Abstract
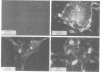
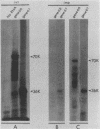
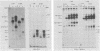
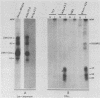
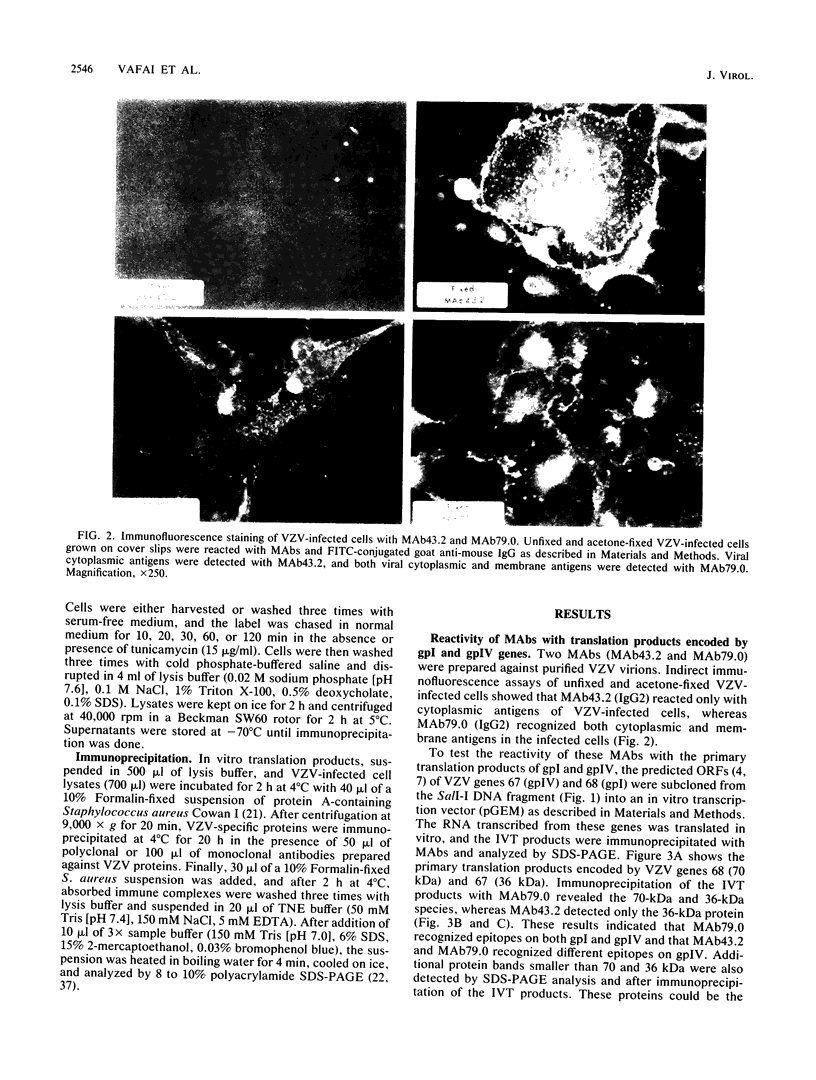
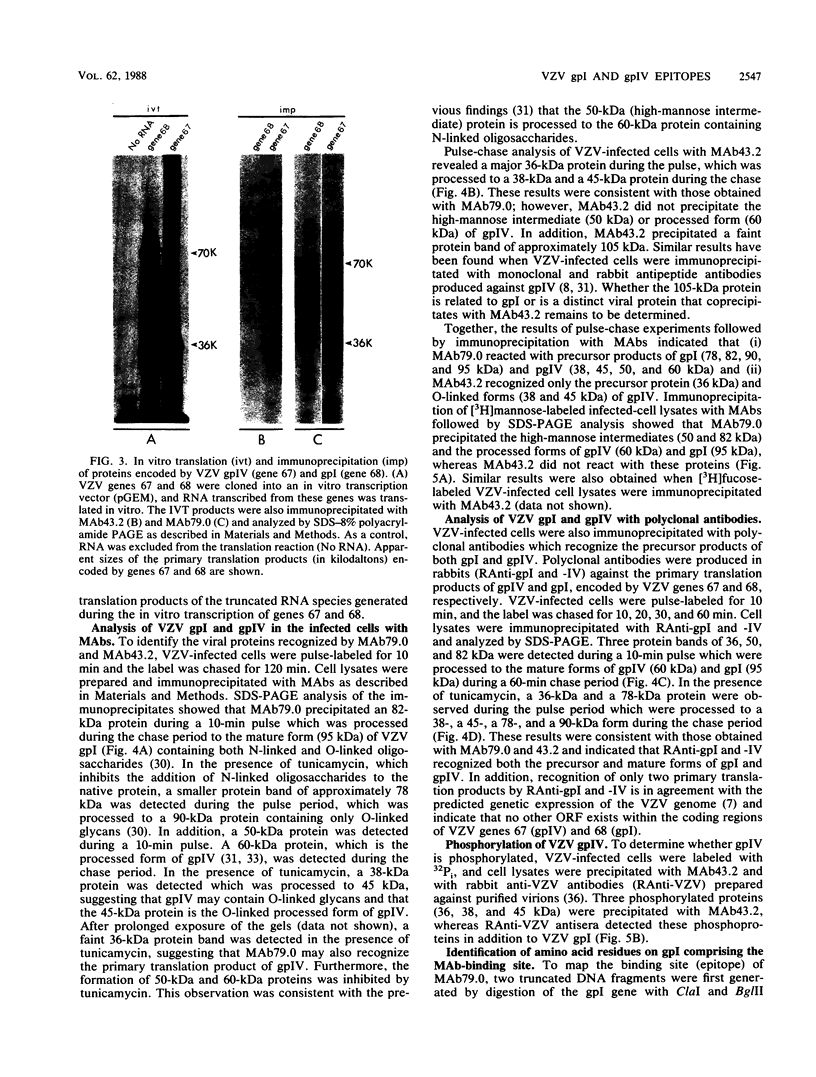
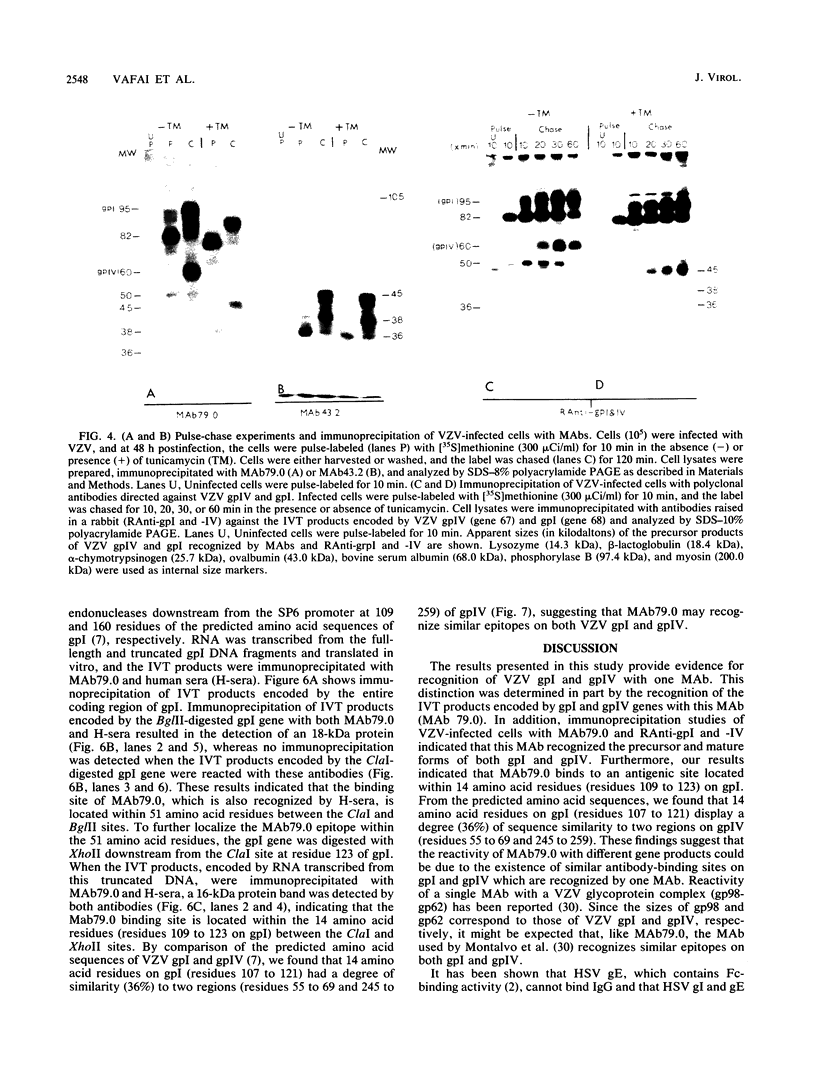
Two monoclonal antibodies, MAb43.2 and MAb79.0, prepared against varicella-zoster virus (VZV) proteins were selected to analyze VZV gpIV and gpI, respectively. MAb43.2 reacted only with cytoplasmic antigens, whereas MAb79.0 recognized both cytoplasmic and membrane antigens in VZV-infected cells. Immunoprecipitation of in vitro translation products with MAb43.2 revealed only proteins encoded by the gpIV gene, whereas MAb79.0 precipitated proteins encoded by the gpIV and gpI genes. Pulse-chase analysis followed by immunoprecipitation of VZV-infected cells indicated reactivity of MAb43.2 with three phosphorylated precursor species of gpIV and reactivity of MAb79.0 with the precursor and mature forms of gpI and gpIV. These results indicated that (i) MAb43.2 and MAb79.0 recognize different epitopes on VZV gpIV, (ii) glycosylation of gpIV ablates recognition by MAb43.2, and (iii) gpIV is phosphorylated. To map the binding site of MAb79.0 on gpI, the pGEM transcription vector, containing the coding region of the gpI gene, was linearized, and three truncated gpI DNA fragments were generated. RNA was transcribed from each truncated fragment by using SP6 RNA polymerase, translated in vitro in a rabbit reticulocyte lysate, and immunoprecipitated with MAb79.0 and human sera. The results revealed the existence of an antibody-binding site within 14 amino acid residues located between residues 109 to 123 on the predicted amino acid sequences of gpI. From the predicted amino acid sequences, 14 residues on gpI (residues 107 to 121) displayed a degree of similarity (36%) to two regions (residues 55 to 69 and 245 to 259) of gp IV. Such similarities may account for the binding of MAb79.0 to both VZV gpI and gpIV.
Full text
PDF



Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Arvin A. M., Kinney-Thomas E., Shriver K., Grose C., Koropchak C. M., Scranton E., Wittek A. E., Diaz P. S. Immunity to varicella-zoster viral glycoproteins, gp I (gp 90/58) and gp III (gp 118), and to a nonglycosylated protein, p 170. J Immunol. 1986 Aug 15;137(4):1346–1351. [PubMed] [Google Scholar]
