Abstract
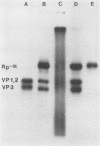
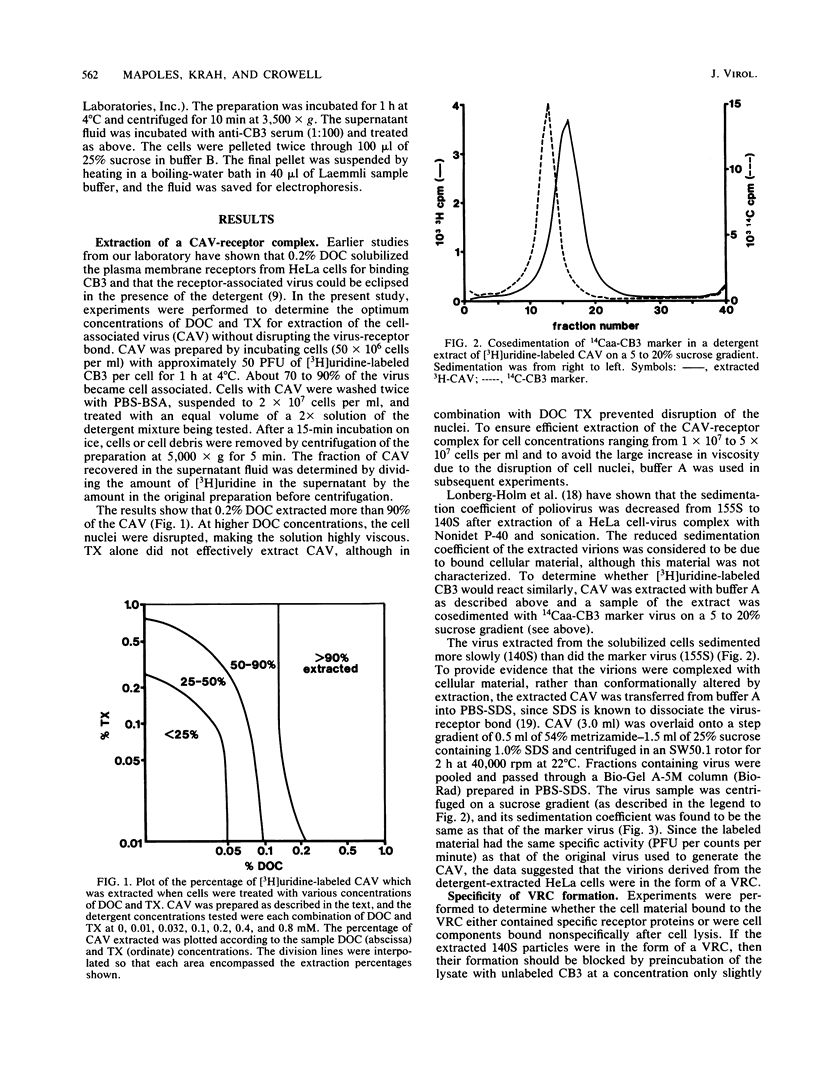
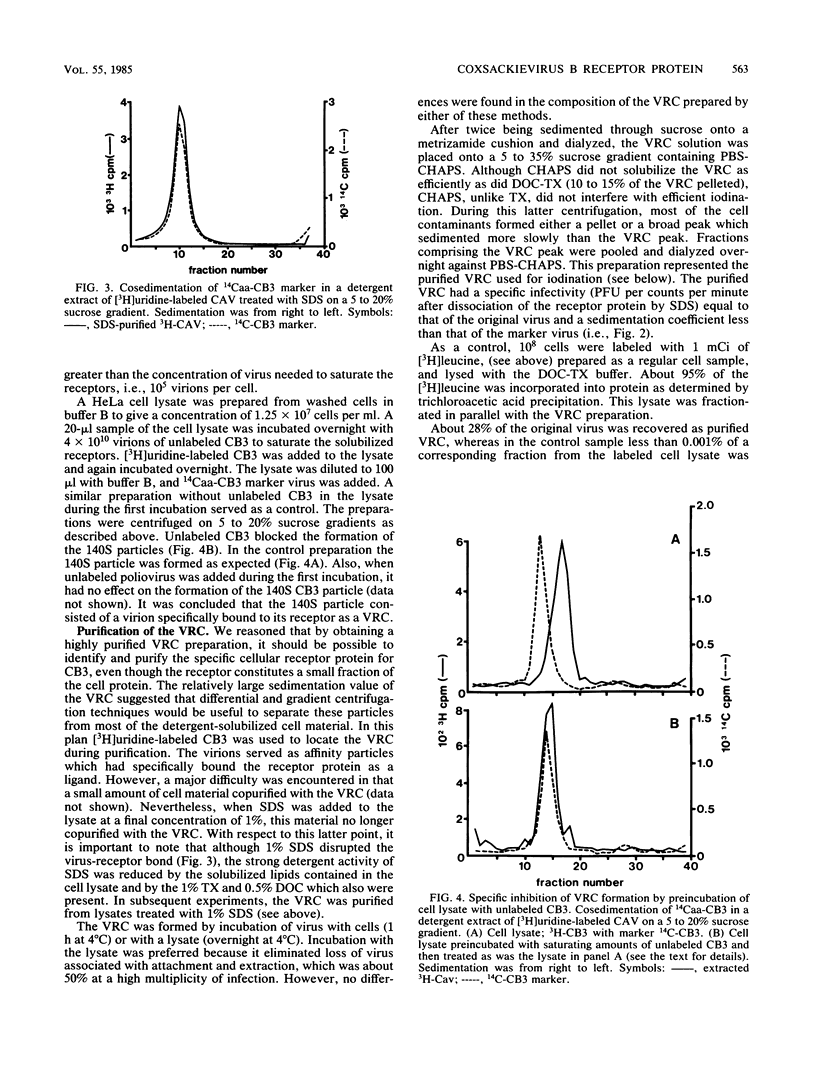
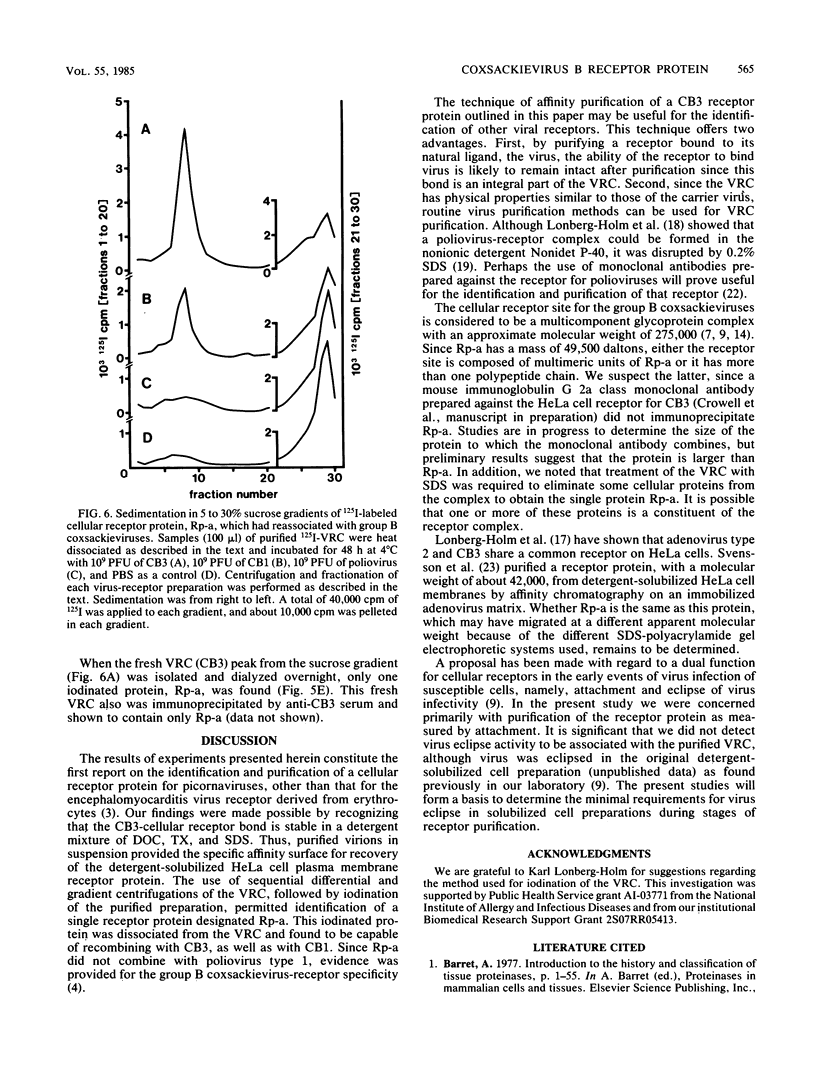
Coxsackievirus B3 (CB3) firmly attaches to HeLa cells, forming a specific complex between the virus and its receptor on the cell surface. We extracted this virus-receptor complex (VRC) with the detergents sodium deoxycholate and Triton X-100. The VRC was identified by its sedimentation coefficient (140S), which was less than that of virions (155S). Formation of the VRC from cell lysates and 3H-CB3 occurred with the same specificity as did attachment of virions to cells, in that its formation was blocked by unlabeled CB3 but not by poliovirus. The VRC was purified 30,000-fold by differential and sucrose gradient centrifugation. Iodination with Na125I revealed that the purified VRC consisted of the normal CB3 proteins and one additional protein (RP-a) with an approximate molecular weight of 49,500. RP-a was eluted from the VRC and was shown to rebind with CB3 and CB1 virions but not with poliovirus type 1. We propose that Rp-a is a protein in the plasma membrane receptor complex which is responsible for the specific recognition and binding of the group B coxsackieviruses.
