Abstract
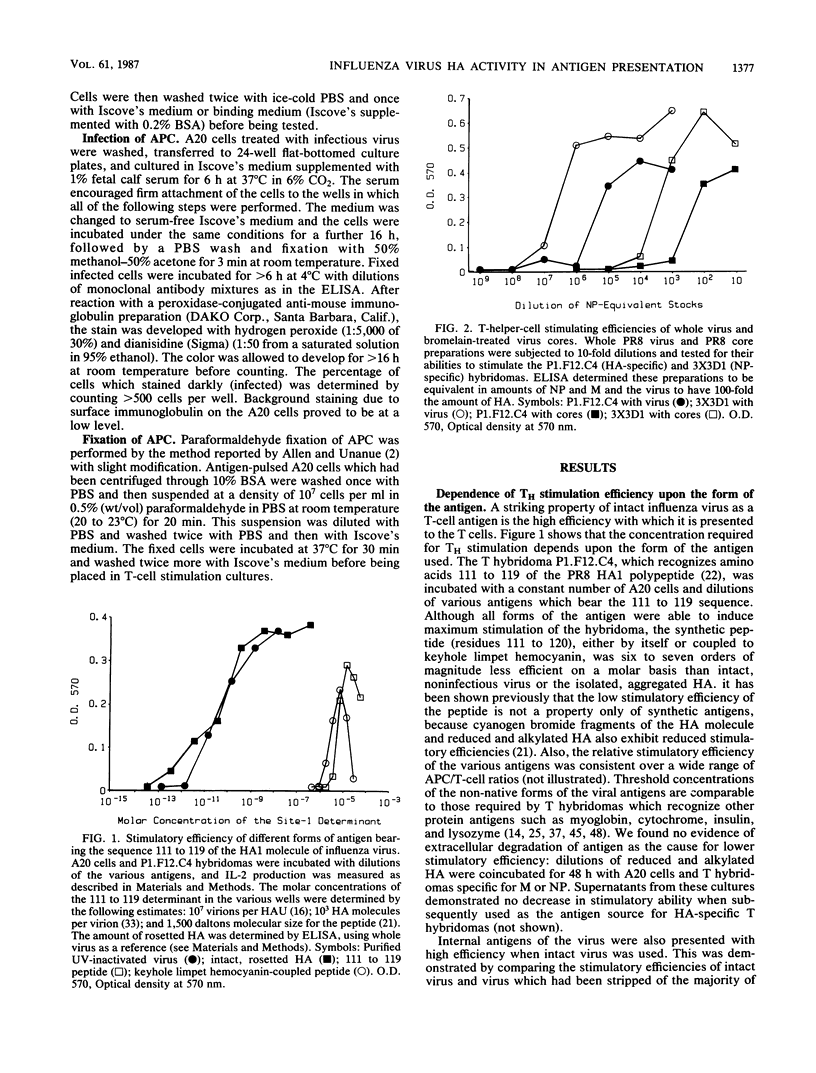
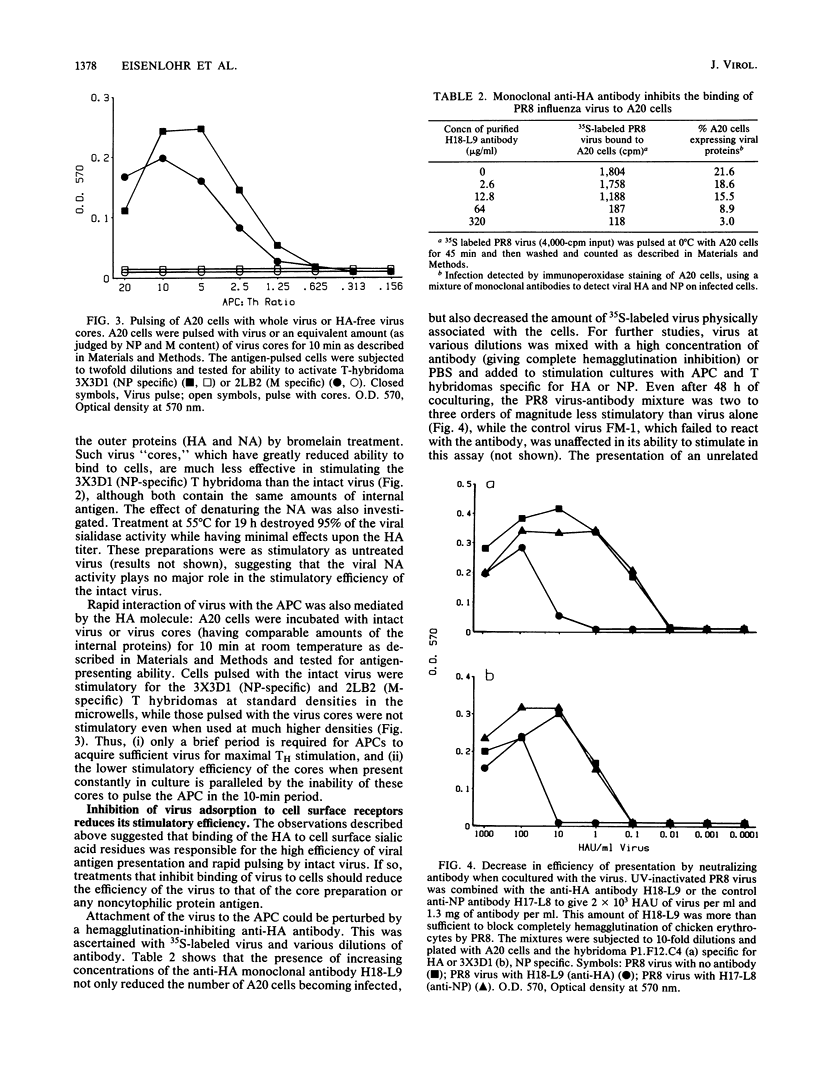
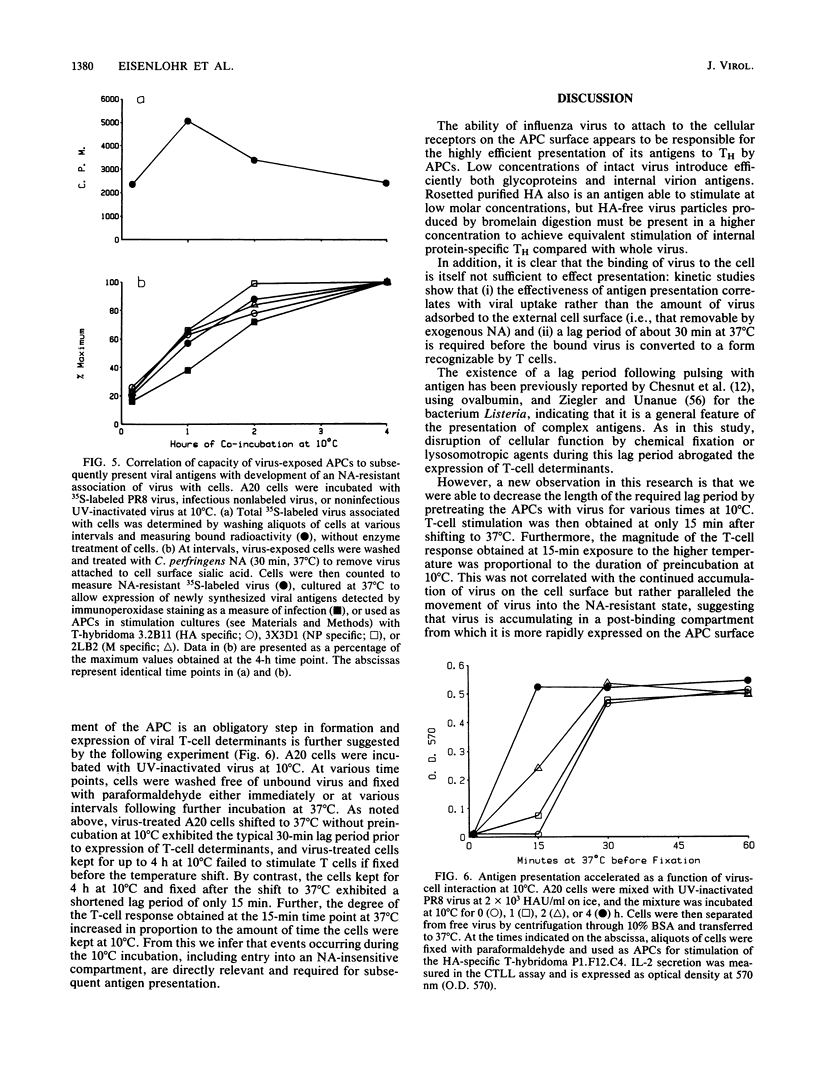
The concentration of antigen required to stimulate influenza virus-specific helper T cells was observed to be dependent upon the antigenic form bearing the relevant determinant: intact, nonreplicative virus was needed only in picomolar amounts, while denatured proteins, protein fragments, or synthetic peptides were required in micromolar concentrations for a threshold level of stimulation. Antigenic efficiency of intact virus was found to result from the attachment of virus to sialic acid residues on the surface of the antigen-presenting cell since spikeless viral particles lacking the hemagglutinin molecule were much less efficient antigens for helper T cells and continuous presence of hemagglutination-inhibiting antihemagglutinin antibodies reduced efficiency of stimulation by intact virus approximately 100-fold for both hemagglutinin and internal virion proteins. Influenza virus associated rapidly with antigen-presenting cells; less than 10 min at 20 degrees C was sufficient to introduce virus for a maximal level of T-cell stimulation. This rapid attachment was blocked by antibodies to the hemagglutinin or by pretreatment of the antigen-presenting cells with neuraminidase to remove the cellular virus receptor. Following viral adsorption by antigen-presenting cells, a lag period of 30 min at 37 degrees C was required for the expression of helper T-cell determinants. One early event identified was the movement of the virus to a neuraminidase-insensitive compartment, which can occur at 10 degrees C, but which was not equivalent to expression of helper T-cell determinants. Preincubation of cells with virus at 10 degrees C for 4 h reduced the lag period of helper T-cell determinant expression to 15 min when these cells were shifted to 37 degrees C, suggesting that transition of the virus to a neuraminidase-resistant state is a required step in presentation of T-cell antigenic determinants.
Full text
PDF





Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Allen P. M., Beller D. I., Braun J., Unanue E. R. The handling of Listeria monocytogenes by macrophages: the search for an immunogenic molecule in antigen presentation. J Immunol. 1984 Jan;132(1):323–331. [PubMed] [Google Scholar]
- Allen P. M., Unanue E. R. Differential requirements for antigen processing by macrophages for lysozyme-specific T cell hybridomas. J Immunol. 1984 Mar;132(3):1077–1079. [PubMed] [Google Scholar]
