Abstract
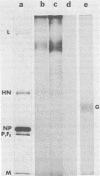
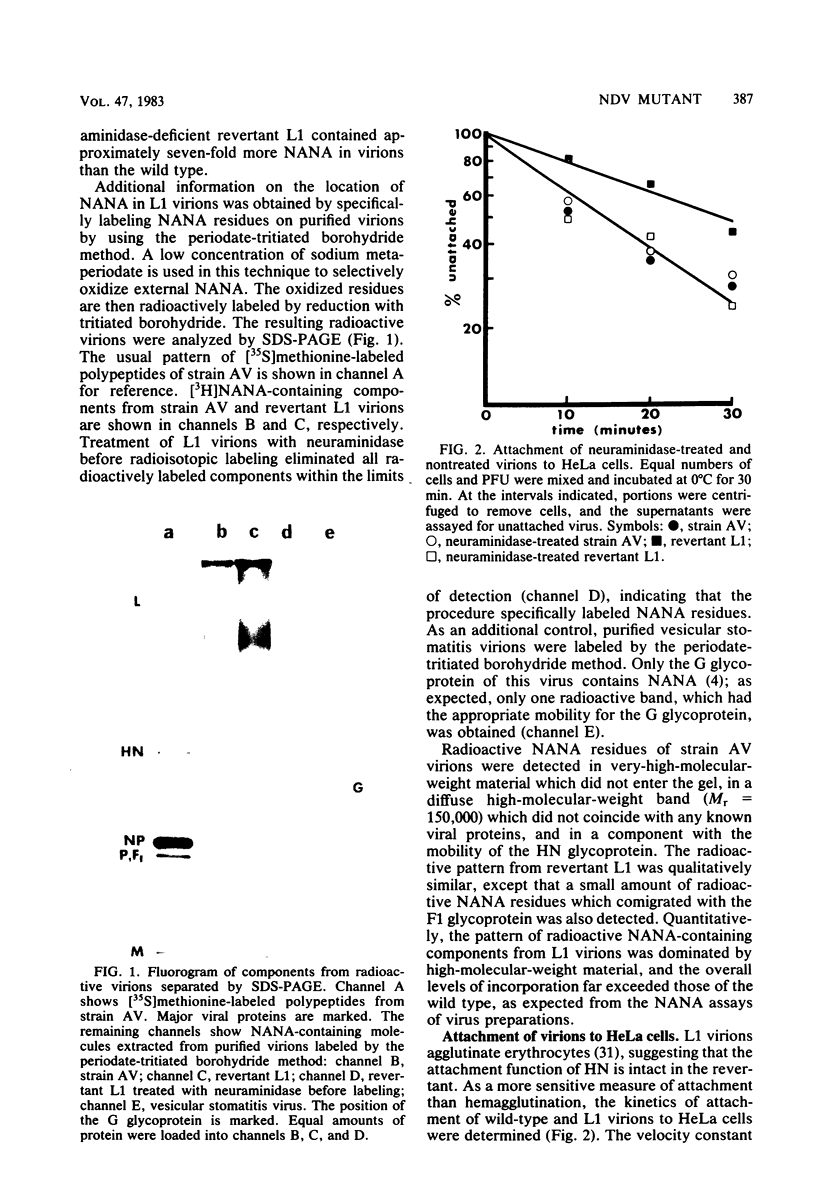
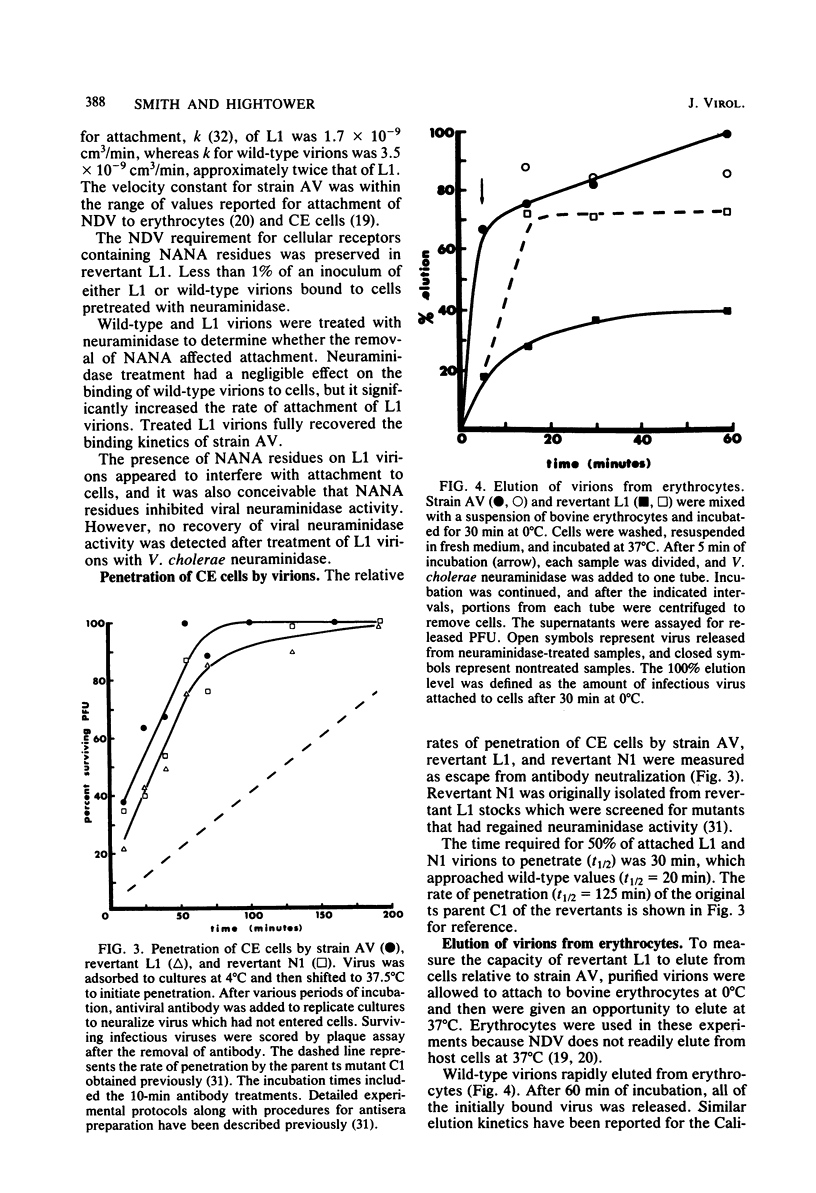
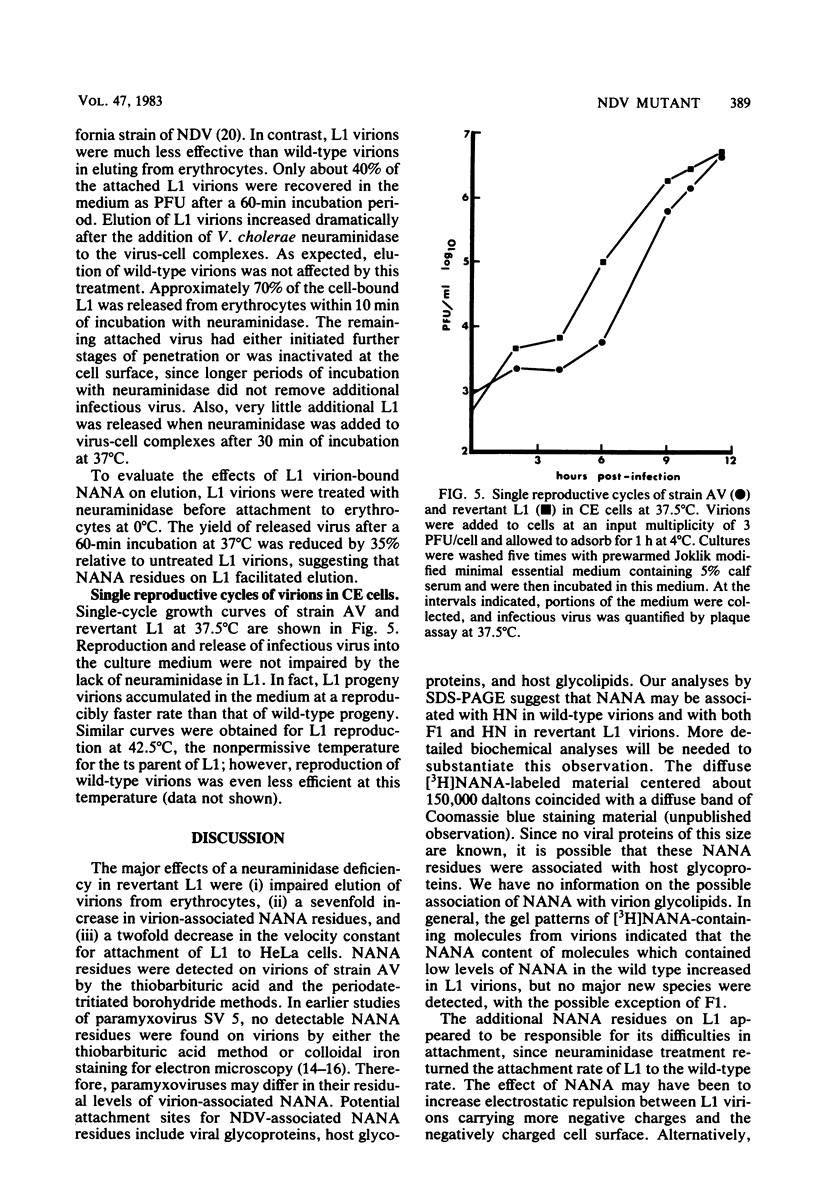
A second-step revertant (L1) of a temperature-sensitive mutant (C1) of Newcastle disease virus agglutinated erythrocytes normally but had less than 3% of the wild-type (strain AV) levels of neuraminidase activity. Revertant L1 had seven times more virion-associated N-acetylneuraminic acid (NANA) than strain AV. NANA residues on purified virions were specifically labeled with periodate and tritiated borohydride. Analyses of radiolabeled L1 virions on sodium dodecyl sulfate-polyacrylamide gels showed that most of the virion-associated NANA was in a high-molecular-weight component with an electrophoretic mobility different from that of any known viral protein. NANA was also detected in molecules with the electrophoretic mobility of the viral glycoproteins HN and F1. Revertant L1 had a twofold lower rate constant of attachment to HeLa cells than that of the wild-type. Treatment of L1 virions with Vibrio cholerae neuraminidase removed the excess NANA and returned L1 attachment kinetics to normal. Revertant N1, which has 10-fold more neuraminidase activity than L1, penetrated host cells at the same rate as L1. L1 was impaired in elution from erythrocytes. Removal of virion-associated NANA exacerbated this defect. Despite a small disadvantage in attachment and a major defect in elution relative to strain AV, revertant L1 enjoyed a slight advantage over the wild-type during a single reproductive cycle in cultured chicken embryo cells.
Full text
PDF



Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Bonner W. M., Laskey R. A. A film detection method for tritium-labelled proteins and nucleic acids in polyacrylamide gels. Eur J Biochem. 1974 Jul 1;46(1):83–88. doi: 10.1111/j.1432-1033.1974.tb03599.x. [DOI] [PubMed] [Google Scholar]
