Abstract
Rhesus macaques (Macaca mulatta) provide well-established models for studying human disease pathogenesis and vaccine development. When challenged with infectious agents macaques exhibit individual differences in susceptibility. An important determinant of these differences is the complement of major histocompatability complex (MHC) class I sequences expressed by each animal. Although previous studies have reported strong associations between MHC expression and disease outcome, a rapid, cost effective method for high resolution MHC genotyping in macaques is lacking. Here, we adapted a modified heteroduplex assay, reference strand mediated conformational analysis (RSCA), to an ABI 3130xl capillary electrophoresis genetic analyzer for macaque MHC class I genotyping. For validation, we investigated the concordance of RSCA genotyping for fourteen MHC class I sequences in twelve Chinese rhesus macaques whose genotypes were established through cDNA cloning and sequencing of MHC class I sequences. We observed a concordance greater than 98% between RSCA and the cloning and sequencing data. Further, RSCA confirmed the presence of MHC haplotype sharing between three macaques as predicted previously by microsatellite analysis. RSCA genotyping of an additional 25 Chinese rhesus macaques demonstrated that the frequency of these fourteen MHC class I sequences ranged from 5 to 32%, with the Mamu-A1*2601 sequence being most common in this cohort. Capillary RSCA genotyping has the potential to enable researchers to rapidly evaluate MHC class I genotypes in rhesus macaques and associate specific MHC sequences with disease susceptibility.
Keywords: RSCA, MHC, Macaca mulatta, Genotyping
Introduction
The rhesus macaque (Macaca mulatta) is the most widely used nonhuman primate in biomedical research primarily due to their physiological and genetic similarity to humans (Gibbs et al. 2007). Functional similarities between the human and macaque major histocompatability complex (MHC) make the macaque an indispensible model for biomedical studies that focus on human disease pathogenesis (Wiseman and O’Connor 2007). MHC gene products play a fundamental role in regulating the host immune response to foreign antigens and expression of specific MHC gene products may significantly influence disease susceptibility and outcome. For example, strong associations have been shown between slower disease progression following infection with human immunodeficiency virus (HIV) or simian immunodeficiency virus (SIV) and specific MHC genes such as HLA-B*27 and HLA-B*57 in humans or Mamu-B*08 and Mamu-B*17 in rhesus macaques, respectively (Kaslow et al. 1996; Bontrop and Watkins 2005; Martin and Carrington 2005; Loffredo et al. 2007). As correlations between macaque MHC genetics and disease susceptibility become increasingly evident, the need for a cost effective method for high resolution MHC genotyping becomes more pronounced.
Cloning and sequencing from rhesus macaque cDNA libraries remains the “gold standard” for high resolution MHC genotyping (Wiseman and O’Connor 2007) However, preparation and screening of cDNA libraries for MHC class I sequences is laborious and prohibitively expensive for comprehensive MHC genotyping of large cohorts of animals, like those used for vaccine studies. More economical techniques such as sequence specific primer (PCR-SSP) and microsatellite PCR analysis are commonly used, but these techniques provide information on only a limited number of known MHC sequences (Kaizu et al. 2007; Penedo et al. 2005).
Several recent studies utilized reference strand mediated conformational analysis (RSCA) to characterize MHC alleles in various nonhuman primates, including MHC-DRB sequences in Aotus monkeys (Baquero et al. 2006) and MHC class I sequences in pigtail, cynomolgus, and rhesus macaques (Smith et al. 2005; Pratt et al. 2006; Krebs et al. 2005; Wiseman et al. 2007; Mee et al. 2008; Tanaka-Takahashi et al. 2007). RSCA resolves heteroduplexes between PCR amplicons and sequence-mismatched reference strands based on their electrophoretic mobility under non-denaturing conditions. RSCA is an effective method for identifying both known and novel MHC sequences and has the added advantage of detecting shared sequences among multiple macaques in a single run. However, difficulties with reproducibility using slab gel platforms and subjective interpretation of electrophoretic profiles have limited the broad application of RSCA as a genotyping method. Additionally, the discontinued technical support for out-dated instrumentation, (e.g., the ABI 377 and Alf Express II), has highlighted a need to adapt RSCA onto a capillary electrophoresis platform.
This study aimed to develop a protocol for capillary RSCA on the ABI 3130xL genetic analyzer as a method for MHC class I genotyping in rhesus macaques. The adaptation of our RSCA protocol was validated with 12 Chinese rhesus macaques whose genotypes were established previously through cDNA cloning and sequencing of MHC class I sequences. An additional 25 Chinese rhesus macaques were screened by capillary RSCA and frequencies of 14 MHC class I sequences were determined.
