Abstract
Background
Little is known about the racial differences in the incidence and progression of HIV-related chronic kidney disease (CKD) that underlie African American–white disparities in HIV-related end-stage renal disease (ESRD).
Methods
In a cohort in Baltimore, Maryland, we measured CKD incidence, glomerular filtration rate (GFR) slope, and progression to ESRD in 3332 African American and 927 white HIV-infected subjects.
Results
A total of 284 subjects developed CKD, 100 (35%) of whom subsequently developed ESRD. African American subjects were at slightly increased risk for incident CKD, compared with white subjects (hazard ratio [HR], 1.9 [95% confidence interval {CI}, 1.2–2.8]). However, once CKD had commenced, the African American subjects developed ESRD markedly faster than did the white subjects (HR, 17.7 [95% CI, 2.5–127.0]), and, correspondingly, their GFR decline after diagnosis of CKD was 6-fold more rapid (P < .001). In the subset of African American subjects for whom kidney-biopsy data were available, progression to ESRD was significantly faster than that in white subjects with CKD, irrespective of the presence of HIV-associated nephropathy.
Conclusions
The results of this study suggest that African American–white disparities in HIV-related ESRD are explained predominantly by a more aggressive natural disease history in African Americans and less by racial differences in CKD incidence.
Chronic kidney disease (CKD) and end-stage renal disease (ESRD) are important comorbid conditions that are prevalent in HIV-infected individuals [1]. Data from the United States Renal Data System indicate that the risk of development of ESRD is ~50-fold higher in HIV-infected African Americans than in HIV-infected whites [2]. However, because data collection in this database begins at the initiation of renal replacement therapy, this resource is less useful for characterizing the course of events prior to ESRD. Analyses of data from HIV cohort studies have assessed the natural history of CKD but have been limited by nonstandardized definitions [3, 4] and small numbers of ESRD events [4–6].
Little information is available regarding the racial differences in HIV-related CKD epidemiology that underlie African American–white disparities in ESRD. For example, it is not known if racial differences in HIV-related ESRD are explained primarily by differences in the incidence of CKD, in progression rates to ESRD, or by a combination of the two. Additionally, the degree to which racial differences in ESRD may be explained by confounding factors, such as injection drug use and use of highly active antiretroviral therapy (HAART) or angiotensin blockade, is also undefined. To address these issues, we analyzed incident CKD and progression to ESRD in a well-characterized, predominantly African American and white, HIV cohort in Baltimore, Maryland.
SUBJECTS AND METHODS
Cohorts and subjects
The Johns Hopkins HIV Clinical Cohort includes data from >5000 subjects who, from 1990 onward, have received primary care in the clinic [7]. Information from clinical records was reviewed and abstracted by trained technicians onto structured data-collection forms and then was entered into a relational database. Comprehensive demographic, clinical, laboratory, pharmaceutical, and psychosocial data were collected at times corresponding to enrollment by the clinic and at 6-month intervals thereafter. Clinical diagnoses of AIDS-associated opportunistic conditions, other selected conditions (including initiation of renal replacement therapy), and deaths were routinely abstracted since cohort inception. Subjects provided written informed consent, and the study was approved by the Johns Hopkins Medicine Institutional Review Board.
Individuals were included in the present analysis if they (1) were non-Hispanic African American or white, (2) had not started renal replacement therapy prior to enrollment, (3) had an estimated glomerular filtration rate (GFR) [8] >15 mL/min/1.73 m2 at enrollment, and (4) had at least 2 GFR estimates, separated by 3 months, after enrollment. A chart review to abstract additional clinical and pharmacologic data was conducted in subjects with CKD.
Definitions
AIDS was defined according to the 1993 Centers for Disease Control and Prevention classification guidelines [9] (including a CD4 count <200 cells/mm3). The pre-HAART and HAART eras were defined as the calendar periods before and after 1 January 1996, respectively. Diabetes was defined as a prior clinical diagnosis of diabetes, prescription of insulin or oral antidiabetic medications, or a measured serum glucose concentration >200 mg/dL. Hypertension was defined as a prior clinical diagnosis of hypertension, prescription of antihypertensive medication, or, on at least 2 separate occasions, systolic blood pressure >140 mmHg or diastolic blood pressure >90 mmHg.
All serum creatinine measurements that were obtained in clinical practice from cohort subjects were available (Jaffe method prior to February 2003, enzymatic method thereafter; Roche Diagnostics). To avoid undue influence from periods of acute illness when serum creatinine may have been measured frequently, a reduced data set was generated that comprised only values measured closest in time to quarterly anniversaries after enrollment. GFR was estimated by the 4-variable Modification of Diet in Renal Disease equation [8]. CKD was diagnosed on the basis of at least 2 GFR measurements <60 mL/min/1.73 m2 that were separated by >3 months [8]. To increase the specificity this diagnosis, we also required that >50% of GFR values, after the index value, be <60 mL/min/1.73 m2. Kidney disease was considered to be prevalent if the GFR at enrollment was <60 mL/min/1.73 m2 and if the aforementioned criteria were also met; it was considered to be incident if the GFR was >60 mL/min/1.73 m2 at enrollment but the criteria for CKD were later met during follow-up. ESRD was diagnosed on the basis the initiation of renal replacement therapy.
In subjects with CKD, we retrospectively abstracted additional clinical data (e.g., diabetes status and hypertension status) and laboratory data from medical charts and the electronic patient record, data that were not available for the cohort as a whole. Estimated 24-h urine-protein excretion was defined as the higher of either the urine protein (or albumin):creatinine concentration ratio, from a timed or random urine sample, or the total measured protein from a 24-h urine collection. Semiquantitative urine-protein concentrations were measured by Clinitek or Atlas (after 1 January 2002) automated assays (Bayer). When data on estimated 24-h urine-protein excretion were available, they were used to categorize proteinuria into 3 grades: low (<1 g/24 h), intermediate (1–3 g/24 h), and high (≥3 g/24 h). When no quantitative proteinuria data were available, the proteinuria grade was defined on the basis of the semiquantitative urine-protein measurement—low (negative or trace proteinuria), intermediate (1+ or 2+ proteinuria), or high (≥3+ proteinuria)—closest in time to the onset of CKD.
