Abstract
Apolipoprotein A-V (apoA-V) is present in low amounts in plasma and has been found to modulate triacylglycerol levels in humans and in animal models. ApoA-V displays affinity for members of the low density lipoprotein receptor (LDL-R) gene family, known as the classical lipoprotein receptors, including LRP1 and SorLA/LR11. In addition to LDL-A binding repeats, the mosaic receptor SorLA/LR11 also possesses a Vps10p domain. Here we show that apoA-V also binds to sortilin, a receptor from the Vsp10p domain gene family that lacks LDL-A repeats. Binding of apoA-V to sortilin was competed by neurotensin, a ligand that binds specifically to the Vps10p domain. To investigate the biological fate of receptor-bound apoA-V, binding experiments were conducted with cultured human embryonic kidney cells transfected with either SorLA/LR11 or sortilin. Compared with nontransfected cells, apoA-V binding to SorLA/LR11- and sortilin-expressing cells was markedly enhanced. Internalization experiments, live imaging studies, and fluorescence resonance energy transfer analyses demonstrated that labeled apoA-V was rapidly internalized, co-localized with receptors in early endosomes, and followed the receptors through endosomes to the trans-Golgi network. The observed decrease of fluorescence signal intensity as a function of time during live imaging experiments suggested ligand uncoupling in endosomes with subsequent delivery to lysosomes for degradation. This interpretation was supported by experiments with 125I-labeled apoA-V, demonstrating clear differences in degradation between transfected and nontransfected cells. We conclude that apoA-V binds to receptors possessing LDL-A repeats and Vsp10p domains and that apoA-V is internalized into cells via these receptors. This could be a mechanism by which apoA-V modulates lipoprotein metabolism in vivo.
APOA5 is localized in the apolipoprotein APOA4/APOC3/APOA1 gene cluster on human chromosome 11q23 (1). Transgenic mice expressing human apolipoprotein A-V (apoA-V) have decreased plasma triacylglycerol (TG)2 levels, whereas APOA5 knock-out mice show increased plasma TG levels. Genetic variation in the human APOA5 locus correlate with changes in plasma lipoprotein levels (2–4), and a common polymorphism in APOA5 is significantly associated with increased risk for the metabolic syndrome (5, 6). Mutations in the APOA5 gene, leading to truncated apoA-V devoid of lipid-binding domains, have been demonstrated to cause severe hyperlipidemia if present in patients in the homozygous state (7).
The mechanism whereby apoA-V exerts its effect on plasma TG levels is unknown. Animal studies and in vitro experiments have given rise to two different hypotheses for the function of apoA-V in vivo, inhibition of VLDL-TG production (8) or modulation of lipoprotein lipase (LPL) activity (8–11). Adenovirus-mediated gene transfer of human apoA-V into apoE-deficient mice decreased plasma TG levels as well as total plasma cholesterol levels without affecting LPL activity (12). In vitro experiments support a role for apoA-V as an activator of LPL but only if the lipase is bound to heparan sulfate proteoglycans. Lookene et al. (13) showed that apoA-V binds to heparin and facilitates binding of chylomicrons and VLDL to heparin by bridging the lipoproteins to the glycan chains. In accordance, Merkel et al. found that apoA-V stimulates lipolysis when the lipase is bound to heparan sulfate proteoglycans on microtiter plates (9).
ApoA-V is present in plasma on chylomicrons, VLDL, and high density lipoprotein but in minute amounts compared with other apolipoproteins (14). Values range between 150 and 200 ng/ml. These levels correspond to less than 1% of the level of apoC-III and would be sufficient for approximately one molecule of apoA-V per 20 VLDL particles in plasma (15). The physiological relevance of apoA-V modulation of LPL-mediated lipolysis of TG-rich lipoproteins has therefore been questioned. Another as yet unexplained observation is that apoA-V levels in human plasma are positively correlated with plasma TG levels (16, 17).
ApoA-V was previously shown to bind to receptors of the low density lipoprotein receptor (LDL-R) gene family (18). Both lipid-free apoA-V and apoA-V dimyristoylphosphatidylcholine (DMPC) complexes bound to LRP1 (LDL receptor-related protein I) and to the mosaic receptor SorLA/LR11. ApoA-V was able to bridge chylomicrons to these receptors, implying that apoA-V has a role in receptor-mediated clearance of TG-rich lipoprotein remnants (18). These results were supported by the finding that avian apoA-V binds to the LDL receptor homologue, LR8 (19) and that VLDL particles from apoa5–/– mice display lower affinity for the LDL-R than VLDL from wild-type mice (20). In addition, human apoA-V was recently shown to bind to the newly discovered LPL-binding protein GPIHBP1 (glycosylphosphatidylinositol-anchored high density lipoprotein-binding protein 1) (21).
SorLA/LR11 (250 kDa) and sortilin (110 kDa) are both Type-1 receptors that belong to the Vps10p domain (Vps10p-D) receptor family (22, 23). Sortilin is present within a novel locus on chromosome 1p13.3 and appears to be strongly related to LDL cholesterol (24, 25) and coronary artery disease (26). Besides the N-terminal Vps10p-D that is present in all five Vps10-D receptors, SorLA/LR11 (from now on referred to as SorLA) also shares structural similarities with members of the LDL-R gene family (27). The Vps10p-D contains a propeptide that is removed by the endopeptidase, furin, in the trans-Golgi network (TGN), and the propeptide is essential for normal processing of sortilin (28). SorLA and sortilin are multiligand receptors, and among the ligands that bind to both receptors are receptor-associated protein (RAP), apoE, and LPL.
