Abstract
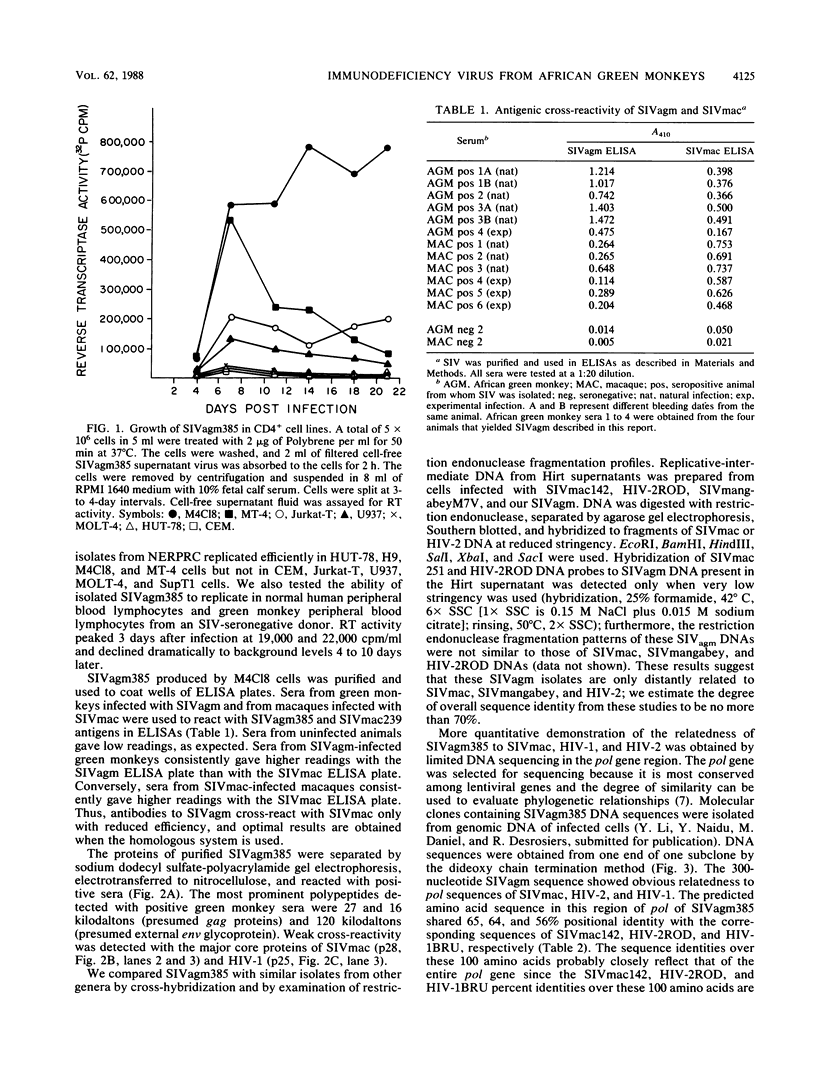
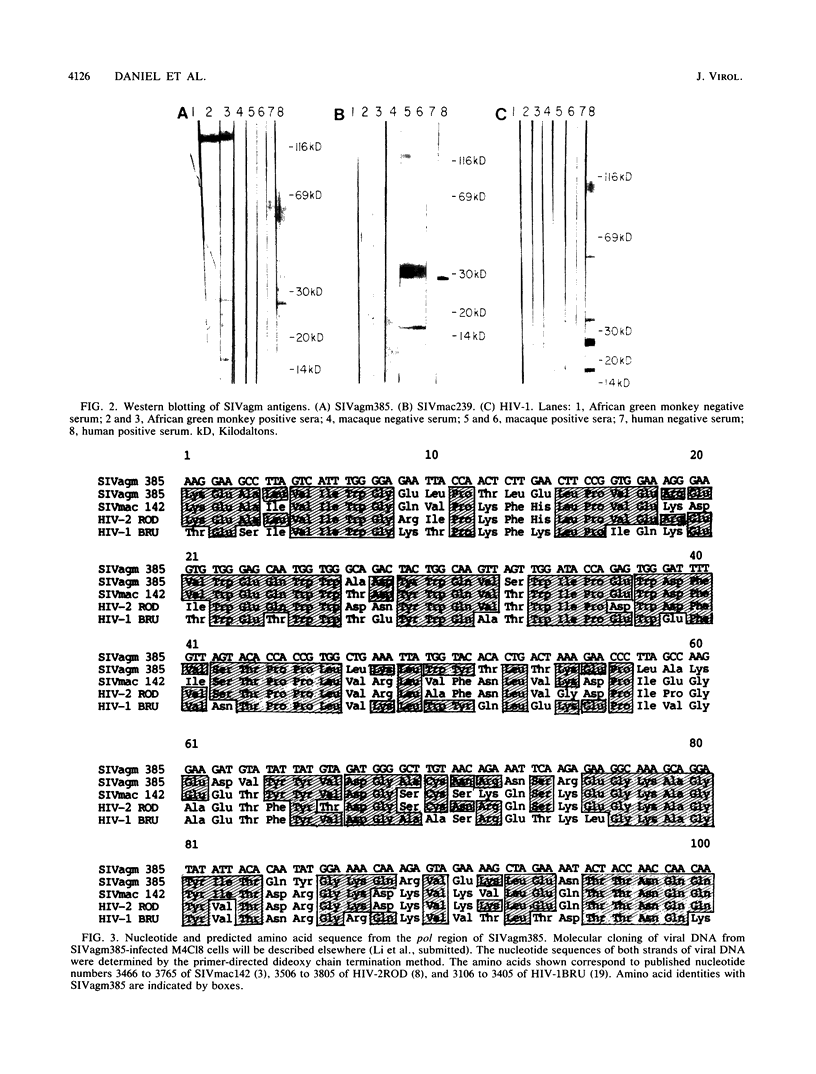
Simian immunodeficiency virus (SIV) was isolated from the total peripheral blood mononuclear cell population and the monocyte-macrophage adherent cell population of three seropositive green monkeys originating from Kenya. SIV from these African green monkeys (SIVagm) was isolated and continuously produced with the MOLT-4 clone 8 (M4C18) cell line but not with a variety of other cells including HUT-78, H9, CEM, MT-4, U937, and uncloned MOLT-4 cells. Once isolated, these SIVagm isolates were found to replicate efficiently in M4C18, SupT1, MT-4, U937, and Jurkat-T cells but much less efficiently if at all in HUT-78, H9, CEM, and MOLT-4 cells. The range of CD4+ cells fully permissive for replication of these SIVagm isolates thus differs markedly from that of previous SIV isolates from macaques (SIVmac). These SIVagm isolates had a morphogenesis and morphology like that of human immunodeficiency virus (HIV) and other SIV isolates. Antigens of SIVagm and SIVmac cross-reacted by comparative enzyme-linked immunosorbent assay only with reduced efficiency, and optimal results were obtained when homologous antibody and antigen were used. Western blotting (immunoblotting) of purified preparations of SIVagm isolate 385 (SIVagm385) revealed major viral proteins of 120, 27, and 16 kilodaltons (kDa). The presumed major core protein of 27 kDa cross-reacted antigenically with the corresponding proteins of SIVmac (28 kDa) and HIV-1 (24 kDa) by Western blotting. Hirt supernatant replicative-intermediate DNA prepared from cells freshly infected with SIVagm hybridized to SIVmac and HIV-2 DNA probes. Detection of cross-hybridizing DNA sequences, however, required very low stringency, and the restriction endonuclease fragmentation patterns of SIVagm were not similar to those of SIVmac and HIV-2. The nucleotide sequence of a portion of the pol gene of SIVagm385 revealed amino acid identities of 65% with SIVmac142, 64% with HIV-2ROD, and 56% with HIV-1BRU; SIVagm385 is thus related to but distinct from previously described primate lentiviruses SIVmac, HIV-1, and HIV-2. Precise information on the genetic makeup of these and other SIV isolates will possibly lead to better understanding of the history and evolution of these viruses and may provide insight into the origin of viruses that cause acquired immunodeficiency syndrome in humans.
Full text
PDF



Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Alizon M., Wain-Hobson S., Montagnier L., Sonigo P. Genetic variability of the AIDS virus: nucleotide sequence analysis of two isolates from African patients. Cell. 1986 Jul 4;46(1):63–74. doi: 10.1016/0092-8674(86)90860-3. [DOI] [PubMed] [Google Scholar]