Materials and Methods
Animals
Peripheral blood mononuclear cell (PBMC) samples were obtained from 37 Chinese-origin rhesus macaques (ChRh01–18, 22–33, 35–41) from Covance (Alice, TX). Nucleic acids from the 37 PBMC samples were isolated using the Qiagen AllPrep DNA/RNA Mini purification kit (Qiagen, Valencia, CA, USA). MHC genotypes for twelve macaques (ChRh01–12) were determined by cloning and sequencing and haplotyped using microsatellite analysis as previously described (Karl et al. 2007). The additional 25 macaques (ChRh13–18, 22–33, 35–41) were genotyped by RSCA alone in this present study.
Generation of custom DNA sizing standard
A custom internal ROX-labeled sizing standard was generated for RSCA on the 3130xL instrument using amplicons from the vector pcDNA3.1+ (Invitrogen Life Technologies, Carlsbad, CA, USA). A universal ROX-labeled forward primer ROX-pcDNA3.1+ (5′-[ROX]AGACAATCGGCTGCTCTGAT) and one of a series of unlabeled reverse oligonucleotides were used to generate PCR amplicons that increased approximately 30 bp in size (Table 1). Primers used in this study were purified by standard de-salting only. PCR of each amplicon was performed using high-fidelity Phusion™ DNA polymerase (New England BioLabs, Ipswich, MA, USA), 5ng pcDNA 3.1+ and 3.2μM of primers. Each reaction was carried out under the following PCR conditions: 98°C for an initial 30 seconds followed by 24 cycles of 5 seconds at 98°C, 1 second at 60°C, and 20 seconds at 72°C, then a final extension for 5 minutes at 72°C. PCR products were purified by electrophoresis through a 1% agarose gel and a QiaQuick™ centrifuge PCR clean up kit (Qiagen). Purified products were normalized to 10ng/μL and pooled to generate an internal size standard. Pooled amplicons were diluted 1:20 in nuclease free water (Integrated DNA Technologies, Coralville, IA, USA). 16μL of pooled amplicons were mixed with 4uL heteroduplex to make a final 20μL sample volume.
Table 1.
Custom Internal Size Standard Primers.
| Primer | Sequence | Product Size (Bp) |
|---|---|---|
| Forward Primer | 5′-ROX-AGACAATCGGCTGCTCTGAT | - |
| Reverse Primer | 5′-CTCGTCCTGCAGTTCATTCA | 110 |
| 5′-GTGGCCAGCCACGATAGC | 136 | |
| 5′-CACAGCTGCGCAAGGAAC | 161 | |
| 5′-CTTCCCGCTTCAGTGACAAC | 187 | |
| 5′-GCCCAATAGCAGCCAGTC | 206 | |
| 5′-GCAAGGTGAGATGACAGGAGA | 244 | |
| 5′-GCCATGATGGATACTTTCTCG | 271 | |
| 5′-GTAGCCGGATCAAGCGTATG | 310 | |
| 5′-TTCGCTTGGTGGTCGAAT | 334 | |
| 5′-AGTACGTGCTCGCTCGATG | 359 | |
| 5′-TCATCCTGATCGACAAGACC | 391 | |
| 5′-AGCCCCTGATGCTCTTCGT | 415 | |
| 5′-CTTGAGCCTGGCGAACAGT | 446 | |
| 5′-CCATGGGTCACGACGAGAT | 487 | |
| 5′-ACCATGATATTCGGCAAGCA | 514 | |
| 5′-GCCACAGTCGATGAATCCAG | 551 | |
| 5′-AGCCAACGCTATGTCCTGAT | 593 | |
| 5′-TCTTCAGCAATATCACGGGTA | 613 | |
| 5′-CCGTAAAGCACGAGGAAGC | 655 | |
| 5′-AAGGCGATAGAAGGCGATG | 698 |
A single ROX-labeled forward primer and a series of 20 reverse primers were used to generate amplicons of varying length using the pcDNA3.1+ plasmid as a template for PCR. Amplicon lengths are indicated to the right of each reverse primer. PCR products were purified, normalized to 10ng/uL and pooled to generate a custom internal size standard for 3130xL capillary electrophoresis RSCA.
cDNA synthesis and PCR of Chinese rhesus macaque MHC class I sequences
Synthesis of complementary DNA (cDNA) was performed using the Superscript™ III First-Strand Synthesis System for RT-PCR (Invitrogen). The highly polymorphic MHC class I exon 2/exon 3 region was amplified from the cDNA using high-fidelity Phusion™ DNA polymerase (New England BioLabs), phosphorylated forward primer 5P Refstrand (5′-[Phos]GCTACGTGGACGACACGC), and reverse primer 3′Short-RSCA (5′ TTCAGGGCGATGTAATCC). Amplicons were generated and purified under the conditions described above for custom sizing standard. Purified products were treated with λ Exonuclease (New England BioLabs) to generate single stranded antisense products for heteroduplex formation.
