Abstract
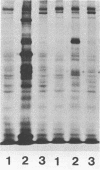
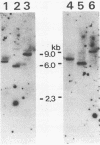
A new avian transforming retrovirus, NK24, was isolated from a chicken with a nephroblastoma. This transforming virus induced fibrosarcomas with osteogenic cell proliferation and nephroblastomas in vivo and transformed fibroblast cells in vitro. From extracts of NK24-transformed cells, anti-gag serum immunoprecipitated a 100-kilodalton nonglycosylated protein with no detectable protein kinase activity. An NK24 provirus present in infected quail cells was molecularly cloned and subjected to nucleotide sequence analysis. The genome of NK24 was 5.3 kilobases long and had a 1,126-base-pair sequence of cellular origin in place of a viral sequence of avian leukosis virus containing the 3' half of the gag gene and the 5' half of the pol gene. Although the entire env gene was retained, it appeared to be inactive, possibly owing to the loss of function of its splice acceptor site as a result of a second deletion of 1,598 bases in the 3' half of the pol gene that extended to the acceptor site. Nucleotide sequence analysis revealed that the NK24 virus contained the fos gene, previously identified as the oncogene of FBJ and FBR murine osteosarcoma viruses. Unlike the v-fos gene products of FBJ and FBR, which suffer a structural alteration at their carboxyl termini, the NK24 v-fos gene product seemed to have the same carboxyl-terminal structure as the chicken c-fos gene product. A comparison of the structures of the products of the NK24 v-fos and mouse c-fos genes suggested that the fos gene product consists of highly conserved regions and relatively divergent regions.
Full text
PDF







Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- BURMESTER B. R., WALTER W. G., GROSS M. A., FONTES A. K. The oncogenic spectrum of two pure strains of avian leukosis. J Natl Cancer Inst. 1959 Aug;23:277–291. [PubMed] [Google Scholar]
- CARR J. G. Kidney carcinomas of the fowl induced by the MH2 reticuloendothelioma virus. Br J Cancer. 1960 Mar;14:77–82. doi: 10.1038/bjc.1960.9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- CARR J. G. Renal adenocarcinoma induced by fowl leukaemia virus. Br J Cancer. 1956 Jun;10(2):379–383. doi: 10.1038/bjc.1956.43. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Curran T., Peters G., Van Beveren C., Teich N. M., Verma I. M. FBJ murine osteosarcoma virus: identification and molecular cloning of biologically active proviral DNA. J Virol. 1982 Nov;44(2):674–682. doi: 10.1128/jvi.44.2.674-682.1982. [DOI] [PMC free article] [PubMed] [Google Scholar]
- DeLorbe W. J., Luciw P. A., Goodman H. M., Varmus H. E., Bishop J. M. Molecular cloning and characterization of avian sarcoma virus circular DNA molecules. J Virol. 1980 Oct;36(1):50–61. doi: 10.1128/jvi.36.1.50-61.1980. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Finkel M. P., Biskis B. O., Jinkins P. B. Virus induction of osteosarcomas in mice. Science. 1966 Feb 11;151(3711):698–701. doi: 10.1126/science.151.3711.698. [DOI] [PubMed] [Google Scholar]
- Fujiwara K. T., Ashida K., Nishina H., Iba H., Miyajima N., Nishizawa M., Kawai S. The chicken c-fos gene: cloning and nucleotide sequence analysis. J Virol. 1987 Dec;61(12):4012–4018. doi: 10.1128/jvi.61.12.4012-4018.1987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Graf T., Beug H. Avian leukemia viruses: interaction with their target cells in vivo and in vitro. Biochim Biophys Acta. 1978 Nov 17;516(3):269–299. doi: 10.1016/0304-419x(78)90011-2. [DOI] [PubMed] [Google Scholar]
