Abstract
The ability of tuberculosis patients to recognize Mycobacterium vaccae-specific antigens before starting chemotherapy and according to disease severity was analyzed. We report that the M. vaccae cell wall skeleton fraction triggers more enhanced cytokine production than the whole bacterium. Moreover, a tendency was observed for a lower gamma interferon/interleukin-10 ratio in patients with cavitary disease induced by M. vaccae antigens.
The saprophytic Mycobacterium vaccae is being tested as an immunotherapeutic agent in tuberculosis (TB). It is supposed to boost a cross-protective immune response to epitopes shared with Mycobacterium tuberculosis. However, the efficiency of a single injected dose of M. vaccae varied between different clinical trials (14). The causes of this variability are not clear, but the immune status background of the patients (11, 14) or differences in the disease severity (6) could have been influential. Evidence from some of these trials suggests that M. vaccae immunotherapy induces an enhancement of protective gamma interferon (IFN-γ) levels, together with a reduction in circulating interleukin-10 (IL-10) levels (3). In fact, a reverse relationship between the M. tuberculosis antigen-induced IFN-γ/IL-10 ratio and disease severity in TB (5) and also a direct relationship between the IFN-γ/IL-10 ratio with TB cure (1, 12) have been demonstrated in a large-scale analysis. However, no data are available on the ability of the immune systems of TB patients to recognize M. vaccae-specific antigens before starting anti-TB chemotherapy and according to TB severity.
Herein, we analyze the IFN-γ- and IL-10-induced responses in peripheral blood mononuclear cells (PBMC) of TB patients, using both M. tuberculosis and, for the first time, M. vaccae and its cell wall skeleton (CWS) fraction. We have recently demonstrated that the CWS fraction elicits a prominent recall IFN-γ response in splenocyte cultures from mice with TB in comparison with the whole bacterium (10).
Blood samples were collected from adult pulmonary TB patients admitted to the Tuberculosis Unit-Infectious Diseases Service of the Bellvitge Hospital (Spain), before starting chemotherapy. In all cases the disease was confirmed by isolation of M. tuberculosis in cultures. All patients were tuberculin skin test positive and had not been previously Mycobacterium bovis BCG vaccinated. None of the patients had human immunodeficiency virus coinfection, M. tuberculosis reinfection, or other diseases that could affect the immune response. According to the presence of lung cavitation at the time of chest X-ray examination, the patients were classified as affected by noncavitary (n = 4 patients) or cavitary (n = 9 patients) pulmonary TB. Ten blood samples were also taken from healthy tuberculin skin test-negative employees at the hospital. The study was approved by the Ethics Committee of Clinical Investigation of Bellvitge Hospital, and written informed consent was obtained from all participants.
Cells of a stable rough variant of M. vaccae strain ATCC 15483T and M. tuberculosis H37Rv (ATCC 27294T) were heat killed and diluted as described previously (9). The CWS fraction was obtained as described previously (10). All antigens were frozen at −40°C until use. Isolated PBMC were cultured at 2 × 106 cells/ml in Iscove's modified Dulbecco's medium containing 10% fetal bovine serum, 100 U/ml penicillin, and 100 μg/ml streptomycin and were stimulated using phytohemagglutinin (Sigma, St. Louis, MO) at 1:300 as a positive control, M. tuberculosis (10 μg/ml), M. vaccae (10 μg/ml), CWS (5 μg/ml), or medium alone as a negative control. Culture supernatants were collected after 48 h of culture (for IL-10 quantification) or 120 h (for IFN-γ), and cytokine concentrations were measured by enzyme-linked immunosorbent assays using commercially available kits (Mabtech AB, Stockholm, Sweden).
Cytokine levels were compared using a two-tailed Mann-Whitney rank sum or Wilcoxon signed rank test, linear regression analysis, and Spearman's rank correlation, as indicated in the figure legends and table footnotes. A P value of <0.05 was considered significant.
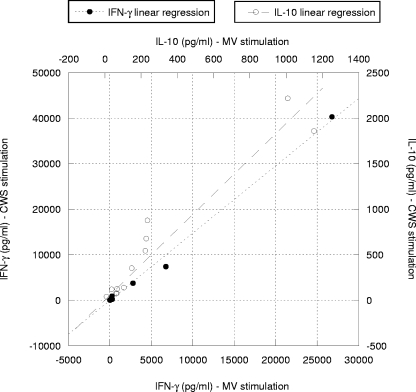
Analyzing the response for each TB patient individually, we observed a strong positive correlation between M. tuberculosis- and M. vaccae-induced IFN-γ production (r = 0.937; P < 0.001) and IL-10 production (r = 0.923; P < 0.001) and also between M. tuberculosis- and CWS-induced IFN-γ production (r = 0.916; P < 0.001) and IL-10 production (r = 0.895; P < 0.001). When comparing M. vaccae- and CWS-induced IFN-γ or IL-10 production, there was again a significant positive correlation in each TB patient (Fig. 1). However, although each patient reacted in the same way to both M. vaccae and CWS, remarkably, the CWS not only retained the immune-stimulatory ability of the whole bacterium but also triggered enhanced cytokine production in TB patients (Table 1). In all cases the cytokine levels in cultures from healthy controls were lower than those obtained from TB patients (Table 1).
