Abstract
The influence of protein phosphorylation on the kinetics of cytochrome c oxidase was investigated by applying Western blotting, mass spectrometry, and kinetic measurements with an oxygen electrode. The isolated enzyme from bovine heart exhibited serine, threonine, and/or tyrosine phosphorylation in various subunits, except subunit I, by using phosphoamino acid-specific antibodies. The kinetics revealed slight inhibition of oxygen uptake in the presence of ATP, as compared with the presence of ADP. Mass spectrometry identified the phosphorylation of Ser-34 at subunit IV and Ser-4 and Thr-35 at subunit Va. Incubation of the isolated enzyme with protein kinase A, cAMP, and ATP resulted in serine and threonine phosphorylation of subunit I, which was correlated with sigmoidal inhibition kinetics in the presence of ATP. This allosteric ATP-inhibition of cytochrome c oxidase was also found in rat heart mitochondria, which had been rapidly prepared in the presence of protein phosphatase inhibitors. The isolated rat heart enzyme, prepared from the mitochondria by blue native gel electrophoresis, showed serine, threonine, and tyrosine phosphorylation of subunit I. It is concluded that the allosteric ATP-inhibition of cytochrome c oxidase, previously suggested to keep the mitochondrial membrane potential and thus the reactive oxygen species production in cells at low levels, occurs in living cells and is based on phosphorylation of cytochrome c oxidase subunit I.
Phosphorylation of mitochondrial proteins has become of general interest since the role of mitochondria in apoptosis and degenerative diseases became evident. During the past ten years many protein kinases and phosphatases, mostly known to occur outside of mitochondria, have also been identified in mitochondria or are translocated to mitochondria after activation (1–6). In addition, an increasing number of phosphorylated proteins, including subunits of complexes I-V of the mitochondrial oxidative phosphorylation system, have been identified (7–9). Of particular interest is the phosphorylation of cytochrome c oxidase (CcO)1, the terminal, and rate-limiting enzyme of the respiratory chain (complex IV) (10). CcO is composed of three mitochondrial DNA-encoded subunits, forming the catalytic core of the enzyme, and ten nuclear-encoded subunits with regulatory functions. The crystal structure of the bovine heart enzyme forms a dimer (11, 12), and supercomplexes of CcO with complex III (cytochrome c reductase) and complex I (NADH dehydrogenase) have been identified in mitochondrial membranes (13–15). The complicated structure of the mammalian enzyme contrasts the bacterial CcO containing only 2–4 subunits (16, 17). The additional subunits in eukaryotes are suggested to regulate CcO activity, either by binding effectors or by chemical modification, like glycosylation and phosphorylation. Ten high-affinity binding sites for ADP have been identified in the isolated bovine heart enzyme, seven of which are exchanged by ATP at high ATP/ADP ratios (18, 19). Exchange of bound ADP by ATP at subunit VIa-H (heart type) was shown to decrease the H+/e−-stoichiometry of reconstituted CcO from bovine heart (20). Exchange of bound ADP by ATP at subunit IV induces the allosteric ATP-inhibition (21), which is prevented by 3,5-diiodothyronine, after its binding to subunit Va (22). At high ATP/ADP ratios the allosteric ATP-inhibition results in sigmoidal inhibition curves, when oxygen consumption is measured at increasing cytochrome c concentrations. This feedback inhibition of CcO was suggested to keep the membrane potential ΔΨm and ROS production of mitochondria at low levels (16, 23, 24), based on the dependence of ROS production on ΔΨm (25) and saturation of ATPase activity at low ΔΨm (<120 mV) (26). Mitochondrial respiration, and thus CcO activity, is also inhibited at high ATP/ADP ratios through high ΔΨm values, known as “respiratory control” (27, 28). The allosteric ATP-inhibition represents a second mechanism of respiratory control (29), which is independent of ΔΨm (30). The allosteric ATP-inhibition, however, is lost when the enzyme is dephosphorylated (31, 32), possibly at Ser-441 of bovine heart CcO subunit I (23).
Phosphorylation of CcO was first demonstrated by Steenaart and Shore (33) at subunit IV by in vitro incubation of mitochondrial membranes with [γ-32P]ATP. Incubation of isolated bovine heart CcO with PKA (protein kinase A), cAMP, and [γ-32P]ATP resulted in labeling of subunits I, II, and Vb (31). After activation of PKCɛ in cardiac myocytes with phorbol ester, in vitro phosphorylation of a membrane fraction with [γ-32P]ATP revealed phosphorylation of CcO subunit IV (34). In later studies, binding of PKCɛ to CcO subunit IV is demonstrated accompanied by enhanced CcO activity (35). Hüttemann and coworkers (36) described phosphorylation of CcO subunit I at Tyr-304. The phosphorylation was obtained in liver cells or tissues after activation with glucagon or forskolin via the cAMP/PKA signal pathway and was accompanied by enhanced allosteric inhibition kinetics of the enzyme. The conversion of the cAMP signal, normally leading to phosphorylation of serine/threonine residues into activation of a tyrosine kinase, was assumed to involve the protein kinase A anchor protein AKAP121 (37). Avadhani and coworkers demonstrated phosphorylation of CcO subunits I, IV, and Vb after ischemia of Langendorff perfused rabbit hearts (38). This phosphorylation was prevented by the presence of the PKA inhibitor H89 in the perfusion medium. In a subsequent study the group identified the phosphorylation sites in rabbit heart CcO at Ser-115 and Ser-116 of subunit I, Thr-52 of subunit IV, and Ser-40 of subunit Vb (39). All of these results contrast previous analysis of isolated bovine heart CcO by matrix-assisted laser desorption/ionization mass spectrometry (MALDI-MS), where for all subunits only masses corresponding to the determined amino acid sequences are obtained (40). The authors concluded that the 13 subunits of CcO are not post-translationally modified by phosphorylation or glycosylation.
In the present study we compared the kinetics of CcO from bovine heart, with its phosphorylation pattern using antibodies against phosphoserine, phosphothreonine, and phosphotyrosine. The enzyme was purified from mitochondria, which had been isolated in the presence of 25 mm NaF, an unspecific inhibitor of protein phosphatases. In the isolated bovine heart enzyme, three phosphorylated amino acids have been identified by mass spectrometry, i.e. Ser-34 of subunit IV and Ser-4 and Thr-35 of subunit Va. Incubation of isolated CcO with PKA, cAMP, and ATP resulted in phosphorylation of CcO subunit I at serine and threonine, accompanied by induction of allosteric ATP-inhibition. Rapid isolation of mitochondria from rat heart in the presence of various protein phosphatase inhibitors resulted in CcO kinetics with allosteric ATP-inhibition and phosphorylation of subunit I at serine, threonine, and tyrosine. The results indicate that the allosteric ATP-inhibition of CcO is present under in vivo conditions, but is immediately lost during stress, such as death of an animal.
EXPERIMENTAL PROCEDURES
Isolation of Mitochondria—
Mitochondria were isolated either from fresh or frozen bovine hearts. The hearts were obtained from the slaughterhouse about two hours after animal death, transported on ice, and either directly used for preparation of mitochondria or frozen at −80 °C. Frozen hearts were thawed overnight on ice, cut into cubes, minced in a meat grinder, and mixed in a commercial blender for 3 × 10 s at maximal speed in 3 volumes of a medium containing 250 mm sucrose, 20 mm Hepes, pH 7.4, 2 mm EGTA, which was supplemented with 25 mm NaF, 1–5 mm sodium vanadate, and 10 nm okadaic acid, when indicated. The pH was readjusted during homogenization with 2 m Tris. The homogenate was centrifuged for 10 min at 650 × g, and from the supernatant mitochondria were centrifuged down for 15 min at 16,000 × g. Mitochondria were washed once in the same medium.