Preparation of fluorescently labeled reference strands
Fluorescently labeled reference strands were generated from MHC class I clones containing Mamu-B*07 (U41829), Mamu-A1*7402 (EU622036), or Mafa-B*430101 (AY958140). Sequence clones were amplified using a labeled forward primer, FAM-Refstrand-F (5′-[FAM]CTACGTGGACGACACGC) and 3′Short-RSCA-P (5′-[Phos]TTCAGGGCGATGTAATCC). Fluorescently labeled reference strands were amplified, purified, and exonuclease treated as described above to generate single stranded sense products for heteroduplex formation.
Selecting MHC class I sequences for the RSCA screening panel
Fourteen cloned MHC class I cDNAs were selected for the RSCA genotyping screening panel based on their identification in two or more of twelve previously screened Chinese rhesus macaques (Karl et al. 2007). These fourteen sequences were amplified from plasmid clones under PCR conditions described above using the 5P-Refstrand and 3′Short-RSCA primers. Products were purified and exonuclease treated as described above with resultant single stranded DNA products being added directly into a heteroduplex reaction. Screening panel clones were hybridized with each of three fluorescently labeled reference strands then combined into two separate pools of seven clones each per reference strand based on their respective electrophoretic mobility (Table 2).
Table 2.
MHC class I screening panel sequences pooled based on reference strand specific migration rates.
| Reference Strand | |||
|---|---|---|---|
| Mamu-B*07 | Mamu-Al*7402 | Mafa-B*43 | |
| Pool 1 | B*6502 | A7*0103 | B*8301 |
| A2*050202 | A2*050202 | B*9001 | |
| Al*5702 | Al*2202 | B*8701 | |
| A7*0103 | B*8701 | B*2401 | |
| B*6902 | Al*2601 | A2*050202 | |
| B*4802 | B*9001 | Al*1103 | |
| B*8301 | B*6902 | A7*0103 | |
|
|
|||
| Pool 2 | B*8701 | Al*1103 | B*4802 |
| Al*1103 | Al*5702 | A2*2601 | |
| B*1702 | B*6502 | B*6902 | |
| Al *2202 | B*2401 | Al *2202 | |
| B*2401 | B*4802 | A1*57Q2 | |
| Al*2601 | B*8301 | B*6502 | |
| B*9001 | B*17Q2 | B*17Q2 | |
The fourteen screening panel sequences exhibit distinct characteristic electrophoretic mobilities when hybridized to each of three reference strand sequences. Based on their relative mobilities, screening panel sequences were divided into two pools per reference strand, increasing the number of sequences that can be identified in a single run.
Heteroduplex formation
To form the heteroduplexes, single stranded FAM-labeled reference strands were mixed 1:5 with single stranded screening panel sequence clones or single stranded cDNA PCR products. Annealing reactions were carried out under the following conditions: 95°C for 4 minutes, 56°C for 5 minutes, and 15°C for 5 minutes. Heteroduplexed products were then purified using the Invitrogen ChargeSwitch® PCR clean up kit (Invitrogen). Four μL of heteroduplexed sample was mixed in 16μL custom ROX-labeled internal size standard and analyzed on the ABI 3130xL.
Electrophoresis
RSCA genotyping was performed on an Applied Biosystems (ABI) 3130xL genetic analyzer (Applied BioSystems, Foster City, CA, USA) managed by Data Collection software (Applied BioSystems) under the following run conditions: Oven Temperature: 18°C; Poly Fill Vol: 10000 Steps; Current Stability: 5.0 μAmps; PreRun Voltage: 15kV; PreRun Time: 180; Injection Voltage: 1.6kV; Injection Time: 15seconds (s); Voltage Number of Steps: 30 nk; Voltage Step Interval: 15 s; Data Delay Time: 200 s; Run Voltage: 4kV; Run Time: 14000 s. Fluorescently labeled heteroduplexes underwent electrophoresis through a 5% non-denaturing conformational analysis polymer (CAP) prepared by mixing 12.7g of 9% CAP (Applied BioSystems), 5.5g Urea (Fluka), 1.7g ddH20, and 1.1g 20x TTE (National Diagnostics, Atlanta, GA, USA) for 45 minutes followed by filtering through a 0.22μM filter (Nalgene, Rochester, NY, USA) (Mattocks et al. 2007). Introduction of the custom 5% CAP onto the 3130xL platform led to an instrument pre-run voltage check failure due to the change in polymer electric resistance. To overcome this issue, additional code was written into the instrument startup program to circumvent the pre-run voltage check (Applied BioSystems, personal communication; Mattocks et al. 2007). The ABI 3130xL can simultaneously resolve fluorescently labeled heteroduplexes for 16 capillaries. For this study, a single injection series contained one reference strand specific screening panel in quadruplicate and four macaques in triplicate. A 50cm array was used to improve assay resolution as compared to the standard 36cm array (Arguello and Madrigal 1999). The instrument was buffered with 1x TTE buffer (National Diagnostics) and its spectral calibration was established using Dye Set D (Applied BioSystems).
