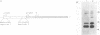Abstract
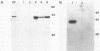
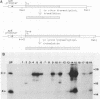
The DNA sequence of the ground squirrel hepatitis virus (GSHV) genome predicts the existence of several proteins in addition to the major surface (S) and core antigens. These include the pre-S1 and pre-S2 proteins, initiated at sites within the open reading frame preceding and continuous with the coding region for the S gene product, and the X protein, the putative product of an independent reading frame. Using an antibody directed against a peptide predicted by codons 130 to 143 of the pre-S1 reading frame, we identified a 43-kilodalton product of the pre-S1 coding region in preparations of GSHV surface antigen purified from the sera of infected animals. In addition, by immunoprecipitation of S- and pre-S-specific in vitro translation products with ground squirrel sera obtained after GSHV infection, we determined that antibodies arise to both S and pre-S determinants. The antibody response to pre-S includes, in some cases, reactivity to pre-S1-specific domains and is not always associated with an anti-S response. Similarly, by production of the viral X gene product in vitro followed by immunoprecipitation with ground squirrel sera, we showed that antibodies to this viral gene product also arise during infection, indicating that X antigenic determinants are synthesized during viral infection and are recognized by the host immune system.
Full text
PDF







Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Dane D. S., Cameron C. H., Briggs M. Virus-like particles in serum of patients with Australia-antigen-associated hepatitis. Lancet. 1970 Apr 4;1(7649):695–698. doi: 10.1016/s0140-6736(70)90926-8. [DOI] [PubMed] [Google Scholar]
- Enders G. H., Ganem D., Varmus H. Mapping the major transcripts of ground squirrel hepatitis virus: the presumptive template for reverse transcriptase is terminally redundant. Cell. 1985 Aug;42(1):297–308. doi: 10.1016/s0092-8674(85)80125-2. [DOI] [PubMed] [Google Scholar]
- Feitelson M. A., Marion P. L., Robinson W. S. Core particles of hepatitis B virus and ground squirrel hepatitis virus. I. Relationship between hepatitis B core antigen- and ground squirrel hepatitis core antigen-associated polypeptides by sodium dodecyl sulfate-polyacrylamide gel electrophoresis and tryptic peptide mapping. J Virol. 1982 Aug;43(2):687–696. doi: 10.1128/jvi.43.2.687-696.1982. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Feitelson M. A., Marion P. L., Robinson W. S. The nature of polypeptides larger in size than the major surface antigen components of hepatitis b and like viruses in ground squirrels, woodchucks, and ducks. Virology. 1983 Oct 15;130(1):76–90. doi: 10.1016/0042-6822(83)90119-8. [DOI] [PubMed] [Google Scholar]
- Galibert F., Chen T. N., Mandart E. Nucleotide sequence of a cloned woodchuck hepatitis virus genome: comparison with the hepatitis B virus sequence. J Virol. 1982 Jan;41(1):51–65. doi: 10.1128/jvi.41.1.51-65.1982. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ganem D., Greenbaum L., Varmus H. E. Virion DNA of ground squirrel hepatitis virus: structural analysis and molecular cloning. J Virol. 1982 Oct;44(1):374–383. doi: 10.1128/jvi.44.1.374-383.1982. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ganem D., Weiser B., Barchuk A., Brown R. J., Varmus H. E. Biological characterization of acute infection with ground squirrel hepatitis virus. J Virol. 1982 Oct;44(1):366–373. doi: 10.1128/jvi.44.1.366-373.1982. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gerlich W. H., Feitelson M. A., Marion P. L., Robinson W. S. Structural relationships between the surface antigens of ground squirrel hepatitis virus and human hepatitis B virus. J Virol. 1980 Dec;36(3):787–795. doi: 10.1128/jvi.36.3.787-795.1980. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Heermann K. H., Goldmann U., Schwartz W., Seyffarth T., Baumgarten H., Gerlich W. H. Large surface proteins of hepatitis B virus containing the pre-s sequence. J Virol. 1984 Nov;52(2):396–402. doi: 10.1128/jvi.52.2.396-402.1984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kay A., Mandart E., Trepo C., Galibert F. The HBV HBX gene expressed in E. coli is recognised by sera from hepatitis patients. EMBO J. 1985 May;4(5):1287–1292. doi: 10.1002/j.1460-2075.1985.tb03774.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kozak M. Comparison of initiation of protein synthesis in procaryotes, eucaryotes, and organelles. Microbiol Rev. 1983 Mar;47(1):1–45. doi: 10.1128/mr.47.1.1-45.1983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lerner R. A., Green N., Alexander H., Liu F. T., Sutcliffe J. G., Shinnick T. M. Chemically synthesized peptides predicted from the nucleotide sequence of the hepatitis B virus genome elicit antibodies reactive with the native envelope protein of Dane particles. Proc Natl Acad Sci U S A. 1981 Jun;78(6):3403–3407. doi: 10.1073/pnas.78.6.3403. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Machida A., Kishimoto S., Ohnuma H., Baba K., Ito Y., Miyamoto H., Funatsu G., Oda K., Usuda S., Togami S. A polypeptide containing 55 amino acid residues coded by the pre-S region of hepatitis B virus deoxyribonucleic acid bears the receptor for polymerized human as well as chimpanzee albumins. Gastroenterology. 1984 May;86(5 Pt 1):910–918. [PubMed] [Google Scholar]