Isolation of Bovine Heart CcO—
CcO was isolated as described previously (41). In short, mitochondria were extracted with Triton X-114 and ultracentrifuged. The CcO pellet was dissolved in Triton X-100 followed by DEAE-Sephadex chromatography and ammonium sulfate precipitation in the presence of 1% sodium cholate.
Treatment of Isolated CcO—
To remove bound cholate and to supplement the enzyme with cardiolipin, CcO from bovine heart was dialyzed overnight against 50 mm potassium phosphate, pH 7.4, 1% Tween 20, 1 mm EDTA, 0.5 mm ATP. The sample contained 5 μm CcO and 1% cardiolipin. 2.5 μm of dialyzed enzyme was incubated for 40 min at 30 °C with 200 units/ml PKA, 50 μm cAMP, 5 mm ATP, 10 mm ascorbate, 0.2 μm cytochrome c in 50 mm potassium phosphate, pH 7.4, 5 mm MgSO4, 1% Tween 20.
Isolation of CcO by Blue Native Gel Electrophoresis—
The procedure of Schägger and von Jagow was applied (42, 43). Mitochondria from rat heart (10 mg/ml) were homogenized in 50 mm NaCl, 50 mm Bistris, 2 mm 6-aminocaproic acid, 1 mm EDTA, pH 7.0, and dodecylmaltoside was added at a concentration of 2%. After 10 min the solution was centrifuged at 20000 × g for 20 min. To the supernatant 5% glycerol and 0.25% Coomassie Blue R-250 (Sigma-Aldrich) were added, and 11 μl of the mixture were loaded per sample well to an acrylamide gel in a small gel electrophoresis apparatus (10 × 10 × 0.1 cm). A gradient separation gel was applied from 6 to 13% polyacrylamide (48-g acrylamide and 1.5 g bisacrylamide/100 ml). After 2 h electrophoresis at 200 V complex IV of the respiratory chain was excised from the gel, incubated in 1% SDS for 15 min, and put at the position of stacking gel on glass plates for the second dimension by SDS-PAGE (44). After positioning the spacers and covering the second glass plates the acrylamide solution was poured through the gaps, and after polymerization the stacking gel was added.
Western Blotting—
The purified enzyme or the CcO complex from blue native gel electrophoresis was separated by SDS-PAGE as described previously (44) in a small gel electrophoresis apparatus, and part of the gels was stained with Coomassie Brilliant Blue R-250. The other part was blotted onto polyvinylidene difluoride membranes by “semi-dry-blotting” at room temperature (45), and phosphoproteins were detected with monoclonal antibodies against phosphoserine (Sampler kit, 1:1000; Biomol, Hamburg, Germany), phosphothreonine (Sampler kit, 1:1000; Biomol) and phosphotyrosine (clone 4G10; Upstate). The polyvinylidene difluoride membranes were blocked with 1:10 dilution of Roti Block buffer (Carl Roth GmbH & Co. KG, Karlsruhe, Germany) at room temperature for 90 min under shaking. Antibodies were reacted for 1 h at room temperature and washed 4× with phosphate-buffered saline/0.1% Tween 20, followed by reaction for 60 min with a 1:10000 dilution of horseradish peroxidase-conjugated goat anti-mouse IgG (Invitrogen). The bound monoclonal antibodies were detected using a Pierce® ECL Western blotting substrate (Thermo Scientific) and visualized on x-ray film. Stripping off of the antibodies was performed by heating the blots for 30 min at 60 °C under shaking with 2% SDS, 62.5 mm Tris-HCl, pH 6.8, 100 mm mercaptoethanol, followed by 4× washing with phosphate-buffered saline/0.1% Tween 20. After blocking, as described above, the blots were reacted with monoclonal antibodies against CcO subunits I (1:10000) and subunit IV (1:30000) from Molecular Probes (Göttingen).
Measurement of CcO Activity—
The kinetics of CcO activity were measured polarographically at 25 °C using an Oxygraph System (Hansatech, Norfolk, UK) in a volume of 0.5 ml. Mitochondria, or the isolated enzyme, were dissolved in 50 mm potassium phosphate, pH 7.4, 2 mm EGTA, 5 mm MgSO4, and 1% Tween 20. Oxygen consumption was measured with increasing concentrations of cytochrome c (0.2–100 μm) in the presence of 15 mm ascorbate and either 5 mm ADP or 5 mm ATP and a regenerating system consisting of 10 mm phosphoenolpyruvate and 200 units/ml pyruvate kinase.
Enzymatic Digestion of the CcO Complex—
Aliquots of 100 μg DEAE-Sepharose-enriched CcO samples were dried in a vacuum centrifuge and solubilized in 40 μl of guanidinium hydrochloride (GuHCl) digestion buffer. For digests with sequencing grade-modified trypsin (Promega Corporation, Madison, WI), a buffer consisting of 5 m GuHCl, 50 mm ammonium bicarbonate (pH 8.3) was used and 5 m GuHCl, 100 mm Tris, 10 mm CaCl2 (pH 7.8) in the case of digests with sequencing grade-modified chymotrypsin (Roche, Penzberg, Germany). Cysteines were reduced for 10 min at 70 °C with 5 μl of 200 mm dithiothreitol solution and alkylated for 1 h at 45 °C with 4 μl of 1 m iodoacetamide according to Immler et al. (46). To scavenge remaining alkylation reagent, further 5 μl of the reducing reagent was added followed by 1 h incubation at 45 °C. The samples were diluted to bring the GuHCl concentration to 1 m and digested with 1/20 of the quantity of CcO protein by weight with chymotrypsin or trypsin (both diluted in digestion buffer). Digestion was allowed to take place for 16 h at 37 °C.
TiO2 Enrichment of Phosphorylated Peptides—
The identification of phosphorylated peptides required their enrichment with TiO2 material in self-made columns. The column preparation and slightly differing sample treatments are described earlier (47, 48). The enzymatic digests of the CcO samples were diluted with a 5-fold volume of TiO2-loading solution (10% lactic acid, 5% trifluoroacetic acid, 80% acetonitril (ACN)) and loaded on a 2–200 μl of Tip (Eppendorf, Hamburg, Germany) equipped with a C8 membrane plug (3 m Empore™ C8 extraction disc; IVA Analysentechnik, Meerbusch, Germany) packed ∼2 mm high with TiO2 material (5 μm, Titansphere; GL Sciences, Torrance, CA). The flow-through was collected and used for the analysis of non-phosphorylated peptides. The TiO2-bound peptides were washed with 10 μl of TiO2 loading solution, 20 μl of washing solution (1% trifluoroacetic acid, 80% ACN), and 5 μl of H2O. Peptides were eluted with 20 μl of elution buffer (125 mm ammonium bicarbonate, 50 mm ammonium phosphate, pH 10.5) according to Mazanek et al. (48). C8 membrane plug remaining peptides were eluted with 5 μl of 30% CAN, and the collected eluate was immediately acidified with 2.5-μl formic acid (FA, 100%) and dried in a vacuum centrifuge.