FIG. 1.
Correlation of IFN-γ and IL-10 production in TB patients for each stimulus (M. vaccae and CWS antigens). A significant correlation was found between IFN-γ and IL-10 production after in vitro stimulation with the M. vaccae or CWS antigen. This was confirmed by Spearman's rank correlation coefficients (r = 0.93 and P < 0.001 for IFN-γ production; r = 0.972 and P < 0.001 for IL-10 production).
TABLE 1.
IFN-γ and IL-10 production in PBMC cultures from pulmonary TB patients and healthy subjects
| Subject(s) | Cytokine production induced by indicated antigen
|
IFN-γ/IL-10 production ratiob
|
TB form | |||||||
|---|---|---|---|---|---|---|---|---|---|---|
| IFN-γ (pg/ml)a
|
IL-10 (pg/ml)a
|
|||||||||
| M. tuberculosis | M. vaccae | CWS | M. tuberculosis | M. vaccae | CWS | M. tuberculosis | M. vaccae | CWS | ||
| TB patients | ||||||||||
| A | 162,250.4 | 26,786.6 | 40,313.7 | 2,407.0 | 1,008.9 | 2,218.3 | 67.4 | 26.6 | 18.2 | Cavitary |
| B | 32,222.2 | 2,791.5 | 3,768.4 | 424.0 | 148.4 | 352.6 | 76.0 | 18.8 | 10.7 | Noncavitary |
| C | 22,423.7 | 6,742.5 | 7,394.9 | 379.3 | 228.7 | 675.9 | 59.1 | 29.5 | 10.9 | Noncavitary |
| D | 3,143.0 | 258.2 | 922.0 | 210.0 | 68.0 | 125.3 | 15.0 | 3.8 | 7.4 | Noncavitary |
| E | 1,001.0 | 270.9 | 229.9 | 256.4 | 63.6 | 73.4 | 3.9 | 4.3 | 3.1 | Cavitary |
| F | 2,758.8 | 47.8 | 74.7 | 2,272.4 | 1,153.0 | 1,859.9 | 1.2 | 0.0 | 0.0 | Noncavitary |
| G | 1,423.1 | 94.5 | 185.8 | 740.4 | 223.3 | 543.4 | 1.9 | 0.4 | 0.3 | Cavitary |
| H | 526.2 | 2.8 | 26.6 | 187.7 | 105.9 | 138.9 | 2.8 | 0.0 | 0.2 | Cavitary |
| I | 325.9 | 13.8 | 7.9 | 861.4 | 235.2 | 876.1 | 0.4 | 0.1 | 0.0 | Cavitary |
| J | 155.8 | 0.9 | 0.0 | 45.3 | 10.0 | 37.6 | 3.4 | 0.1 | 0.0 | Cavitary |
| K | 27.8 | 0.7 | 1.0 | 131.5 | 66.2 | 75.1 | 0.2 | 0.0 | 0.0 | Cavitary |
| L | 25.9 | 0.0 | 10.6 | 85.8 | 37.5 | 119.4 | 0.3 | 0.0 | 0.1 | Cavitary |
| Medianc | 1,212.0&,¶ | 71.1 | 130.3* | 317.9& | 127.1 | 245.8* | ||||
| Healthy subjects (median) | 61.5 | 20.6 | 10.7 | 101.3 | 57.8 | 79.7 | ||||
Minimum detectable concentrations were 2 pg/ml for IFN-γ and 0.5 pg/ml for IL-10.
Ratios are the average IFN-γ values divided by the IL-10 value for each individual (5).
Median levels of CWS-induced cytokines were significantly increased compared to those produced by M. vaccae (*, P < 0.05). Median levels of M. tuberculosis-induced cytokines were higher than those produced by M. vaccae (&, P < 0.001) or CWS (¶, P < 0.001), measured using the two-tailed Wilcoxon signed rank test.