Statistical analysis
Baseline characteristics of white and African American subjects with CKD were compared by Fisher’s exact test or the Wilcoxon rank-sum test. Risk factors for CKD were assessed by Poisson regression and Cox proportional-hazards models. Baseline risk factors included sex, race, CD4 cell count stratum at enrollment, hepatitis C antibody status, and hepatitis B antigen status. Age, calendar period, and AIDS status were treated as time-dependent covariates, with individual subjects’ data being allowed to contribute follow-up time to more than one stratum of these variables. HIV RNA data were not included because the relevant assay was not used in the present study’s cohort prior to 1996. Similarly, we did not include diabetes or hypertension in the cohort-wide analysis of factors associated with CKD because these variables were not abstracted in the cohort until approximately 1998. Variables in the final adjusted model were selected, in a stepwise manner, from all covariates considered in univariate analysis, with factors sequentially removed from the model if P > .2.
GFR slopes were derived from quarterly GFR estimates spanning the period from diagnosis of CKD to the earliest of either (1) the last available GFR, (2) a GFR <15 mL/min/1.73 m2 with no subsequent rebound toward normal, or (3) initiation of renal replacement therapy. GFR slopes were estimated by a random-effects linear model. Factors associated with progression to ESRD were assessed in subjects with CKD by Kaplan-Meier plots and Cox proportional-hazards models. The proportional-hazards assumption was checked by assessment of Schoenfeld residuals [10] and log-log plots. All analyses were conducted by use of Stata software (version 10; Stata).
RESULTS
A total of 4259 individuals—3332 (78%) of whom were African American and 927 (22%) of whom were white—met the inclusion criteria for the study and provided 18,778 person-years of follow-up (table 1). The mean follow-up time was 4.5 years, in both African American and white subjects. Compared with these included subjects, the 875 excluded individuals were more likely (10% vs. 0; P < .001) to be Hispanic, Asian, or unknown race and were slightly younger (median age, 37 vs. 38 years; P = .02) at enrollment. However, sex distribution and injection drug use in the included subjects were not statistically significantly different from those in the excluded subjects (data not shown).
Table 1.
Characteristics of HIV-infected cohort subjects overall and of those with chronic kidney disease (CKD), stratified by race, in Baltimore, Maryland, 1990–2004.
| Subjects with CKD
|
||||||||
|---|---|---|---|---|---|---|---|---|
| Characteristic | Cohort overall (n = 4259) | White (n = 31) | African American (n = 253) | Pa | ||||
| Age at enrollment, median (IQR), years | 38 (32–43) | 44 (36–48) | 39 (34–46) | .13 | ||||
| Sex, no. (%) of subjects | .70 | |||||||
| Male | 2887 (68) | 20 (65) | 152 (60) | |||||
| Female | 1372 (32) | 11 (35) | 101 (40) | |||||
| Injection drug–use history, no. (%) of subjects | 1998 (47) | 10 (32) | 158 (62) | .002 | ||||
| Hepatitis C antibody status, no. (%) of subjects | .001 | |||||||
| Negative | 1803 (50) | 19 (76) | 97 (42) | |||||
| Positive | 1785 (50) | 6 (24) | 134 (58) | |||||
| Hepatitis B surface–antigen status, no. (%) of subjects | ||||||||
| Negative | 2914 (92) | 22 (96) | 200 (93) | 1.00 | ||||
| Positive | 272 (8) | 1 (4) | 16 (7) | |||||
| Serum creatinine concentration at enrollment, median (IQR), mg/dL | 0.9 (0.8–1.1) | 1.0 (0.8–1.4) | 1.2 (0.9–2.4) | .007 | ||||
| Glomerular filtration rate at enrollment, mL/min/1.73 m2, median (IQR) | 104 (85–123) | 85 (63–96) | 76 (51–101) | .38 | ||||
| Subjects with CKD | ||||||||
| Type of case, no. (%) of subjects | ||||||||
| Prevalent at enrollment | 3 (10) | 71 (28) | .03 | |||||
| Incident during follow-up | 28 (90) | 182 (72) | ||||||
| HIV-treatment era,b no. (%) of subjects | ||||||||
| Pre-HAART | 6 (19) | 65 (26) | .52 | |||||
| HAART | … | 25 (81) | 188 (74) | |||||
| Age at CKD, median (IQR), years | … | 46 (40–54) | 41 (37–49) | .04 | ||||
| CD4 count at CKD, cells/mm3, median (IQR) | … | 239 (117–417) | 122 (24–328) | .04 | ||||
| Nadir CD4 count prior to CKD, cells/mm3, median (IQR) | … | 104 (26–243) | 80 (14–247) | .46 | ||||
| CDC category C opportunistic condition prior to CKD, no. (%) of subjects | … | 20 (65) | 136 (54) | .34 | ||||
| Diabetes mellitus, no. (%) of subjects | … | 2 (6) | 24 (10) | .75 | ||||
| Hypertension, no. (%) of subjects | … | 10 (32) | 105 (46) | .18 | ||||
| Antiretroviral exposure prior to CKD, no. (%) of subjects | … | |||||||
| None | 3 (10) | 60 (24) | ||||||
| NRTI only | 6 (19) | 89 (35) | .009 | |||||
| PI or NNRTI | 22 (71) | 104 (41) | ||||||
| Serum creatinine concentration, median (IQR), mg/dL | … | 1.5 (1.4–1.6) | 1.9 (1.6–2.4) | <.001 | ||||
| Glomerular filtration rate, mLmin/1.73 m2, median (IQR) | … | 52 (45–56) | 45 (33–53) | .005 | ||||
| Urine proteinuria (dipstick), no. (%) of subjects | ||||||||
| Negative or trace | 16 (59) | 46 (19) | ||||||
| 1+ | 6 (22) | 31 (13) | <.001 | |||||
| 2+ | 5 (19) | 39 (16) | ||||||
| ≥3+ | 0 | 121 (51) | ||||||
| Estimated 24-h urine protein excretion, median (IQR), g/dL | … | 0.5 (0.2–1.2) | 2.7 (1.2–8.1) | <.001 | ||||
| Proteinuria grade,c no. (%) of subjects | … | |||||||
| Low | 16 (59) | 53 (22) | <.001 | |||||
| Intermediate | 10 (37) | 80 (33) | ||||||
| High | 1 (4) | 111 (45) | ||||||
| Serum albumin, median (IQR), g/dL | … | 4.1 (3.6–4.5) | 3.3 (2.7–3.7) | <.001 | ||||
| Hemoglobin, median (IQR), g/dL | … | |||||||
| Male | 13.1 (10.5–15.3) | 10.9 (9.5–12.9) | .02 | |||||
| Female | 11.8 (9.3–13.4) | 10.3 (8.6–11.7) | .10 | |||||
NOTE. CDC, Centers for Disease Control and Prevention; HAART, highly active antiretroviral therapy; IQR, interquartile range; NNRTI, nonnucleoside reverse-transcriptase inhibitor; NRTI, nucleoside reverse-transcriptase inhibitor; PI, protease inhibitor.