- Hagino-Yamagishi K., Ikawa S., Kawai S., Hihara H., Yamamoto T., Toyoshima K. Characterization of two strains of avian sarcoma virus isolated from avian lymphatic leukosis virus-induced sarcomas. Virology. 1984 Sep;137(2):266–275. doi: 10.1016/0042-6822(84)90218-6. [DOI] [PubMed] [Google Scholar]
- Hattori M., Sakaki Y. Dideoxy sequencing method using denatured plasmid templates. Anal Biochem. 1986 Feb 1;152(2):232–238. doi: 10.1016/0003-2697(86)90403-3. [DOI] [PubMed] [Google Scholar]
- Hayward W. S., Neel B. G., Astrin S. M. Activation of a cellular onc gene by promoter insertion in ALV-induced lymphoid leukosis. Nature. 1981 Apr 9;290(5806):475–480. doi: 10.1038/290475a0. [DOI] [PubMed] [Google Scholar]
- Henikoff S. Unidirectional digestion with exonuclease III creates targeted breakpoints for DNA sequencing. Gene. 1984 Jun;28(3):351–359. doi: 10.1016/0378-1119(84)90153-7. [DOI] [PubMed] [Google Scholar]
- Kawai S., Hanafusa H. Plaque assay for some strains of avian leukosis virus. Virology. 1972 Apr;48(1):126–135. doi: 10.1016/0042-6822(72)90120-1. [DOI] [PubMed] [Google Scholar]
- Kawai S., Koyama T. Characterization of a Rous sarcoma virus mutant defective in packaging its own genomic RNA: biological properties of mutant TK15 and mutant-induced transformants. J Virol. 1984 Jul;51(1):147–153. doi: 10.1128/jvi.51.1.147-153.1984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kawai S., Nishizawa M. New procedure for DNA transfection with polycation and dimethyl sulfoxide. Mol Cell Biol. 1984 Jun;4(6):1172–1174. doi: 10.1128/mcb.4.6.1172. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kawai S., Nishizawa M., Yamamoto T., Toyoshima K. Cell transformation by a virus containing a molecularly constructed gag-erbB fused gene. J Virol. 1987 May;61(5):1665–1669. doi: 10.1128/jvi.61.5.1665-1669.1987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kawai S., Segawa K., Yamamoto T. Antisera from mice and rats incubated with syngeneic Rous sarcoma cells. Jpn J Exp Med. 1980 Oct;50(5):339–349. [PubMed] [Google Scholar]
- Lee C. K., Chan E. W., Reilly C. A., Jr, Pahnke V. A., Rockus G., Finkel M. P. In vitro properties of FBR murine osteosarcoma virus. Proc Soc Exp Biol Med. 1979 Oct;162(1):214–220. doi: 10.3181/00379727-162-40650. [DOI] [PubMed] [Google Scholar]
- Levy J. A., Hartley J. W., Rowe W. P., Huebner R. J. Studies of FBJ osteosarcoma virus in tissue culture. I. Biologic characteristics of the "C"-type viruses. J Natl Cancer Inst. 1973 Aug;51(2):525–539. [PubMed] [Google Scholar]
- Meijlink F., Curran T., Miller A. D., Verma I. M. Removal of a 67-base-pair sequence in the noncoding region of protooncogene fos converts it to a transforming gene. Proc Natl Acad Sci U S A. 1985 Aug;82(15):4987–4991. doi: 10.1073/pnas.82.15.4987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Miles B. D., Robinson H. L. High-frequency transduction of c-erbB in avian leukosis virus-induced erythroblastosis. J Virol. 1985 May;54(2):295–303. doi: 10.1128/jvi.54.2.295-303.1985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Miller A. D., Curran T., Verma I. M. c-fos protein can induce cellular transformation: a novel mechanism of activation of a cellular oncogene. Cell. 1984 Jan;36(1):51–60. doi: 10.1016/0092-8674(84)90073-4. [DOI] [PubMed] [Google Scholar]
