Abstract
The incidence of enterotoxigenic Escherichia coli diarrhea among Egyptian children was 1.5 episodes per child per year and accounted for 66% of all first episodes of diarrhea after birth. The incidence increased from 1.7 episodes per child per year in the first 6 months of life to 2.3 in the second 6 months and declined thereafter.
Enterotoxigenic Escherichia coli (ETEC) has been recognized as the most common cause of infectious diarrhea in infants and young children in developing countries (14). Manifestations of ETEC infection run the spectrum from asymptomatic infection to severe dehydrating diarrheal illness. The virulence of ETEC is attributed to its ability to colonize the intestinal epithelium via fimbrial adhesive factors and express secretogenic enterotoxins of a heat-labile (LT) and/or a heat-stable (ST) variety (10).
The epidemiology of ETEC diarrhea was studied in a cohort of Egyptian children in two villages located in the Nile Delta. Children under the age of 24 months at the start of the study and all subsequent new-birth infants were monitored until the age of 36 months or the end of the study was reached. Surveillance for diarrhea was conducted by twice-weekly home visits in which fecal specimens were collected when loose stools were reported (9). For infants, a detailed dietary history (with reference to breastfeeding and the introduction of supplementary liquids and solids) was obtained at each visit. Standard methods were used to detect Salmonella, Shigella, Campylobacter, Vibrionaceae, rotavirus, and astrovirus (8, 9, 12). Rectal swabs were plated on McConkey's medium, and GM1-ganglioside enzyme-linked immunosorbent assays (11, 13) were used to evaluate five lactose-positive colonies for both LT and ST expression.
Diarrheal episodes were defined as beginning on the first day of loose stools after at least three consecutive nondiarrheal days and were considered to have ended when followed by 3 days without diarrhea (9). An episode was classified as an ETEC episode when ETEC was detected at any time during the episode. Children were considered breastfed if they had received any breast milk. To analyze the “time to first episode” of all-cause diarrhea or ETEC diarrhea, Kaplan Meier (KM) curves were constructed for new-birth infants to obtain the distribution of the times to first episode (5). Since exclusively breastfed children usually have loose stools that may be misclassified as diarrhea and no pathogens were isolated in 73% of the diarrheal episodes in the first 3 months of life, the analysis of the time to first episode was restricted to episodes associated with pathogens (ETEC, Campylobacter, Shigella, Salmonella, rotavirus, or astrovirus). Poisson regression models using generalized estimating equations were fitted to adjust for confounding variables (6).
In a cohort of 397 children with 211 new-birth infants, 3,477 episodes of diarrhea were detected over 3 years, resulting in an incidence of 5.5 episodes of diarrhea per child per year. Attack rates were highest during infancy (8.1 episodes per child per year). With an incidence of 1.5 episodes per child per year, ETEC was the most common cause of diarrhea; the next most common causes of diarrhea were Campylobacter (0.6 episodes per child per year) and Shigella, rotavirus, and astrovirus (approximately 0.2 episodes per child per year for each). The incidence of ETEC diarrhea increased from 1.7 episodes per child per year in the first 6 months of life to 2.3 episodes per child per year in the second 6 months and declined thereafter (Table 1).
TABLE 1.