For comparisons between white and African American subjects.
HAART era is defined as beginning 1 January 1996.
For definition, see text.
Characteristics of subjects with CKD, stratified by race, are shown in table 1. Compared with white subjects with CKD, African American subjects with CKD were significantly more likely to have prevalent (rather than incident) disease, to be younger, to have a history of injection drug use, and to be seropositive for hepatitis C. African American and white subjects with CKD had similar nadir CD4 cell counts and were similarly likely to have had an opportunistic infection; however, CD4 cell counts at the time of diagnosis of CKD were significantly lower in African American subjects than in white subjects. Compared with white subjects, African American subjects were more likely to be anti-retroviral naive or to have used nucleoside reverse-transcriptase inhibitors only and were less likely to have received HAART.
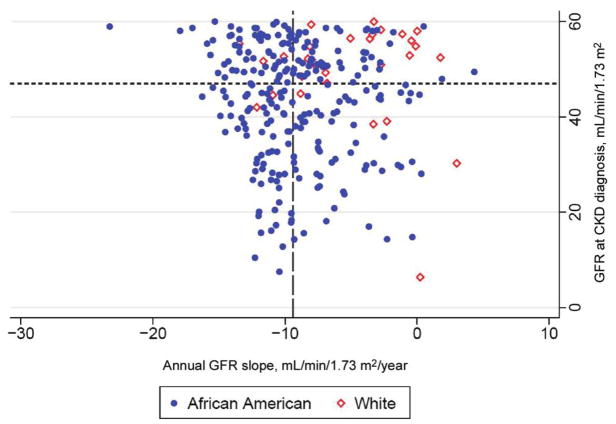
Compared with white subjects At diagnosis of CKD, African Americans had significantly higher serum creatinine and urinary protein excretion, and significantly lower GFR, serum albumin, and serum hemoglobin, than did whites. The median number of GFR estimates subsequent to diagnosis of CKD was 4 (interquartile range [IQR], 3–9), with no significant difference by race (P = .88). The average time between GFR estimates was 95 days (IQR, 76–128 days). When adjusted for baseline GFR, the annual GFR slopes after diagnosis of CKD were −9.2 mL/min/1.73 m2/year (95% confidence interval [CI] −10.6 to −7.9 mL/min/1.73 m2/year) and −1.5 mL/min/1.73 m2/year (95% CI, −5.0 to 2.0 mL/min/1.73 m2/year) in African American and white subjects, respectively (difference, −7.8 mL/min/1.73 m2/year [95% CI, −11.5 to −4.0 mL/min/1.73 m2/year]; P < .001). When an indicator variable for the change in creatinine-measurement method (in February 2003) was included in the models, there was no substantive alteration in the estimates or inferences (data not shown). Figure 1 shows a scatterplot in which each subject is represented in terms of GFR at diagnosis of CKD and subsequent GFR slope.
Figure 1.
Scatterplot showing distribution of HIV-infected subjects with chronic kidney disease, in terms of their glomerular filtration rate (GFR) at diagnosis and their subsequent annualized GFR slope. The figure is divided into quadrants based on the median GFR at baseline (47 mL/min/1.73 m2 [short-dashed horizontal line] ) and the median GFR slope (long-dashed vertical line, −9.4 mL/min/1.73 m2/year). White subjects are overrepresented in the upper-right quadrant (least-aggressive disease) and are underrepresented in the lower-left quadrant (most-aggressive disease).
Of 4185 subjects without CKD at enrollment, the disease developed in 210, for an overall incidence rate of 11.2 cases (95% CI, 9.8 –12.8 cases) per 1000 person-years. Table 2 shows univariate and adjusted associations for the development of CKD. In the multivariate model, factors significantly associated with increased risk of CKD included African American race, female sex, increasing age, presence of AIDS, lower CD4 cell count at enrollment, history of injection drug use, and earlier calendar time.
Table 2.