- Baucke R. B., Spear P. G. Membrane proteins specified by herpes simplex viruses. V. Identification of an Fc-binding glycoprotein. J Virol. 1979 Dec;32(3):779–789. doi: 10.1128/jvi.32.3.779-789.1979. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Berns A., van den Ouweland A., Quint W., van Oirschot J., Gielkens A. Presence of markers for virulence in the unique short region or repeat region or both of pseudorabies hybrid viruses. J Virol. 1985 Jan;53(1):89–93. doi: 10.1128/jvi.53.1.89-93.1985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Davison A. J. DNA sequence of the US component of the varicella-zoster virus genome. EMBO J. 1983;2(12):2203–2209. doi: 10.1002/j.1460-2075.1983.tb01724.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Davison A. J., Edson C. M., Ellis R. W., Forghani B., Gilden D., Grose C., Keller P. M., Vafai A., Wroblewska Z., Yamanishi K. New common nomenclature for glycoprotein genes of varicella-zoster virus and their glycosylated products. J Virol. 1986 Mar;57(3):1195–1197. doi: 10.1128/jvi.57.3.1195-1197.1986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Davison A. J., McGeoch D. J. Evolutionary comparisons of the S segments in the genomes of herpes simplex virus type 1 and varicella-zoster virus. J Gen Virol. 1986 Apr;67(Pt 4):597–611. doi: 10.1099/0022-1317-67-4-597. [DOI] [PubMed] [Google Scholar]
- Davison A. J., Scott J. E. The complete DNA sequence of varicella-zoster virus. J Gen Virol. 1986 Sep;67(Pt 9):1759–1816. doi: 10.1099/0022-1317-67-9-1759. [DOI] [PubMed] [Google Scholar]
- Davison A. J., Waters D. J., Edson C. M. Identification of the products of a varicella-zoster virus glycoprotein gene. J Gen Virol. 1985 Oct;66(Pt 10):2237–2242. doi: 10.1099/0022-1317-66-10-2237. [DOI] [PubMed] [Google Scholar]
- Edson C. M., Hosler B. A., Poodry C. A., Schooley R. T., Waters D. J., Thorley-Lawson D. A. Varicella-zoster virus envelope glycoproteins: biochemical characterization and identification in clinical material. Virology. 1985 Aug;145(1):62–71. doi: 10.1016/0042-6822(85)90201-6. [DOI] [PubMed] [Google Scholar]
- Edson C. M., Hosler B. A., Waters D. J. Varicella-zoster virus gpI and herpes simplex virus gE: phosphorylation and Fc binding. Virology. 1987 Dec;161(2):599–602. doi: 10.1016/0042-6822(87)90157-7. [DOI] [PubMed] [Google Scholar]