- Ashman R. B., Müllbacher A. A T helper cell for anti-viral cytotoxic T-cell responses. J Exp Med. 1979 Nov 1;150(5):1277–1282. doi: 10.1084/jem.150.5.1277. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Askonas B. A., Mullbacher A., Ashman R. B. Cytotoxic T-memory cells in virus infection and the specificity of helper T cells. Immunology. 1982 Jan;45(1):79–84. [PMC free article] [PubMed] [Google Scholar]
- Benacerraf B. Role of MHC gene products in immune regulation. Science. 1981 Jun 12;212(4500):1229–1238. doi: 10.1126/science.6165083. [DOI] [PubMed] [Google Scholar]
- Bos E. S., van der Doelen A. A., van Rooy N., Schuurs A. H. 3,3',5,5' - Tetramethylbenzidine as an Ames test negative chromogen for horse-radish peroxidase in enzyme-immunoassay. J Immunoassay. 1981;2(3-4):187–204. doi: 10.1080/15321818108056977. [DOI] [PubMed] [Google Scholar]
- Brand C. M., Skehel J. J. Crystalline antigen from the influenza virus envelope. Nat New Biol. 1972 Aug 2;238(83):145–147. doi: 10.1038/newbio238145a0. [DOI] [PubMed] [Google Scholar]
- Buck C. A., Warren L. The repair of the surface structure of animal cells. J Cell Physiol. 1976 Oct;89(2):187–200. doi: 10.1002/jcp.1040890202. [DOI] [PubMed] [Google Scholar]
- Burns W., Billups L. C., Notkins A. L. Thymus dependence of viral antigens. Nature. 1975 Aug 21;256(5519):654–656. doi: 10.1038/256654a0. [DOI] [PubMed] [Google Scholar]
- Butchko G. M., Armstrong R. B., Ennis F. A. Specificity studies on the proliferative response of thymus-derived lymphocytes to influenza viruses. J Immunol. 1978 Dec;121(6):2381–2385. [PubMed] [Google Scholar]
- Chesnut R. W., Grey H. M. Antigen presenting cells and mechanisms of antigen presentation. Crit Rev Immunol. 1985;5(3):263–316. [PubMed] [Google Scholar]
- Corradin G., Chiller J. M. Lymphocyte specificity to protein antigens. II. Fine specificity of T-cell activation with cytochrome c and derived peptides as antigenic probes. J Exp Med. 1979 Feb 1;149(2):436–447. doi: 10.1084/jem.149.2.436. [DOI] [PMC free article] [PubMed] [Google Scholar]
- DONALD H. B., ISAACS A. Counts of influenza virus particles. J Gen Microbiol. 1954 Jun;10(3):457–464. doi: 10.1099/00221287-10-3-457. [DOI] [PubMed] [Google Scholar]
- DeFreitas E. C., Chesnut R. W., Grey H. M., Chiller J. M. Macrophage-dependent activation of antigen-specific T cells requires antigen and a soluble monokine. J Immunol. 1983 Jul;131(1):23–29. [PubMed] [Google Scholar]