To optimize the conditions for mass spectrometry (MS) the peptide samples were desalted by an additional purification step: TiO2-enriched phosphorylated peptides were desalted using reverse phase poros medium R3 (PerSeptive Biosystems, Framingham, MA) packed ∼2 mm high in Microloader tips (Eppendorf) equipped with C8 membrane plugs. Washing was performed with 20-μl ACN (100%), and equilibration was done with 20 μl of 5% FA. Peptides were solubilized in 20 μl of 5% FA, bound to the column material, washed two times with 20 μl of 5% FA, and eluted with 20 μl of reverse phase elution buffer (5% FA, 80% ACN). As the volume of the flow-through peptide sample was too high to use, those tips desalting was done in an additional step using 1-ml Chromabond C18 cartridges (Machery-Nagel, Düren, Germany). Prior to sample loading cartridges were washed with 1-ml ACN (100%) and equilibrated with 1 ml of 5% FA. Afterward, peptides were bound to the column material, washed two times with 1 ml of 5% FA and eluted with 0.5 ml of reverse phase elution buffer. Finally all samples were dried once more to remove ACN, solubilized in 15 μl of 5% FA, and analyzed by nanoLC-ESI-MS/MS.
NanoLC-ESI-MS/MS Analysis—
The nano liquid chromatography-electrospray ionization-MS/MS analyses were done using the Ultimate™ 3000 HPLC system (Dionex LC Packings, Idstein, Germany) online coupled to the HCTultra PTM Discovery System™ ion trap mass spectrometer (Bruker Daltonics, Bremen, Germany). Peptides were first separated by reverse phase nanoLC on a 75-μm inner diameter × 150 mm C18 PepMap column (Dionex LC Packings) with pre-concentration for 10 min with 0.1% trifluoroacetic acid on a μ-precolumn (300-μm inner diameter × 1 mm, C18 PepMap, Dionex LC Packings). For peptide separation a solvent system consisting of 0.1% (v/v) FA (solvent A) and 0.1% (v/v) FA, 84% (v/v) ACN (solvent B) was used. The gradient was 5–30% solvent B in 89 min, 30–95% solvent B in 5 min, held at 95% solvent B for 5 min before the column was conditioned for the next run for 15 min with 5% solvent B.
The ion trap mass spectrometer was equipped with a nano-electrospray ion source and distal-coated SilicaTips™ (FS360–20-10-D-20; New Objective Inc., Woburn, MA). The peptides were analyzed in the positive mode after ionization with 1.0–1.4 kV source voltage. The three most abundant preferentially more than doubly charged precursor ions of a MS scan were selected by the MS control software (Esquiere Control V. 6.1; Bruker Daltonics) for CID-MS/MS (collision induced dissociation-MS/MS) experiments. TiO2-enriched phosphorylated peptides were detected with neutral loss scans including the automatic detection of fragment ions explaining CID-MS/MS-generated dissociation of phosphoric acid with losses of m/z 49.0 (doubly charged ions) and m/z 32.7 (triply charged ions). The precursor masses of these ions were selected for ETD (electron transfer dissociation) fragmentation experiments, and the masses explaining the neutral losses were additionally fragmented with CID. The peptides of the flow-through (not trapped with TiO2) were analyzed with the same liquid chromatography parameters but only peptides with a charge-state >2 were selected for CID- as well as ETD-MS/MS experiments.
Data Analysis—
For identification of the CcO subunits and their phosphorylation sites the MS output files (mgf format) of the TiO2 enrichment and flow-through were generated with the Data Analysis software V. 3.4 (Bruker Daltonics) and merged separately for CID-MS/MS as well as ETD-MS/MS spectra. These combined data sets were compared with the mammalian protein sub-database of NCBI nr decoy (2007.11.09; 6340306 sequences in major database) using the Mascot® V. 2.2 search algorithm. The following search parameters were selected: peptide mass accuracy of 0.6 Da (mono-isotopic), fragment mass accuracy of 0.2 Da (mono-isotopic), variable modification because of oxidation of methionine, carbamidomethylation of cysteine, and phosphorylation of serine, threonine, and tyrosine, maximal two missed cleavage sites as well as three missed cleavage sites for a further search. For positive protein identification a minimum of two unique peptides, adequately explained by additional manual interpretation of the respective fragment ion spectra and by theoretical fragmentation with the MS Product software tool were required, which also equated to at least 5% sequence coverage of the respective protein.
RESULTS
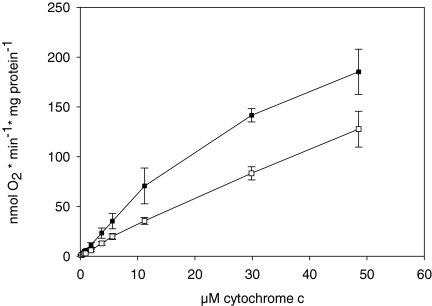
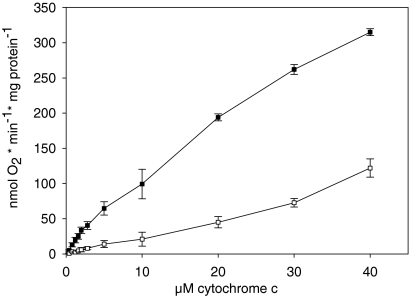
The kinetics of CcO in isolated mitochondria from fresh bovine heart tissue, measured polarographically at increasing cytochrome c concentrations in the presence of 5 mm ADP or 5 mm ATP, are presented in Fig. 1. To prevent dephosphorylation of CcO during preparation of mitochondria, the isolation medium contained 25 mm NaF, an unspecific inhibitor of protein phosphatases, 1 mm vanadate, an inhibitor of phosphoprotein tyrosine phosphatases, and 2 mm EGTA, which complexed calcium, an activator of some protein phosphatases. No allosteric ATP-inhibition but hyperbolic saturation kinetics was obtained in both cases, although with decreased activity in the presence of ATP.
Fig. 1.
Kinetics of CcO in isolated bovine heart mitochondria. Mitochondria were isolated in the presence of 25 mm NaF, 1 mm vanadate, and 2 mm EGTA. Oxygen consumption was measured at increasing concentrations of cytochrome c in the presence of 5 mm ADP (closed squares) and 5 mm ATP and an ATP regenerating system (open squares). The average of three determinations is shown.
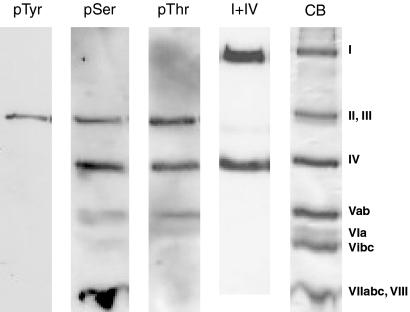
From the mitochondria, used in Fig. 1 for activity measurements, CcO was isolated by our standard procedure including Triton X-114/X-100 extraction, DEAE-Sepharose chromatography and ammonium sulfate precipitation in the presence of 1% cholate (41). With the purified enzyme Western blots were performed with phosphoamino acid-specific antibodies, as shown in Fig. 2. The position of CcO subunits is presented in the Coomassie Blue-stained part of the gel, presented at the right side of the figure (CB). After stripping off the antibodies, further staining of the blots with antibodies against CcO subunits I and IV was performed to identify the subunit positions on the blots. Phosphorylation is seen in subunit II/III at tyrosine, serine, and threonine. Further phosphorylation at serine and threonine is detectable in subunits IV and Vab. Threonine phosphorylation is also found at subunit VIa and serine phosphorylation at subunits VIbc. A strong serine phosphorylation is visualized at subunits VIIabc/VIII. No phosphorylation was found at subunit I.
Fig. 2.