Using surface plasmon resonance (SPR) spectroscopy, we examined the binding of apoA-V to sortilin and investigated the effects of other known sortilin ligands on this interaction. Furthermore, interaction between human chylomicrons and sortilin was demonstrated. Binding and internalization studies in human embryonic kidney (HEK) 293 cells transfected with sortilin or SorLA showed that apoA-V undergoes receptor-mediated endocytosis. The endocytotic itinerary of apoA-V was investigated and characterized.
EXPERIMENTAL PROCEDURES
Materials—Soluble sortilin was expressed in CHO-K1 cells and purified on a RAP-Sepharose 4B column as previously described (29). ApoC-II was purified from human plasma (30). RAP was expressed as a His6-tagged fusion protein in Bl21 (DE3) cells and purified using a Talon metal affinity resin according to the manufacturer's manual (Clontech). Neurotensin (NT) and bovine serum albumin fraction V were purchased from Sigma, and heparin was obtained from Leo Pharma (Malmö, Sweden). LRP1 was purified as previously stated (18).
DNA Constructs, Cell Lines, and Transfection—HEK293 cells were cultured in Dulbecco's modified Eagle's medium (Bio-Whittaker, Veriers, Belgium) supplemented with 10% fetal calf serum (Invitrogen). Construction of pcDNA3.1(–) zeo plasmids harboring wild type or mutant SorLA and sortilin generation of stable transfectants have been described (29, 31).
ApoA-V-DMPC Disks—Recombinant human apoA-V was prepared, isolated, and incorporated into DMPC disks, as described elsewhere (32). FLAG-tagged recombinant apoA-V and DMPC disks were prepared similarly (33). The apoA-V-DMPC disks were dialyzed in 20 mm Hepes buffer, pH 7.4, 150 mm NaCl, 2.5 mm CaCl using tubes with a cut-off of 14 kDa (SpectraPor). An Alexa Fluor 488 protein labeling kit from Invitrogen was used to label apoA-V when the protein was present in DMPC disks. For degradation studies, apoA-V-DMPC discs were labeled with 125I using IODO-BEADs (Pierce) according to the manufacturer's instructions. The labeling corresponded to 260 cpm/ng apoA-V protein.
Internalization and Ligand Binding—Alexa Fluor 488-conjugated apoA-V or 125I-labeled apoA-V (∼95,000 cpm/well) was incubated with HEK cells (wild type or transfected with SorLA or sortilin constructs) in 300 μl of Dulbecco's modified Eagle's medium (+ 10% fetal calf serum) for 90 min at 4 °C. Unbound Alexa Fluor 488-conjugated apoA-V was removed, and the cells were then either directly fixed with 4% paraformaldehyde to visualize binding of the ligand to the cell surface or further incubated at 37 °C for given periods of time to study internalization. Unbound 125I-labeled apoA-V remaining in the medium was removed and precipitated with 12.5% tricloroacetic acid to estimate degradation. Cell-associated tracer was released from the plastic dish with 1 m NaOH and counted directly without precipitation.
Immunocytochemistry on cells stably transfected with SorLA or sortilin was made with polyclonal antibodies raised in rabbits against the SorLA Vps10p domain or monoclonal antibodies raised in mouse against the sortilin-Vps10p domain. Cell organelles were visualized using rabbit-anti-Lamp-1 (GLA7; Hybridoma Bank, Iowa), mouse anti-MPR300 (2C2; personal gift from Prof. Regina Pohlmann, Münster, Germany), or goat-anti-TGN-46 (Serotec, Norway). All secondary Alexa Fluor 488-conjugated immunoglobolins were from Invitrogen.
Microscopy of stained fixed cells and live imaging were carried out using a confocal LSM510 META laser-scanning microscope from Zeiss. A ×63 or ×40 water objective lens (numerical aperture 1.2) was used in all recordings. Brightness and contrast were adjusted using NIH ImageJ software.
Fluorescence Resonance Energy Transfer (FRET)—For FRET measurements, cultured HEK293 cell samples transfected with SorLA or sortilin were incubated with apoA-V488 for 20 min and then incubated with anti-SorLA or anti-sortilin, followed by Alexa 568-labeled secondary antibodies. FRET measurements and analysis were performed according to the sensitized acceptor emission PFRET procedure using the associated ImageJ PFRET plugin developed by the group of Periasamy et al. (34, 35). Donor and acceptor fluorescence signals were detected in the 501–533 nm and 565–651 nm spectral regions, and images were acquired with zoom 4 and line average 8 (scan speed 7). 10 donor (a), acceptor (d), and FRET (b and c) images were acquired for each donor- and acceptor-only sample, whereas 13–15 e, f, and g images were acquired for the double-labeled samples (for nomenclature, see Ref. 34). For calculation of the apparent FRET effeciencies, Eapp, the PFRET program contains an option for selection of lower and upper thresholds (bounds) on intensity values in the e, f, and g images acquired for double-labeled samples. For those images, upper bounds were set one unit lower than the (eventual) saturated intensity. Lower bounds were empirically chosen as 15 and 20 intensity units for sortilin-ApoA-V and Sorla-ApoA-V, respectively; this choice was judged to represent the best compromise between an increasing amount of noisy (high) Eapp values appearing at low e, f, and g signal levels versus an increasing loss of information about FRET behavior in cellular regions with low staining efficiency.