- Dunn W. A., Hubbard A. L., Aronson N. N., Jr Low temperature selectively inhibits fusion between pinocytic vesicles and lysosomes during heterophagy of 125I-asialofetuin by the perfused rat liver. J Biol Chem. 1980 Jun 25;255(12):5971–5978. [PubMed] [Google Scholar]
- Fazekas de St Groth, Webster R. G. Disquisitions of Original Antigenic Sin. I. Evidence in man. J Exp Med. 1966 Sep 1;124(3):331–345. doi: 10.1084/jem.124.3.331. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gerhard W., Hackett C., Melchers F. The recognition specificity of a murine helper T cell for hemagglutinin of influenza virus A/PR/8/34. J Immunol. 1983 May;130(5):2379–2385. [PubMed] [Google Scholar]
- Gillis S., Ferm M. M., Ou W., Smith K. A. T cell growth factor: parameters of production and a quantitative microassay for activity. J Immunol. 1978 Jun;120(6):2027–2032. [PubMed] [Google Scholar]
- Hackett C. J., Dietzschold B., Gerhard W., Ghrist B., Knorr R., Gillessen D., Melchers F. Influenza virus site recognized by a murine helper T cell specific for H1 strains. Localization to a nine amino acid sequence in the hemagglutinin molecule. J Exp Med. 1983 Aug 1;158(2):294–302. doi: 10.1084/jem.158.2.294. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hackett C. J., Hurwitz J. L., Dietzschold B., Gerhard W. A synthetic decapeptide of influenza virus hemagglutinin elicits helper T cells with the same fine recognition specificities as occur in response to whole virus. J Immunol. 1985 Aug;135(2):1391–1394. [PubMed] [Google Scholar]
- Hurwitz J. L., Hackett C. J., McAndrew E. C., Gerhard W. Murine TH response to influenza virus: recognition of hemagglutinin, neuraminidase, matrix, and nucleoproteins. J Immunol. 1985 Mar;134(3):1994–1998. [PubMed] [Google Scholar]
- Hurwitz J. L., Herber-Katz E., Hackett C. J., Gerhard W. Characterization of the murine TH response to influenza virus hemagglutinin: evidence for three major specificities. J Immunol. 1984 Dec;133(6):3371–3377. [PubMed] [Google Scholar]
- Infante A. J., Atassi M. Z., Fathman C. G. T cell clones reactive with sperm whale myoglobin. Isolation of clones with specificity for individual determinants on myoglobin. J Exp Med. 1981 Nov 1;154(5):1342–1356. doi: 10.1084/jem.154.5.1342. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Iscove N. N., Melchers F. Complete replacement of serum by albumin, transferrin, and soybean lipid in cultures of lipopolysaccharide-reactive B lymphocytes. J Exp Med. 1978 Mar 1;147(3):923–933. doi: 10.1084/jem.147.3.923. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jakway J. P., Shevach E. M. Stimulation of T-cell activation by UV-treated, antigen-pulsed macrophages: evidence for a requirement for antigen processing and interleukin 1 secretion. Cell Immunol. 1983 Aug;80(1):151–162. doi: 10.1016/0008-8749(83)90102-8. [DOI] [PubMed] [Google Scholar]
- Kakiuchi T., Chesnut R. W., Grey H. M. B cells as antigen-presenting cells: the requirement for B cell activation. J Immunol. 1983 Jul;131(1):109–114. [PubMed] [Google Scholar]
- Katz J. M., Laver W. G., White D. O., Anders E. M. Recognition of influenza virus hemagglutinin by subtype-specific and cross-reactive proliferative T cells: contribution of HA1 and HA2 polypeptide chains. J Immunol. 1985 Jan;134(1):616–622. [PubMed] [Google Scholar]
- Kawamura H., Berzofsky J. A. Enhancement of antigenic potency in vitro and immunogenicity in vivo by coupling the antigen to anti-immunoglobulin. J Immunol. 1986 Jan;136(1):58–65. [PubMed] [Google Scholar]
- Kim K. J., Kanellopoulos-Langevin C., Merwin R. M., Sachs D. H., Asofsky R. Establishment and characterization of BALB/c lymphoma lines with B cell properties. J Immunol. 1979 Feb;122(2):549–554. [PubMed] [Google Scholar]
- Lamb J. R., Eckels D. D., Lake P., Woody J. N., Green N. Human T-cell clones recognize chemically synthesized peptides of influenza haemagglutinin. Nature. 1982 Nov 4;300(5887):66–69. doi: 10.1038/300066a0. [DOI] [PubMed] [Google Scholar]
- Lanzavecchia A. Antigen-specific interaction between T and B cells. Nature. 1985 Apr 11;314(6011):537–539. doi: 10.1038/314537a0. [DOI] [PubMed] [Google Scholar]
- Laver W. G., Webster R. G. Preparation and immunogenicity of an influenza virus hemagglutinin and neuraminidase subunit vaccine. Virology. 1976 Feb;69(2):511–522. doi: 10.1016/0042-6822(76)90481-5. [DOI] [PubMed] [Google Scholar]
- Londei M., Lamb J. R., Bottazzo G. F., Feldmann M. Epithelial cells expressing aberrant MHC class II determinants can present antigen to cloned human T cells. Nature. 1984 Dec 13;312(5995):639–641. doi: 10.1038/312639a0. [DOI] [PubMed] [Google Scholar]
- Maizels R. M., Clarke J. A., Harvey M. A., Miller A., Sercarz E. E. Epitope specificity of the T cell proliferative response to lysozyme: proliferative T cells react predominantly to different determinants from those recognized by B cells. Eur J Immunol. 1980 Jul;10(7):509–515. doi: 10.1002/eji.1830100705. [DOI] [PubMed] [Google Scholar]