- Burge B. W., Huang A. S. Comparison of membrane protein glycopeptides of Sindbis virus and vesicular stomatitis virus. J Virol. 1970 Aug;6(2):176–182. doi: 10.1128/jvi.6.2.176-182.1970. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Collins P. L., Hightower L. E., Ball L. A. Transcription and translation of Newcastle disease virus mRNA's in vitro. J Virol. 1978 Oct;28(1):324–336. doi: 10.1128/jvi.28.1.324-336.1978. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Drzeniek R. Substrate specificity of neuraminidases. Histochem J. 1973 May;5(3):271–290. doi: 10.1007/BF01004994. [DOI] [PubMed] [Google Scholar]
- Gahmberg C. G., Andersson L. C. Selective radioactive labeling of cell surface sialoglycoproteins by periodate-tritiated borohydride. J Biol Chem. 1977 Aug 25;252(16):5888–5894. [PubMed] [Google Scholar]
- Hightower L. E., Bratt M. A. Protein synthesis in Newcastle disease virus-infected chicken embryo cells. J Virol. 1974 Apr;13(4):788–800. doi: 10.1128/jvi.13.4.788-800.1974. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Huang R. T., Rott R., Wahn K., Klenk H. D., Kohama T. The function of the neuraminidase in membrane fusion induced by myxoviruses. Virology. 1980 Dec;107(2):313–319. doi: 10.1016/0042-6822(80)90299-8. [DOI] [PubMed] [Google Scholar]
- Klenk H. D., Caliguiri L. A., Choppin P. W. The proteins of the parainfluenza virus SV5. II. The carbohydrate content and glycoproteins of the virion. Virology. 1970 Oct;42(2):473–481. doi: 10.1016/0042-6822(70)90290-4. [DOI] [PubMed] [Google Scholar]
- Klenk H. D., Choppin P. W. Glycosphingolipids of plasma membranes of cultured cells and an enveloped virus (SV5) grown in these cells. Proc Natl Acad Sci U S A. 1970 May;66(1):57–64. doi: 10.1073/pnas.66.1.57. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Klenk H. D., Compans R. W., Choppin W. P. An electron microscopic study of the presence or absence of neuraminic acid in enveloped viruses. Virology. 1970 Dec;42(4):1158–1162. doi: 10.1016/0042-6822(70)90368-5. [DOI] [PubMed] [Google Scholar]