- Benveniste R. E., Arthur L. O., Tsai C. C., Sowder R., Copeland T. D., Henderson L. E., Oroszlan S. Isolation of a lentivirus from a macaque with lymphoma: comparison with HTLV-III/LAV and other lentiviruses. J Virol. 1986 Nov;60(2):483–490. doi: 10.1128/jvi.60.2.483-490.1986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chakrabarti L., Guyader M., Alizon M., Daniel M. D., Desrosiers R. C., Tiollais P., Sonigo P. Sequence of simian immunodeficiency virus from macaque and its relationship to other human and simian retroviruses. Nature. 1987 Aug 6;328(6130):543–547. doi: 10.1038/328543a0. [DOI] [PubMed] [Google Scholar]
- Daniel M. D., Letvin N. L., King N. W., Kannagi M., Sehgal P. K., Hunt R. D., Kanki P. J., Essex M., Desrosiers R. C. Isolation of T-cell tropic HTLV-III-like retrovirus from macaques. Science. 1985 Jun 7;228(4704):1201–1204. doi: 10.1126/science.3159089. [DOI] [PubMed] [Google Scholar]
- Daniel M. D., Letvin N. L., Sehgal P. K., Hunsmann G., Schmidt D. K., King N. W., Desrosiers R. C. Long-term persistent infection of macaque monkeys with the simian immunodeficiency virus. J Gen Virol. 1987 Dec;68(Pt 12):3183–3189. doi: 10.1099/0022-1317-68-12-3183. [DOI] [PubMed] [Google Scholar]
- Daniel M. D., Letvin N. L., Sehgal P. K., Schmidt D. K., Silva D. P., Solomon K. R., Hodi F. S., Jr, Ringler D. J., Hunt R. D., King N. W. Prevalence of antibodies to 3 retroviruses in a captive colony of macaque monkeys. Int J Cancer. 1988 Apr 15;41(4):601–608. doi: 10.1002/ijc.2910410421. [DOI] [PubMed] [Google Scholar]
- Fukasawa M., Miura T., Hasegawa A., Morikawa S., Tsujimoto H., Miki K., Kitamura T., Hayami M. Sequence of simian immunodeficiency virus from African green monkey, a new member of the HIV/SIV group. Nature. 1988 Jun 2;333(6172):457–461. doi: 10.1038/333457a0. [DOI] [PubMed] [Google Scholar]
- Gonda M. A., Braun M. J., Carter S. G., Kost T. A., Bess J. W., Jr, Arthur L. O., Van der Maaten M. J. Characterization and molecular cloning of a bovine lentivirus related to human immunodeficiency virus. 1987 Nov 26-Dec 2Nature. 330(6146):388–391. doi: 10.1038/330388a0. [DOI] [PubMed] [Google Scholar]
- Guyader M., Emerman M., Sonigo P., Clavel F., Montagnier L., Alizon M. Genome organization and transactivation of the human immunodeficiency virus type 2. Nature. 1987 Apr 16;326(6114):662–669. doi: 10.1038/326662a0. [DOI] [PubMed] [Google Scholar]
- Hahn B. H., Kong L. I., Lee S. W., Kumar P., Taylor M. E., Arya S. K., Shaw G. M. Relation of HTLV-4 to simian and human immunodeficiency-associated viruses. Nature. 1987 Nov 12;330(6144):184–186. doi: 10.1038/330184a0. [DOI] [PubMed] [Google Scholar]