Annual age-specific incidence rates (episodes per child per year) of all-cause diarrhea, ETEC diarrhea, and diarrhea due to other enteropathogens in Abu Homos, Egypt, from 1995 to 1998
| Agea | Incidence rate in episodes/child/yr of diarrhea (no. of cases) caused by:
|
||||||||
|---|---|---|---|---|---|---|---|---|---|
| All causes | All ETEC types | LT-ETECb | ST-ETECb | LTST-ETECb | Campylobacter | Shigella | Rotavirus | Astrovirus | |
| 0-5 | 7.33 (681) | 1.67 (155) | 0.53 (49) | 0.91 (85) | 0.18 (17) | 0.87 (81) | 0.08 (7) | 0.19 (18) | 0.38 (35) |
| 6-11 | 8.81 (913) | 2.26 (234) | 0.62 (64) | 1.24 (129) | 0.31 (32) | 1.48 (153) | 0.24 (25) | 0.38 (39) | 0.40 (41) |
| 12-17 | 6.40 (731) | 1.99 (227) | 0.51 (58) | 1.11 (127) | 0.28 (32) | 0.58 (66) | 0.42 (48) | 0.26 (30) | 0.18 (21) |
| 18-23 | 4.61 (542) | 1.35 (159) | 0.26 (31) | 0.87 (102) | 0.20 (24) | 0.29 (34) | 0.24 (28) | 0.13 (15) | 0.14 (16) |
| 24-35 | 2.92 (610) | 0.76 (158) | 0.17 (35) | 0.48 (101) | 0.11 (22) | 0.15 (32) | 0.12 (26) | 0.08 (16) | 0.05 (10) |
| All ages | 5.46 (3,477) | 1.47 (933) | 0.37 (237) | 0.85 (544) | 0.20 (127) | 0.57 (366) | 0.21 (134) | 0.19 (118) | 0.19 (123) |
Child age in months at the time of follow-up.
Results do not include findings for ETECs in which different colonies from the same fecal specimen express different toxins.
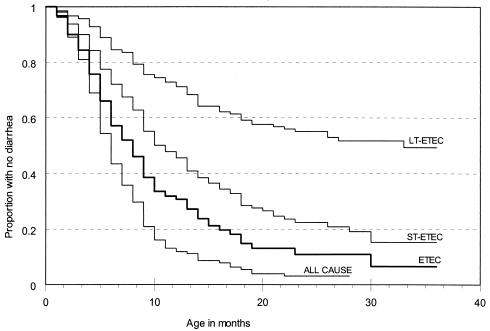
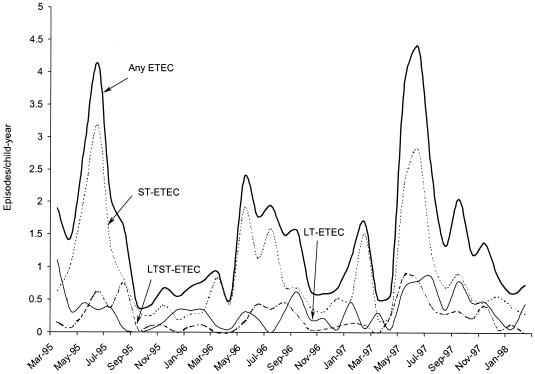
Among 211 new-birth infants, the median time to the first episode of pathogen-associated diarrhea was 6 months (95% confidence interval [CI], 5 to 7 months) and about 90% of the cohort experienced a first episode of pathogen-associated diarrhea by 14 months of age (Fig. 1). ETEC diarrhea accounted for 66% of all first episodes. The median time to a first episode of ETEC diarrhea was 8 months (95% CI, 7 to 9 months), and the KM curve for the time to first ETEC episode closely paralleled the KM curve for the time to first diarrheal episode associated with any pathogen (Fig. 1). In the first 3 years of life, approximately 84% of the cohort had at least one episode of ST-ETEC diarrhea, with a median time to first episode of 11 months (95% CI, 9 to 13 months) (Fig. 1). In contrast, only 50% of the children had an episode of LT-ETEC diarrhea by 3 years of age (Fig. 1). The incidence of ETEC diarrhea was consistently higher during the warmer season between April and September. While ST-ETEC diarrhea was more frequent in the warmer months (relative rate [RR] = 2.67 [95% CI, 2.22 to 3.21; P < 0.001]), LT-ETEC was present at similar levels throughout the year (Fig. 2). The incidence for males was higher than that for females (RR = 1.33 [95% CI, 1.12 to 1.58; P < 0.01]), and breastfeeding was not associated with the risk of ETEC diarrhea (RR = 0.93 [95% CI, 0.75 to 1.14]).