- Ellis R. W., Keller P. M., Lowe R. S., Zivin R. A. Use of a bacterial expression vector to map the varicella-zoster virus major glycoprotein gene, gC. J Virol. 1985 Jan;53(1):81–88. doi: 10.1128/jvi.53.1.81-88.1985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Forghani B., Schmidt N. J., Myoraku C. K., Gallo D. Serological reactivity of some monoclonal antibodies to varicella-zoster virus. Arch Virol. 1982;73(3-4):311–317. doi: 10.1007/BF01318084. [DOI] [PubMed] [Google Scholar]
- Gilden D. H., Shtram Y., Friedmann A., Wellish M., Devlin M., Cohen A., Fraser N., Becker Y. Extraction of cell-associated varicella-zoster virus DNA with triton X-100-NaCl. J Virol Methods. 1982 May;4(4-5):263–275. doi: 10.1016/0166-0934(82)90073-8. [DOI] [PubMed] [Google Scholar]
- Gilden D. H., Wroblewska Z., Kindt V., Warren K. G., Wolinsky J. S. Varicella-zoster virus infection of human brain cells and ganglion cells in tissue culture. Arch Virol. 1978;56(1-2):105–117. doi: 10.1007/BF01317286. [DOI] [PubMed] [Google Scholar]
- Grose C., Edwards D. P., Friedrichs W. E., Weigle K. A., McGuire W. L. Monoclonal antibodies against three major glycoproteins of varicella-zoster virus. Infect Immun. 1983 Apr;40(1):381–388. doi: 10.1128/iai.40.1.381-388.1983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ito M., Ihara T., Grose C., Starr S. Human leukocytes kill varicella-zoster virus-infected fibroblasts in the presence of murine monoclonal antibodies to virus-specific glycoproteins. J Virol. 1985 Apr;54(1):98–103. doi: 10.1128/jvi.54.1.98-103.1985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Johnson D. C., Feenstra V. Identification of a novel herpes simplex virus type 1-induced glycoprotein which complexes with gE and binds immunoglobulin. J Virol. 1987 Jul;61(7):2208–2216. doi: 10.1128/jvi.61.7.2208-2216.1987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Johnson D. C., Spear P. G. O-linked oligosaccharides are acquired by herpes simplex virus glycoproteins in the Golgi apparatus. Cell. 1983 Mar;32(3):987–997. doi: 10.1016/0092-8674(83)90083-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Keller P. M., Neff B. J., Ellis R. W. Three major glycoprotein genes of varicella-zoster virus whose products have neutralization epitopes. J Virol. 1984 Oct;52(1):293–297. doi: 10.1128/jvi.52.1.293-297.1984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Keller P. M., Neff B. J., Ellis R. W. Three major glycoprotein genes of varicella-zoster virus whose products have neutralization epitopes. J Virol. 1984 Oct;52(1):293–297. doi: 10.1128/jvi.52.1.293-297.1984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Laemmli U. K. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature. 1970 Aug 15;227(5259):680–685. doi: 10.1038/227680a0. [DOI] [PubMed] [Google Scholar]