- Malynn B. A., Wortis H. H. Role of antigen-specific B cells in the induction of SRBC-specific T cell proliferation. J Immunol. 1984 May;132(5):2253–2258. [PubMed] [Google Scholar]
- Marsh M., Helenius A., Matlin K., Simons K. Binding, endocytosis, and degradation of enveloped animal viruses. Methods Enzymol. 1983;98:260–266. doi: 10.1016/0076-6879(83)98153-3. [DOI] [PubMed] [Google Scholar]
- Matlin K. S., Reggio H., Helenius A., Simons K. Infectious entry pathway of influenza virus in a canine kidney cell line. J Cell Biol. 1981 Dec;91(3 Pt 1):601–613. doi: 10.1083/jcb.91.3.601. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mosmann T. Rapid colorimetric assay for cellular growth and survival: application to proliferation and cytotoxicity assays. J Immunol Methods. 1983 Dec 16;65(1-2):55–63. doi: 10.1016/0022-1759(83)90303-4. [DOI] [PubMed] [Google Scholar]
- Rock K. L., Benacerraf B., Abbas A. K. Antigen presentation by hapten-specific B lymphocytes. I. Role of surface immunoglobulin receptors. J Exp Med. 1984 Oct 1;160(4):1102–1113. doi: 10.1084/jem.160.4.1102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shastri N., Malissen B., Hood L. Ia-transfected L-cell fibroblasts present a lysozyme peptide but not the native protein to lysozyme-specific T cells. Proc Natl Acad Sci U S A. 1985 Sep;82(17):5885–5889. doi: 10.1073/pnas.82.17.5885. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shimonkevitz R., Kappler J., Marrack P., Grey H. Antigen recognition by H-2-restricted T cells. I. Cell-free antigen processing. J Exp Med. 1983 Aug 1;158(2):303–316. doi: 10.1084/jem.158.2.303. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Solinger A. M., Ultee M. E., Margoliash E., Schwartz R. H. T-lymphocyte response to cytochrome c. I. Demonstration of a T-cell heteroclitic proliferative response and identification of a topographic antigenic determinant on pigeon cytochrome c whose immune recognition requires two complementing major histocompatibility complex-linked immune response genes. J Exp Med. 1979 Oct 1;150(4):830–848. doi: 10.1084/jem.150.4.830. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Steinman R. M., Mellman I. S., Muller W. A., Cohn Z. A. Endocytosis and the recycling of plasma membrane. J Cell Biol. 1983 Jan;96(1):1–27. doi: 10.1083/jcb.96.1.1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Streicher H. Z., Berkower I. J., Busch M., Gurd F. R., Berzofsky J. A. Antigen conformation determines processing requirements for T-cell activation. Proc Natl Acad Sci U S A. 1984 Nov;81(21):6831–6835. doi: 10.1073/pnas.81.21.6831. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thomas J. W., Danho W., Bullesbach E., Föhles J., Rosenthal A. S. Immune response gene control of determinant selection. III. Polypeptide fragments of insulin are differentially recognized by T but not by B cells in insulin immune guinea pigs. J Immunol. 1981 Mar;126(3):1095–1100. [PubMed] [Google Scholar]
- Unanue E. R. Antigen-presenting function of the macrophage. Annu Rev Immunol. 1984;2:395–428. doi: 10.1146/annurev.iy.02.040184.002143. [DOI] [PubMed] [Google Scholar]
- Virelizier J. L., Postlethwaite R., Schild G. C., Allison A. C. Antibody responses to antigenic determinants of influenza virus hemagglutinin. I. Thymus dependence of antibody formation and thymus independence of immunological memory. J Exp Med. 1974 Dec 1;140(6):1559–1570. doi: 10.1084/jem.140.6.1559. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Walden P., Nagy Z. A., Klein J. Induction of regulatory T-lymphocyte responses by liposomes carrying major histocompatibility complex molecules and foreign antigen. Nature. 1985 May 23;315(6017):327–329. doi: 10.1038/315327a0. [DOI] [PubMed] [Google Scholar]
- Ware C. F., Reade J. L., Der L. C. A rat anti-mouse kappa chain specific monoclonal antibody, 187.1.10: purification, immunochemical properties and its utility as a general second-antibody reagent. J Immunol Methods. 1984 Nov 16;74(1):93–104. doi: 10.1016/0022-1759(84)90371-5. [DOI] [PubMed] [Google Scholar]
- Yelton D. E., Desaymard C., Scharff M. D. Use of monoclonal anti-mouse immunoglobulin to detect mouse antibodies. Hybridoma. 1981;1(1):5–11. doi: 10.1089/hyb.1.1981.1.5. [DOI] [PubMed] [Google Scholar]
- Yewdell J. W., Frank E., Gerhard W. Expression of influenza A virus internal antigens on the surface of infected P815 cells. J Immunol. 1981 May;126(5):1814–1819. [PubMed] [Google Scholar]
- Yoshimura A., Ohnishi S. Uncoating of influenza virus in endosomes. J Virol. 1984 Aug;51(2):497–504. doi: 10.1128/jvi.51.2.497-504.1984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ziegler K., Unanue E. R. Identification of a macrophage antigen-processing event required for I-region-restricted antigen presentation to T lymphocytes. J Immunol. 1981 Nov;127(5):1869–1875. [PubMed] [Google Scholar]