FIG. 1.
Time to first infection resulting in diarrhea due to any pathogen isolated (ALL CAUSE), any ETEC, ST-ETEC, or LT-ETEC in a cohort of 211 new-birth infants in Abu Homos, Egypt, from 1995 to 1998.
FIG. 2.
Monthly incidence (episodes per child per year) of ETEC diarrhea in Abu Homos, Egypt, from 1995 to 1998 (with a smoothing fit to illustrate monthly variability).
An incidence of 1.5 episodes per child per year of ETEC diarrhea in the first 3 years of life ranks among the highest rates reported (1, 15). ETEC was the principal identified cause of diarrhea among the study children, with an incidence eight times that of rotavirus, astrovirus, or Shigella. Additionally, among the pathogens evaluated, ETEC was also the most common organism isolated from the first episode of diarrhea experienced by a study child. Therefore, preventing ETEC diarrhea at an early age can have a significant impact on the burden of illness due to diarrhea during infancy.
Breastfeeding has been associated with a reduced risk of diarrhea, with evidence that secretory immunoglobulin A antibodies in breast milk could protect against ETEC diarrhea in children (3, 4). Unlike the findings from a study in Mexico (7), this study did not find any evidence that breastfeeding protects against LT-ETEC diarrhea. Findings of no protection against ETEC diarrhea were also reported in Bangladesh and at a different field site in the Nile Delta (1, 2). This lack of protection may be a possible explanation for the majority of first episodes of diarrhea associated with ETEC in this population.
ETEC continues to be a significant cause of diarrhea in the Nile Delta, and better interventions are clearly needed to reduce the high level of disease burden in early life. Although major improvements in hygiene and sanitation have been successful elsewhere, advances in this realm do not appear to be imminently attainable within this setting. Therefore, as endorsed by the World Health Organization, immunization against ETEC should be considered a public health priority in such settings (16). Since the risk of ETEC is greatest during infancy, any candidate vaccine must confer immunity at a very young age and should ideally be integrated into the routine immunization schedule.
Acknowledgments
Financial support for the study was provided by the Naval Medical Research and Development Command (Work Unit no. M00101.HIX.3421 and M00101.PIX.3270), the National Institute of Child Health and Human Development (Interagency Agreement Y1-HD-0026-01), the World Health Organization Global Program for Vaccines and Immunization, and the World Health Organization Control of Diarrheal Diseases Programme.
We thank Sahar Abd El Samad, Manal El Sayed, and the staff of the Abu Homos Field Research Center for their contributions to field and laboratory work and Mahmoud Abu El Nasr of the Egyptian Ministry of Health and Population for his advice and support. In addition, we thank Robert Bradley Sack, Louis Bourgeois, Stephen Gange, and Nathaniel Pierce for their valuable comments and suggestions.
The opinions and assertions contained herein are the private ones of the authors and are not to be construed as official or as reflecting the views of the U.S. Department of Health and Human Services, the U.S. Department of Defense, the World Health Organization, or the Egyptian Ministry of Health.