- Lomniczi B., Watanabe S., Ben-Porat T., Kaplan A. S. Genetic basis of the neurovirulence of pseudorabies virus. J Virol. 1984 Oct;52(1):198–205. doi: 10.1128/jvi.52.1.198-205.1984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Longnecker R., Chatterjee S., Whitley R. J., Roizman B. Identification of a herpes simplex virus 1 glycoprotein gene within a gene cluster dispensable for growth in cell culture. Proc Natl Acad Sci U S A. 1987 Jun;84(12):4303–4307. doi: 10.1073/pnas.84.12.4303. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Low M. G., Saltiel A. R. Structural and functional roles of glycosyl-phosphatidylinositol in membranes. Science. 1988 Jan 15;239(4837):268–275. doi: 10.1126/science.3276003. [DOI] [PubMed] [Google Scholar]
- McGeoch D. J., Dolan A., Donald S., Rixon F. J. Sequence determination and genetic content of the short unique region in the genome of herpes simplex virus type 1. J Mol Biol. 1985 Jan 5;181(1):1–13. doi: 10.1016/0022-2836(85)90320-1. [DOI] [PubMed] [Google Scholar]
- Mettenleiter T. C., Lukàcs N., Rziha H. J. Pseudorabies virus avirulent strains fail to express a major glycoprotein. J Virol. 1985 Oct;56(1):307–311. doi: 10.1128/jvi.56.1.307-311.1985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Montalvo E. A., Grose C. Varicella zoster virus glycoprotein gpI is selectively phosphorylated by a virus-induced protein kinase. Proc Natl Acad Sci U S A. 1986 Dec;83(23):8967–8971. doi: 10.1073/pnas.83.23.8967. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Montalvo E. A., Parmley R. T., Grose C. Structural analysis of the varicella-zoster virus gp98-gp62 complex: posttranslational addition of N-linked and O-linked oligosaccharide moieties. J Virol. 1985 Mar;53(3):761–770. doi: 10.1128/jvi.53.3.761-770.1985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Namazue J., Campo-Vera H., Kitamura K., Okuno T., Yamanishi K. Processing of virus-specific glycoproteins of varicella zoster virus. Virology. 1985 May;143(1):252–259. doi: 10.1016/0042-6822(85)90112-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Neidhardt H., Schröder C. H., Kaerner H. C. Herpes simplex virus type 1 glycoprotein E is not indispensable for viral infectivity. J Virol. 