For further quantitative FRET analysis, ROIs of size 5 × 5, 4 × 6, or 6 × 4 pixels were defined automatically (SorLA-apoA-V) or manually (sortilin-apoA-V) within e images based on the criteria of an average e signal of at least 25 within the ROIs and a fulfillment of the lower bound settings for at least 80% of the pixels within an ROI. Since negative Eapp values are rounded up to zero by the PFERT program, we decided to exclude those values from the results.
Surface Plasmon Resonance—Binding studies were performed on a Biacore 2000 (Biacore, Uppsala, Sweden) using CM5 sensor chips. Sortilin and bovine serum albumin were immobilized using the Biacore amine coupling kit according to the manufacturer's instructions. Hepes (20 mm, pH 7.4) containing 150 mm NaCl and 2.5 mm CaCl was used as running buffer. In studies of interactions in the absence of lipids, 0.05% (v/v) surfactant P20 was used. The temperature was 25 °C, and the flow was maintained at 5 μl/min. ApoA-V was dialyzed against running buffer and inhibitors were diluted in running buffer just prior to injection. Reference flow cells containing a corresponding amount of covalently bound bovine serum albumin was used as previously described (18). Sensorgrams were analyzed using the BIAevaluation software version 3.2 (Biacore, Uppsala, Sweden).
RESULTS
Binding of ApoA-V to Sortilin Studied by SPR—Binding of apoA-V-DMPC disks to sortilin, covalently bound to Biacore sensor chips, was studied during a 6-min injection (Fig. 1). To evaluate the nature of the interaction, competition experiments were conducted using known ligands for sortilin: RAP and NT. Heparin was also used, because it is known to bind to apoA-V and prevent its interaction with members of the LDL-R gene family (18). Previous studies have shown that RAP interaction with receptor-covered sensor chips elicits a detector response. Therefore, this ligand was injected prior to the apoA-V-DMPC disks. In this case, RAP prevented binding of apoA-V (Fig. 1). Instead, there was a decreased response, probably corresponding to dissociation of RAP from the receptor-bound chip surface. In contrast to RAP, the small molecule NT induces only a weak SPR detector response. Thus, NT could be co-injected with apoA-V, contributing a negligible addition to the response signal. Co-injection of NT and apoA-V-DMPC did not give rise to any response (Fig. 1). Since the apoA-V interaction with sortilin was prevented by RAP and was competed by NT, it may be concluded that apoA-V interacts with the Vps10p domain of sortilin. By the same token, when heparin was co-injected with apoA-V-DMPC disks, no binding to sortilin was detected (Fig. 1). Thus, we conclude that the receptor-binding site in apoA-V overlaps with or is located close to the heparin-binding region.
FIGURE 1.
Binding of ApoA-V-DMPC to sortilin studied by SPR. CM5 sensor chips with immobilized sortilin (17 fmol/mm2) in the active flow cells were used for binding studies. An equivalent amount of immobilized bovine serum albumin was used in a reference flow cell. Identical injections of apoA-V-DMPC disks were made with or without pre/co-injections of NT (846 nm), RAP (7.7 fmol/mm2), or heparin (10 units/ml).
To further study the interaction between the apoA-V-DMPC disks and sortilin, NT was injected after apoA-V-DMPC disks were bound to the receptor. In this case, a rapid dissociation of bound apoA-V from the sensor chip surface occurred (Fig. 2A). Compared with the slow dissociation of apoA-V-DMPC disks from sortilin in the absence of NT (Fig. 1), these data show that NT competes with apoA-V for binding to sortilin. Given the nature of NT binding to sortilin, we conclude that apoA-V binds to the receptor's Vps10p domain in a specific manner.
FIGURE 2.
Neurotensin causes dissociation of apoA-V-DMPC disks and chylomicrons from sortilin. Sensor chip surfaces were prepared as in Fig. 1. A, apoA-V-DMPC was injected to a response corresponding to a bound mass of 0.53 ng/mm2. Subsequently, 30 μl of NT (846 nm) was injected (diluted in running buffer), and the dissociation of apoA-V-DMPC from sortilin was recorded. B, identical injections of chylomicrons were made with and without co-injections of NT (846 nm).
The dissociation constant (KD) for the interaction of lipid-free apoA-V with sortilin was determined in the presence of 0.05% (v/v) surfactant P20 and was found to be 350 × 10–9 m. This was ∼3 times higher than the KD for the interaction of apoA-V with LRP1 determined under the same conditions and consistent with what was previously reported (18).
The role of sortilin as a lipoprotein receptor was investigated using human chylomicrons from an apoC-II-deficient patient by SPR. The chylomicrons contained relatively high amounts of apoA-V3 and bound to sortilin-covered sensor chips in a concentration-dependent manner. The binding of chylomicrons was fully competed, as in the case of apoA-V-DMPC disks, by NT (Fig. 2B). Due to higher nonspecific binding to the control surface, the sensorgram displayed a weak inverted response curve when NT was used to compete chylomicron binding.