Western blots and Coomassie Blue staining (CB) of CcO from bovine heart, purified by standard procedures from mitochondria isolated in the presence of 25 mm NaF, 1 mm sodium vanadate, and 2 mm EGTA. Mitochondria were isolated immediately after obtaining the heart from the slaugtherhouse. Antibodies against phosphotyrosine (pTyr), phosphoserine (pSer), phosphothreonine (pThr), and CcO subunits I and IV were applied, as indicated.
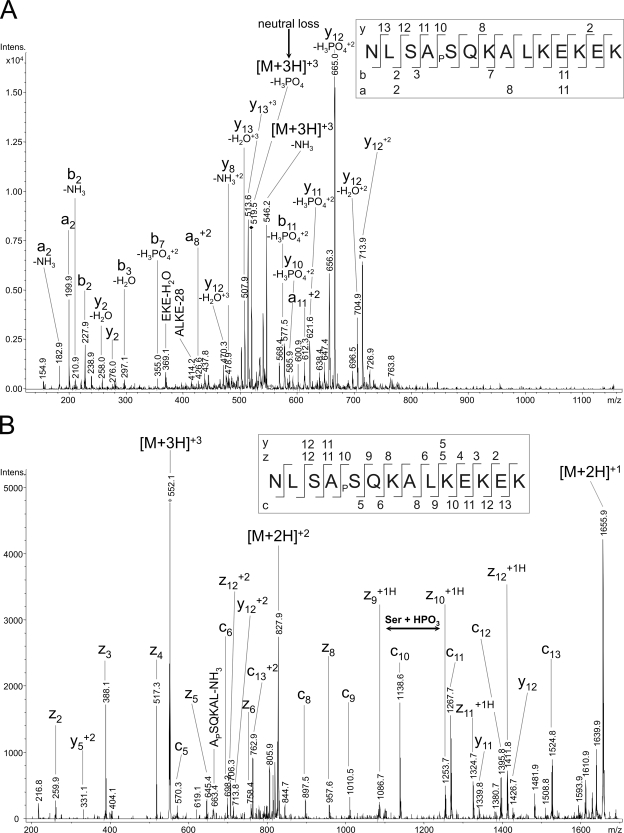
To confirm protein phosphorylation, the purified enzyme was analyzed by nanoLC-ESI-MS/MS. We were able to identify one phosphorylated serine residue (Ser-34) in subunit IV as well as a serine and a threonine phosphorylation site of subunit Va (Ser-4, Thr-35). The proteins were identified with a sequence coverage of 61% (subunit IV, gi 1942989) and 72% (subunit Va, gi 1942990). Fig. 3 shows both spectra (A, CID; B, ETD) unequivocally identifying phosphorylated Ser-34 in subunit IV after protease cleavage with trypsin and TiO2 enrichment of phosphopeptides. Corresponding peptides were identified as doubly and triply charged ions with two and three missed cleavage sites and Mascot scores for the processed ETD spectra of up to 74. The analyzed peptide ion with the sequence NLSAPSQKALKEKEK includes two possible serine phosphorylation sites. For localization of the correct phosphorylation site we were able to generate fragment ion spectra by CID as well as ETD. The obtained fragment ion spectrum of m/z 555.2 of the triply charged precursor ion explains the neutral loss of phosphoric acid from phosphoserine (Fig. 3A), which is typical for CID fragmentation experiments. Fragment ions can be unequivocally assigned indicating the phosphorylation at Ser-34 by the ions y10+2 and y11+2 both with loss of phosphoric acid. Furthermore, the b3 ion excludes a phosphorylation of Ser-32. The subsequent analysis by ETD complemented the CID data with the z11 and z12 ions as well as the y11 and y12 ions also excluding phosphorylation of Ser-32 (Fig. 3B). The difference between the C-terminal z9 + 1H and z10 + 1H ions (both singly charged with additional hydrogen) clearly explains a phosphorylated Ser-34.
Fig. 3.
NanoLC-ESI-MS/MS analysis of the CcO subunit IV. The MS/MS spectra (A, CID and B, ETD) was unambiguously assigned to the peptide NLSAPSQKALKEKE. Localization of Ser-34 as site of phosphorylation was possible after fragmentation of the triply charged precursor ion m/z 552.2. A, the CID-MS/MS spectrum contains a CID-typical neutral loss of phosphoric acid from the phosphoserine of the precursor ion. The neutral loss ions y10 and y12 as well as the b3 ion indicates a phosphorylated Ser-34. B, both, z9 + 1H and z10 + 1H (both single charged with additional hydrogen) in the ETD-MS/MS experiment additionally confirm the findings of CID fragmentation.
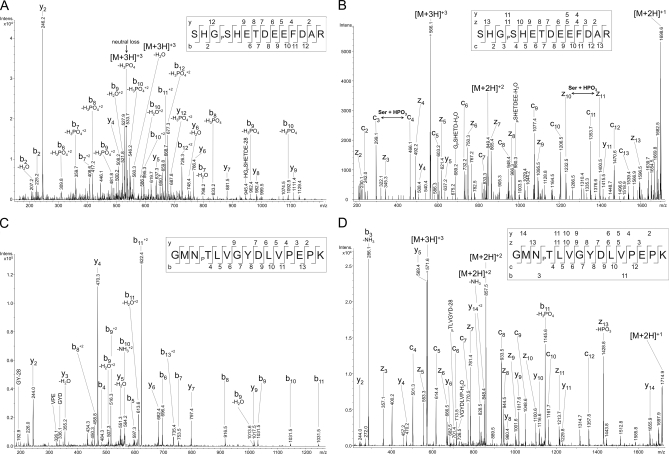
Two further phosphorylated amino acids were identified in subunit Va. These are Ser-4 identified in TiO2-enriched samples and Thr-35 identified in the TiO2 flow-through, as presented in Fig. 4. Phosphorylation at Ser-4 was observed after enzymatic cleavage with chymotrypsin and analysis of the doubly charged precursor ion (see supplemented data, Fig. 1) as well as after cleavage with trypsin for doubly and triply charged precursor ions; a spectrum of the latter is presented in Fig. 4, A and B. This peptide ion of the sequence SHGPSHETDEEFDAR contains several possible phosphorylation sites, and the combination of CID and ETD again allows an unambiguous localization of the present phosphorylation site. The CID fragment ion spectrum of the triply charged precursor ion with m/z 566.3 shows a neutral loss of phosphoric acid from the phosphoserine of the precursor ion providing a conclusive characterization of the C-terminal part (from His-5). The fragmentation pattern excludes a phosphorylation at Thr-7 (Fig. 4A). Discrimination between Ser-1 and Ser-4 phosphorylation is easily possible on the basis of the measured ETD spectrum (Fig. 4B, Mascot score 109). It contains a nearly complete clarification of the peptide amino acid sequence including the Ser-4 phosphorylation by the ions c3 and c4 as well as z10, z11, and y11. Thr-35 phosphorylation is identified in spectra of the triply charged peptide ion GMNPTLVGYDLVPEPK (m/z 571.6) containing an additional possible phosphorylation site at Tyr-39 (Fig. 4, C and D). In contrast to the previously described CID spectra no neutral loss of phosphoric acid is detected for CID fragments of this peptide ion but two abundant ions the y4 ion and the corresponding b11+2 ion explain a breakage at the labile peptide bond at Pro-43 (Fig. 4C). The tyrosine phosphorylation is excluded by the generation of b7 and b8 ions and additionally by the formation of y7 and y9. Furthermore, the versatile ETD fragment ions give a good explanation of the amino acid sequence including c7 and c8 ions and the corresponding z7 and z8 ions explaining the state of the tyrosine and the c4, z13, and y14 ions proving the phosphorylation of Thr-35 (Fig. 4D, Mascot score 72).
