Abstract
Deposits in macular human Bruch’s membrane (BrM) increase with age and have been postulated to be associated with age-related maculopathy. We used two ultrastructural methods to compare these deposits by electron microscopy in macular and peripheral BrM of eight eyes from donors 63–86 years of age. Quick-freeze/deep-etch (QFDE) was used to prepare replicas that showed the ultrastructure of deposits, and osmium—tannic acid—paraphenylenediamine (OTAP) was used to preserve small extracellular lipid particles. We found that an accumulation of lipoprotein-like particles (LLPs) occurred in the peripheral BrM just as it does in the macular region, but with perhaps a somewhat slower time course. The ”lipid wall,” reported in macular BrM, was also found occasionally in the peripheral regions. The same processes that lead to age-related accumulation of LLPs in macular BrM appear to also occur in the peripheral regions.
Keywords: AMD, basal lamina, collagen, elastin, lipoprotein, morphology
INTRODUCTION
Age-related maculopathy (ARM) is defined clinically by the appearance of drusen and retinal pigment epithelium (RPE) changes in the macula. However, drusen are also frequently seen in the periphery of normal eyes and in some eyes with ARM.1,2 The question arises as to whether the same disease process that occurs in the macular region might also be occurring in the periphery.
The most prominent histopathologic lesions seen in the earlier stages of ARM involve Bruch’s membrane (BrM), the thin connective tissue that lies between the RPE and the choriocapillaris, the blood supply of the photoreceptors. BrM undergoes a variety of age-related changes. These include increased deposition of lipids3–5 that is likely related to the age-related accumulation of lipoprotein-like particles (LLPs) seen in macular BrM using the quick-freeze/deep-etch technique (QFDE).6,7 The age-related accumulation of LLPs in the macula leads to the formation of a lipid wall that could inhibit transport through this region. The deposition of lipids in BrM correlates well with the dramatic age-related increase in hydraulic resistivity of this tissue.8,9
Our more recent examination of the age-related changes in BrM using QFDE determined that deposition of LLPs (most 60–80 nm in diameter but up to 300 nm) and other small granules (10 nm in diameter) in macular BrM appears to begin during the fourth decade of life in or near the elastic layer. Then, in the next 2–4 decades of life, the inner collagenous layer (ICL) is progressively filled, leading eventually to the formation of a lipid wall between the ICL and the basal lamina of the RPE.7
Increased age-related deposition of lipids has also been reported in peripheral BrM.4,5,10 In this study, we use QFDE and osmium—tannic acid—paraphenylenediamine (OTAP) to compare macular and peripheral deposition of LLPs and ultrastructural changes in BrM of aged eyes. Our goal is to determine if the same age-related process that occurs in macular BrM also occurs in the peripheral region.
MATERIALS AND METHODS
Eye Tissues
Human eyes without grossly visible drusen larger than 63 μm in diameter, pigmentary changes, or other chorioretinal pathology were obtained from the Alabama Eye Bank and preserved within 4 hr postmortem. Eyes from donors with diabetes or receiving artificial respiration longer than 5 days were excluded. A total of eight eyes were examined in this study from donors with ages of 63, 64, 72, 76, 78, 80, 84, and 86 years. Due to technical problems, tissue from the 86-year-old eye was processed for OTAP only.
The posterior segment of the normal eyes was preserved by immersion in 0.1 M phosphate buffer solution with 2.5% glutaraldehyde and 1% paraformaldehyde for at least 24 hr after the removal of anterior segment. Macular samples were taken from a 8 × 8 mm square centered on the fovea; peripheral samples were taken from an 8 × 8 mm square centered 13 mm temporal to the fovea. The samples, including retina, BrM, choroid, and sclera, were then dissected into 2 × 2 mm square blocks for QFDE or thin-sectioning transmission electron microscopy (TEM) processing.
QFDE Processing
For eyes examined by QFDE, the retina was removed using fine forceps. The chorioretinal juncture (RPE, BrM, and choriocapillaris) was then lifted carefully from the sclera and replaced. This facilitated later separation at this location. The RPE-BrM-choroid-sclera complex was slammed frozen in liquid nitrogen (-196°C) in a Leica EM MM80E Freeze Slammer (Leica Microsystems Inc., Bannockburn, IL, USA). The frozen tissue, with the RPE side facing up, was then transferred to a freeze-fracture deep-etch chamber (Cressington CFE-60; Cressington Scientific Instruments Ltd., Watford, UK) held at a vacuum of 10–7 mbar.
