaks in the COSY spectrum. Similarly, both of the crosspeaks to C-11 (36.9 ppm) were absent from the HMQC spectrum. By contrast, cyclization of (1R)-[1-2H]-FPP (1b) gave 2b lacking H-11anti (δ 1.47) while displaying a broad singlet for H-11syn shifted upfield to δ 1.36 due to isotope effect of the geminal deuterium. 2b also had a single H-11syn/C-11 crosspeak in the HSQC spectrum, shifted upfield 0.4 ppm in the 1 3C dimension to 36.5 ppm by the {2H1 3C} isotope effect.1 1 Similarly, the sample of 2c derived from (1S)-[1-2H]-FPP (1c) lacked H-11syn, while displaying H-11anti as a broadened doublet (J= 5.3 Hz, δ 1.45). The COSY spectrum of 2c showed the expected H-11anti/H-1 crosspeak, while the HSQC spectrum retained a single crosspeak between H-11anti and C-11 (36.3 ppm). The combined labeling results are completely consistent with the stereochemical model illustrated in Scheme 2, confirming the expected net retention of configuration in the displacement of the pyrophosphate group of FPP that reflects the intermediacy of (3R)-NPP (3).
Scheme 2.
Mechanism of the cyclization of farnesyl diphosphate (1) to epi-isozizaene (2)
Supplementary Material
Supporting Information Available: Sequence alignments, expression plasmid construction, protein expression and assay, NMR spectroscopic data for 2, including labeling by deuterated FPP and full citation for Ref 5. This material is available free of charge via the Internet at http://pubs.acs.org. See any current masthead page for ordering information and Web access instructions.
Acknowledgments
We thank Dr. Tun-Li Shen for assistance in obtaining the mass spectra, Prof. Keith Chater of the John Innes Centre for a gift of S. coelicolor cosmid SC7E4, Dr. Gregory L. Challis of the University of Warwick for providing S. coelicolor M145, and Prof. Niels H. Andersen of the University of Washington for providing the original NMR and IR spectra of (−)-epi-isozizaene and (+)-isozizaene. This work was supported by NIH grant GM30301 to DEC.
References
- 1.Hodgson DA. Adv Microb Physiol. 2000;42:47–238. doi: 10.1016/s0065-2911(00)42003-5. [DOI] [PubMed] [Google Scholar]
- 2.Cane DE, Oliver JS, Harrison PHM, Abell C, Hubbard BR, Kane CT, Lattman R. J Am Chem Soc. 1990;112:4513–4524. [Google Scholar]
- 3.(a) Cane DE, Sohng JK, Lamberson CR, Rudnicki SM, Wu Z, Lloyd MD, Oliver JS, Hubbard BR. Biochemistry. 1994;33:5846–5857. doi: 10.1021/bi00185a024. [DOI] [PubMed] [Google Scholar]; Lesburg CA, Zhai G, Cane DE, Christianson DW. Science. 1997;277:1820–1824. doi: 10.1126/science.277.5333.1820. [DOI] [PubMed] [Google Scholar]; (b) Tetzlaff CN, You Z, Cane DE, Takamatsu S, Omura S, Ikeda H. Biochemistry. 2006 doi: 10.1021/bi060419n. in press. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Hopwood DA. Microbiology. 1999;145:2183–2202. doi: 10.1099/00221287-145-9-2183. [DOI] [PubMed] [Google Scholar]
- 5.Hopwood DA, et al. Nature. 2002;417:141–147. [Google Scholar]
- 6.(a) Cane DE, Watt RM. Proc Natl Acad Sci U S A. 2003;100:1547–1551. doi: 10.1073/pnas.0337625100. [DOI] [PMC free article] [PubMed] [Google Scholar]; He X, Cane DE. J Am Chem Soc. 2004;126:2678–2679. doi: 10.1021/ja039929k. [DOI] [PubMed] [Google Scholar]; (b) Gust B, Challis GL, Fowler K, Kieser T, Chater KF. Proc Natl Acad Sci U S A. 2003;100:1541–1546. doi: 10.1073/pnas.0337542100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Substitution of 10 mM Mn2+ or Fe3+ for Mg2+gave relative rates of formation of 2 of 72% and 85%, respectively. Other divalent cations were significantly less effective: Zn2+ (9%), Fe2+ (8%), Co2+ (4%), Cu2+ (4%), and Ni2= (1%).
- 8.The observed coupling constants were fully consistent with the conformation of 2 calculated using MM+ with HyperChem 7.5 and HyperNMR. See Supporting Information for detailed NMR assignments and spectra.
- 9.Andersen NH, Smith SE, Ohta Y. J Chem Soc, Chem Commun. 1973:447–448.The 1H NMR spectrum of enzymatically-generated 2 matched that of synthetic 2, and was readily distinguished from that of isozizaene. See Supporting Information for details. (b) Rearrangement of (−)-α-cedrene in superacid also gives (−)-epi-isozizaene, whose reported 1H and 13C NMR spectra are identical to those determined for enzymatically-generated 2. ( Polovinka MP, Korchagina DV, Shcherbukhin VV, Gatilov YV, Rybalova TV, Zefirov NS, Barkhash VA. Tetrahedron Lett. 1995;36:8093–8096.Because Andersen had shown, however, that the secondary methyl group of epi-isozizaene can epimerize under strong acid conditions, the configuration of the C-15 methyl group could not be assumed a priori to be the same as that in (−)-α-cedrene.
-
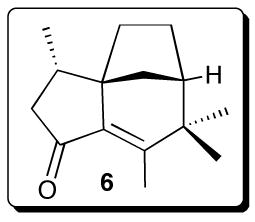
10.Although epi-isozizaene (2) has not been previously reported as a natural product, the corresponding 5-keto derivative, albaflavenone (6), of unassigned absolute configuration, has recently been isolated from a species of S. albidoflavus ( Gurtler H, Pedersen R, Anthoni U, Christophersen C, Nielsen PH, Wellington EMH, Pedersen C, Bock K. J Antibiot. 1994;47:434–439. doi: 10.7164/antibiotics.47.434.Albaflavenone is reported to have antibiotic activity and an “earthy, camphor-like” odor. We have as yet been unable to detect either 2 or albaflavenone by GC-MS in the volatile extracts of either submerged or surface cultures of S. coelicolor M145.
- 11.Garson MJ, Staunton J. Chem Soc Rev. 1979:539–561. [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Supporting Information Available: Sequence alignments, expression plasmid construction, protein expression and assay, NMR spectroscopic data for 2, including labeling by deuterated FPP and full citation for Ref 5. This material is available free of charge via the Internet at http://pubs.acs.org. See any current masthead page for ordering information and Web access instructions.


