Abstract
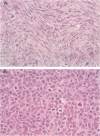
Subcutaneous injection of wild-type simian virus 40 into Syrian hamsters normally induces fibrosarcomas at the injection site. We showed that subcutaneous injection of three different small t deletion mutants (dl884, dl883, and dl890) led to the formation of abdominal reticulum cell sarcomas (lymphomas) in about 15% of the animals bearing tumors. The remainder of the tumors were fibrosarcomas occurring with prolonged latencies at the site of injection. We postulated that, in the absence of an active small t protein, which is thought to have cell growth-promoting properties, the mutant virus preferentially transforms rapidly proliferating lymphoid cells.
Full text
PDF


Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Bouck N., Beales N., Shenk T., Berg P., di Mayorca G. New region of the simian virus 40 genome required for efficient viral transformation. Proc Natl Acad Sci U S A. 1978 May;75(5):2473–2477. doi: 10.1073/pnas.75.5.2473. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Diamandopoulos G. T. Induction of lymphocytic leukemia, lymphosarcoma, reticulum cell sarcoma, and osteogenic sarcoma in the Syrian golden hamster by oncogenic DNA simian virus 40. J Natl Cancer Inst. 1973 May;50(5):1347–1365. doi: 10.1093/jnci/50.5.1347. [DOI] [PubMed] [Google Scholar]
- Diamandopoulos G. T., McLane M. F. Effect of host age, virus dose, and route of inoculation on tumor incidence, latency, and morphology in Syrian hamsters inoculated intravenously with oncogenic DNA simian virus 40. J Natl Cancer Inst. 1975 Aug;55(2):479–482. [PubMed] [Google Scholar]
- Dixon K., Ryder B. J., Burch-Jaffe E. Enhanced metastasis of tumours induced by a SV40 small T deletion mutant. Nature. 1982 Apr 15;296(5858):672–675. doi: 10.1038/296672a0. [DOI] [PubMed] [Google Scholar]
- EDDY B. E., BORMAN G. S., BERKELEY W. H., YOUNG R. D. Tumors induced in hamsters by injection of rhesus monkey kidney cell extracts. Proc Soc Exp Biol Med. 1961 May;107:191–197. doi: 10.3181/00379727-107-26576. [DOI] [PubMed] [Google Scholar]
- EDDY B. E., BORMAN G. S., GRUBBS G. E., YOUNG R. D. Identification of the oncogenic substance in rhesus monkey kidney cell culture as simian virus 40. Virology. 1962 May;17:65–75. doi: 10.1016/0042-6822(62)90082-x. [DOI] [PubMed] [Google Scholar]
- EDDY B. E. SIMIAN VIRUS 40 (SV-40): AN ONCOGENIC VIRUS. Prog Exp Tumor Res. 1964;4:1–26. doi: 10.1159/000385971. [DOI] [PubMed] [Google Scholar]
- GIRARDI A. J., SWEET B. H., SLOTNICK V. B., HILLEMAN M. R. Development of tumors in hamsters inoculated in the neonatal period with vacuolating virus, SV-40. Proc Soc Exp Biol Med. 1962 Mar;109:649–660. doi: 10.3181/00379727-109-27298. [DOI] [PubMed] [Google Scholar]
- Lewis A. M., Jr, Martin R. G. Oncogenicity of simian virus 40 deletion mutants that induce altered 17-kilodalton t-proteins. Proc Natl Acad Sci U S A. 1979 Sep;76(9):4299–4302. doi: 10.1073/pnas.76.9.4299. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Martin R. G., Chou J. Y. Simian virus 40 functions required for the establishment and maintenance of malignant transformation. J Virol. 1975 Mar;15(3):599–612. doi: 10.1128/jvi.15.3.599-612.1975. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Martin R. G., Setlow V. P., Edwards C. A. Roles of the simian virus 40 tumor antigens in transformation of Chinese hamster lung cells: studies with simian virus 40 double mutants. J Virol. 1979 Sep;31(3):596–607. doi: 10.1128/jvi.31.3.596-607.1979. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Martin R. G., Setlow V. P., Edwards C. A., Vembu D. The roles of the simian virus 40 tumor antigens in transformation of Chinese hamster lung cells. Cell. 1979 Jul;17(3):635–643. doi: 10.1016/0092-8674(79)90271-x. [DOI] [PubMed] [Google Scholar]
- Seif R., Martin R. G. Growth state of the cell early after infection with simian virus 40 determines whether the maintenance of transformation will be A-gene dependent or independent. J Virol. 1979 Aug;31(2):350–359. doi: 10.1128/jvi.31.2.350-359.1979. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shenk T. E., Carbon J., Berg P. Construction and analysis of viable deletion mutants of simian virus 40. J Virol. 1976 May;18(2):664–671. doi: 10.1128/jvi.18.2.664-671.1976. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sleigh M. J., Topp W. C., Hanich R., Sambrook J. F. Mutants of SV40 with an altered small t protein are reduced in their ability to transform cells. Cell. 1978 May;14(1):79–88. doi: 10.1016/0092-8674(78)90303-3. [DOI] [PubMed] [Google Scholar]
- Tegtmeyer P. Function of simian virus 40 gene A in transforming infection. J Virol. 1975 Mar;15(3):613–618. doi: 10.1128/jvi.15.3.613-618.1975. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thimmappaya B., Shenk T. Nucleotide sequence analysis of viable deletion mutants lacking segments of the simian virus 40 genome coding for small t antigen. J Virol. 1979 Jun;30(3):668–673. doi: 10.1128/jvi.30.3.668-673.1979. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Topp W. C., Rifkin D. B., Sleigh M. J. SV40 mutants with an altered small-t protein are tumorigenic in newborn hamsters. Virology. 1981 Jun;111(2):341–350. doi: 10.1016/0042-6822(81)90338-x. [DOI] [PubMed] [Google Scholar]