To reveal BrM, the tissue block was fractured at a very low oblique angle and then etched at -95°C for 25 min. After etching, a replica of the surface of the tissue was produced by rotary shadowing using a platinum/carbon mixture at 20° to the tissue surface and then backed by carbon. The replica and tissue were placed into a digestion solution (water/bleach = 1:1). The sclera was removed soon after the start of immersion into the solution. The replica and the remained chorioretinal juncture were immersed in the solution for at least 2 hr to remove the tissue. The replica pieces were then washed in deionized water, picked up by copper hexagonal grids, air dried, and then examined under TEM (JEOL100 CX; JEOL USA, Peabody, MA). Micrographs of the replica were scanned (AGFA Duoscan T2500; AGFA USA, Ridgefield Park, NJ, USA) in TIFF mode.
Thin-Sectioning TEM
For each eye, one or two fixed tissue blocks from both the macular and peripheral regions each were also postfixed using OTAP to preserve neutral lipid components.11,12 Tissue postfixed by the OTAP method was treated with 1% osmium in 0.1 M sodium cacodylate buffer (2.5 hr), 1% tannic acid (gallotannin, C14H10O9, 30 min), 1% sodium sulfate (Na2SO4, 5 min), 70% ethanol (3 × 5 min), and 1% paraphenylenediamine in 70% ethanol (30 min).12 Tissues were dehydrated, infiltrated, and embedded in PolyBed 812 (Polysciences, Warrington, PA, USA). A 1-μm-thick section was stained with 1% toluidine-blue-O in 2% sodium borohydrate. Thin sections were examined using a JEOL1200 EXII (JEOL USA) electron microscope. Representative negatives of thin sections (Kodak EM film 4489) were scanned with a PowerLook 1100 scanner and Umax Magiscan 4.5 (Umax Technology, Dallas, TX, USA).
As previously described,12 use of the OTAP method resulted in a gradation of lipid preservation. A few sections showed excellent lipid preservation, but most sections showed “continuous rather than dichotomous range of preservation quality.” In sections in which LLP interiors were empty, we refer to the micrographs as thin-sections rather than as OTAP images, as the lipids were not preserved in those sections. The LLPs were still apparent, but only the borders of the particles were electron dense. Note that in both the OTAP and thin-section images, we show these images as negatives to match the contrast polarity of the QFDE images (holes appear dark in all images).
RESULTS
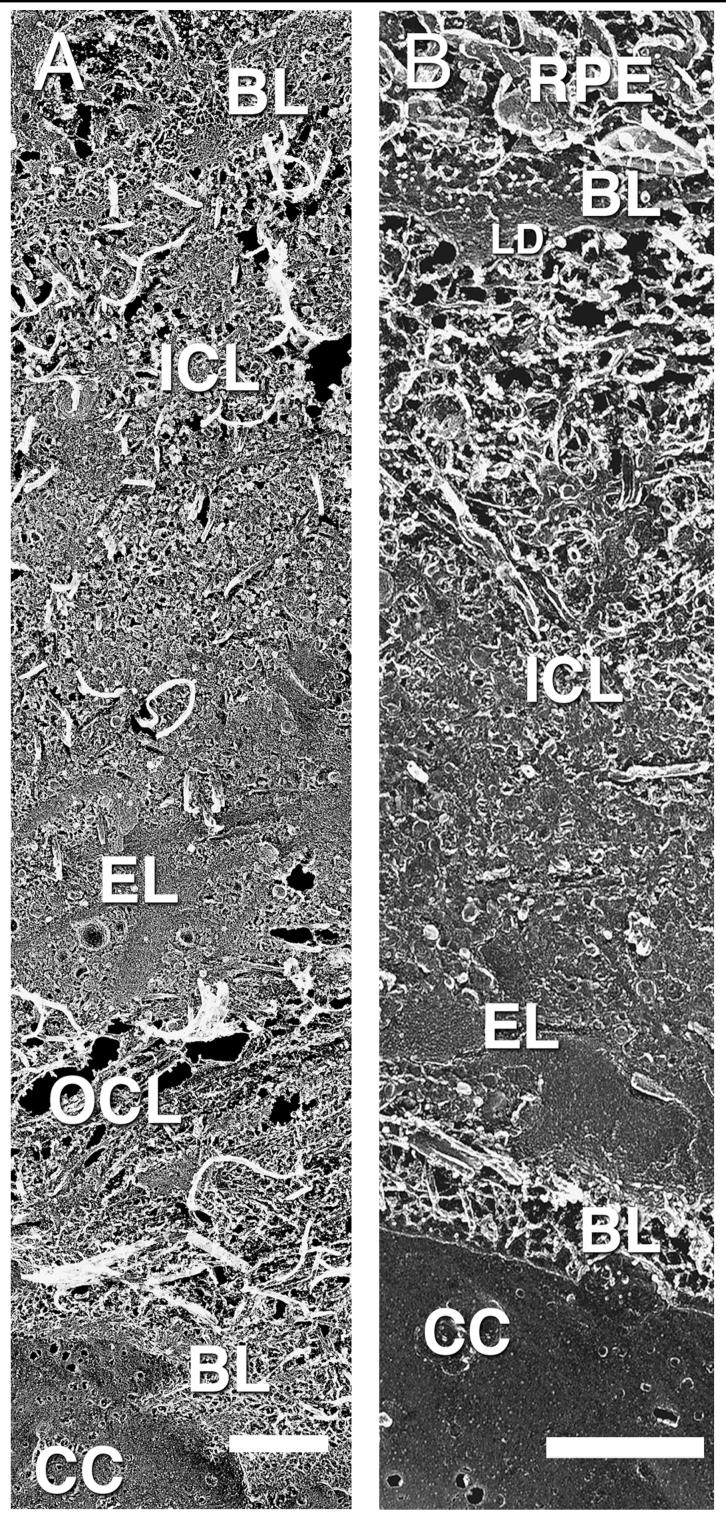