Analysis
Raw .fsa files were extracted from the 3130xL using Data Collection software (Applied Biosystems). The electrophoretic mobility of fluorescently labeled heteroduplexes was converted into peak profiles and analyzed using the Dax software package (Van Mierlo Software Consultancy, Netherlands). Macaque heteroduplexes were run in triplicate and compared to intra-run screening pool heteroduplexes. Peaks with an apparent base pair size of ± 0.5% of the screening pool heteroduplexes were scored positive. A minimum threshold for fluorescence intensity set at 10 times the run noise level was established for Dax analysis to avoid error associated with minor peaks created by the formation of alternative secondary structures.Macaques that scored positive with the ± 0.5% tolerance for a given sequence with at least two of the three reference strands were considered positive in final genotype reporting.
Results and Discussion
Adaptation of RSCA onto the 3130xl platform for MHC class I genotyping
Run conditions and the non-denaturing polymer mixture were established on the 3130xL based on a conformational sensitive capillary electrophoresis application note from the National Genetics Reference Laboratory, Wessex, UK (Mattocks et al. 2007). Commercially available internal sizing standards have been employed previously to reduce RSCA intra- and inter-experiment variability (Krebs et al. 2005; Kennedy et al. 2005). During the initial adaptation, the commercially available internal size standards MegaBACE ET900-ROX (GE Healthcare) and GeneScan 600-LIZ (Applied Biosystems) exhibited significant fluorescent signal decay while samples were in queue to be processed, severely limiting RSCA automation and throughput (Fig. 1). Using a series of fluorescently labeled amplicons (Table 1), we generated a custom internal size standard that maintained strong fluorescent signal intensity over a 24-hour period. This custom size standard, included in all capillary lanes, acts as a scaffold for establishing electrophoretic mobility and was used by the Dax software package (Van Mierlo Software Consultancy) for automated alignment of samples in all 16 capillaries to normalize inter- and intra-experiment variation (data not shown).
Figure 1. Degradation of commercial size standard signal intensity on the 3130xL platform.
Commercially available size standards GeneScan 600-LIZ and MegaBACE ET900-ROX exhibited significant degradation in signal intensity after being in queue for 8 hours, severely hindering data analysis and sample throughput. The commercially available internal sizing standards and our custom designed internal size standard were analyzed on the ABI 3130xL after waiting in queue for 8 hours and the relative signal intensities were compared.
Establishing a screening panel for RSCA genotyping
Fourteen rhesus macaque MHC class I cDNA clones were selected as a screening panel for RSCA genotyping based on their identification in multiple macaques (Karl et al. 2007). The highly polymorphic exon 2/exon 3 region for all fourteen MHC class I sequences were aligned with the sequence for Mamu-B*07 reference strand using the LaserGene MegAlign program (DNASTAR, Madison, WI, USA) to illustrate the wide range of sequence variation among these sequences. For example, the heteroduplex between Mamu-B*1702 and the Mamu-B*07 reference strand contains 22 mismatched base pairs. These fourteen sequences were hybridized with a reference strand to generate screening panel heteroduplexes. To reduce genotyping errors and ambiguities due to co-migration of different MHC class I sequences (Baquero et al. 2006; Kennedy et al. 2005; Krebs et al. 2005; Pratt et al. 2006; Tanaka-Takahashi et al. 2007), we used three diverse reference strands for heteroduplex formation: Mamu-A1*7402, Mamu-B*07, and Mafa-B*430101 (Fig. 2). The screening panel heteroduplex migration rates were established through a series of RSCA runs on the 3130xL (Fig. 3). We found an inter-run variance of less than 0.5% total apparent base pair mobility for 41/42 (98%) screening panel heteroduplexes. The only exception was the Mamu-A1*7402/Mamu-B*6902 heteroduplex whose mobility varied 0.69% between runs. Based on their relative mobilities, screening panel clones were divided into two pools per reference strand to increase the number of MHC class I sequences that can be screened for in a single RSCA run (Table 2).
Figure 2. Use of three reference strands to vary heteroduplex migration rates for improved genotyping accuracy.
