Abstract
Background
The location of osteosarcoma in the metaphysis as well as the age of the patients during the most rapid tumour growth suggest that factors related to skeletal growth are involved in the pathogenesis of this tumour. In this aspect this study aims to detect somatostatin receptors in human osteosarcomas and correlate this finding with the clinical outcome of the tumour.
Patients and methods
Immunohistochemical staining for the presence of somatostatin receptors as well as overall survival and disease free survival rates were retrospectively studied in twenty-nine osteosarcoma patients.
Results
Four osteosarcomas with several aggressive biologic behaviour expressed somatostatin receptors. In these four young patients the event free rate was 0% and the overall survival rate was 50% at 4, 3 years. In contrast the event free survival rate of the twenty-five patients with negative somatostatin receptor status was 72% with an overall survival rate of 76% at 4,3 years.
Conclusion
The present study demonstrates the existence of somatostatin receptors in human osteosarcoma. Tumours expressing somatostatin receptors seemed to be aggressive with a very low disease free and overall survival rate compared to osteosarcoma with negative receptor status.
Background
Osteosarcoma is the most common primary malignant tumour of bone, with the exception of multiple myeloma. It represents approximately 15% of all biopsy-analyzed primary bone tumours [1,2]. It is most common in males and occurs primarily in the second decade of life. The most common location sites are the metaphysis of bone [3,4]. The age of the patients, coinciding with the adolescent growth spurt as well as the location of tumour sites has led to the syllogism that factors related to skeletal growth are involved in the pathogenesis of this tumour [5-7]. Previous studies maintain that treatment with growth hormone and somatostatin affects the growth of osteosarcoma in animal models [8-10]. Somatostatin is believed to exert antiproliferative effects on tumour cells through receptor-mediated stimulation of tyrosine phosphatase and inhibition of other endogenous growth factors, like growth hormone and insuline-like growth factor 1 [11,12]. In this respect, the presence of somatostatin receptors in human osteosarcoma may have a diagnostic, prognostic and therapeutic value [13].
In this study we aim to detect somatostatin receptors in human osteosarcomas and correlate this finding with the clinical outcome of the tumour.
Patients and methods
Twenty-nine patients with primary osteosarcoma who were treated at the authors' institution between 1997 and 2006 were included in this study. Fourteen patients were female and fifteen were male. The average age at the time of diagnosis was 27.03 years (range 16–49 years) (Table 1). Preoperative evaluation included precision imaging techniques (plain radiographs, computed tomography and MRI of the lesion, computed tomography of the chest and full body scan with Tc99m). Distribution of anatomic tumour sites was as described in Table 1. The therapeutic protocol included neoadjuvant chemotherapy in all patients with high-dose methotrexate [14-16]. During preoperative chemotherapy one patient died, while we operated on twenty-eight patients aiming at wide resection margins.
Table 1.
Sex, Age, Location, Surgical Treatment, Outcome and GH receptor status of 29 patients with osteosarcoma.