- Mizusawa S., Nishimura S., Seela F. Improvement of the dideoxy chain termination method of DNA sequencing by use of deoxy-7-deazaguanosine triphosphate in place of dGTP. Nucleic Acids Res. 1986 Feb 11;14(3):1319–1324. doi: 10.1093/nar/14.3.1319. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nishizawa M., Mayer B. J., Takeya T., Yamamoto T., Toyoshima K., Hanafusa H., Kawai S. Two independent mutations are required for temperature-sensitive cell transformation by a Rous sarcoma virus temperature-sensitive mutant. J Virol. 1985 Dec;56(3):743–749. doi: 10.1128/jvi.56.3.743-749.1985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ogura H., Gelderblom H., Bauer H. Isolation of avian nephroblastoma virus from avian myeloblastosis virus by the infectious DNA technique. Intervirology. 1974;4(2):69–76. doi: 10.1159/000149845. [DOI] [PubMed] [Google Scholar]
- Payne G. S., Bishop J. M., Varmus H. E. Multiple arrangements of viral DNA and an activated host oncogene in bursal lymphomas. Nature. 1982 Jan 21;295(5846):209–214. doi: 10.1038/295209a0. [DOI] [PubMed] [Google Scholar]
- Raines M. A., Lewis W. G., Crittenden L. B., Kung H. J. c-erbB activation in avian leukosis virus-induced erythroblastosis: clustered integration sites and the arrangement of provirus in the c-erbB alleles. Proc Natl Acad Sci U S A. 1985 Apr;82(8):2287–2291. doi: 10.1073/pnas.82.8.2287. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schwartz D. E., Tizard R., Gilbert W. Nucleotide sequence of Rous sarcoma virus. Cell. 1983 Mar;32(3):853–869. doi: 10.1016/0092-8674(83)90071-5. [DOI] [PubMed] [Google Scholar]
- Shibuya M., Hanafusa T., Hanafusa H., Stephenson J. R. Homology exists among the transforming sequences of avian and feline sarcoma viruses. Proc Natl Acad Sci U S A. 1980 Nov;77(11):6536–6540. doi: 10.1073/pnas.77.11.6536. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Smith R. E., Moscovici C. The oncogenic effects of nontransforming viruses from avian myeloblastosis virus. Cancer Res. 1969 Jul;29(7):1356–1366. [PubMed] [Google Scholar]
- Stehelin D., Guntaka R. V., Varmus H. E., Bishop J. M. Purification of DNA complementary to nucleotide sequences required for neoplastic transformation of fibroblasts by avian sarcoma viruses. J Mol Biol. 1976 Mar 5;101(3):349–365. doi: 10.1016/0022-2836(76)90152-2. [DOI] [PubMed] [Google Scholar]
- Sukegawa J., Semba K., Yamanashi Y., Nishizawa M., Miyajima N., Yamamoto T., Toyoshima K. Characterization of cDNA clones for the human c-yes gene. Mol Cell Biol. 1987 Jan;7(1):41–47. doi: 10.1128/mcb.7.1.41. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Van Beveren C., Enami S., Curran T., Verma I. M. FBR murine osteosarcoma virus. II. Nucleotide sequence of the provirus reveals that the genome contains sequences acquired from two cellular genes. Virology. 1984 May;135(1):229–243. doi: 10.1016/0042-6822(84)90133-8. [DOI] [PubMed] [Google Scholar]
- Van Beveren C., van Straaten F., Curran T., Müller R., Verma I. M. Analysis of FBJ-MuSV provirus and c-fos (mouse) gene reveals that viral and cellular fos gene products have different carboxy termini. Cell. 1983 Apr;32(4):1241–1255. doi: 10.1016/0092-8674(83)90306-9. [DOI] [PubMed] [Google Scholar]
- Ward J. M., Young D. M. Histogenesis and Morphology of periosteal sarcomas induced by FBJ virus in NIH Swiss mice. Cancer Res. 1976 Nov;36(11 Pt 1):3985–3992. [PubMed] [Google Scholar]
- Westaway D., Papkoff J., Moscovici C., Varmus H. E. Identification of a provirally activated c-Ha-ras oncogene in an avian nephroblastoma via a novel procedure: cDNA cloning of a chimaeric viral-host transcript. EMBO J. 1986 Feb;5(2):301–309. doi: 10.1002/j.1460-2075.1986.tb04213.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yamamoto T., Hihara H., Nishida T., Kawai S., Toyoshima K. A new avian erythroblastosis virus, AEV-H, carries erbB gene responsible for the induction of both erythroblastosis and sarcomas. Cell. 1983 Aug;34(1):225–232. doi: 10.1016/0092-8674(83)90153-8. [DOI] [PubMed] [Google Scholar]