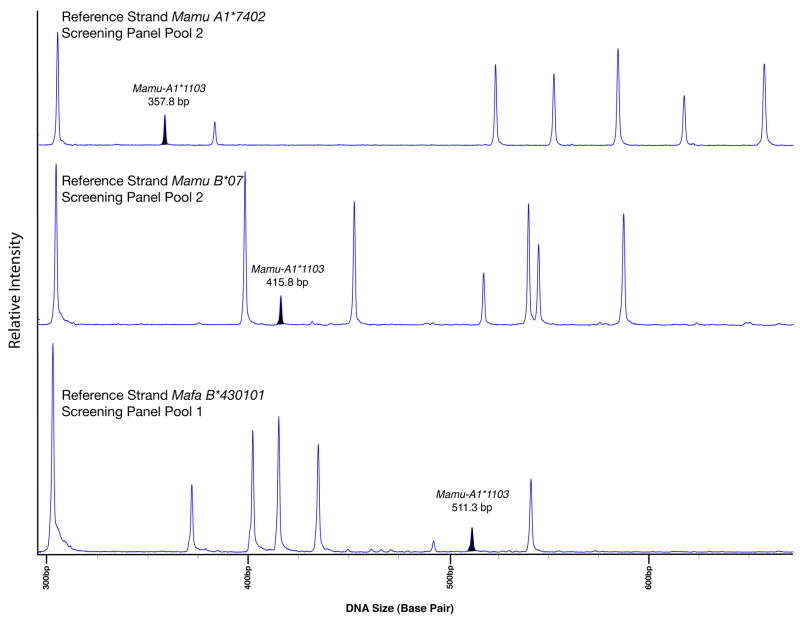
The heteroduplexes formed when screening panel sequences are hybridized with a reference strand exhibit characteristic electrophoretic mobilities. Instances in which these heteroduplexes co-migrate make genotyping more difficult. To improve genotyping resolution and overcome genotyping errors and ambiguities, the screening panel clones were hybridized to each of three distinct reference strands: Mamu-A1*7402, Mamu-B*07, and Mafa-B*430101. This example illustrates the variation in migration rate for screening panel sequence Mamu-A1*1103 when heteroduplexed with Mamu-A1*7402 (357.8 bp), Mamu-B*07 (415.8 bp), and Mafa-B*430101 (511.3 bp). The prominent peak at 304 bp is the Mamu-B*07 reference strand homoduplex.
Figure 3. Migration values for fourteen screening panel MHC class I sequences bound to each of three fluorescently labeled reference strands.
The relative electrophoresis mobility of each screening panel MHC sequence changes dramatically depending on the reference strand used for heteroduplex formation. Migrations rates for all fourteen MHC class I sequences were determined by averaging the heteroduplex mobility from three independent instrument runs. The relative migration rates for each heteroduplex were taken into account when pooling sequences for screening panels.
Validation of RSCA capillary electrophoresis MHC class I genotyping
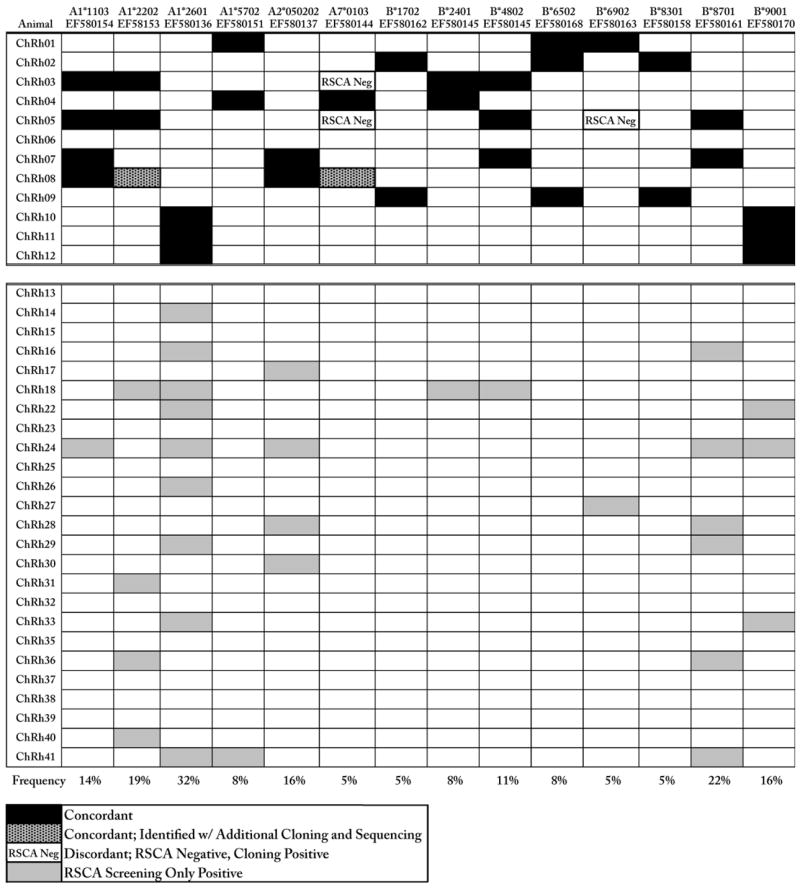
To validate the ability of RSCA to identify the 14 screening panel sequences in animals samples, cDNA PCR amplicons from twelve Chinese rhesus macaques, previously used for MHC class I sequence discovery (Karl et al. 2008), were hybridized to the same three fluorescently labeled reference strands used to establish the screening panel. Capillary RSCA of the resultant heteroduplexes was compared against the appropriate screening panels to analyze and score each animal as described above. RSCA genotyping results were then compared to the previous cloning and sequencing data and we found a 98% concordance (165/168) between these two methods. Eight-eight percent of these positive RSCA scores (30/34) were concordant in all three reference strands. RSCA yielded only three false negatives for 168 expected genotypes (Table 3). The screening panel sequence Mamu-A7*0103 was discordant in two out of the three of the false negatives and therefore may be a poor candidate for future RSCA genotyping studies when hybridized with these specific reference strands. The discordant sequence Mamu-B*6902 has a single nucleotide mismatch under one of the amplification primers that may have influenced our ability to consistently genotype for this sequence (Supplemental Fig 1). Further investigation into the preparation and electrophoresis of the discordant sequences is needed to identify any possible sources underlying the validation typing discordance, however the transcription level of these sequences may also play a significant role in determining the ability of RSCA to accurately genotype. Additional cloning and sequencing performed on ChRh08 confirmed the presence of two sequences, Mamu-A1*2202 and Mamu-A7*0103 that were detected by capillary electrophoresis RSCA but had been missed previously due to limited cloning and sequencing.