| Sex | Age | Location (Site) | Surgical Treatment | Oncologic outcome | GH receptor status | |
| 1 | M | 28 | Thoracic Spine | LSS | DOD (Died On Disease) |
|
| 2 | F | 16 | Distal femur | LSS | NED (No Evident Disease) |
|
| 3 | F | 19 | Proximal Tibia | Amputation | DOD | + |
| 4 | F | 17 | Proximal Tibia | Amputation | DOD | + |
| 5 | F | 39 | Distal Femur | LSS | DOD | |
| 6 | M | 28 | Distal Femur | Amputation | NED | |
| 7 | M | 16 | Distal Fibula | LSS | NED | |
| 8 | M | 22 | Distal Femur | LSS | DOD | |
| 9 | F | 18 | Distal Femur | LSS | DOD | |
| 10 | F | 27 | Distal Femur | LSS | NED | |
| 11 | F | 18 | Distal Femur | Amputation | NED | |
| 12 | F | 35 | Proximal Tibia | LSS | NED | |
| 13 | M | 16 | Distal Femur | LSS | NED | |
| 14 | M | 24 | Proximal Humerus | LSS | Disease Progression (Pulmonary metastases) |
+ |
| 15 | M | 18 | Proximal Tibia | LSS | NED | |
| 16 | M | 32 | Distal Tibia | LSS | NED | |
| 17 | F | 34 | Hip | Died during chemotherapy | DOD | |
| 18 | F | 49 | Proximal Tibia | LSS | NED | |
| 19 | F | 24 | Distal Femur | LSS | NED | |
| 20 | M | 44 | Proximal Humerus | LSS | DOD | |
| 21 | F | 39 | Distal Femur | LSS | Disease Progression (Local recurrence) |
|
| 22 | F | 25 | Distal Femur | LSS | NED | |
| 23 | M | 18 | Distal Femur | LSS | NED | |
| 24 | M | 44 | Distal Femur | LSS | NED | |
| 25 | M | 40 | Distal Femur | LSS | NED | |
| 26 | M | 20 | Proximal Humerus | LSS | NED | |
| 27 | M | 30 | Proximal Humerus | LSS | NED | |
| 28 | M | 16 | Proximal Tibia | LSS | Disease Progression (Pulmonary metastases) |
+ |
| 29 | F | 28 | Proximal Tibia | LSS | NED |
Twenty-four patients underwent a limb salvage procedure, while in four patients amputation was the only surgical option in order to achieve adequate local control.
Disease-free and overall survival was recorded in all patients (table 2).
Table 2.
Disease free and overall survival rate at 4, 48 years, in 29 patients with osteosarcoma.
| Frequency | Percent | |
| NED (No Evident Disease) |
18 | 62,0 |
| Disease progression | 3 | 10,4 |
| DOD (Died On Disease) |
8 | 27,6 |
| Total | 29 | 100,0 |
Histological specimens were available for all patients and were reviewed by one experienced pathologist (I.I.). The resected specimens were sliced coronally or axially or both to represent the largest portion of the tumour. The slices were fixed in 10% neutral buffered formaldehyde solution and embedded separately in paraffin. The sections were stained with haematoxylin and eosin and were used for immunohistochemistry. Polyclonal Rabbit Anti-Human somatostatin was used (Dako, Denmark) [17-19] in order to detect the presence of somatostatin receptors [20,21]. The study was approved by the Metaxa Anticancer Hospital Ethical & Scientific Committee.
Results
Somatostatin receptors were expressed in four osteosarcoma's that exhibited aggressive features (figure 1 and 2). These four tumours appeared in young patients (table 3) with an aggressive biologic behaviour having an event-free rate of 0% and an overall survival rate of 50% at 4.3 years (table 4). In contrast, the event-free survival rate of the twenty-five patients with negative growth hormone receptor status was 72% with an overall survival rate of 76% at 4.3 years.
Figure 1.
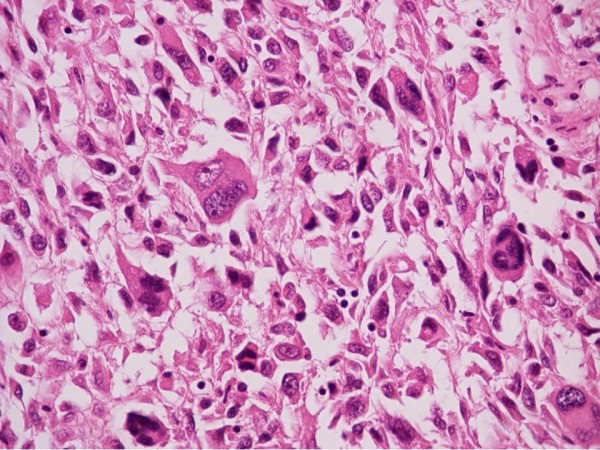
Osteosarcoma somatostatin negative. Magnification ×400. This case of an osteosarcoma had no somatostatin receptors. Immunohistochemistry staining with somatostatin did not produce any reaction.