REFERENCES
- 1.Abu-Elyazeed, R., T. F. Wierzba, A. S. Mourad, L. F. Peruski, B. A. Kay, M. Rao, A. M. Churilla, A. L. Bourgeois, A. K. Mortagy, S. M. Kamal, S. J. Savarino, J. R. Campbell, J. R. Murphy, A. Naficy, and J. D. Clemens. 1999. Epidemiology of enterotoxigenic Escherichia coli diarrhea in a pediatric cohort in a periurban area of lower Egypt. J. Infect. Dis. 179:382-389. [DOI] [PubMed] [Google Scholar]
- 2.Clemens, J. D., M. R. Rao, J. Chakraborty, M. Yunus, M. Ali, B. Kay, F. P. L. van Loon, A. Naficy, and D. A. Sack. 1997. Breastfeeding and the risk of life-threatening enterotoxigenic Escherichia coli diarrhea in Bangladeshi infants and children. Pediatrics 100:E2. [DOI] [PubMed] [Google Scholar]
- 3.Cruz, J. R., L. Gil, F. Cano, P. Caceres, and G. Pareja. 1988. Breast milk anti-Escherichia coli heat-labile toxin IgA antibodies protect against toxin-induced infantile diarrhea. Acta Paediatr. Scand. 77:658-662. [DOI] [PubMed] [Google Scholar]
- 4.Holmgren, J., L. A. Hanson, B. Carlson, B. S. Lindblad, and J. Rahimtoola. 1976. Neutralizing antibodies against Escherichia coli and Vibrio cholerae enterotoxins in human milk from a developing country. Scand. J. Immunol. 5:867-871. [DOI] [PubMed] [Google Scholar]
- 5.Kalbfleisch, J. D., and R. L. Prentice. 1980. The statistical analysis of failure time data. John Wiley & Sons, Inc., New York, N.Y.
- 6.Liang, K. Y., and S. L. Zeger. 1986. Longitudinal data analysis using generalized linear models. Biometrika 73:13-22. [Google Scholar]
- 7.Long, K. Z., J. W. Wood, E. Vasquez Gariby, K. M. Weiss, J. J. Mathewson, F. J. de la Cabada, H. L. DuPont, and R. A. Wilson. 1994. Proportional hazards analysis of diarrhea due to enterotoxigenic Escherichia coli and breast feeding in a cohort of urban Mexican children. Am. J. Epidemiol. 139:193-205. [DOI] [PubMed] [Google Scholar]
- 8.Murray, P. R., E. J. Baron, M. A. Pfaller, F. C. Tenover, and R. H. Yolken. 1995. Manual of clinical microbiology, 6th ed. American Society for Microbiology, Washington, D.C.
- 9.Naficy, A. B., M. R. Rao, J. L. Holmes, R. Abu-Elyazeed, S. J. Savarino, T. F. Wierzba, R. W. Frenck, S. S. Monroe, R. I. Glass, and J. D. Clemens. 2000. Astrovirus diarrhea in Egyptian children. J. Infect. Dis. 182:685-690. [DOI] [PubMed] [Google Scholar]
- 10.Nataro, J. P., and J. B. Kaper. 1998. Diarrheagenic Escherichia coli. Clin. Microbiol. Rev. 11:142-201. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Sanchez, J., J. Holmgren, and A. M. Svennerholm. 1990. Recombinant fusion protein for simple detection of Escherichia coli heat-stable enterotoxin by GM1 enzyme-linked immunosorbent assay. J. Clin. Microbiol. 28:2175-2177. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Skirrow, M. B. 1977. Campylobacter enteritis: a new disease. Br. Med. J. 2:9-11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Svennerholm, A. M., and G. Wiklund. 1983. Rapid GM1-enzyme-linked immunosorbent assay with visual reading for identification of Escherichia coli heat-labile enterotoxin. J. Clin. Microbiol. 17:596-600. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Svennerholm, A. M., and J. Holmgren. 1995. Oral vaccines against cholera and enterotoxigenic Escherichia coli diarrhea. Adv. Exp. Med. Biol. 8:1623-1628. [PubMed] [Google Scholar]
- 15.Viboud, G. I., M. J. Jouve, N. Binsztein, M. Vergara, M. Rivas, M. Quiroga, and A. M. Svennerholm. 1999. Prospective cohort study of enterotoxigenic Escherichia coli infections in Argentinean children. J. Clin. Microbiol. 37:2829-2833. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.World Health Organization. 1999. New frontiers in the development of vaccines against enterotoxinogenic (ETEC) and enterohaemorrhagic (EHEC) E. coli infections. Part I. Wkly. Epidemiol. Rec. 74:98-101. [PubMed] [Google Scholar]