Table 3.
The concordance of RSCA genotyping for 12 previously screened Chinese rhesus macaques and the frequency of 14 MHC class I alleles in 37 Chinese rhesus macaques.
|
The concordance of RSCA genotyping with cDNA cloning and sequencing was determined for 12 Chinese rhesus macaques (upper panel). The frequencies of the 14 screening panel sequences were determined using an additional 25 Chinese rhesus macaques (bottom row). Nine macaques (ChRh13, 15, 23, 25, 32, 35, 37–39) did not express any of the 14 screening panel clones.
MHC class I haplotype sharing
RSCA profiles for three Chinese rhesus macaques (ChRh10, 11, and 12) previously shown to be haploidentical by microsatellite analysis were examined for potential sharing of MHC class I sequences (Karl et al. 2008). All three macaques were positive for Mamu-A1*2601 and Mamu-B*9001 by capillary electrophoresis RSCA, as predicted by cloning and sequencing. Moreover, these macaques exhibited nearly identical peak profiles for each reference strand (Fig. 4). All three macaques shared at least five unknown heteroduplexes with each reference strand (Fig. 4, data not shown). Additional cloning and sequencing of the RSCA cDNA PCR products suggests that at least two of these shared sequences are Mamu-A4*1403 and a Mamu-I sequence. The ability to identify both known and unknown shared MHC class I sequences in rhesus macaques highlights the versatility of capillary RSCA genotyping.
Figure 4. Capillary RSCA identifies three haploidentical Chinese rhesus macaques.
Capillary electrophoresis RSCA confirmed previously reported haplotype sharing between three Chinese rhesus macaques (ChRh10–12). RSCA heteroduplex profiles for the three macaques are aligned below the Mamu-B*07 reference strand screening panel. Unknown shared heteroduplexes (A–E) are indicated with open arrows.
Frequency of 14 MHC class I sequences in 37 Chinese rhesus macaques
After validating our capillary electrophoresis RSCA method with Chinese rhesus macaques of known MHC class I genotypes, we determined the frequency of the fourteen MHC class I screening panel sequences in a total of 37 Chinese rhesus macaques (Table 3). Our results show that MHC class I frequencies range between 5 and 32% with two MHC class I sequences identified in more than 20% of this cohort; Mamu-A1*2601 was present in 32% and Mamu-B*8701 was identified in 22% of the macaques genotyped. Interestingly, the Mamu-A1*2601 cDNA has been previously described in rhesus macaque of both Indian and Chinese origin (Otting et al., 2005; Otting et al. 2007; Karl et al. 2008). Given the largely distinct allelic repertoire of these geographically distinct macaque populations, this suggests that Mamu-A1*2601 is an unusually well conserved allele and merits further study.