Figure 2.
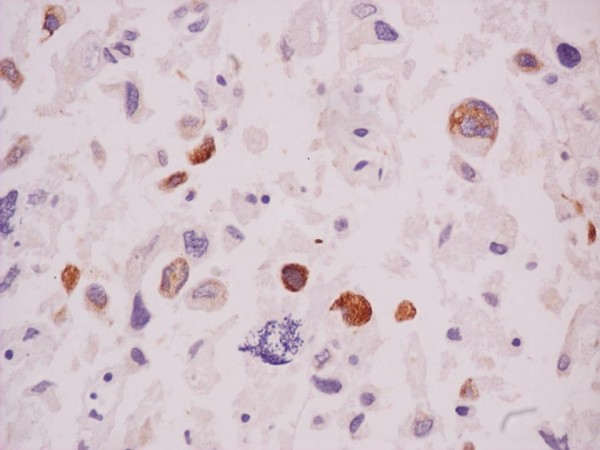
Osteosarcoma somatostatin positive. Magnification ×630. In this case staining with somatostatin produced a reaction appearing with an orange zone around the nuclei. This case of osteosarcoma is expressing somatostatin receptors.
Table 3.
Mean Age of patients with positive staining vs. patients with negative staining for receptors of Growth Hormone.
| Patients with Positive staining for receptors of Growth Hormone | Patients with Negative staining for receptors of Growth Hormone | |
| AGE (MEAN/RANGE) | 19/16–24 years | 28,32/16–49 years |
Table 4.
Disease free and overall survival rate at 4, 48 years in patients with positive staining vs. patients with negative staining for receptors of Growth Hormone.
| Patients with Positive staining for receptors of Growth Hormone (Frequency/Percent) |
Patients with Negative staining for receptors of Growth Hormone (Frequency/Percent) |
|
| NED (No Evident Disease) |
0/0,0 | 18/72,0 |
| Disease progression | 2/50,0 | 1/4,0 |
| DOD (Died On Disease) |
2/50,0 | 6/24,0 |
| Total | 4/100,0 | 25/100,0 |
Case one represents a woman, 19-years-old, with a right proximal tibia tumour, stage II B+ on Enneking's staging system [22]. She underwent neoadjuvant chemotherapy followed by femoral amputation. Histological examination revealed grade II osteosarcoma with osteoblastic, as well as chondroblastic areas and 80% tumour necrosis. Two years later there was a local recurrence in the stump of the sciatic nerve, which was treated with hip disarticulation and chemotherapy. Four years post-operative, this patient presented lung metastases, was treated with chemotherapy and eventually died after 1 year. In our retrospective histological study somatostatin receptors were detected.
Discussion
The use of neoadjuvant chemotherapy in the treatment protocol of osteosarcoma in the late 70's improved disease-free survival, giving a cure rate of 60%–70% for patients with nonmetastatic osteosarcoma of the extremities at presentation [23-25].
Little is known about the aetiology and pathogenesis of this tumour. Genetic predisposition, viral aetiology, irradiation and alkylating agents have been suggested in the pathogenesis of osteosarcoma [3,26,27]. Nowadays, molecular biology seems to be the next step in understanding pathogenesis and improving survival of osteosarcoma. Tumour location in the metaphysis as well as the age of the patients coinciding with the period of rapid body growth suggest that factors related to skeletal growth are involved in the pathogenesis of this tumour.
Somatostatin is characterized as a hormone which inhibits the release of growth hormone from the anterior pituitary gland [28]. The present study demonstrates the existence of somatostatin receptors in human osteosarcoma. Further research is necessary to demonstrate the importance of this finding and its clinical relevance, since there is also evidence from animal studies that treatment with growth hormone and somatostatin affects the growth of osteosarcoma in animal models [8-10]. There is also one study in pediatric patients having metastatic osteosarcoma treated with somatostatin analogue (OncoLar) which shows that the levels of Insulin-like growth factor-1 were reduced. However, this study did not yield significant clinical results [29].