1987 Feb;61(2):600–603. doi: 10.1128/jvi.61.2.600-603.1987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Okuno T., Yamanishi K., Shiraki K., Takahashi M. Synthesis and processing of glycoproteins of Varicella-Zoster virus (VZV) as studied with monoclonal antibodies to VZV antigens. Virology. 1983 Sep;129(2):357–368. doi: 10.1016/0042-6822(83)90175-7. [DOI] [PubMed] [Google Scholar]
- Petrovskis E. A., Timmins J. G., Post L. E. Use of lambda gt11 to isolate genes for two pseudorabies virus glycoproteins with homology to herpes simplex virus and varicella-zoster virus glycoproteins. J Virol. 1986 Oct;60(1):185–193. doi: 10.1128/jvi.60.1.185-193.1986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vafai A., Wellish M., Gilden D. Polypeptides encoded by varicella-zoster virus unique short sequences. Virus Res. 1986 Jul;5(1):67–76. doi: 10.1016/0168-1702(86)90066-3. [DOI] [PubMed] [Google Scholar]
- Vafai A., Wellish M., Wroblewska Z., Cisco M., Gilden D. Induction of antibody against in vitro translation products encoded by varicella-zoster virus glycoprotein genes. Virus Res. 1987 Jun;7(4):325–333. doi: 10.1016/0168-1702(87)90046-3. [DOI] [PubMed] [Google Scholar]
- Vafai A., Wroblewska Z., Wellish M., Green M., Gilden D. Analysis of three late varicella-zoster virus proteins, a 125,000-molecular-weight protein and gp1 and gp3. J Virol. 1984 Dec;52(3):953–959. doi: 10.1128/jvi.52.3.953-959.1984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Waxham M. N., Merz D. C., Wolinsky J. S. Intracellular maturation of mumps virus hemagglutinin-neuraminidase glycoprotein: conformational changes detected with monoclonal antibodies. J Virol. 1986 Aug;59(2):392–400. doi: 10.1128/jvi.59.2.392-400.1986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wroblewska Z., Devlin M., Reilly K., van Trieste H., Wellish M., Gilden D. H. The production of varicella Zoster virus antiserum in laboratory animals. Brief report. Arch Virol. 1982;74(2-3):233–238. doi: 10.1007/BF01314717. [DOI] [PubMed] [Google Scholar]
- Wroblewska Z., Gilden D., Green M., Devlin M., Vafai A. Affinity-purified varicella-zoster virus glycoprotein gp1/gp3 stimulates the production of neutralizing antibody. J Gen Virol. 1985 Aug;66(Pt 8):1795–1799. doi: 10.1099/0022-1317-66-8-1795. [DOI] [PubMed] [Google Scholar]