Capillary electrophoresis RSCA for MHC class I genotyping
Current techniques for MHC class I genotyping in rhesus macaques are either high resolution but expensive or low resolution and limited to only known sequences. Although previous studies have used RSCA for MHC genotyping, cumbersome assay preparation and the loss of support for slab gel instrumentation necessitate a need for further assay optimization. Our results demonstrate that capillary RSCA on the widely available ABI 3130xL is an effective tool for simultaneously typing entire cohorts of Chinese rhesus macaques for multiple known and unknown MHC class I sequences.
RSCA can easily be expanded to screen for additional known MHC class I sequences and the ability to pool screening panel sequences drastically increases the breadth of information gleaned from a single instrument run. In addition, the ability of RSCA to detect the presence of both known and unknown MHC sequences is particularly useful for genotyping nonhuman primates less studied than Indian rhesus macaques, as well as other model organisms that lack complete MHC sequence databases. However, the current setup for capillary RSCA does have limitations. The use of non-denaturing polymer for capillary RSCA is an inconvenience for investigators who currently use Performance Optimized Polymer for sequencing and fragment analysis applications on the 3130xL platform. To perform these assays on a single instrument, the capillary array must be thoroughly flushed and filled with the appropriate polymer when switching between assays.
The ability of capillary RSCA to screen multiple macaques within a single run for both known and unknown MHC class I sequences makes it especially appealing for researchers who intend to screen for shared MHC genotypes in large macaque cohorts. Our successful adaptation of capillary electrophoresis RSCA for MHC class I genotyping of Chinese rhesus macaques is an important step forward in the development of increased throughput methods for high resolution MHC genotyping of nonhuman primates.
Supplementary Material
Acknowledgments
This publication was made possible in part by grant number P51 RR000167 and grant number R24RR021745 from the National Center for Research Resources (NCRR), a component of the National Institutes of Health (NIH), to the Wisconsin National Primate Research Center, University of Wisconsin—Madison. This research was conducted in part at a facility constructed with support from Research Facilities Improvement Program grant numbers RR15459-01 and RR020141-01. This work was supported by NIAID contract number HHSN266200400088C and NIH grant 1R21AI068488-01A2. This publication’s contents are solely the responsibility of the authors and do not necessarily represent the official views of NCRR or NIH.
The authors would like to thank Shelby O’Connor, Justin Greene, Leo Spaceman, Jason Weinfurter, and other members of the O’Connor lab for their helpful discussions. We thank Nel Otting, Natasja de Groot, and the IMGT for assigning uniform allele nomenclature.
References
- Arguello JR, Madrigal JA. HLA typing by Reference Strand Mediated Conformation Analysis (RSCA) Rev Immunogenet. 1999;1(2):209–219. [PubMed] [Google Scholar]
- Baquero JE, Miranda S, Murillo O, Mateus H, Trujillo E, Suarez C, Patarroyo ME, Parra-Lopez C. Reference strand conformational analysis (RSCA) is a valuable tool in identifying MHC-DRB sequences in three species of Aotus monkeys. Immunogenetics. 2006;58(7):590–597. doi: 10.1007/s00251-006-0101-x. [DOI] [PubMed] [Google Scholar]
- Bontrop RE, Watkins DI. MHC polymorphism: AIDS susceptibility in non-human primates. Trends Immunol. 2005;26(4):227–233. doi: 10.1016/j.it.2005.02.003. [DOI] [PubMed] [Google Scholar]
- Gibbs RA, Rogers J, Katze MG, Bumgarner R, Weinstock GM, Mardis ER, Remington KA, Strausberg RL, Venter JC, Wilson RK, et al. Evolutionary and biomedical insights from the rhesus macaque genome. Science. 2007;316(5822):222–234. doi: 10.1126/science.1139247. [DOI] [PubMed] [Google Scholar]
- Kaizu M, Borchardt GJ, Glidden CE, Fisk DL, Loffredo JT, Watkins DI, Rehrauer WM. Molecular typing of major histocompatibility complex class I alleles in the Indian rhesus macaque which restrict SIV CD8+ T cell epitopes. Immunogenetics. 2007;59(9):693–703. doi: 10.1007/s00251-007-0233-7. [DOI] [PubMed] [Google Scholar]
- Karl JA, Wiseman RW, Campbell KJ, Blasky AJ, Hughes AL, Ferguson B, Read DS, O’Connor DH. Identification of MHC class I sequences in Chinese-origin rhesus macaques. Immunogenetics. 2007;60(1):37–46. doi: 10.1007/s00251-007-0267-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kaslow RA, Carrington M, Apple R, Park L, Munoz A, Saah AJ, Goedert JJ, Winkler C, O’Brien SJ, Rinaldo C, et al. Influence of combinations of human major histocompatibility complex genes on the course of HIV-1 infection. Nat Med. 1996;2(4):405–411. doi: 10.1038/nm0496-405. [DOI] [PubMed] [Google Scholar]
- Kennedy LJ, Quarmby S, Fretwell N, Martin AJ, Jones PG, Jones CA, Ollier WE. High-resolution characterization of the canine DLA-DRB1 locus using reference strand-mediated conformational analysis. J Hered. 2005;96(7):836–842. doi: 10.1093/jhered/esi112. [DOI] [PubMed] [Google Scholar]
- Krebs KC, Jin Z, Rudersdorf R, Hughes AL, O’Connor DH. Unusually high frequency MHC class I alleles in Mauritian origin cynomolgus macaques. J Immunol. 2005;175(8):5230–5239. doi: 10.4049/jimmunol.175.8.5230. [DOI] [PubMed] [Google Scholar]
- Loffredo JT, Bean AT, Beal DR, Leon EJ, May GE, Piaskowski SM, Furlott JR, Reed J, Musani SK, Rakasz EG, et al. Patterns of CD8+ Immunodominance May Influence the Ability of Mamu-B*08-positive Macaques to Naturally Control SIVmac239 Replication. J Virol. 2007;82(4):1723–1738. doi: 10.1128/JVI.02084-07. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mattocks C, White H, Owen N, Ward D. Conformational Sensitive Capillary Electroporesis, National Genetics Reference Labortory. 2007 www.ngrl.org.uk/wessex.