To our knowledge, there is only one study on humans in the literature with 18 osteosarcoma patients where the authors investigated somatostatin receptors by virtue of scintigraphy. In this study a very high incidence of patients with somatostatin receptors was found (up to 75%). The authors found higher incidence in non-metastatic patients and concluded that there is a possible relation between the somatostatin receptors presence and the biological behaviour of the tumour. [30]
A limitation to our study is the small number of specimens that were analyzed, which makes statistical analysis unfeasible; however, because of the novelty of our study and since the tumours expressing somatostatin receptors had a more deleterious course with a very low disease-free and overall survival rate compared to osteosarcoma with negative receptor status, even though the percentage (14%) was much lower than that in the Rizzoli study [30], we believe that this finding should be thoroughly evaluated and investigated with further studies.
Conclusion
In this study we detected somatostatin receptors in human osteosarcomas. This finding seems to have a prognostic value, predicating a severe aggressive biologic behaviour of the tumour as well as possible therapeutic implications.
Competing interests
The authors declare that they have no competing interests.
Authors' contributions
MI drafted the manuscript and carried out the design of the study and performed. II carried out the immunohistochemical studies. PJP, IP and SK participated in the design and coordination of the study and helped to draft the manuscript. All authors read and approved the final manuscript.
Acknowledgments
Acknowledgements
The authors would like to thank Panou Christina for text editing (email:christinepanou@yahoo.com)
Contributor Information
Markos Ioannou, Email: markosioannou@yahoo.gr.
Panayiotis J Papagelopoulos, Email: pjp@hol.gr.
Ioannis Papanastassiou, Email: jpapa73@yahoo.gr.
Ioanna Iakovidou, Email: yian_kyr@vivodinet.gr.
Stamatios Kottakis, Email: dmytas@gmail.com.
Nikolaos Demertzis, Email: stavrosmitas@gmail.com.
References
- Campanacci M. Bone and soft tissue tumors: clinical features, imaging, pathology and treatment. 2. Padova: Wein: Springer-Verlag; 1999. [Google Scholar]
- Whelan JS. Osteosarcoma. Eur J Cancer. 1997;33:1611–1618. doi: 10.1016/S0959-8049(97)00251-7. discussion 1618–1619. [DOI] [PubMed] [Google Scholar]
- Huvos AG. Bone tumors: diagnosis, treatment and prognosis. 2. Philadelphia; London: W.B. Saunders; 1991. [Google Scholar]
- Bacci G, Longhi A, Versari M, Mercuri M, Briccoli A, Picci P. Prognostic factors for osteosarcoma of the extremity treated with neoadjuvant chemotherapy: 15-year experience in 789 patients treated at a single institution. Cancer. 2006;106:1154–1161. doi: 10.1002/cncr.21724. [DOI] [PubMed] [Google Scholar]
- Cotterill SJ, Wright CM, Pearce MS, Craft AW. Stature of young people with malignant bone tumors. Pediatr Blood Cancer. 2004;42:59–63. doi: 10.1002/pbc.10437. [DOI] [PubMed] [Google Scholar]
- James RA, Dymock RB. Osteosarcoma associated with acromegaly: a case report. Pathology. 1976;8:157–159. doi: 10.3109/00313027609094442. [DOI] [PubMed] [Google Scholar]