Incidence of Chronic kidney disease (CKD) incidence and associated factors in HIV-infected cohort subjects without CKD at enrollment, Baltimore, Maryland, 1990–2004
| Unadjusted model
|
Adjusted modela |
||||||
|---|---|---|---|---|---|---|---|
| Characteristic | Person-years | CKD cases, no. | CKD rate/1000 person-years | IRR (95% CI) | P | IRR (95% CI) | P |
| Overall | 18,778 | 210 | 11.2 | … | |||
| Sex | |||||||
| Male | 12,344 | 127 | 10.3 | 1.0 | 1.0 | ||
| Female | 6435 | 83 | 12.9 | 1.25 (0.95–1.65) | .11 | 1.50 (1.08–2.07) | .015 |
| Race | |||||||
| White | 4162 | 28 | 6.7 | 1.0 | 1.0 | ||
| African American | 14,616 | 182 | 12.5 | 1.85 (1.24–2.76) | .002 | 1.65 (1.00–2.71) | .048 |
| Age, years | |||||||
| <45 | 13,591 | 133 | 9.8 | 1.0 | 1.0 | ||
| 45–55 | 4258 | 58 | 13.6 | 1.39 (1.02–1.89) | .036 | 1.45 (1.01–2.09) | .044 |
| >55 | 930 | 19 | 20.4 | 2.08 (1.29–3.38) | .003 | 3.47 (2.07–5.81) | <.001 |
| AIDSb | |||||||
| No | 8414 | 43 | 5.1 | 1.0 | 1.0 | ||
| Yes | 10,365 | 167 | 16.1 | 3.15 (2.25–4.41) | <.001 | 2.80 (1.80–4.39) | <.001 |
| CD4 count at cohort enrollment, cells/mm3 | |||||||
| ≥300 | 10,189 | 70 | 6.9 | 1.0 | 1.0 | ||
| 100–299 | 4770 | 67 | 14.0 | 2.04 (1.46–2.86) | <.001 | 1.37 (0.91–2.05) | .14 |
| <100 | 3,751 | 73 | 19.5 | 2.83 (2.04–3.93) | <.001 | 1.68 (1.09–2.60) | .018 |
| Injection drug use | |||||||
| No | 9663 | 78 | 8.1 | 1.0 | 1.0 | ||
| Yes | 9116 | 132 | 14.5 | 1.79 (1.36–2.37) | <.001 | 1.73 (1.25–2.41) | .001 |
| Hepatitis C antibody status | |||||||
| Negative | 8082 | 83 | 10.3 | 1.0 | … | ||
| Positive | 8505 | 108 | 12.7 | 1.24 (0.93–1.65) | .15 | … | |
| Hepatitis B surface antigen status | |||||||
| Negative | 13,731 | 169 | 12.3 | 1.0 | … | ||
| Positive | 1140 | 10 | 8.8 | 0.71 (0.38–1.35) | .30 | … | |
| Calendar period | |||||||
| Before 1996 | 3334 | 46 | 13.8 | 1.0 | 1.0 | ||
| 1996–1998 | 4133 | 54 | 13.1 | 0.95 (0.64–1.40) | .79 | 0.81 (0.52–1.28) | .37 |
| 1999–2001 | 5780 | 64 | 11.1 | 0.80 (0.55–1.17) | .26 | 0.69 (0.44–1.08) | .11 |
| ≥2002 | 5531 | 46 | 8.3 | 0.60 (0.40–0.91) | .015 | 0.49 (0.30–0.79) | .004 |
NOTE. CI, confidence interval; IRR, incidence rate ratio.
Adjusted for factors indicated. Factors for the adjusted model were selected by stepwise selection; factors for which P > .2 were removed from the adjusted model.
Defined according to Centers for Disease Control and Prevention 1993 criteria, including opportunistic condition or CD4 count <200 cells/mm3.
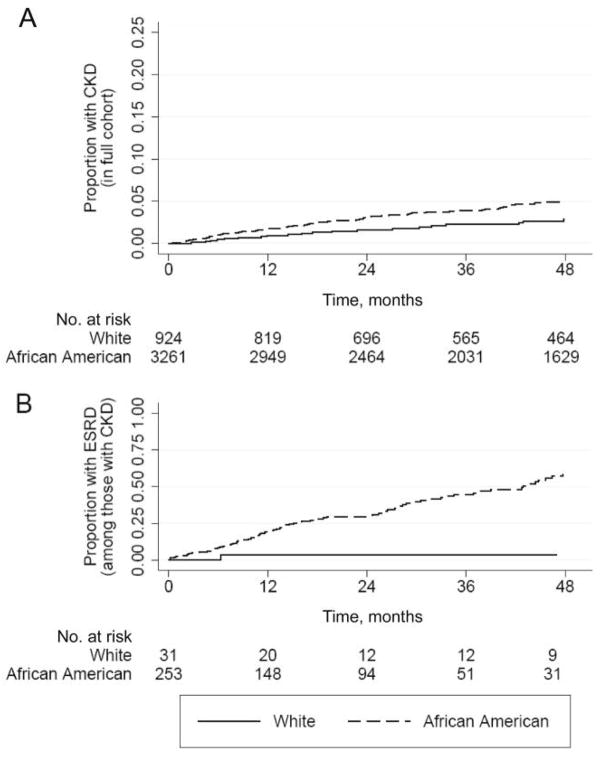
Of the 284 subjects with prevalent or incident CKD, 100 (35%) developed ESRD. An additional 5 subjects (all African American) developed ESRD without having previously met the criteria for CKD and therefore were not included in the analysis; these 5 subjects either developed ESRD after acute kidney injury or, prior to presenting with ESRD, had a prolonged hiatus from cohort follow-up. The numbers of African American and white subjects with CKD who progressed to ESRD were 99 (39%) and 1 (3%), respectively (P < .001, by log-rank test). Kaplan-Meier plots shown in figure 2 contrast the racial difference in the incidence of CKD in the cohort overall (hazard ratio [HR], 1.9 [95% CI 1.2–2.8] for African Americans vs. whites) to the racial difference in the progression to ESRD (HR, 17.7 [95% CI, 2.5–127.0], for African Americans vs. whites) in those with CKD. The associations between other factors and progression to ESRD are shown in table 3. In addition to African American race, younger age, baseline GFR, annual GFR slope, serum albumin concentration, degree of proteinuria, and nonuse of angiotensin-converting–enzyme inhibitor (ACE-I) or angiotensin II–receptor blocker (ARB) were statistically significantly associated with progression to ESRD. Multivariate analysis was not performed because only a single white subject with CKD progressed to ESRD, which made point estimates for race unstable in adjusted analyses.
Figure 2.
Kaplan-Meier plots showing (A) time to onset of chronic kidney disease (CKD) in HIV-infected subjects free of CKD at enrollment (hazard ratio, 1.9 [95% confidence interval, 1.2–2.8]; P = .002, by log-rank test) and (B) time to end-stage renal disease (ESRD) in subjects with CKD (hazard ratio, 17.7 [95% confidence interval, 2.5–127.0]; P = .001, by log-rank test).
Table 3.