Fig. 4.
NanoLC-ESI-MS/MS analysis of CcO subunit Va. Phosphoserine-4 in CcO subunit Va was unequivocally localized by a combination of CID (A) and ETD (B) of the triply charged precursor ion SHGPSHETDEEFDAR m/z 566.3. The spectrum shows CID-typical neutral loss of phosphoric acid from the precursor ion. The neutral loss containing ions b6+2 and y12+2 indicate the phosphorylation of Ser-4. Furthermore, the occurrence of b2 and y8 ion excludes phosphorylation at Ser-1 and Thr-7. The ETD-MS/MS spectrum additionally confirms a clear localization of Ser-4 phosphorylation by the presence of N-terminal c3 and c4 ions as well as the C-terminal z10 and z11 ions. Additionally, phosphorylated Thr-35 was localized in the triply charged precursor ion GMNPTLVGYDLVPEPK m/z 571.6 using a combination of CID (C) and ETD (D). The CID-MS/MS spectrum shows two abundant ions, the y4 ion and the corresponding N-terminal b11+2 ion, pointing to breakage at the labile peptide bondage in front of Pro-43. The tyrosine phosphorylation (Tyr-39) was excluded by the occurrence of b7, b8, y7, and y9 ions. Furthermore, ETD fragmentation resulted in an easily interpretable spectrum giving a good explanation of the amino acid sequence including the c7 and c8 ions and the corresponding z7 and z8 ions. On the basis of the fragmentation pattern Tyr-39 could be excluded to be phosphorylated, and the c4, z13, and y14 ions explicitly confirm a phosphorylation of Thr-35.
To analyze the influence of the unspecific phosphatase inhibitor NaF mitochondria were also isolated from frozen bovine heart in the presence and absence of 25 mm NaF, and CcO was purified from the mitochondria by standard procedures. The kinetics of the purified enzymes, measured in the presence of 5 mm ADP and 5 mm ATP, indicated for both enzyme preparations hyperbolic curves with no inhibition by ATP (not shown). The lack of inhibition of activity in the presence of ATP, as compared with Fig. 1, suggests additional loss of phosphorylation of CcO subunits during storage of the heart. The Western blots of the two enzymes indicated tyrosine and serine phosphorylation at subunit II/III and serine and threonine phosphorylation at subunit IV in both enzyme preparations. Phosphorylation of serine and threonine at subunits VIIabc, however, was only seen in the enzyme from mitochondria isolated in the presence of NaF (not shown), indicating loss of this phosphorylation during isolation of bovine heart mitochondria in the absence of NaF. In these two CcO preparations no phosphorylation of subunit I and of subunits Vab and VIabc, as found in Fig. 2 for CcO prepared from fresh tissue, was found, indicating loss of the phosphorylations during storage at −80 °C and/or during freezing and thawing of the bovine heart.
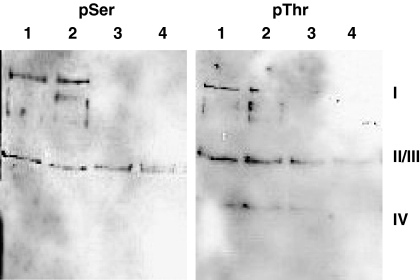
In previous studies the allosteric ATP-inhibition was induced in purified CcO from bovine heart by incubation with PKA, cAMP, and ATP (32). To identify the phosphorylation sites related to the allosteric ATP-inhibition, we incubated isolated CcO with PKA, cAMP, and ATP and measured the kinetics as described in Fig. 5. Before incubation part of the enzyme was dialyzed overnight in the presence of cardiolipin to remove bound cholate and to supplement the enzyme with cardiolipin, which could have been lost during the preparation of CcO. Incubation with PKA induced the allosteric ATP-inhibition as seen in Fig. 5, A and B, characterized by low respiration rates in the presence of ATP at low concentrations of cytochrome c. Incubation of CcO in the absence of PKA resulted in little difference between the activity measured in the presence of ATP or ADP (Fig. 5, C and D). This result does not prove that PKA induces the allosteric ATP-inhibition of CcO in vivo, it could as well be induced in vivo by another, yet unknown, mitochondria specific serine/threonine protein kinase. The Western blots of the two enzymes with antibodies against phosphoserine and phosphothreonine are presented in Fig. 6. Incubation of CcO with PKA results in additional phosphorylation of subunit I on serine and threonine. Approaches to identify the phosphorylated amino acids by mass spectrometry, however, were so far unsuccessful. This is mostly because of the strong hydrophobic nature of subunit I containing 13 hydrophobic transmembrane helices as well as its poor accessibility for the common proteases used for MS analysis.
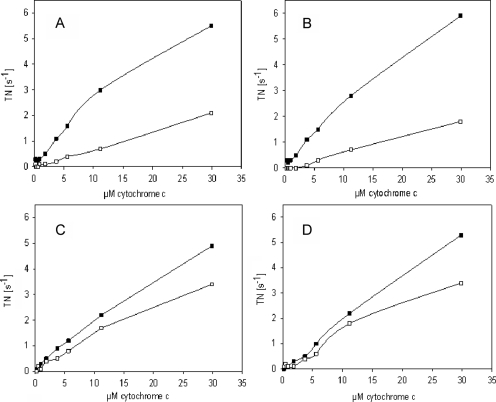
Fig. 5.
Incubation of isolated CcO from bovine heart with PKA, cAMP, and ATP induces the allosteric ATP-inhibition. CcO was incubated for 40 min at 30 °C with (A and B) or without PKA (C and D), as described under “Experimental Procedures.” Before incubation part of the enzyme was dialyzed overnight with cardiolipin (A and C). Oxygen consumption was measured at increasing concentrations of cytochrome c in the presence of 5 mm ADP (closed squares) and 5 mm ATP and an ATP regenerating system (open squares). The turnover number (TN) is presented as μmol cytochrome c × μmol heme aa3−1 × s−1.
Fig. 6.
Treatment of isolated CcO from bovine heart with PKA, cAMP, and ATP leads to serine (pSer) and threonine phosphorylation (pThr) of subunit I. CcO was incubated for 40 min at 30 °C with (1, 2) or without PKA (3, 4), as described under “Experimental Procedures.” Part of the enzyme was dialyzed overnight with cardiolipin (1, 3).