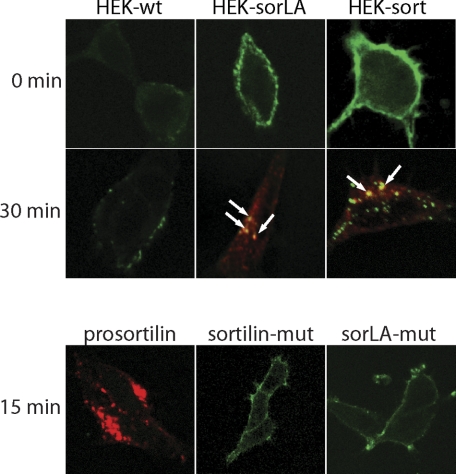
Binding and Endocytosis of ApoA-V-DMPC Disks by Cells Expressing SorLA and Sortilin—To determine if apoA-V undergoes receptor-mediated endocytosis, we investigated cell surface binding of Alexa Fluor 488-labeled apoA-V (apoA-V488)-DMPC disks to HEK293 cells. Binding of apoA-V to control nontransfected HEK293 cells at 4 °C was low. The low level of binding observed is probably due to interaction with heparan sulfate proteoglycan structures on the cell surfaces (Fig. 3, top panels, 0 min). On the other hand, HEK cells transfected with SorLA (HEK-SorLA) or sortilin (HEK-sort) displayed markedly enhanced fluorescence intensity. Internalization was observed in HEK-SorLA and HEK-sort upon incubation of the cells at 37 °C for 30 min (Fig. 3, middle panels). Furthermore, a high degree of co-localization between apoA-V488 and both receptors was detected (yellow, indicated by white arrows). By contrast, with nontransfected HEK cells, no apoA-V488 internalization was observed.
FIGURE 3.
ApoA-V-DMPC disks are bound and internalized by HEK293 cells overexpressing SorLA or sortilin. ApoA-V-DMPC-488 disks (green) were incubated with HEK293 cells transfected with the different receptors. The top two panels show binding to the cell surface after incubation at 4 °C (time 0), and the middle panels show binding after incubation at 37 °C for 30 min. The bottom panels show loss of binding to cells expressing prosortilin and hampered internalization to cells expressing receptor mutants modified in the internalization motif. Double staining with monoclonal anti-SorLA and anti-sortilin and Alexa Fluor 568-labeled secondary antibodies (red) was applied for the images showing 30-min internalization and prosortilin-transfected cells. wt, wild type.
To investigate the specificity of the observed interactions, control experiments were performed using cell lines expressing previously defined nonfunctional receptor variants. Internalization of apoA-V488 DMPC disks was investigated in cells expressing a variant sortilin construct mutated in the furin cleavage site, termed prosortilin (HEK-prosort) (28). Unlike sortilin, prosortilin is unable to bind ligands. The results of this experiment (Fig. 3) reveal staining for prosortilin (in red) but no internalization of apoA-V488 (in green) after 15 min at 37 °C. In the middle and right lower panels (Fig. 3), apoA-V488 was incubated with HEK cells expressing two additional receptor variants, one of SorLA (HEKSorLA-mut) and the other of sortilin (HEKsortilin-mut). These receptor constructs were mutated in their C-terminal cytoplasmic domains, resulting in markedly slower rates of endocytosis. The HEKsortilin-mut is altered in the tyrosine internalization motif (YXXL to AXXA) and in the dileucine motif (LL to AA) (29). The HEKSorLA-mut refers to a mutated amino acid cluster DD, EDDED, which is involved in internalization (31). No significant internalization of apoA-V488 was observed after incubation for 15 min with cells expressing these mutant receptors.
As a negative control, we generated DMPC disks containing Alexa 488-labeled apoC-II (apoC-II488). In addition, DMPC liposomes without protein were also taken through to the Alexa 488 labeling protocol (DMPC488). Experiments with cultured HEK cells showed that apoC-II488 did not interact with any of the cells investigated, and the DMPC particles alone did not show any surface binding and internalization (data not shown).
FRET Results—By means of FRET, it is possible to check for a direct interaction between donor- and acceptor-labeled protein partners of interest. The Eapp probability value calculated by means of the PFRET method is proportional to the molecular FRET efficiency E and is positive for donor-acceptor pairs that are within a distance of less than 10 nm from each other.
In Fig. 4 (A and B), examples are shown of donor (e), acceptor (g), and FRET (f) images as well as the resulting Eapp image for double-Alexa-labeled SorLA-apoA-V and sortilin-ApoA-V samples, indicating the apparent FRET probability Eapp (%) in each pixel of the image. FRET is seen to take place mainly at the membrane of vesicles. The absence of FRET inside of the vesicles is real (i.e. not an artifact related to removal of saturated pixels). The constant dependence of Eapp as a function of acceptor signal, as seen in the scatter plot for Sor-LA-ApoA-V, points toward a specific interaction (36). In this plot, each data point represents the average Eapp value obtained within each of a total of 1299 different ROIs distributed over 13 images with at least 1 cell/image. The straight line represents the best fit to the data, resulting in a slope statistically equal to zero (p = 0.53). For sortilin-apoA-V, the slope is significantly different from zero (p = 0.001), but the fitted curve is far from passing through the origin, as would be the case for a purely random interaction (767 ROIs evaluated). The average Eapp value based on all ROIs was estimated to be 3.3 ± 2.9% (mean and one S.D.) for SorLA-apoA-V and 2.7 ± 2.7% for sortilin-apoA-V. As can be seen from the Eapp versus acceptor signal scatter plots and Eapp histograms, the Eapp values do not exhibit a normal distribution, as would be expected if only one type of interaction with a well defined intramolecular distance were present. This might suggest the presence of at least two different subpopulations, one of which could represent a bridging of two apoA-Vs in one DMPC disk to two receptors.
FIGURE 4.
Fluorescence resonance energy transfer results showing receptor-apoA-V FRET interactions in vesicular regions in the cytosol of SorLA- and sortilin-expressing HEK293 cells. Energy transfer is registered as blue regions in the FRET analysis images. As donor-acceptor FRET pair, Alexa 488 and 568 were used. The constant behavior of the apparent FRET probability, Eapp, as a function of acceptor signal level within ROIs of size 20–25 pixels, is indicative of specific interactions. A, analyses of SorLA-HEK cells; B, analyses of sortilin-HEK cells.