Factors associated with progression to end-stage renal disease (ESRD) in HIV-infected subjects with chronic kidney disease (CKD), Baltimore, Maryland, 1990–2004.
| Characteristic | Total subjects, no | Subjects with ESRD, no. (%) | Unadjusted HR (95% CI) | P |
|---|---|---|---|---|
| All CKD types | 284 | 100 (35) | … | … |
| Prevalent at enrollment | 74 | 33 (45) | 1.0 | |
| Incident during follow-up | 210 | 67 (32) | 0.9 (0.6–1.4) | .70 |
| Sex | ||||
| Male | 172 | 61 (35) | 1.0 | |
| Female | 112 | 39 (35) | 1.0 (0.7–1.5) | .94 |
| Race | ||||
| White | 31 | 1 (3) | 1.0 | |
| African American | 253 | 99 (39) | 17.7 (2.5–127.0) | .004 |
| Injection drug use | ||||
| No | 116 | 42 (36) | 1.0 | |
| Yes | 168 | 58 (35) | 1.1 (0.7–1.6) | .70 |
| Age | ||||
| ≤40 years | 122 | 55 (45) | 1.0 | |
| >40 years | 162 | 45 (28) | 0.5 (0.3–0.7) | <.001 |
| AIDSa | ||||
| No | 96 | 37 (57) | 1.0 | |
| Yes | 188 | 63 (47) | 0.9 (0.7–1.3) | .61 |
| CD4 at CKD onset | ||||
| ≥200 cells/mm3 | 113 | 39 (34) | 1.0 | |
| <200 cells/mm3 | 166 | 61 (37) | 1.4 (0.9–2.0) | .14 |
| Treatment erab | ||||
| Pre-HAART | 71 | 23 (32) | 1.0 | |
| HAART | 213 | 77 (36) | 1.1 (0.7–1.8) | .64 |
| Baseline GFR | ||||
| Below the medianc | 139 | 43 (31) | 1.0 | |
| Less than or equal to the medianc | 145 | 57 (39) | 1.7 (1.1–2.5) | .013 |
| Annual GFR slope | ||||
| 1st quartile | 70 | 41 (59) | 1.0 | |
| 2nd quartile | 69 | 32 (46) | 0.8 (0.5–1.2) | .29 |
| 3rd quartile | 70 | 12 (17) | 0.2 (0.1–0.3) | <.001 |
| 4th quartile | 69 | 9 (13) | 0.04 (0.01–0.08) | <.001 |
| Albumin at onset of CKD | ||||
| ≤3.5 g/dL | 153 | 67 (44) | 1.0 | |
| >3.5 g/dL | 131 | 33 (25) | 0.3 (0.2–0.4) | <.001 |
| Hemoglobin at onset of CKD | ||||
| Less than or equal to the mediand | 144 | 41 (28) | 1.0 | |
| Below the mediand | 140 | 59 (42) | 1.5 (1.0–2.2) | .07 |
| Proteinuria gradee | ||||
| Low | 69 | 13 (19) | 1.0 | |
| Intermediate | 90 | 25 (28) | 2.3 (1.2–4.5) | .017 |
| High | 112 | 59 (53) | 6.2 (3.3–11.6) | <.001 |
| Diabetes | ||||
| No | 252 | 88 (35) | 1.0 | |
| Yes | 26 | 9 (35) | 0.9 (0.4–1.8) | .74 |
| Hypertension | ||||
| No | 142 | 43 (30) | 1.0 | |
| Yes | 115 | 43 (37) | 1.0 (0.6–1.5) | .97 |
| Use of ACE-I or ARB | ||||
| No | 185 | 66 (36) | 1.0 | |
| Yes | 66 | 21 (32) | 0.5 (0.3–0.8) | .006 |
| Data unavailable | 33 | 13 (39) | 1.0 (0.5–1.8) | .96 |
| Antiretroviral therapy | ||||
| None or NRTI only | 90 | 26 (29) | 1.0 | |
| HAART | 194 | 74 (38) | 0.9 (0.6–1.4) | .64 |
NOTE. ACE-I, angiotensin converting–enzyme inhibitor, ARB, angiotensin II–receptor blocker; CI, confidence interval; GFR, glomerular filtration rate; HAART, highly active antiretroviral therapy; NRTI, nucleoside reverse-transcriptase inhibitor.
Defined according to Centers for Disease Control and Prevention 1993 criteria, including opportunistic condition or CD4 count <200 cells/mm3.
HAART era is defined as beginning 1 January 1996.
Median GFR at diagnosis of CKD was 47 mL/min/1.73 m2.
Stratified by sex.
For definition, see text.
In subjects with CKD, kidney-biopsy samples were obtained from 16% (5 of 31) of the whites and 27% (68/253) of the African Americans (P = .28). Histopathologic findings from the biopsy samples are shown in table 4. Consistent with prior reports [11–13], HIV-associated nephropathy (HIVAN) was found exclusively in African Americans. In African American subjects for whom biopsy samples were available, 63% (17 of 27) of those with HIVAN progressed to ESRD, compared with 46% (19 of 41) of those with non-HIVAN histopathology (HR, 3.1 [95% CI, 1.6–6.2]; P = .001). Compared with white subjects (1 of 31 of whom progressed to ESRD), African American subjects were significantly more likely to progress to ESRD, irrespective of whether they had biopsy samples showing HIVAN (HR, 45 [95% CI, 6–343]; P < .001), had biopsy samples showing non-HIVAN histopathology (HR, 14 [95% CI, 2–109]; P = .009), or were not biopsied (63 of 185 who were not biopsied developed ESRD; HR, 16 [95% CI, 2–117]; P = .006).
Table 4.
Lesions identified in kidney-biopsy samples from HIV-infected subjects with chronic kidney disease, Baltimore, Maryland, 1990 –2004.
| Histopathologic lesion | White (n = 5) | African American (n = 68) |
|---|---|---|
| HIV-associated nephropathy | 0 | 27 (37) |
| Focal segmental glomerulosclerosis | 3 (60) | 16 (24) |
| Diabetic glomerulosclerosis | 0 | 4 (6) |
| Hypertensive nephrosclerosis | 1 (20) | 3 (4) |
| Lupus-like nephritis | 0 | 3 (4) |
| Acute interstitial nephritis | 0 | 3 (4) |
| Glomerulonephritisa | 0 | 4 (6) |
| Amyloidosis | 0 | 1 (1) |
| Other or nondiagnostic | 1 (20) | 7 (10) |
NOTE. Data are no. (%) of subjects.
Includes postinfectious, membranous, membranoproliferative, and immune-complex types (not otherwise specified).