Our results also showed different patterns of responses to M. vaccae and CWS antigens among TB patients. Seven out of 12 patients showed impaired IFN-γ production compared to IL-10 production, i.e., samples F to L (Table 1), showing an IFN-γ/IL-10 index lower than 1. However, the remainder of the patients (samples A to E) had an index higher than 1. When we correlated these results with the radiographic manifestation of the disease, a tendency towards a lower IFN-γ/IL-10 ratio in patients with cavitary disease was observed, i.e., 85.7% of the patients (six out of seven) with a ratio lower than 1 had cavitation in their lungs (Table 1).
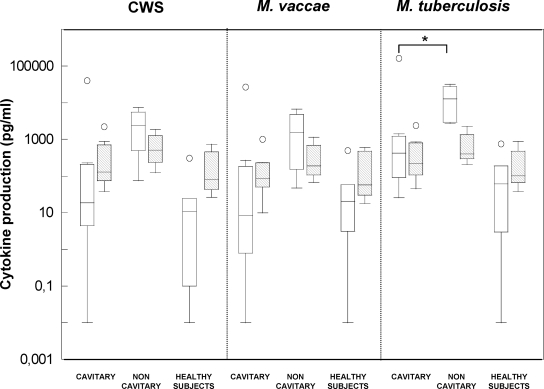
A clear increase in cytokine levels in noncavitary versus cavitary TB patients was observed (Fig. 2). Although the median ratio showed a log difference among groups, the differences were statistically significant only in the case of M. tuberculosis-induced IFN-γ detection (Fig. 2). Although the specific cytokine response against M. vaccae antigens in cavitary and noncavitary TB patients has not been studied before, diminished Th1 responses to M. tuberculosis antigens in PBMC from TB patients have been directly correlated with the severity of the disease (2, 8, 13, 15). Our findings further support these previous observations (Fig. 2). This is an interesting point because one of the initially reported benefits of M. vaccae immunotherapy was the improvement in chest radiographic healing or cavity closure (14). Johnson et al. (7) specifically analyzed the effect of one injected dose of M. vaccae on radiographic response in three large trials and did not observe any benefit. On the contrary, studies in which three or five doses of M. vaccae were administered showed its efficacy for healing and closing cavities in TB patients (4, 14). These previous studies together with our results lead us to believe that patients showing an impaired M. vaccae-induced IFN-γ response in vitro will not respond to one dose of M. vaccae administration in vivo. Therefore, the in vitro analysis of the M. vaccae-specific immune responses in each TB patient could be indicative of the outcome of the in vivo administration, which may be of interest in understanding the unsuccessful results and the requirement of many injected doses among patients whose Th1-type responses are decreased.
FIG. 2.
IFN-γ and IL-10 production in pulmonary TB patients and healthy controls in response to CWS, M. vaccae, or M. tuberculosis antigens. The boxes extend from the 25th to the 75th percentiles, with a line at the median, and the whiskers show the highest and lowest values. Empty boxes represent IFN-γ production and scattered boxes show IL-10 production. A dot represents one out-of-order value. Median levels of M. tuberculosis-induced IFN-γ in noncavitary TB patients were significantly higher than those produced in cavitary TB patients (*, P < 0.05; measured using the two-tailed Mann-Whitney rank sum test).
Although larger studies are needed to confirm these preliminary findings, our results showed that the CWS fraction appeared to be a possible immunotherapeutic agent candidate, confirming the previous results obtained in mouse experiments (10). Furthermore, the results suggest an association between the in vitro reactions to M. vaccae antigens with disease severity, suggesting a different capability to respond to in vivo immunotherapy.
Acknowledgments
We are deeply indebted to Lucia González, Mar Lázaro, Luisa Luquin, Gabriel Rufí, and Miguel Santín of the Hospital Universitari de Bellvitge for their help in collecting the samples. G. Rufí passed away in December 2005.
This work was supported by grants from the Ministerio de Ciencia y Tecnología (SAF2002-00514), the Generalitat de Catalunya (SGR2005-00956), and the Fundació La Marató de TV3 (041230/1).
Footnotes
Published ahead of print on 2 July 2008.