The Endocytic Route of ApoA-V—Previous studies have demonstrated that SorLA and sortilin are rapidly internalized from the cell surface (29, 31). In accord with these data, we found that apoA-V488 bound to the surfaces of HEK-SorLA or HEK-sortilin and was internalized within minutes (for live imaging, see (supplemental Figs. 1 and 2). When apoA-V488 was incubated with HEK-SorLA or HEK-sortilin for 30 min at 37 °C (Fig. 5), internalization was detected. ApoA-V488 fluorescence co-localized with Lamp-1, MPR300, and TGN-46 (markers for lysosomes, late endosomes, and TGN, respectively). This suggests that, with cells expressing either SorLA or sortilin, apoA-V488 is first internalized to early endosomes. Following this, the ligand is sorted in a retrograde fashion to late endosomes and TGN. While passing the late endosomes, a portion of the ligand is uncoupled and sent to lysosomes for degradation. Experiments with prolonged internalization time indicated that little or no recycling of apoA-V occurs (supplemental Fig. 1 and 2).
FIGURE 5.
The endocytic route for apoA-V visualized by markers for lysosomes, late endosomes, and the trans-Golgi network. HEK-SorLA and HEK-sortilin cells were stained for intracellular markers for lysosomes (Lamp-1), late endosomes (MPR300), and TGN (TGN46), all shown in red, after internalization of apoA-V488 (green). Co-localization is shown in yellow (white arrows).
Studies of Degradation of 125I-Labeled ApoA-V in HEK Cells—To quantify the internalization process, 125I-labeled apoA-V-DMPC disks (125I-apoA-V) were incubated with cells at 37 °C for 45 or 90 min (Fig. 6). In the case of non-transfected HEK293 cells, lower amounts of the tracer had been degraded at 45 min compared with cells transfected with SorLA or sortilin. This difference was more pronounced at the longer incubation time. At 90 min, HEK-SorLA and HEK-sortilin had degraded about 2.5-fold more of the 125I-apoA-V tracer compared with control HEK cells. Higher levels of cell-associated 125I-apoA-V were recorded with HEK-SorLA and HEK-sortilin (data not shown), supporting the previous finding that apoA-V binds to the overexpressed receptors to a higher degree than to the endogenous expressed receptors present at the HEK293 cells.
FIGURE 6.
Degradation of 125I-labeled apoA-V-DMPC disks in cells expressing SorLA and sortilin. ApoAV-DMPC disks labeled with 125I were incubated at 37 °C with HEK293 cells expressing SorLA and sortilin; as a control, wild type HEK293 cells were used. Degradation of the tracer were measured at two different time points, 45 min (gray bars) and 90 min (black bars).
DISCUSSION
In this paper, we demonstrate that apoA-V DMPC disks bind to SorLA and sortilin on cell surfaces and that apoA-V is rapidly internalized. FRET data show that apoA-V is localized in very close proximity to either SorLA or sortilin at the membrane in intracellular vesicles. This is a strong indication that these two Vps10 domain receptors are responsible for the endocytic function of apoA-V. After 30 min of internalization, apoA-V accumulated in TGN-46-, Lamp-1-, and MPR300-positive vesicles, whereas nontransfected HEK293 cells internalized little or no apoA-V. The SorLA- and sortilin-mediated transport of apoA-V is in accordance with previous studies of the routing of SorLA and sortilin (29, 31, 37). AP-2-mediated endocytosis goes rapidly through early endosomes, and the cargo is then relocated to TGN by interacting with the retromer complex. It is not clear if sortilin and SorLA go through late endosomes on their way from early endosomes to TGN, but once they are located in TGN, they are engaged in TGN-endosome transport. In the endosomes, apoA-V can be uncoupled from the receptors and sent to lysosomes for degradation. Recycling of SorLA and sortilin has not been reported. This is in contrast to LDL-R and LRP1, which are rapidly recycled using an NPXY tyrosine-based internalization motif (27).
We have previously shown that apoA-V interacts with LRP1 and SorLA and that apoA-V can mediate binding of chylomicrons to these receptors (18). In the present study, we demonstrate binding of apoA-V also to sortilin. Thus, apoA-V can bind to endocytotic receptors from two different gene families; the LDL-R gene family and the Vps10p domain family. SorLA has typical LDL-A repeats and has been shown to share many ligands with LDL-R gene family members. In addition, SorLA has a Vps10p domain, also found in sortilin. Sortilin, on the other hand, contains a Vps10p domain but lacks LDL-A repeats present in LDL-R family members (22, 23). Although sortilin is involved in signaling by proneurotrophins, sortilin binds a range of lipoprotein-related proteins (e.g. LPL, apoE, and, as shown in the present paper, apoA-V). The SPR experiments indicate apoA-V interacts specifically with the Vps10p domain of sortilin, since binding was fully competed by neurotensin. Based on past and present results, we conclude that apoA-V probably has a common binding domain for both types of receptors. As with LRP1 and SorLA (18), apoA-V binding to sortilin was competed by RAP and heparin. Further experiments demonstrated that apoA-V interacts with sortilin in both lipid-free and lipid-bound state.