- Harada S., Purtilo D. T., Koyanagi Y., Sonnabend J., Yamamoto N. Sensitive assay for neutralizing antibodies against AIDS-related viruses (HTLV-III/LAV). J Immunol Methods. 1986 Sep 27;92(2):177–181. doi: 10.1016/0022-1759(86)90163-8. [DOI] [PubMed] [Google Scholar]
- Kanki P. J., Alroy J., Essex M. Isolation of T-lymphotropic retrovirus related to HTLV-III/LAV from wild-caught African green monkeys. Science. 1985 Nov 22;230(4728):951–954. doi: 10.1126/science.2997923. [DOI] [PubMed] [Google Scholar]
- Kanki P. J., Kurth R., Becker W., Dreesman G., McLane M. F., Essex M. Antibodies to simian T-lymphotropic retrovirus type III in African green monkeys and recognition of STLV-III viral proteins by AIDS and related sera. Lancet. 1985 Jun 8;1(8441):1330–1332. doi: 10.1016/s0140-6736(85)92818-1. [DOI] [PubMed] [Google Scholar]
- Kestler H. W., 3rd, Li Y., Naidu Y. M., Butler C. V., Ochs M. F., Jaenel G., King N. W., Daniel M. D., Desrosiers R. C. Comparison of simian immunodeficiency virus isolates. Nature. 1988 Feb 18;331(6157):619–622. doi: 10.1038/331619a0. [DOI] [PubMed] [Google Scholar]
- Kikukawa R., Koyanagi Y., Harada S., Kobayashi N., Hatanaka M., Yamamoto N. Differential susceptibility to the acquired immunodeficiency syndrome retrovirus in cloned cells of human leukemic T-cell line Molt-4. J Virol. 1986 Mar;57(3):1159–1162. doi: 10.1128/jvi.57.3.1159-1162.1986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kornfeld H., Riedel N., Viglianti G. A., Hirsch V., Mullins J. I. Cloning of HTLV-4 and its relation to simian and human immunodeficiency viruses. Nature. 1987 Apr 9;326(6113):610–613. doi: 10.1038/326610a0. [DOI] [PubMed] [Google Scholar]
- Ohta Y., Masuda T., Tsujimoto H., Ishikawa K., Kodama T., Morikawa S., Nakai M., Honjo S., Hayami M. Isolation of simian immunodeficiency virus from African green monkeys and seroepidemiologic survey of the virus in various non-human primates. Int J Cancer. 1988 Jan 15;41(1):115–122. doi: 10.1002/ijc.2910410121. [DOI] [PubMed] [Google Scholar]
- Sanger F., Nicklen S., Coulson A. R. DNA sequencing with chain-terminating inhibitors. Proc Natl Acad Sci U S A. 1977 Dec;74(12):5463–5467. doi: 10.1073/pnas.74.12.5463. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wain-Hobson S., Sonigo P., Danos O., Cole S., Alizon M. Nucleotide sequence of the AIDS virus, LAV. Cell. 1985 Jan;40(1):9–17. doi: 10.1016/0092-8674(85)90303-4. [DOI] [PubMed] [Google Scholar]
- Wendler I., Bienzle U., Hunsmann G. Neutralizing antibodies and the course of HIV-induced disease. AIDS Res Hum Retroviruses. 1987 Summer;3(2):157–163. doi: 10.1089/aid.1987.3.157. [DOI] [PubMed] [Google Scholar]