- Pizzo PA, Poplack DG. Principles and practice of pediatric oncology. 4. Philadelphia; London: Lippincott Williams & Wilkins; 2001. [Google Scholar]
- Pinski J, Schally AV, Halmos G, Szepeshazi K, Groot K. Somatostatin analog RC-160 inhibits the growth of human osteosarcomas in nude mice. Int J Cancer. 1996;65:870–874. doi: 10.1002/(SICI)1097-0215(19960315)65:6<870::AID-IJC27>3.0.CO;2-6. [DOI] [PubMed] [Google Scholar]
- Conzemius MG, Graham JC, Haynes JS, Graham CA. Effects of treatment with growth hormone and somatostatin on efficacy of diammine [1,1-cyclobutane dicarboxylato (2-)-0,0']-(SP-4-2) in athymic rats with osteosarcoma. Am J Vet Res. 2000;61:646–650. doi: 10.2460/ajvr.2000.61.646. [DOI] [PubMed] [Google Scholar]
- Khanna C, Prehn J, Hayden D, Cassaday RD, Caylor J, Jacob S, Bose SM, Hong SH, Hewitt SM, Helman LJ. A randomized controlled trial of octreotide pamoate long-acting release and carboplatin versus carboplatin alone in dogs with naturally occurring osteosarcoma: evaluation of insulin-like growth factor suppression and chemotherapy. Clin Cancer Res. 2002;8:2406–2412. [PubMed] [Google Scholar]
- Koper JW, Markstein R, Kohler C, Kwekkeboom DJ, Avezaat CJ, Lamberts SW, Reubi JC. Somatostatin inhibits the activity of adenylate cyclase in cultured human meningioma cells and stimulates their growth. J Clin Endocrinol Metab. 1992;74:543–547. doi: 10.1210/jc.74.3.543. [DOI] [PubMed] [Google Scholar]
- Ganz MB, Pachter JA, Barber DL. Multiple receptors coupled to adenylate cyclase regulate Na-H exchange independent of cAMP. J Biol Chem. 1990;265:8989–8992. [PubMed] [Google Scholar]
- Reubi JC, Laissue J, Krenning E, Lamberts SW. Somatostatin receptors in human cancer: incidence, characteristics, functional correlates and clinical implications. J Steroid Biochem Mol Biol. 1992;43:27–35. doi: 10.1016/0960-0760(92)90184-K. [DOI] [PubMed] [Google Scholar]
- Daw NC, Billups CA, Rodriguez-Galindo C, McCarville MB, Rao BN, Cain AM, Jenkins JJ, Neel MD, Meyer WH. Metastatic osteosarcoma. Cancer. 2006;106:403–412. doi: 10.1002/cncr.21626. [DOI] [PubMed] [Google Scholar]
- Meyers PA, Heller G, Healey J, Huvos A, Lane J, Marcove R, Applewhite A, Vlamis V, Rosen G. Chemotherapy for nonmetastatic osteogenic sarcoma: the Memorial Sloan-Kettering experience. J Clin Oncol. 1992;10:5–15. doi: 10.1200/JCO.1992.10.1.5. [DOI] [PubMed] [Google Scholar]
- Bacci G, Ferrari S, Mercuri M, Longhi A, Fabbri N, Galletti S, Forni C, Balladelli A, Serra M, Picci P. Neoadjuvant chemotherapy for osteosarcoma of the extremities in patients aged 41–60 years: outcome in 34 cases treated with adriamycin, cisplatinum and ifosfamide between 1984 and 1999. Acta Orthop. 2007;78:377–384. doi: 10.1080/17453670710013960. [DOI] [PubMed] [Google Scholar]
- Alumets J, Sundler F, Hakanson R. Distribution, ontogeny and ultrastructure of somatostatin immunoreactive cells in the pancreas and gut. Cell Tissue Res. 1977;185:465–479. doi: 10.1007/BF00220651. [DOI] [PubMed] [Google Scholar]
- Erlandsen SL, Hegre OD, Parsons JA, McEvoy RC, Elde RP. Pancreatic islet cell hormones distribution of cell types in the islet and evidence for the presence of somatostatin and gastrin within the D cell. J Histochem Cytochem. 1976;24:883–897. doi: 10.1177/24.7.60437. [DOI] [PubMed] [Google Scholar]