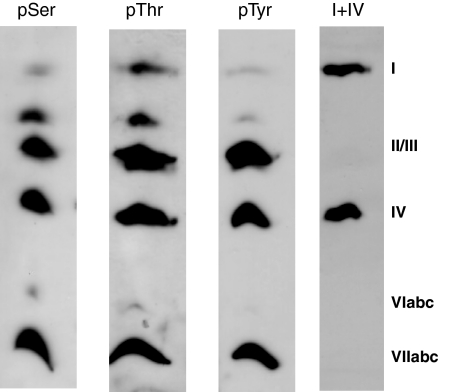
To prove that the allosteric ATP-inhibition is present under in vivo conditions, we rapidly prepared mitochondria from rat heart immediately after the death of the animal in the presence of 25 mm NaF, 5 mm sodium vanadate, 10 nm okadaic acid, 2 mm EGTA, and 0.2% bovine serum albumin and measured the CcO kinetics without delay. As shown in Fig. 7, a strong inhibition of CcO activity is obtained in the presence of ATP, but the curve has no clear sigmoidal shape. This could be due to 1) a mixture of enzymes with and without ATP-inhibition, 2) a mixture of CcO complexes differing in their phosphorylation pattern, thus leading to multiple mixed kinetic behavior. The Western blots of the enzyme, purified from the rat heart mitochondria by blue native gel electrophoresis (42, 43), indicate phosphorylation of serine, threonine, and tyrosine of CcO subunits II/III, IV, and VIIabc/VIII. In contrast to Fig. 2, phosphorylation of serine, threonine, and tyrosine at subunit I was detected (Fig. 8). The immunoreactive spot between subunits I and II/III does not represent a subunit of CcO. According to mass spectrometric analysis of the bovine CcO the α- and β-subunits of the bc1 complex were identified in corresponding one-dimensional SDS-PAGE separated protein bands. This result, together with the in vitro phosphorylation of CcO subunit I by PKA (Figs. 5 and 6), clearly demonstrates the relationship between the allosteric ATP-inhibition and phosphorylation of CcO subunit I, which apparently occurs in living cells, but is lost under stress (e.g. hypoxic stress).
Fig. 7.
Kinetics of CcO in rat heart mitochondria, isolated immediately after death of the animal in the presence of 25 mm NaF, 5 mm vanadate, 10 nm okadaic acid, 2 mm EGTA, and 0.2% bovine serum albumin. Oxygen consumption was measured at increasing concentrations of cytochrome c in the presence of 5 mm ADP (closed squares) and 5 mm ATP and an ATP regenerating system (open squares). The average of three determinations is shown.
Fig. 8.
Western blot of rat heart CcO, isolated by blue native gel electrophoresis, with antibodies against phosphoserine (pSer), phosphothreonine (pThr), phosphotyrosine (pTyr), and CcO subunits I and IV. The immunoreactive spot between subunits I and II/III does not correspond to a CcO subunit.
DISCUSSION
We have identified three new phosphorylation sites in isolated CcO from bovine heart, contrasting the previous view that the bovine heart enzyme may not be phosphorylated (38). Most phosphorylation sites, however, appear to be rapidly lost after death of the animal (e.g. due to hypoxic stress). The identified phosphorylation sites at subunit IV (Ser-34) and subunit Va (Ser-4 and Thr-35) are only obtained under certain conditions including the use of protein phosphatase inhibitors in the medium for isolation of mitochondria. The applied methods do not allow estimating the percentage of phosphorylation of these amino acids. The phosphorylation sites at subunit Va have only been found in CcO prepared from fresh heart tissue (Fig. 2). After storage of the heart at −80 °C for several weeks these phosphorylations are lost (data not shown). The identified phosphorylated amino acids are located at the matrix side of CcO, as presented in the crystal structure of the bovine heart enzyme in Fig. 9. They are apparently not related to the allosteric ATP-inhibition (21, 30), which appears to involve phosphorylation of subunit I (see Figs. 6 and 8). Their function could be to modify supercomplex formation of respiratory chain complexes (13–15) or to change the binding affinity of CcO to specific proteins. In fact, the binding of at least seven proteins/enzymes to specific subunits of CcO have been described (binding of voltage-dependent anion channel to subunit I, epidermal growth factor receptor-pY845 to subunit II, HBx (viral protein) to subunit III, PKCɛ to subunit IV, nitric oxide synthase to subunit Va, subunit RIα of PKA to subunit Vb, androgen receptor to subunit Vb, for review see (8). Phosphorylation of Tyr-304 in subunit I of bovine liver CcO is shown to enhance the allosteric kinetics of CcO in the presence of ADP and ATP (36). The allosteric ATP-inhibition, however, is characterized by sigmoidal inhibition curves only in the presence of ATP, not in the presence of ADP, if oxygen consumption is measured at increasing cytochrome c concentrations (23). Therefore we postulate that for the allosteric ATP-inhibition of CcO phosphorylation of other amino acids than Tyr-304 in subunit I are responsible, including serine and/or threonine phosphorylation of subunit I (Fig. 8). The presented data support the previous postulate that the allosteric ATP-inhibition of CcO is turned off under stress (e.g. death of an animal) via dephosphorylation of CcO subunit I at serine and/or threonine (23, 24). The physiological significance of this mechanism is suggested to increase the rate of ATP synthesis under stress, which, however, is accompanied by increased formation of reactive oxygen species (8, 23, 24). Further work is required to identify the phosphorylated amino acid(s) at CcO subunit I.
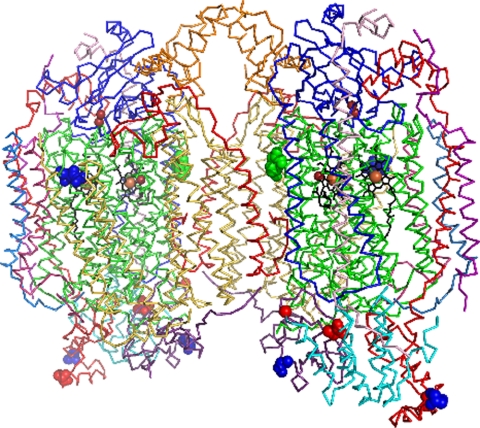
Fig. 9.
Crystal structure of dimeric bovine heart CcO (12) with indication of known phosphorylation sites. Polypeptides above subunit I, presented in green and mainly located within the lipid membrane space, are located in the intermembrane space, those below subunit I in the matrix space. The phosphorylation sites are visualized in both monomers by bold atoms of the corresponding amino acids: Tyr-304 of subunit I (36) in green; Ser-115 + Ser-116 of subunit I; Ser-52 (in rabbit = Thr-52) in subunit IV; and Ser-40 in subunit Vb (39) in blue; Ser-34 in subunit IV, and Ser-4 (indicated by the Cα atom of His-5 because Ser-4 is not in the crystal structure) and Thr-35 in subunit Va (this paper) in red. Subunit II is shown in dark blue. The transmembrane subunit IV and subunit VIa, located at the interface between the two monomers, are presented in red, subunit Va in light blue, and subunit Vb (and VIII) in lila. The two heme a groups are in black, with iron and copper atoms in brown. The two copper atoms of the CuA site and the copper atom at heme a3 are smaller than the iron atoms in heme a and heme a3.
The results of this paper indicate multiple phosphorylation sites to occur in CcO in living cells, which, at least in part, participate in regulation of activity. The phosphorylation sites appear to be reversibly phosphorylated, depending on the metabolic state. In fact, at least 10 of the 13 subunits of CcO have been found to be phosphorylated at serine and/or threonine, depending on the isolation conditions of the enzyme.2 Multiple phosphorylation sites could be envisioned in mammalian CcO based on consensus sequences for PKA-dependent phosphorylation sites at serine and threonine residues according to Pearson and Kemp (49). From the consensus sequences 53 sites for PKA-dependent phosphorylation of serine and threonine occur within the amino acid sequences of the 13 subunits of bovine heart CcO (11 in subunit I, 10 in II, 3 in III, 6 in IV, 3 in Va, 3 in Vb, 4 in VIa, 3 in VIb, 2 in VIc, 2 in VIIa, 1 in VIIb, 1 in VIIc, and 4 in VIII). It remains to be investigated which of these possible phosphorylation sites of CcO subunits are phosphorylated in living cells and what will be their physiological function.
Supplementary Material
Acknowledgments
We thank Lars Essen, Daniel Kress, and Simon Reitz for preparing Fig. 9. The technical assistance of Petra Weber is gratefully acknowledged.