DISCUSSION
In HIV-infected individuals, the risk of ESRD is ~50-fold higher in African Americans than in whites [2]. However, the racial differences in the natural history of CKD that underlie this discrepancy have not been well characterized. We assessed the incidence of CKD, subsequent GFR slope, and progression to ESRD in a large, long-running HIV cohort, composed mostly of African American and white subjects. In unadjusted analysis, African Americans were found to be 1.9-fold more likely to develop CKD than were white subjects; however, when adjusted for sex, age, AIDS status, injection drug use, and calendar time, the HR decreased to 1.65 and was of borderline statistical significance. In contrast, the unadjusted risk for progression to ESRD in subjects with CKD was nearly 20-fold higher in African Americans than in whites, although a caveat worth noting is that the 95% CIs for this point estimate are wide. These data suggest that, although the risk of CKD is slightly higher in HIV-infected African Americans than in whites, the preponderance of the markedly increased risk of ESRD in the former group is explained by a more rapid progression of CKD, once commenced. This view was corroborated both by the higher prevalence of aggressive disease features in African Americans with CKD than in whites with CKD (e.g., 45% vs. 4%, respectively, had high-grade proteinuria) and by the difference in GFR decline, which was >6-fold more rapid in African Americans than in whites.
The pathology of HIV-associated CKD is heterogeneous, with a wide range of pathologic lesions being identified in autopsy [12] and biopsy series [11, 14, 15]. However, a collapsing variant of focal segmental glomerulosclerosis, termed “HIVAN,” occurs nearly exclusively in individuals of African descent [11–13] and has been associated with an aggressive course and rapid progression to ESRD [16, 17]. The present study found that, in the subset of African American subjects with CKD who had kidney biopsy data, the risk of progression to ESRD was 3-fold-higher in those with HIVAN than in those with other histopathologic lesions. It is clear that the racial predilection of HIVAN accounts for a portion of the excess risk of CKD progression that we observed in African American versus white subjects. However, we also found that, in subjects with CKD, the risk of progression to ESRD was significantly higher in African American subjects with non-HIVAN histopathology than in white subjects. These results suggest that a substantial racial difference in CKD progression exists independent of HIVAN. It should be noted that kidney biopsies were performed at the discretion of consulting nephrologists and that the biopsied group may be a biased subset (e.g., more-rapidly progressive disease, greater medical adherence, etc.) of those with CKD.
The present study’s finding that African American race was a substantially stronger risk factor for progression of CKD to ESRD than for development of CKD accords with epidemiological findings for the general US population [18, 19]; for example, Hsu et al., using data from the third National Health and Nutrition Examination Survey and the United States Renal Data System, found that, although the prevalence of CKD was similar in African American subjects and white subjects, the risk of progression from CKD to ESRD was 5-fold higher in the former group [18]. Genetic susceptibility to kidney failure has been hypothesized on the basis of familial clustering of ESRD, in several settings [20–22].
In the present study, increasing age was associated with a significantly higher risk of CKD but with a significantly lower risk of progression to ESRD once CKD had developed. However, the association between age and progression to ESRD may have been confounded by the fact that, at the time of diagnosis of CKD, the African American subjects were younger than the white subjects; when adjusted for race, the association between age and ESRD was no longer statistically significant (data not shown). AIDS status was the strongest risk factor for incident CKD. Immunologic deterioration, as indicated by prior opportunistic condition or a CD4 count <200 cellss/mm3, may potentiate the onset of CKD. Finally, we found that the incidence of CKD declined in successive calendar periods but that the risk of progression to ESRD was similar during the pre-HAART and HAART eras and was not associated with the type of antiretroviral therapy being used at the onset of the disease (table 3). Others have reported that HAART is associated with slowed progression to ESRD in patients with HIVAN but not in those with non-HIVAN histopathology [11, 23].
Although the present study has notable strengths, it also has limitations that should be noted. First, it was conducted in a single urban center, and therefore its results may not necessarily be generalizable to other settings. However, the study’s well-characterized cohort, which includes nearly 19,000 person-years of follow-up in African American and white HIV-infected individuals, offers an exceptional opportunity to evaluate racial differences in the epidemiology of CKD.
Second, although the cohort included ~1000 white subjects, only 1 progressed to ESRD; thus, the CIs for the relative race-specific HRs of progression to ESRD were wide, and adjustment for other covariates was not possible. This finding is consistent with the low rates of CKD and, particularly, ESRD that have been observed in predominantly white-European HIV cohorts [24, 25]. Future research of racial differences in HIV-related CKD in a multicohort consortium would be of interest.
Third, our diagnosis of CKD (stage 3 or higher) was based on GFR estimated, on the basis of serum creatinine concentrations, by the Modification of Diet in Renal Disease equation, which has not been validated with a gold-standard measure of GFR in HIV-infected individuals. However, this equation was derived from a large sample of African American and white subjects with CKD [26] and both the National Kidney Foundation practice guidelines [8] and the HIV Medicine Association of the Infectious Diseases Society of America [27] recommend it as the preferred method of GFR estimation in clinical practice. To add specificity to our diagnosis of CKD, we required (1) that at least 2 GFR measurements be <60 mL/min/1.73 m2 and separated by >3 months and (2) that >50% of all GFR measurements (which followed the index <60 measurement) also be <60 mL/min/1.73 m2; this was done to avoid classification of subjects as having CKD who may have had 2 episodes of acute renal failure but normal renal function otherwise.
The present study has several implications. First, it serves to highlight the importance of CKD and ESRD in HIV-infected African Americans, who are disproportionately affected by the HIV epidemic. In 2005, African Americas constituted just 12% of the general population but accounted for 49% of newly diagnosed HIV infections [28]. Moreover, little is known about the burden of HIV-related CKD in sub-Saharan Africa, where >60% of the world’s HIV-infected individuals reside. As patients live longer because of HAART, it will be important to monitor HIV-related CKD in developed and developing areas of the world.
Second, the high prevalence and incidence of CKD that we observed strongly support the incorporation of screening into routine HIV care, as has been recommended in recently published guidelines [27]. The results of the present study imply that ACE-I/ARB substantially reduces the risk of progression to ESRD in patients with HIV-related CKD. Identification of CKD at an early stage is likely to maximize the benefits of therapy. Because serum creatinine– based GFR estimates are insensitive markers of early kidney disease [29], annual urinalysis or quantitation of the urine protein:creatinine ratio [27] should be viewed as a quality-of-care indicator in HIV treatment centers.