- Parsons JA, Erlandsen SL, Hegre OD, McEvoy RC, Elde RP. Central and peripheral localization of somatostatin. Immunoenzyme immunocytochemical studies. J Histochem Cytochem. 1976;24:872–882. doi: 10.1177/24.7.60436. [DOI] [PubMed] [Google Scholar]
- Srkalovic G, Cai RZ, Schally AV. Evaluation of receptors for somatostatin in various tumors using different analogs. J Clin Endocrinol Metab. 1990;70:661–669. doi: 10.1210/jcem-70-3-661. [DOI] [PubMed] [Google Scholar]
- Pinski J, Schally AV, Halmos G, Szepeshazi K, Groot K, O'Byrne K, Cai RZ. Effects of somatostatin analogue RC-160 and bombesin/gastrin-releasing peptide antagonists on the growth of human small-cell and non-small-cell lung carcinomas in nude mice. Br J Cancer. 1994;70:886–892. doi: 10.1038/bjc.1994.415. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Enneking WF. A system of staging musculoskeletal neoplasms. Clin Orthop Relat Res. 1986:9–24. [PubMed] [Google Scholar]
- Bacci G, Ferrari S, Bertoni F, Ruggieri P, Picci P, Longhi A, Casadei R, Fabbri N, Forni C, Versari M, Campanacci M. Long-term outcome for patients with nonmetastatic osteosarcoma of the extremity treated at the istituto ortopedico rizzoli according to the istituto ortopedico rizzoli/osteosarcoma-2 protocol: an updated report. J Clin Oncol. 2000;18:4016–4027. doi: 10.1200/JCO.2000.18.24.4016. [DOI] [PubMed] [Google Scholar]
- Fuchs N, Bielack SS, Epler D, Bieling P, Delling G, Korholz D, Graf N, Heise U, Jurgens H, Kotz R, et al. Long-term results of the co-operative German-Austrian-Swiss osteosarcoma study group's protocol COSS-86 of intensive multidrug chemotherapy and surgery for osteosarcoma of the limbs. Ann Oncol. 1998;9:893–899. doi: 10.1023/A:1008391103132. [DOI] [PubMed] [Google Scholar]
- Provisor AJ, Ettinger LJ, Nachman JB, Krailo MD, Makley JT, Yunis EJ, Huvos AG, Betcher DL, Baum ES, Kisker CT, Miser JS. Treatment of nonmetastatic osteosarcoma of the extremity with preoperative and postoperative chemotherapy: a report from the Children's Cancer Group. J Clin Oncol. 1997;15:76–84. doi: 10.1200/JCO.1997.15.1.76. [DOI] [PubMed] [Google Scholar]
- Finkel MP, Reilly CA, Jr, Biskis BO. Pathogenesis of radiation and virus-induced bone tumors. Recent Results Cancer Res. 1976:92–103. doi: 10.1007/978-3-642-80997-2_7. [DOI] [PubMed] [Google Scholar]
- Swaney JJ. Familial osteogenic sarcoma. Clin Orthop Relat Res. 1973:64–68. doi: 10.1097/00003086-197311000-00010. [DOI] [PubMed] [Google Scholar]
- Guyton AC, Hall JE. Textbook of medical physiology. 11. Edinburgh: Elsevier Saunders; Oxford: Elsevier Science [distributor]; 2006. [Google Scholar]
- Mansky PJ, Liewehr DJ, Steinberg SM, Chrousos GP, Avila NA, Long L, Bernstein D, Mackall CL, Hawkins DS, Helman LJ. Treatment of metastatic osteosarcoma with the somatostatin analog OncoLar: significant reduction of insulin-like growth factor-1 serum levels. J Pediatr Hematol Oncol. 2002;24:440–446. doi: 10.1097/00043426-200208000-00007. [DOI] [PubMed] [Google Scholar]
- Ferrari S, Dondi M, Fanti S, Zoboli S, Giacomini S, Mercuri M, Bacci G. Somatostatin receptor (SSTR) scintigraphy in patients with osteosarcoma. Cancer Biother Radiopharm. 2003;18:847–851. doi: 10.1089/108497803770418391. [DOI] [PubMed] [Google Scholar]