Footnotes
Published, MCP Papers in Press, June 9, 2008, DOI 10.1074/mcp.M800137-MCP200
The abbreviations used are: CcO, cytochrome c oxidase; ACN, acetonitril; FA, formic acid; ESI, electrospray ionization; CID, collision-induced dissociation; ETD, electron transfer dissociation; PKA, protein kinase A; MS, mass spectrometry; ROS, reactive oxygen species.
S. Vogt, A. Rhiel, and B. Kadenbach, unpublished results.
This work was supported by the Deutsche Forschungsgemeinschaft (Project Ka 192/40-1 and Marburger Herzstiftung). The costs of publication of this article were defrayed in part by the payment of page charges. This article must therefore be hereby marked “advertisement” in accordance with 18 U.S.C. Section 1734 solely to indicate this fact.
The amino acid sequence of this protein can be accessed through NCBI Protein Database under NCBI accession number 2007.11.09.
REFERENCES
- 1.Papa, S., Sardanelli, A. M., Scacco, S., and Technikova-Dobrova, Z. ( 1999) cAMP-dependent protein kinase and phosphoproteins in mammalian mitochondria. An extension of the cAMP-mediated intracellular signal transduction. FEBS Lett. 444, 245–249 [DOI] [PubMed] [Google Scholar]
- 2.Thompson, M. ( 2002) Evidence of undiscovered cell regulatory mechanisms: phosphoproteins and protein kinases in mitochondria. Cell. Mol. Life Sci. 59, 213–219 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Goldenthal, M. J., and Marín-García, J. ( 2004) Mitochondrial signaling pathways: a receiver/integrator organelle. Mol. Cell. Biochem. 262, 1–16 [DOI] [PubMed] [Google Scholar]
- 4.Horbinski, C., and Chu, C. T. ( 2005) Kinase signaling cascades in the mitochondrion: a matter of life or death. Free Radic. Biol. Med. 38, 2–11 [DOI] [PubMed] [Google Scholar]
- 5.Sardanelli, A. M., Signorile, A., Nuzzi, R., Rasmo, D. D., Technikova-Dobrova, Z., Drahot, Z., Occhiell, A., Pica, A., and Papa, S. ( 2006) Occurrence of A-kinase anchor protein and associated cAMP-dependent protein kinase in the inner compartment of mammalian mitochondria. FEBS Lett. 580, 5690–5696 [DOI] [PubMed] [Google Scholar]
- 6.Boneh, A. ( 2006) Regulation of mitochondrial oxidative phosphorylation by second messenger-mediated signal transduction mechanisms. Cell. Mol. Life Sci. 63, 1236–1248 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Pagliarini, D. J., and Dixon, J. E. ( 2006) Mitochondrial modulation: reversible phosphorylation takes center stage? Trends Biochem. Sci. 31, 26–34 [DOI] [PubMed] [Google Scholar]
- 8.Vogt, S., Rhiel, A., Koch, V., and Kadenbach, B. ( 2007) Regulation of oxidative phosphorylation by inhibition of its enzyme complexes via reversible phosphorylation. Curr. Enzyme Inhib. 3, 189–206 [Google Scholar]
- 9.Hüttemann, M., Lee, I., Samavati, L., Yu, H., and Doan, J. W. ( 2007) Regulation of mitochondrial oxidative phosphorylation through cell signaling. Biochim. Biophys. Acta 1773, 1701–1720 [DOI] [PubMed] [Google Scholar]
- 10.Villani, G., Greco, M., Papa, S., and Attardi, G. ( 1998) Low reserve of cytochrome c oxidase capacity in vivo in the respiratory chain of a variety of human cell types. J. Biol. Chem. 273, 31829–31836 [DOI] [PubMed] [Google Scholar]
- 11.Tsukihara, T., Aoyama, H., Yamashita, E., Tomizaki, T., Yamaguchi, H., Shinzawa-Itoh, K., Nakashima, R., Yaono, R., and Yoshikawa, S. ( 1996) The whole structure of the 13-subunit oxidized cytochrome c oxidase at 2.8 A. Science 272, 1136–1144 [DOI] [PubMed] [Google Scholar]
- 12.Shinzawa-Itoh, K., Aoyama, H., Muramoto, K., Terada, H., Kurauchi, T., Tadehara, Y., Yamasaki, A., Sugimura, T., Kurono, S., Tsujimoto, K., Mizushima, T., Yamashita, E., Tsukihara, T., and Yoshikawa, S. ( 2007) Structures and physiological roles of 13 integral lipids of bovine heart cytochrome c oxidase. EMBO J. 26, 1713–1725 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Wittig, I., Carrozzo, R., Santorelli, F. M., and Schägger, H. ( 2006) Supercomplexes and subcomplexes of mitochondrial oxidative phosphorylation. Biochim. Biophys. Acta 1757, 1066–1072 [DOI] [PubMed] [Google Scholar]
- 14.Schäfer, E., Seelert, H., Reifschneider, N. H., Kraus, e, F., Dencher, N. A., and Vonck, J. ( 2006) Architecture of active mammalian respiratory chain supercomplexes. J. Biol. Chem. 281, 15370–15375 [DOI] [PubMed] [Google Scholar]
- 15.Boekema, E. J., and Braun, H. P. ( 2007) Supramolecular structure of the mitochondrial oxidative phosphorylation system. J. Biol. Chem. 282, 1–4 [DOI] [PubMed] [Google Scholar]
- 16.Ludwig, B., Bender, E., Arnold, S., Hüttemann, M., Lee, I., and Kadenbach, B. ( 2001) Cytochrome c oxidase and the regulation of oxidative phosphorylation. Chem. Bio. Chem. 2, 392–403 [DOI] [PubMed] [Google Scholar]
- 17.Richter, O. M., and Ludwig, B. ( 2003) Cytochrome c oxidase - structure, function, and physiology of a redox-driven molecular machine. Rev. Physiol. Biochem. Pharmacol. 147, 47–74 [DOI] [PubMed] [Google Scholar]
- 18.Napiwotzki, J., Shinzawa-Itoh, K., Yoshikawa, S., and Kadenbach, B. ( 1997) ATP and ADP bind to cytochrome c oxidase and regulate its activity. Biol. Chem. 378, 1013–1021 [DOI] [PubMed] [Google Scholar]
- 19.Napiwotzki, J., and Kadenbach, B. ( 1998) Extramitochondrial ATP/ADP-ratios regulate cytochrome c oxidase activity via binding to the cytosolic domain of subunit IV. Biol. Chem. 379, 335–339 [DOI] [PubMed] [Google Scholar]
- 20.Frank, V., and Kadenbach, B. ( 1996) Regulation of the H+/e−-stoichiometry of cytochrome c oxidase from bovine heart by intraliposomal ATP/ADP ratios. FEBS Lett. 382, 121–124 [DOI] [PubMed] [Google Scholar]
- 21.Arnold, S., and Kadenbach, B. ( 1997) Cell respiration is controlled by ATP, an allosteric inhibitor of cytochrome c oxidase. Eur. J. Biochem. 249, 350–354 [DOI] [PubMed] [Google Scholar]
- 22.Arnold, S., Goglia, F., and Kadenbach, B. ( 1998) 3,5-diiodothyronine binds to subunit Va of cytochrome c oxidase and abolishes the allosteric inhibition of respiration by ATP. Eur. J. Biochem. 252, 325–330 [DOI] [PubMed] [Google Scholar]