- Machida A., Kishimoto S., Ohnuma H., Miyamoto H., Baba K., Oda K., Nakamura T., Miyakawa Y., Mayumi M. A hepatitis B surface antigen polypeptide (P31) with the receptor for polymerized human as well as chimpanzee albumins. Gastroenterology. 1983 Aug;85(2):268–274. [PubMed] [Google Scholar]
- Mandart E., Kay A., Galibert F. Nucleotide sequence of a cloned duck hepatitis B virus genome: comparison with woodchuck and human hepatitis B virus sequences. J Virol. 1984 Mar;49(3):782–792. doi: 10.1128/jvi.49.3.782-792.1984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Marion P. L., Knight S. S., Salazar F. H., Popper H., Robinson W. S. Ground squirrel hepatitis virus infection. Hepatology. 1983 Jul-Aug;3(4):519–527. doi: 10.1002/hep.1840030408. [DOI] [PubMed] [Google Scholar]
- Marion P. L., Oshiro L. S., Regnery D. C., Scullard G. H., Robinson W. S. A virus in Beechey ground squirrels that is related to hepatitis B virus of humans. Proc Natl Acad Sci U S A. 1980 May;77(5):2941–2945. doi: 10.1073/pnas.77.5.2941. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Meyers M. L., Trepo L. V., Nath N., Sninsky J. J. Hepatitis B virus polypeptide X: expression in Escherichia coli and identification of specific antibodies in sera from hepatitis B virus-infected humans. J Virol. 1986 Jan;57(1):101–109. doi: 10.1128/jvi.57.1.101-109.1986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Milich D. R., McNamara M. K., McLachlan A., Thornton G. B., Chisari F. V. Distinct H-2-linked regulation of T-cell responses to the pre-S and S regions of the same hepatitis B surface antigen polypeptide allows circumvention of nonresponsiveness to the S region. Proc Natl Acad Sci U S A. 1985 Dec;82(23):8168–8172. doi: 10.1073/pnas.82.23.8168. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Milich D. R., Thornton G. B., Neurath A. R., Kent S. B., Michel M. L., Tiollais P., Chisari F. V. Enhanced immunogenicity of the pre-S region of hepatitis B surface antigen. Science. 1985 Jun 7;228(4704):1195–1199. doi: 10.1126/science.2408336. [DOI] [PubMed] [Google Scholar]
- Moriarty A. M., Alexander H., Lerner R. A., Thornton G. B. Antibodies to peptides detect new hepatitis B antigen: serological correlation with hepatocellular carcinoma. Science. 1985 Jan 25;227(4685):429–433. doi: 10.1126/science.2981434. [DOI] [PubMed] [Google Scholar]
- Persing D. H., Varmus H. E., Ganem D. A frameshift mutation in the pre-S region of the human hepatitis B virus genome allows production of surface antigen particles but eliminates binding to polymerized albumin. Proc Natl Acad Sci U S A. 1985 May;82(10):3440–3444. doi: 10.1073/pnas.82.10.3440. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Peterson D. L. Isolation and characterization of the major protein and glycoprotein of hepatitis B surface antigen. J Biol Chem. 1981 Jul 10;256(13):6975–6983. [PubMed] [Google Scholar]
- Peterson D. L., Nath N., Gavilanes F. Structure of hepatitis B surface antigen. Correlation of subtype with amino acid sequence and location of the carbohydrate moiety. J Biol Chem. 1982 Sep 10;257(17):10414–10420. [PubMed] [Google Scholar]
- Robinson W. S., Lutwick L. I. The virus of hepatitis, type B. (Second of two parts). N Engl J Med. 1976 Nov 25;295(22):1232–1236. doi: 10.1056/NEJM197611252952206. [DOI] [PubMed] [Google Scholar]
- Schaeffer E., Snyder R. L., Sninsky J. J. Identification and localization of pre-s-encoded polypeptides from woodchuck and ground squirrel hepatitis viruses. J Virol. 1986 Jan;57(1):173–182. doi: 10.1128/jvi.57.1.173-182.1986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Seeger C., Ganem D., Varmus H. E. Nucleotide sequence of an infectious molecularly cloned genome of ground squirrel hepatitis virus. J Virol. 1984 Aug;51(2):367–375. doi: 10.1128/jvi.51.2.367-375.1984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Seeger C., Ganem D., Varmus H. E. The cloned genome of ground squirrel hepatitis virus is infectious in the animal. Proc Natl Acad Sci U S A. 1984 Sep;81(18):5849–5852. doi: 10.1073/pnas.81.18.5849. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Siddiqui A., Marion P. L., Robinson W. S. Ground squirrel hepatitis virus DNA: molecular cloning and comparison with hepatitis B virus DNA. J Virol. 1981 Apr;38(1):393–397. doi: 10.1128/jvi.38.1.393-397.1981. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stibbe W., Gerlich W. H. Structural relationships between minor and major proteins of hepatitis B surface antigen. J Virol. 1983 May;46(2):626–628. doi: 10.1128/jvi.46.2.626-628.1983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wong D. T., Nath N., Sninsky J. J. Identification of hepatitis B virus polypeptides encoded by the entire pre-s open reading frame. J Virol. 1985 Jul;55(1):223–231. doi: 10.1128/jvi.55.1.223-231.1985. [DOI] [PMC free article] [PubMed] [Google Scholar]