Third, the present study found that the incidence of CKD decreased in sequential calendar periods, coincident with temporal improvements in antiretroviral therapy [30]. These findings suggest that initiation of HAART be considered earlier during HIV disease than is suggested by current treatment guidelines [31]. This point may be particularly applicable to HIV-infected African Americans, who, compared with whites, tend to start therapy later during the course of the disease [32, 33] and are at substantially higher risk of progressing to ESRD. Results from the SMART study, in which CD4 cell– guided treatment interruptions were compared with continuous HAART, indicate (1) that fatal and nonfatal renal-disease outcomes are significantly more common in the treatment-interruption arm than in the continuous-therapy arm [34], and (2) that the risk that study outcomes are associated with treatment interruptions is larger in African Americans than in whites [35].
In summary, the present study found that racial differences in the incidence of CKD in HIV-infected subjects were relatively small, with AIDS status being the strongest risk factor identified as being associated with new onset of CKD; in contrast, once CKD had commenced, its progression was much more rapid in African Americans than in whites, as demonstrated by both a significantly faster decline in GFR and an 18-fold-higher risk of progression to ESRD. The incidence of CKD declined in sequential calendar periods as HIV treatments improved, and ACE-I/ARB was significantly associated with a lower risk of progression to ESRD.
Acknowledgments
National Institutes of Health (grants R01DA11602, R21AA15032 and K23DA15616 to R.D.M., and K24DA00432 to G.M.L.). The funding source had no role in the design, analysis, or interpretation of data for this study.
Footnotes
Potential conflicts of interest: none reported.
Presented in part: Conference on Retroviruses and Opportunistic Infections, Boston, Massachusetts, 3–6 February 2008 (abstract 972).
References
- 1.Schwartz EJ, Szczech LA, Ross MJ, Klotman ME, Winston JA, Klotman PE. Highly active antiretroviral therapy and the epidemic of HIV+ end-stage renal disease. J Am Soc Nephrol. 2005;16:2412–20. doi: 10.1681/ASN.2005040340. [DOI] [PubMed] [Google Scholar]
- 2.Eggers PW, Kimmel PL. Is there an epidemic of HIV Infection in the US ESRD program? J Am Soc Nephrol. 2004;15:2477–85. doi: 10.1097/01.ASN.0000138546.53152.A7. [DOI] [PubMed] [Google Scholar]
- 3.Krawczyk CS, Holmberg SD, Moorman AC, Gardner LI, McGwin G., Jr Factors associated with chronic renal failure in HIV-infected ambulatory patients. AIDS. 2004;18:2171–8. doi: 10.1097/00002030-200411050-00009. [DOI] [PubMed] [Google Scholar]
- 4.Gardner LI, Holmberg SD, Williamson JM, et al. Development of proteinuria or elevated serum creatinine and mortality in HIV-infected women. J Acquir Immune Defic Syndr. 2003;32:203–9. doi: 10.1097/00126334-200302010-00013. [DOI] [PubMed] [Google Scholar]
- 5.Szczech LA, Gange SJ, Van Der Horst C, et al. Predictors of proteinuria and renal failure among women with HIV infection. Kidney Int. 2002;61:195–202. doi: 10.1046/j.1523-1755.2002.00094.x. [DOI] [PubMed] [Google Scholar]
- 6.Gupta SK, Mamlin BW, Johnson CS, Dollins MD, Topf JM, Dube MP. Prevalence of proteinuria and the development of chronic kidney disease in HIV-infected patients. Clin Nephrol. 2004;61:1–6. doi: 10.5414/cnp61001. [DOI] [PubMed] [Google Scholar]
- 7.Moore RD. Understanding the clinical and economic outcomes of HIV therapy: the Johns Hopkins HIV clinical practice cohort. J Acquir Immune Defic Syndr Hum Retrovirol. 1998;17(suppl 1):S38–41. doi: 10.1097/00042560-199801001-00011. [DOI] [PubMed] [Google Scholar]
- 8.Levey AS, Coresh J, Balk E, et al. National Kidney Foundation practice guidelines for chronic kidney disease: evaluation, classification, and stratification. Ann Intern Med. 2003;139:137–47. doi: 10.7326/0003-4819-139-2-200307150-00013. [DOI] [PubMed] [Google Scholar]
- 9.Centers for Disease Control and Prevention. 1993 Revised classification system for HIV infection and expanded surveillance case definition for AIDS among adolescents and adults. MMWR. 1992;41(RR17):1–19. [PubMed] [Google Scholar]
- 10.Schoenfeld D. Partial residuals for the proportional hazards regression model. Biometrika. 1982;69:239–41. [Google Scholar]
- 11.Szczech LA, Gupta SK, Habash R, et al. The clinical epidemiology and course of the spectrum of renal diseases associated with HIV infection. Kidney Int. 2004;66:1145–52. doi: 10.1111/j.1523-1755.2004.00865.x. [DOI] [PubMed] [Google Scholar]
- 12.Shahinian V, Rajaraman S, Borucki M, Grady J, Hollander WM, Ahuja TS. Prevalence of HIV-associated nephropathy in autopsies of HIV-infected patients. Am J Kidney Dis. 2000;35:884–8. doi: 10.1016/s0272-6386(00)70259-9. [DOI] [PubMed] [Google Scholar]
- 13.D’Agati V, Suh JI, Carbone L, Cheng JT, Appel G. Pathology of HIV-associated nephropathy: a detailed morphologic and comparative study. Kidney Int. 1989;35:1358–70. doi: 10.1038/ki.1989.135. [DOI] [PubMed] [Google Scholar]
- 14.Gerntholtz TE, Goetsch SJ, Katz I. HIV-related nephropathy: a South African perspective. Kidney Int. 2006;69:1885–91. doi: 10.1038/sj.ki.5000351. [DOI] [PubMed] [Google Scholar]