- LEVINE S., SAGIK B. P. The interactions of Newcastle disease virus (NDV) with chick embryo tissue culture cells: attachment and growth. Virology. 1956 Feb;2(1):57–68. doi: 10.1016/0042-6822(56)90076-9. [DOI] [PubMed] [Google Scholar]
- Laemmli U. K. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature. 1970 Aug 15;227(5259):680–685. doi: 10.1038/227680a0. [DOI] [PubMed] [Google Scholar]
- Laskey R. A., Mills A. D. Quantitative film detection of 3H and 14C in polyacrylamide gels by fluorography. Eur J Biochem. 1975 Aug 15;56(2):335–341. doi: 10.1111/j.1432-1033.1975.tb02238.x. [DOI] [PubMed] [Google Scholar]
- Marcus P. I., Salb J. M., Schwartz V. G. Nuclear surface N-acetyl neuraminic acid terminating receptors for myxovirus attachment. Nature. 1965 Dec 11;208(5015):1123–1124. doi: 10.1038/2081122b0. [DOI] [PubMed] [Google Scholar]
- Meindl P., Bodo G., Palese P., Schulman J., Tuppy H. Inhibition of neuraminidase activity by derivatives of 2-deoxy-2,3-dehydro-N-acetylneuraminic acid. Virology. 1974 Apr;58(2):457–463. doi: 10.1016/0042-6822(74)90080-4. [DOI] [PubMed] [Google Scholar]
- Palese P., Compans R. W. Inhibition of influenza virus replication in tissue culture by 2-deoxy-2,3-dehydro-N-trifluoroacetylneuraminic acid (FANA): mechanism of action. J Gen Virol. 1976 Oct;33(1):159–163. doi: 10.1099/0022-1317-33-1-159. [DOI] [PubMed] [Google Scholar]
- Palese P., Schulman J. L., Bodo G., Meindl P. Inhibition of influenza and parainfluenza virus replication in tissue culture by 2-deoxy-2,3-dehydro-N-trifluoroacetylneuraminic acid (FANA). Virology. 1974 Jun;59(2):490–498. doi: 10.1016/0042-6822(74)90458-9. [DOI] [PubMed] [Google Scholar]
- Palese P., Tobita K., Ueda M., Compans R. W. Characterization of temperature sensitive influenza virus mutants defective in neuraminidase. Virology. 1974 Oct;61(2):397–410. doi: 10.1016/0042-6822(74)90276-1. [DOI] [PubMed] [Google Scholar]
- Paulson J. C., Weinstein J., Dorland L., van Halbeek H., Vliegenthart J. F. Newcastle disease virus contains a linkage-specific glycoprotein sialidase. Application to the localization of sialic acid residues in N-linked oligosaccharides of alpha 1-acid glycoprotein. J Biol Chem. 1982 Nov 10;257(21):12734–12738. [PubMed] [Google Scholar]