REFERENCES
- 1.Al-Attiyah, R., A. S. Mustafa, A. T. Abal, N. M. Madi, and P. Andersen. 2003. Restoration of mycobacterial antigen-induced proliferation and interferon-gamma responses in peripheral blood mononuclear cells of tuberculosis patients upon effective chemotherapy. FEMS Immunol. Med. Microbiol. 38:249-256. [DOI] [PubMed] [Google Scholar]
- 2.Dlugovitzky, D., M. L. Bay, L. Rateni, G. Fiorenza, L. Vietti, M. A. Farroni, and O. A. Bottasso. 2000. Influence of disease severity on nitrite and cytokine production by peripheral blood mononuclear cells (PBMC) from patients with pulmonary tuberculosis (TB). Clin. Exp. Immunol. 122:343-349. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Dlugovitzky, D., O. Bottasso, J. C. Dominino, E. Valentini, R. Hartopp, M. Singh, C. Stanford, and J. Stanford. 1999. Clinical and serological studies of tuberculosis patients in Argentina receiving immunotherapy with Mycobacterium vaccae (SRL 172). Respir. Med. 93:557-562. [DOI] [PubMed] [Google Scholar]
- 4.Dlugovitzky, D., G. Fiorenza, M. Farroni, C. Bogue, C. Stanford, and J. Stanford. 2006. Immunological consequences of three doses of heat-killed Mycobacterium vaccae in the immunotherapy of tuberculosis. Respir. Med. 100:1079-1087. [DOI] [PubMed] [Google Scholar]
- 5.Jamil, B., F. Shahid, Z. Hasan, N. Nasir, T. Razzaki, G. Dawood, and R. Hussain. 2007. Interferon gamma/IL-10 ratio defines the disease severity in pulmonary and extra pulmonary tuberculosis. Tuberculosis 87:279-287. [DOI] [PubMed] [Google Scholar]
- 6.Johnson, J. L., R. M. Kamya, A. Okwera, A. M. Loughlin, S. Nyole, D. L. Hom, R. S. Wallis, C. S. Hirsch, K. Wolski, J. Foulds, R. D. Mugerwa, and J. J. Ellner. 2000. Randomized controlled trial of Mycobacterium vaccae immunotherapy in non-human immunodeficiency virus-infected Ugandan adults with newly diagnosed pulmonary tuberculosis. J. Infect. Dis. 181:1304-1312. [DOI] [PubMed] [Google Scholar]
- 7.Johnson, J. L., A. J. Nunn, P. B. Fourie, L. P. Ormerod, R. D. Mugerwa, A. Mwinga, C. Chintu, B. Ngwira, P. Onyebujoh, and A. Zumla. 2004. Effect of Mycobacterium vaccae (SRL172) immunotherapy on radiographic healing in tuberculosis. Int. J. Tuberc. Lung Dis. 8:1348-1354. [PubMed] [Google Scholar]
- 8.Mazzarella, G., A. Bianco, F. Perna, D. D'Auria, E. Grella, E. Moscariello, and A. Sanduzzi. 2003. T lymphocyte phenotypic profile in lung segments affected by cavitary and non-cavitary tuberculosis. Clin. Exp. Immunol. 132:283-288. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Rodríguez-Güell, E., G. Agustí, M. Corominas, P.-J. Cardona, I. Casals, T. Parella, M. A. Sempere, M. Luquin, and E. Julián. 2006. The production of a new extracellular putative long-chain saturated polyester by smooth variants of Mycobacterium vaccae interferes with Th1-cytokine production. Antonie van Leeuwenhoek 90:93-108. [DOI] [PubMed] [Google Scholar]
- 10.Rodríguez-Güell, E., G. Agustí, M. Corominas, P. J. Cardona, M. Luquin, and E. Julián. 2008. Mice with pulmonary tuberculosis treated with Mycobacterium vaccae develop strikingly enhanced recall gamma interferon responses to M. vaccae cell wall skeleton. Clin. Vaccine Immunol. 15:893-896. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Rook, G. A., K. Dheda, and A. Zumla. 2005. Immune responses to tuberculosis in developing countries: implications for new vaccines. Nat. Rev. Immunol. 5:661-667. [DOI] [PubMed] [Google Scholar]
- 12.Sahiratmadja, E., B. Alisjahbana, T. de Boer, I. Adnan, A. Maya, H. Danusantoso, R. H. Nelwan, S. Marzuki, J. W. van der Meer, R. van Crevel, E. van de Vosse, and T. H. Ottenhoff. 2007. Dynamic changes in pro- and anti-inflammatory cytokine profiles and gamma interferon receptor signaling integrity correlate with tuberculosis disease activity and response to curative treatment. Infect. Immun. 75:820-829. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Sodhi, A., J. Gong, C. Silva, D. Qian, and P. F. Barnes. 1997. Clinical correlates of interferon gamma production in patients with tuberculosis. Clin. Infect. Dis. 25:617-620. [DOI] [PubMed] [Google Scholar]
- 14.Stanford, J., C. Stanford, and J. Grange. 2004. Immunotherapy with Mycobacterium vaccae in the treatment of tuberculosis. Front. Biosci. 9:1701-1719. [DOI] [PubMed] [Google Scholar]
- 15.Wu, H. P., C. C. Hua, and D. Y. Chuang. 2007. Decreased in vitro interferon-gamma production in patients with cavitary tuberculosis on chest radiography. Respir. Med. 101:48-52. [DOI] [PubMed] [Google Scholar]