- 15.Estrella M, Fine DM, Gallant JE, et al. HIV type 1 RNA level as a clinical indicator of renal pathology in HIV-infected patients. Clin Infect Dis. 2006;43:377–80. doi: 10.1086/505497. [DOI] [PubMed] [Google Scholar]
- 16.Rao TK, Friedman EA, Nicastri AD. The types of renal disease in the acquired immunodeficiency syndrome. N Engl J Med. 1987;316:1062–8. doi: 10.1056/NEJM198704233161705. [DOI] [PubMed] [Google Scholar]
- 17.Laradi A, Mallet A, Beaufils H, Allouache M, Martinez F. HIV-associated nephropathy: outcome and prognosis factors. Groupe d’ Etudes Nephrologiques d’Ile de France. J Am Soc Nephrol. 1998;9:2327–35. doi: 10.1681/ASN.V9122327. [DOI] [PubMed] [Google Scholar]
- 18.Hsu CY, Lin F, Vittinghoff E, Shlipak MG. Racial differences in the progression from chronic renal insufficiency to end-stage renal disease in the United States. J Am Soc Nephrol. 2003;14:2902–7. doi: 10.1097/01.asn.0000091586.46532.b4. [DOI] [PubMed] [Google Scholar]
- 19.McClellan W, Warnock DG, McClure L, et al. Racial differences in the prevalence of chronic kidney disease among participants in the Reasons for Geographic and Racial Differences in Stroke (REGARDS) Cohort Study. J Am Soc Nephrol. 2006;17:1710–5. doi: 10.1681/ASN.2005111200. [DOI] [PubMed] [Google Scholar]
- 20.Freedman BI, Satko SG. Genes and renal disease. Curr Opin Nephrol Hypertens. 2000;9:273–7. doi: 10.1097/00041552-200005000-00011. [DOI] [PubMed] [Google Scholar]
- 21.Schelling JR, Zarif L, Sehgal A, Iyengar S, Sedor JR. Genetic susceptibility to end-stage renal disease. Curr Opin Nephrol Hypertens. 1999;8:465–72. doi: 10.1097/00041552-199907000-00011. [DOI] [PubMed] [Google Scholar]
- 22.Freedman BI, Soucie JM, Stone SM, Pegram S. Familial clustering of end-stage renal disease in blacks with HIV-associated nephropathy. Am J Kidney Dis. 1999;34:254–8. doi: 10.1016/s0272-6386(99)70352-5. [DOI] [PubMed] [Google Scholar]
- 23.Atta MG, Gallant JE, Rahman MH, et al. Antiretroviral therapy in the treatment of HIV-associated nephropathy. Nephrol Dial Transplant. 2006;21:2809–13. doi: 10.1093/ndt/gfl337. [DOI] [PubMed] [Google Scholar]
- 24.Viertel A, Weidmann E, Rickerts V, Scheuermann EH, Geiger H, Brodt H. Renal involvement in HIV-infection: results from the Frankfurt AIDS Cohort Study (FACS) and a review of the literature. Eur J Med Res. 2000;5:185–98. [PubMed] [Google Scholar]
- 25.Mocroft A, Kirk O, Gatell J, et al. Chronic renal failure among HIV-1-infected patients. AIDS. 2007;21:1119–27. doi: 10.1097/QAD.0b013e3280f774ee. [DOI] [PubMed] [Google Scholar]
- 26.Levey AS, Bosch JP, Lewis JB, Greene T, Rogers N, Roth D. A more accurate method to estimate glomerular filtration rate from serum creatinine: a new prediction equation. Modification of Diet in Renal Disease Study Group. Ann Intern Med. 1999;130:461–70. doi: 10.7326/0003-4819-130-6-199903160-00002. [DOI] [PubMed] [Google Scholar]
- 27.Gupta SK, Eustace JA, Winston JA, et al. Guidelines for the management of chronic kidney disease in HIV-infected patients: recommendations of the HIV Medicine Association of the Infectious Diseases Society of America. Clin Infect Dis. 2005;40:1559–85. doi: 10.1086/430257. [DOI] [PubMed] [Google Scholar]
- 28.Centers for Disease Control and Prevention. Vol. 17. Atlanta, GA: Centers for Disease Control and Prevention, US Department of Health and Human Services; 2006. [Accessed 15 August 2006]. HIV/AIDS Surveillance Report, 2005. Available at http://www.cdc.gov/hiv/topics/surveillance/resources/reports. [Google Scholar]
- 29.Stevens LA, Coresh J, Greene T, Levey AS. Assessing kidney function–measured and estimated glomerular filtration rate. N Engl J Med. 2006;354:2473–83. doi: 10.1056/NEJMra054415. [DOI] [PubMed] [Google Scholar]
- 30.Moore RD, Keruly JC, Gebo KA, Lucas GM. An improvement in virologic response to highly active antiretroviral therapy in clinical practice from 1996 through 2002. J Acquir Immune Defic Syndr. 2005;39:195–8. [PubMed] [Google Scholar]
- 31.Panel on Clinical Practices for the Treatment of HIV Infection. US Department of Health and Human Services: 2006. [Accessed 15 July 2007]. Guidelines for the use of antiretroviral agents in HIV-1-infected adults and adolescents. Available at http://www.AIDSinfo.nih.gov. [Google Scholar]
- 32.Shapiro MF, Morton SC, McCaffrey DF, et al. Variations in the care of HIV-infected adults in the United States: results from the HIV Cost and Services Utilization Study. JAMA. 1999;281:2305–15. doi: 10.1001/jama.281.24.2305. [DOI] [PubMed] [Google Scholar]
- 33.Gebo KA, Fleishman JA, Conviser R, et al. Racial and gender disparities in receipt of highly active antiretroviral therapy persist in a multistate sample of HIV patients in 2001. J Acquir Immune Defic Syndr. 2005;38:96–103. doi: 10.1097/00126334-200501010-00017. [DOI] [PubMed] [Google Scholar]
- 34.El Sadr WM, Lundgren JD, Neaton JD, et al. CD4+ count-guided interruption of antiretroviral treatment. N Engl J Med. 2006;355:2283–96. doi: 10.1056/NEJMoa062360. [DOI] [PubMed] [Google Scholar]
- 35.El Sadr W for the SMART Study Group. Inferior clinical outcomes with episodic CD4-guided antiretroviral therapy aimed at drug conservation (DC) in SMART study: consistency of finding in all patient subgroups [abstract WEAB0204]. Program and abstracts of the XVI International AIDS Conference; Toronto, Canada. Available at: http://www.iasociety.org./Default.aspx?pageId=11&abstractId=2195953. [Google Scholar]