SorLA and sortilin participate in many different cellular events. For example, it has been shown that SorLA interacts with the amyloid precurcursor protein (38), a protein that is directly associated with development of Alzheimer's disease. SorLA might play a role in the regulation of formation of lipid-laden macrophages in atherosclerotic plaques (39). Sortilin is involved in neuronal apoptosis through its interactions with proneurotrophins and p75NTR (40). It has also been established that sortilin binds to LPL in intracellular compartments in cultured 3T3-L1 cells (41). In addition, we have demonstrated here that sortilin binds human chylomicrons in vitro. The physiological roles of SorLA and sortilin are, however, far from clarified. Some of their multifunctional properties can be assigned to motifs found in their cytoplamatic domains. Through these motifs, SorLA and sortilin interact with a wide range of cytosolic adaptor complexes involved in endocytic and intracellular trafficking including GGAs, AP-1 and -2, and the retromer complexes (29, 31, 37).
Sortilin is highly expressed in the brain and spinal cord, where it is anticipated to have limited exposure to apoA-V. However, sortilin is also highly expressed in skeletal and heart muscle (23). Here we used sortilin, in conjunction with SorLA, to study whether apoA-V can bind to a receptor containing a Vsp10p domain but no LDL-A repeats. Previously sortilin has mainly been associated with functions of intracellular processing and trafficking (29, 41). Since SorLA and sortilin are primarily not expressed in the liver, whereas apoA-V is solely expressed there (1), it is likely that SorLA and sortilin participate in endocytic functions involving apoA-V rather than its intracellular processing. Interestingly, the avian homologlue of apoA-V is expressed in the brain as well as in liver, kidney, and ovarian follicles (19).
The equilibrium dissociation constants between the complexes formed by apoA-V and LRP1 or SorLA as determined by SPR are consistent with a high affinity interaction (18). It is possible that high affinity for LDL-R gene family receptors (LDLR and LRP1) results in retention of the bulk of the apoA-V in the liver. ApoA-V in circulation, which has probably left the liver as a component of nascent VLDL, might represent a minor fraction of the overall apoA-V pool. We speculate that this cosecretion could explain the positive relationship between apoA-V levels in human plasma and plasma TG levels (16, 17). Based on past and present findings, circulating apoA-V-containing lipoproteins are likely to be recognized by receptors, including heparan sulfate proteoglycans, and thereby rapidly removed. The results of the present study suggest that when the apoA-V-containing lipoproteins bind to SorLA or sortilin expressed in peripheral tissues, they are eventually routed for degradation. This is in contrast to ligands bound to LDLR or LRP1 where some recirculation occurs, as was previously demonstrated for apoE-containing lipoproteins in liver cells (42).
Interestingly, several recent genome-wide association studies point to the sortilin gene as being part of a chromosomal region that contributes significantly to common variations in plasma lipoprotein concentrations, especially to levels of LDL cholesterol (24, 25) and to development of coronary artery disease (26). It remains to be studied if the interactions of apoA-V with sortilin, which we demonstrate here, could have an impact on plasma lipid transport.
Supplementary Material
Acknowledgments
We thank Jennifer Beckstead and Kasuen Wong for the preparation of apoA-V and DMPC disks and Drs. Ulrike Beisiegel and Joerg Heeren (Eppendorf University Hospital, Hamburg, Germany) for generously providing the apoCII-deficient human chylomicrons.
This work was supported, in whole or in part, by National Institutes of Health Grant HL-073061. This work was also supported by Swedish Research Council Grant 12203 and King Gustav V and Queen Victoria Research Foundation. The MIND-Center is sponsored by The Lundbeck Foundation. The costs of publication of this article were defrayed in part by the payment of page charges. This article must therefore be hereby marked “advertisement” in accordance with 18 U.S.C. Section 1734 solely to indicate this fact.
The on-line version of this article (available at http://www.jbc.org) contains supplemental Figs. 1 and 2 and Movies 1 and 2.
Footnotes
The abbreviations used are: TG, triacylglycerol; apo, apolipoprotein; DMPC, l-α-dimyristoylphosphatidylcholine; HEK, human embryonic kidney; LDL, low density lipoprotein; LDL-R, LDL receptor; LPL, lipoprotein lipase; NT, neurotensin; RAP, receptor-associated protein; SPR, surface plasmon resonance; TGN, trans-Golgi network; VLDL, very low density lipoprotein; FRET, fluorescence resonance energy transfer; ROI, region of interest.