- Peeples M. E., Glickman R. L., Bratt M. A. Thermostabilities of virion activities of Newcastle disease virus: evidence that the temperature-sensitive mutants in complementation groups B, BC, and C have altered HN proteins. J Virol. 1983 Jan;45(1):18–26. doi: 10.1128/jvi.45.1.18-26.1983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Portner A. The HN glycoprotein of Sendai virus: analysis of site(s) involved in hemagglutinating and neuraminidase activities. Virology. 1981 Dec;115(2):375–384. doi: 10.1016/0042-6822(81)90118-5. [DOI] [PubMed] [Google Scholar]
- Scheid A., Choppin P. W. Isolation and purification of the envelope proteins of Newcastle disease virus. J Virol. 1973 Feb;11(2):263–271. doi: 10.1128/jvi.11.2.263-271.1973. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Smith G. W., Hightower L. E. Identification of the P proteins and other disulfide-linked and phosphorylated proteins of Newcastle disease virus. J Virol. 1981 Jan;37(1):256–267. doi: 10.1128/jvi.37.1.256-267.1981. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Smith G. W., Hightower L. E. Revertant analysis of a temperature-sensitive mutant of Newcastle disease virus with defective glycoproteins: implication of the fusion glycoprotein in cell killing and isolation of a neuraminidase-deficient hemagglutinating virus. J Virol. 1982 May;42(2):659–668. doi: 10.1128/jvi.42.2.659-668.1982. [DOI] [PMC free article] [PubMed] [Google Scholar]
- TOLMACH L. J. Attachment and penetration of cells by viruses. Adv Virus Res. 1957;4:63–110. doi: 10.1016/s0065-3527(08)60596-5. [DOI] [PubMed] [Google Scholar]
- Tsipis J. E., Bratt M. A. Isolation and preliminary characterization of temperature-sensitive mutants of Newcastle disease virus. J Virol. 1976 Jun;18(3):848–855. doi: 10.1128/jvi.18.3.848-855.1976. [DOI] [PMC free article] [PubMed] [Google Scholar]