- 23.Lee, I., Bender, E., Arnold, S., and Kadenbach, B. ( 2001) Minireview-Hypothesis. New control of mitochondrial membrane potential and ROS formation. Biol. Chem. 382, 1629–1636 [DOI] [PubMed] [Google Scholar]
- 24.Kadenbach, B., Arnold, S., Lee, S., and Hüttemann, M. ( 2004) The possible role of cytochrome c oxidase in stress-induced apoptosis and degenerative diseases. Biochim. Biophys. Acta 1655, 400–408 [DOI] [PubMed] [Google Scholar]
- 25.Liu, S. S. ( 1997) Generating, partitioning, targeting and functioning of superoxide in mitochondria. Biosci. Rep. 17, 259–272 [DOI] [PubMed] [Google Scholar]
- 26.Kaim, G., and Dimroth, P. ( 1999) ATP synthesis by F-type ATP synthase is obligatorily dependent on the transmembrane voltage. EMBO J. 18, 4118–4127 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Chance, B., and Williams, C. M. ( 1955) Respirtatory enzymes in oxidative phosphorylation. III. The steady state. J. Biol. Chem. 217, 405–427 [PubMed] [Google Scholar]
- 28.Nicholls, D. G., and Ferguson, S. J. ( 2002) Bioenergetics 3, pp. 63–66, Academic Press, San Diego
- 29.Kadenbach, B., and Arnold, S. ( 1999) Minireview: a second mechanism of respiratory control. FEBS Lett. 447, 131–134 [DOI] [PubMed] [Google Scholar]
- 30.Arnold, S., and Kadenbach, B. ( 1999) The intramitochondrial ATP/ADP ratio controls cytochrome c oxidase activity allosterically. FEBS Lett. 443, 105–108 [DOI] [PubMed] [Google Scholar]
- 31.Bender, E., and Kadenbach, B. ( 2000) The allosteric ATP-inhibition of cytochrome c oxidase is reversibly switched on by cAMP-dependent phosphorylation. FEBS Lett. 466, 130–134 [DOI] [PubMed] [Google Scholar]
- 32.Lee, I., Bender, E., and Kadenbach, B. ( 2002) Control of mitochondrial membrane potential and ROS formation by reversible phosphorylation of cytochrome c oxidase. Mol. Cell. Biochem. 234/235, 63–70 [PubMed] [Google Scholar]
- 33.Steenaart, N. A. C., and Shore, G. C. ( 1997) Mitochondrial cytochrome c oxidase subunit IV is phosphorylated by an endogenous kinase. FEBS Lett. 415, 294–298 [DOI] [PubMed] [Google Scholar]
- 34.Ogbi, M., Chew, C. S., Pohl, J., Stuchlik, O., Ogbi, S., and Johnson, J. A. ( 2004) Cytochrome c oxidase subunit IV as a marker of protein kinase Cɛ function in neonatal cardiac myocytes: implications for cytochrome c oxidase activity. Biochem. J. 382, 923–932 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Ogbi, M., and Johnson, J. A. ( 2006) Protein kinase Cɛ interacts with cytochrome c oxidase subunit IV and enhances cytochrome c oxidase activity in neonatal cardiac myocyte preconditioning. Biochem. J. 393, 191–199 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Lee, I., Salomon, A. R., Ficarro, S., Mathes, I., Lottspeich, F., Grossman, L. I., and Hüttemann, M. ( 2005) cAMP-dependent tyrosine phosphorylation of subunit I inhibits cytochrome c oxidase activity. J. Biol. Chem. 280, 6094–6100 [DOI] [PubMed] [Google Scholar]
- 37.Feliciello, A., Gottesman, M. E., and Avvedimento, E. V. ( 2005) cAMP-PKA signaling to the mitochondria: protein scaffolds, mRNA and phosphatases. Cell. Signal. 17, 279–287 [DOI] [PubMed] [Google Scholar]
- 38.Prabu, S. K., Anandatheerthavarada, H. K., Raza, H., Srinivasan, S., Spear, J. F., and Avadhani, N. G. ( 2006) Protein kinase A-mediated phosphorylation modulates cytochrome c oxidase function and augments hypoxia and myocardial ischemia-related injury. J. Biol. Chem. 281, 2061–2070 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Fang, J. K., Prabu, S. K., Sepuri, N. B., Raza, H., Anandatheerthavarada, H. K., Galati, D., Spear, J., and Avadhani, N. G. ( 2007) Site-specific phosphorylation of cytochrome c oxidase subunits I, IVi1 and Vb in rabbit hearts subjected to ischemia/reperfusion. FEBS Lett. 581, 1302–1310 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Marx, M. K., Mayer-Posner, F., Souliname, T., and Buse, G. ( 1998) Matrix-assisted lase desorption/ionization mass spectrometry analysis and thiol-group determination of isoforms of bovine cytochrome c oxidase, a hydrophobic multisubunit membrane protein. Anal. Biochem. 256, 192–199 [DOI] [PubMed] [Google Scholar]
- 41.Kadenbach, B., Stroh, A., Ungibauer, M., Kuhn-Nentwig, L., Büge, U., and Jarausch, J. ( 1986) Isozymes of cytochrome c oxidase: characterization and isolation from different tissues. Methods Enzymol. 126, 32–45 [DOI] [PubMed] [Google Scholar]
- 42.Schägger, H., and von Jagow, G. ( 1991) Blue native electrophoresis for isolation of membrane protein complexes in enzymatically active form. Anal. Biochem. 199, 223–231 [DOI] [PubMed] [Google Scholar]
- 43.Wittig, I., Braun, H. P., and Schägger, H. ( 2006) Blue native PAGE. Nature Protoc. 1, 418–428 [DOI] [PubMed] [Google Scholar]
- 44.Kadenbach, B., Jarausch, J., Hartmann, R., and Merle, P. ( 1983) Separation of mammalian cytochrome c oxidase into 13 poly-peptides by a sodium dodecyl sulfate-gel electrophoretic procedure. Anal. Biochem. 129, 517–521 [DOI] [PubMed] [Google Scholar]
- 45.Eckerskorn, C., Mewes, W., Goretzki, H., and Lottspeich, F. ( 1988) A new siliconized-glass fiber as support for protein-chemical analysis of electroblotted proteins. Eur. J. Biochem. 176, 509–519 [DOI] [PubMed] [Google Scholar]
- 46.Immler, D., Greven, S., and Reinemer, P. ( 2006) Targeted proteomics in biomarker validation: detection and quantification of proteins using a multi-dimensional peptide separation strategy. Proteomics 6, 2947–2958 [DOI] [PubMed] [Google Scholar]
- 47.Thingholm, T. E., Jorgensen, T. J., Jensen, O. N., and Larsen, M. R. ( 2006) Highly selective enrichment of phosphorylated peptides using titanium dioxide. Nature Protoc. 1, 1929–1935 [DOI] [PubMed] [Google Scholar]
- 48.Mazanek, M., Mituloviae, G., Herzog, F., Stingl, C., Hutchins, J. R., Peters, J. M., and Mechtler, K. ( 2007) Titanium dioxide as a chemo-affinity solid phase in offline phosphopeptide chromatography prior to HPLC-MS/MS analysis. Nature Protoc. 2, 1059–1069 [DOI] [PubMed] [Google Scholar]
- 49.Pearson, R. B., and Kemp, B. E. ( 1991) Protein kinase phosphorylation site sequences and consensus specificity motifs: tabulations. Met. Enzymol. 200, 62–81 [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.