J. Heeren, personal communication.
References
- 1.Pennacchio, L. A., Olivier, M., Hubacek, J. A., Cohen, J. C., Cox, D. R., Fruchart, J. C., Krauss, R. M., and Rubin, E. M. (2001) Science 294169 –173 [DOI] [PubMed] [Google Scholar]
- 2.van Dijk, K. W., Rensen, P.C., Voshol, P. J., and Havekes, L. M. (2004) Curr. Opin. Lipidol. 15239 –246 [DOI] [PubMed] [Google Scholar]
- 3.Hubacek, J. A. (2005) Clin. Chem. Lab. Med. 43897 –902 [DOI] [PubMed] [Google Scholar]
- 4.Talmud, P. J., Hawe, E., Martin, S., Olivier, M., Miller, G. J., Rubin, E. M., Pennacchio, L. A., and Humphries, S. E. (2002) Hum. Mol. Genet. 113039 –3046 [DOI] [PubMed] [Google Scholar]
- 5.Yamada, Y., Kato, K., Hibino, T., Yokoi, K., Matsuo, H., Segawa, T., Watanabe, S., Ichihara, S., Yoshida, H., Satoh, K., and Nozawa, Y. (2007) Atherosclerosis 191298 –304 [DOI] [PubMed] [Google Scholar]
- 6.Niculescu, L. S., Fruchart-Najib, J., Fruchart, J. C., and Sima, A. (2007) Clin. Chem. Lab. Med. 451133 –1139 [DOI] [PubMed] [Google Scholar]
- 7.Calandra, S., Priore Oliva, C., Tarugi, P., and Bertolini, S. (2006) Curr. Opin. Lipidol. 17122 –127 [DOI] [PubMed] [Google Scholar]
- 8.Schaap, F. G., Rensen, P. C., Voshol, P. J., Vrins, C., van der Vliet, H. N., Chamuleau, R. A., Havekes, L. M., Groen, A. K., and van Dijk, K. W. (2004) J. Biol. Chem. 27927941 –27947 [DOI] [PubMed] [Google Scholar]
- 9.Merkel, M., Loeffler, B., Kluger, M., Fabig, N., Geppert, G., Pennacchio, L. A., Laatsch, A., and Heeren, J. (2005) J. Biol. Chem. 28021553 –21560 [DOI] [PubMed] [Google Scholar]
- 10.Fruchart-Najib, J., Bauge, E., Niculescu, L. S., Pham, T., Thomas, B., Rommens, C., Majd, Z., Brewer, B., Pennacchio, L. A., and Fruchart, J. C. (2004) Biochem. Biophys. Res. Commun. 319397 –404 [DOI] [PubMed] [Google Scholar]
- 11.Sun, G., Bi, N., Li, G., Zhu, X., Zeng, W., Wu, G., Xue, H., and Chen, B. (2006) Chem. Phys. Lipids 14322 –28 [DOI] [PubMed] [Google Scholar]
- 12.Huang, W., Bi, N., Zhang, X., Wang, Y., Chen, B., and Liu, G. (2006) Biochem. Biophys. Res. Commun. 34614 –18 [DOI] [PubMed] [Google Scholar]
- 13.Lookene, A., Beckstead, J. A., Nilsson, S., Olivecrona, G., and Ryan, R. O. (2005) J. Biol. Chem. 28025383 –25387 [DOI] [PubMed] [Google Scholar]
- 14.Ishihara, M., Kujiraoka, T., Iwasaki, T., Nagano, M., Takano, M., Ishii, J., Tsuji, M., Ide, H., Miller, I. P., Miller, N. E., and Hattori, H. (2005) J. Lipid Res. 462015 –2022 [DOI] [PubMed] [Google Scholar]
- 15.Merkel, M., and Heeren, J. (2005) J. Clin. Invest. 1152694 –2696 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Dallinga-Thie, G. M., van Tol, A., Hattori, H., van Vark-van der Zee, L. C., Jansen, H., and Sijbrands, E. J. (2006) Diabetologia 491505 –1511 [DOI] [PubMed] [Google Scholar]
- 17.Vaessen, S. F., Schaap, F. G., Kuivenhoven, J. A., Groen, A. K., Hutten, B. A., Boekholdt, S. M., Hattori, H., Sandhu, M. S., Bingham, S. A., Luben, R., Palmen, J. A., Wareham, N. J., Humphries, S. E., Kastelein, J. J. P., Talmud, P. J., and Khaw, K.-T. (2006) J. Lipid Res. 472064 –2070 [DOI] [PubMed] [Google Scholar]
- 18.Nilsson, S. K., Lookene, A., Beckstead, J. A., Gliemann, J., Ryan, R. O., and Olivecrona, G. (2007) Biochemistry 463896 –3904 [DOI] [PubMed] [Google Scholar]
- 19.Dichlberger, A., Cogburn, L. A., Nimpf, J., and Schneider, W. J. (2007) J. Lipid Res. 481451 –1456 [DOI] [PubMed] [Google Scholar]
- 20.Grosskopf, I., Baroukh, N., Lee, S. J., Kamari, Y., Harats, D., Rubin, E. M., Pennacchio, L. A., and Cooper, A. D. (2005) Arterioscler. Thromb. Vasc. Biol. 252573 –2579 [DOI] [PubMed] [Google Scholar]
- 21.Beigneux, A. P., Davies, B. S., Gin, P., Weinstein, M. M., Farber, E., Qiao, X., Peale, F., Bunting, S., Walzem, R. L., Wong, J. S., Blaner, W. S., Ding, Z. M., Melford, K., Wongsiriroj, N., Shu, X., de Sauvage, F., Ryan, R. O., Fong, L. G., Bensadoun, A., and Young, S. G. (2007) Cell Metab. 5279 –291 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Jacobsen, L., Madsen, P., Moestrup, S. K., Lund, A. H., Tommerup, N., Nykjaer, A., Sottrup-Jensen, L., Gliemann, J., and Petersen, C. M. (1996) J. Biol. Chem. 27131379 –31383 [DOI] [PubMed] [Google Scholar]
- 23.Petersen, C. M., Nielsen, M. S., Nykjaer, A., Jacobsen, L., Tommerup, N., Rasmussen, H. H., Roigaard, H., Gliemann, J., Madsen, P., and Moestrup, S. K. (1997) J. Biol. Chem. 2723599 –3605 [DOI] [PubMed] [Google Scholar]