- Martin MP, Carrington M. Immunogenetics of viral infections. Curr Opin Immunol. 2005;17(5):510–516. doi: 10.1016/j.coi.2005.07.012. [DOI] [PubMed] [Google Scholar]
- Mee E, Badhan A, Karl JA, RW W, Cutler K, Knapp LA, Almond N, O’Connor DH, Rose N. MHC haplotype frequencies in a UK breeding colony of Mauritian cynomolgus macaques mirror those found in a distinct population from the same geographic origin. Journal of Medical Primatology. 2008 doi: 10.1111/j.1600-0684.2008.00299.x. In Press. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Otting N, de Vos-Rouweler AJ, Heijmans CM, de Groot NG, Doxiadis GG, Bontrop RE. MHC class I A region diversity and polymorphism in macaque species. Immunogenetics. 2007;59(5):367–375. doi: 10.1007/s00251-007-0201-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Otting N, Heijmans CM, Noort RC, de Groot NG, Doxiadis GG, van Rood JJ, Watkins DI, Bontrop RE. Unparalleled complexity of the MHC class I region in rhesus macaques. Proc Natl Acad Sci U S A. 2005;102(5):1626–1631. doi: 10.1073/pnas.0409084102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Penedo MC, Bontrop RE, Heijmans CM, Otting N, Noort R, Rouweler AJ, de Groot N, de Groot NG, Ward T, Doxiadis GG. Microsatellite typing of the rhesus macaque MHC region. Immunogenetics. 2005;57(3–4):198–209. doi: 10.1007/s00251-005-0787-1. [DOI] [PubMed] [Google Scholar]
- Pratt BF, O’Connor DH, Lafont BA, Mankowski JL, Fernandez CS, Triastuti R, Brooks AG, Kent SJ, Smith MZ. MHC class I allele frequencies in pigtail macaques of diverse origin. Immunogenetics. 2006;58(12):995–1001. doi: 10.1007/s00251-006-0164-8. [DOI] [PubMed] [Google Scholar]
- Smith MZ, Dale CJ, De Rose R, Stratov I, Fernandez CS, Brooks AG, Weinfurter J, Krebs K, Riek C, Watkins DI, et al. Analysis of pigtail macaque major histocompatibility complex class I molecules presenting immunodominant simian immunodeficiency virus epitopes. J Virol. 2005;79(2):684–695. doi: 10.1128/JVI.79.2.684-695.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tanaka-Takahashi Y, Yasunami M, Naruse T, Hinohara K, Matano T, Mori K, Miyazawa M, Honda M, Yasutomi Y, Nagai Y, et al. Reference strand-mediated conformation analysis-based typing of multiple alleles in the rhesus macaque MHC class I Mamu-A and Mamu-B loci. Electrophoresis. 2007;28(6):918–924. doi: 10.1002/elps.200600586. [DOI] [PubMed] [Google Scholar]
- Wiseman RW, O’Connor DH. Major histocompatibility complex-defined macaques in transplantation research. Transplantation Reviews. 2007;21:17–25. [Google Scholar]
- Wiseman RW, Wojcechowskyj JA, Greene JM, Blasky AJ, Gopon T, Soma T, Friedrich TC, O’Connor SL, O’Connor DH. Simian immunodeficiency virus SIVmac239 infection of major histocompatibility complex-identical cynomolgus macaques from Mauritius. J Virol. 2007;81(1):349–361. doi: 10.1128/JVI.01841-06. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.