Full text
PDF



Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Burness A. T., Pardoe I. U. A sialoglycopeptide from human erythrocytes with receptor-like properties for encephalomyocarditis and influenza viruses. J Gen Virol. 1983 May;64(Pt 5):1137–1148. doi: 10.1099/0022-1317-64-5-1137. [DOI] [PubMed] [Google Scholar]
- CROWELL R. L., SYVERTON J. T. The mammalian cell-virus relationship. VI. Sustained infection of HeLa cells by Coxsackie B3 virus and effect on superinfection. J Exp Med. 1961 Feb 1;113:419–435. doi: 10.1084/jem.113.2.419. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Crowell R. L., Philipson L. Specific alterations of coxsackievirus B3 eluted from HeLa cells. J Virol. 1971 Oct;8(4):509–515. doi: 10.1128/jvi.8.4.509-515.1971. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Crowell R. L. Specific cell-surface alteration by enteroviruses as reflected by viral-attachment interference. J Bacteriol. 1966 Jan;91(1):198–204. doi: 10.1128/jb.91.1.198-204.1966. [DOI] [PMC free article] [PubMed] [Google Scholar]
- DULBECCO R., VOGT M. Plaque formation and isolation of pure lines with poliomyelitis viruses. J Exp Med. 1954 Feb;99(2):167–182. doi: 10.1084/jem.99.2.167. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Krah D. L., Crowell R. L. A solid-phase assay of solubilized HeLa cell membrane receptors for binding group B coxsackieviruses and polioviruses. Virology. 1982 Apr 15;118(1):148–156. doi: 10.1016/0042-6822(82)90328-2. [DOI] [PubMed] [Google Scholar]
- Krah D. L., Crowell R. L. Properties of the deoxycholate-solubilized HeLa cell plasma membrane receptor for binding group B coxsackieviruses. J Virol. 1985 Mar;53(3):867–870. doi: 10.1128/jvi.53.3.867-870.1985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Laemmli U. K. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature. 1970 Aug 15;227(5259):680–685. doi: 10.1038/227680a0. [DOI] [PubMed] [Google Scholar]
- Lonberg-Holm K., Crowell R. L., Philipson L. Unrelated animal viruses share receptors. Nature. 1976 Feb 26;259(5545):679–681. doi: 10.1038/259679a0. [DOI] [PubMed] [Google Scholar]
- Lonberg-Holm K., Gosser L. B., Kauer J. C. Early alteration of poliovirus in infected cells and its specific inhibition. J Gen Virol. 1975 Jun;27(3):329–342. doi: 10.1099/0022-1317-27-3-329. [DOI] [PubMed] [Google Scholar]
- MANDEL B. The use of sodium dodecyl sulfate in studies on the interaction of poliovirus and HeLa cells. Virology. 1962 Jun;17:288–294. doi: 10.1016/0042-6822(62)90119-8. [DOI] [PubMed] [Google Scholar]
- Medappa K. C., McLean C., Rueckert R. R. On the structure of rhinovirus 1A. Virology. 1971 May;44(2):259–270. doi: 10.1016/0042-6822(71)90258-3. [DOI] [PubMed] [Google Scholar]
- Menegus M. A., Hollick G. E. Increased Efficiency of Group B Coxsackievirus Isolation from Clinical Specimens by Use of BGM Cells. J Clin Microbiol. 1982 May;15(5):945–948. doi: 10.1128/jcm.15.5.945-948.1982. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Minor P. D., Pipkin P. A., Hockley D., Schild G. C., Almond J. W. Monoclonal antibodies which block cellular receptors of poliovirus. Virus Res. 1984;1(3):203–212. doi: 10.1016/0168-1702(84)90039-x. [DOI] [PubMed] [Google Scholar]
- Svensson U., Persson R., Everitt E. Virus-receptor interaction in the adenovirus system I. Identification of virion attachment proteins of the HeLa cell plasma membrane. J Virol. 1981 Apr;38(1):70–81. doi: 10.1128/jvi.38.1.70-81.1981. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zajac I., Crowell R. L. Differential inhibition of attachment and eclipse activities of HeLa cells for enteroviruses. J Virol. 1969 Apr;3(4):422–428. doi: 10.1128/jvi.3.4.422-428.1969. [DOI] [PMC free article] [PubMed] [Google Scholar]