- 24.Sandhu, M. S., Waterworth, D. M., Debenham, S. L., Wheeler, E., Papadakis, K., Zhao, J. H., Song, K., Yuan, X., Johnson, T., Ashford, S., Inouye, M., Luben, R., Sims, M., Hadley, D., McArdle, W., Barter, P., Kesaniemi, Y. A., Mahley, R. W., McPherson, R., Grundy, S. M., Wellcome Trust Case Control Consortium, Bingham, S. A., Khaw, K.-T., Loos, R. J. F., Waeber, G., Barroso, I., Strachan, D. P., Deloukas, P., Vollenweider, P., Wareham, N. J., and Mooser, V. (2008) Lancet 371483 –491 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Kathiresan, S., Melander, O., Guiducci, C., Surti, A., Burtt, N. P., Rieder, M. J., Cooper, G. M., Roos, C., Voight, B. F., Havulinna, A. S., Wahlstrand, B., Hedner, T., Corella, D., Tai, E. S., Ordovas, J. M., Berglund, G. Vartiainen, E., Jousilahti, P., Hedblad, B., Taskinen, M. R., Newton-Cheh, C., Salomaa, V., Peltonen, L., Groop, L., Altshuler, D. M., and Orho-Melander, M. (2008) Nat. Genet. 40189 –197 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Samani, N. J., Erdmann, J., Hall, A. S., Hengstenberg, C., Mangino, M., Mayer, B., Dixon, R. J., Meitinger, T., Braund, P., Wichmann, H. E., Barrett, J. H., König, I. R., Stevens, S. E., Szymczak, S., Tregouet, D.-A., Iles, M. M., Pahlke, F., Pollard, H., Lieb, W., Cambien, F., Fischer, M., Ouwehand, W., Blankenberg, S., Balmforth, A. J., Baessler, A., Ball, S. G., Strom, T. M., Boenne, I., Gieger, C., Deloukas, P., Tobin, M. D., Ziegler, A., Thompson, J. R., and Schunkert, H. (2007) N. Engl. J. Med. 357443 –453 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Herz, J., and Strickland, D. K. (2001) J. Clin. Invest. 108779 –784 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Munck Petersen, C., Nielsen, M. S., Jacobsen, C., Tauris, J., Jacobsen, L., Gliemann, J., Moestrup, S. K., and Madsen, P. (1999) EMBO J. 18595 –604 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Nielsen, M. S., Madsen, P., Christensen, E. I., Nykjaer, A., Gliemann, J., Kasper, D., Pohlmann, R., and Petersen, C. M. (2001) EMBO J. 202180 –2190 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Bengtsson-Olivecrona, G., and Olivecrona, T. (1991) Methods Enzymol. 197345 –356 [DOI] [PubMed] [Google Scholar]
- 31.Nielsen, M. S., Gustafsen, C., Madsen, P., Nyengaard, J. R., Hermey, G., Bakke, O., Mari, M., Schu, P., Pohlmann, R., Dennes, A., and Petersen, C. M. (2007) Mol. Cell. Biol. 276842 –6851 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Beckstead, J. A., Oda, M. N., Martin, D. D., Forte, T. M., Bielicki, J. K., Berger, T., Luty, R., Kay, C. M., and Ryan, R. O. (2003) Biochemistry 429416 –9423 [DOI] [PubMed] [Google Scholar]
- 33.Shu, X., Chan, J., Ryan, R. O., and Forte, T. M. (2007) J. Lipid Res. 481445 –1450 [DOI] [PubMed] [Google Scholar]
- 34.Elangovan, M., Wallrabe, H., Chen, Y., Day, R. N., Barroso, M., and Periasamy, A. (2003) Methods 2958 –73 [DOI] [PubMed] [Google Scholar]
- 35.Wallrabe, H., Chen, Y., Periasamy, A., and Barroso, M. (2006) Microsc. Res. Tech. 69196 –206 [DOI] [PubMed] [Google Scholar]
- 36.Wallrabe, H., Elangovan, M., Burchard, A., Periasamy, A., and Barroso, M. (2003) Biophys. J. 85559 –571 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Mari, M., Bujny, M. V., Zeuschner, D., Geerts, W. J., Griffith, J., Petersen, C. M., Cullen, P. J., Klumperman, J., and Geuze, H. J. (2008) Traffic, 9380 –393 [DOI] [PubMed] [Google Scholar]
- 38.Andersen, O. M., Reiche, J., Schmidt, V., Gotthardt, M., Spoelgen, R., Behlke, J., von Arnim, C. A., Breiderhoff, T., Jansen, P., Wu, X., Bales, K. R., Cappai, R., Masters, C. L., Gliemann, J., Mufson, E. J., Hyman, B. T., Paul, S. M., Nykjaer, A., and Willnow, T. (2005) Proc. Natl. Acad. Sci. U. S. A. 10213461 –13466 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Ohwaki, K., Bujo, H., Jiang, M., Yamazaki, H., Schneider, W. J., and Saito, Y. (2007) Arterioscler. Thromb. Vasc. Biol. 271050 –1056 [DOI] [PubMed] [Google Scholar]
- 40.Jansen, P., Giehl, K., Nyengaard, J. R., Teng, K., Lioubinski, O., Sjoegaard, S. S., Breiderhoff, T., Gotthardt, M., Lin, F., Eilers, A., Petersen, C. M., Lewin, G. R., Hempstead, B. L., Willnow, T. E., and Nykjaer, A. (2007) Nat. Neurosci. 101449 –1457 [DOI] [PubMed] [Google Scholar]
- 41.Nielsen, M. S., Jacobsen, C., Olivecrona, G., Gliemann, J., and Petersen, C. M. (1999) J. Biol. Chem. 2748832 –8836 [DOI] [PubMed] [Google Scholar]
- 42.Heeren, J., Beisiegel, U., and Grewal, T. (2006) Arterioscler. Thromb. Vasc. Biol. 26442 –448 [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.