A summary of our finding is found in Table 1. The overall appearances of BrM as seen by QFDE are shown in Figures 1A (macula) and 1B (periphery). The samples are fractured at a very oblique angle to produce these replicas. As such, the distance between the RPE and choriocapillaris is much larger than the typical cross-sectional thickness of BrM. The dense region of basal lamina (BL) of the RPE seen in Figure 1B is the lamina densa (LD); this region is not as apparent in Figure 1A. Collagen fibrils are seen to be abundant in the ICL of both figures. Each image also shows a prominent elastic layer (EL), but the outer collagenous layer (OCL) is not seen in Figure 1B, because the plane of fracture jumps vertically downward from the elastic layer to the basal lamina of the choriocapillaris in this replica. This is not an infrequent occurrence in our replicas. The choriocapillaris and its basal lamina are clearly evident in both macula and periphery. LLPs can be seen as small particulates in both macular and peripheral regions, but they are not easily distinguished at this low magnification. These particles are more easily seen in the higher-magnification QFDE and OTAP images shown below.
TABLE 1.
Summary of Findings
| Layer of BrM |
Macular region |
Peripheral region |
|---|---|---|
| Basal lamina of RPE |
No LLPs present | No LLPs present |
| Lipid wall | Found in some eyes in seventies; found in most eyes in eighties |
Not found as often as in macular region; more often found in eyes in eighties |
| ICL | Progressively filled with LLPs with aging |
Progressively filled with LLPs with aging, but a little slower than in macular region |
| Elastic layer | Layer filled with LLPs |
Layer filled with LLPs |
| OCL | Few LLPs present in older eyes |
Few LLPs present in older eyes |
| Basal lamina of choriocapillaris |
No LLPs present | No LLPs present |
FIGURE 1.
Replica showing macular (A) and peripheral (B) BrM from the RPE at the top of the figures to the choriocapillaris on the bottom. Basal lamina (BL), inner collagenous layer (ICL), elastic layer (EL), outer collagenous layer (OCL), choriocapillaris (CC), lamina densa (LD); 63-year-old eye. Note that panels (A) and (B) are of different magnifications, because the fracture angle is different in (A) than (B). They are included in this illustration because these replicas showed the most complete demonstration of all layers in both regions. Bars are 1 μm.
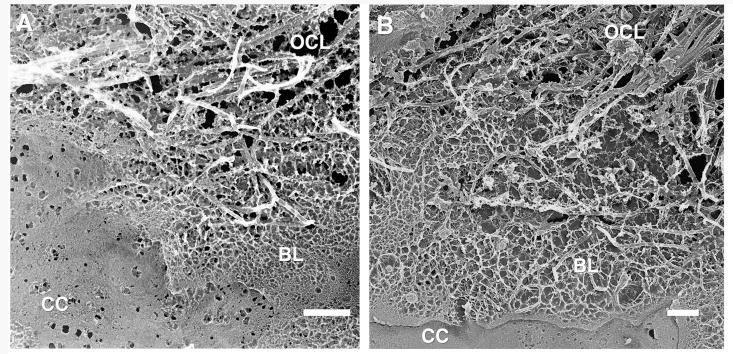
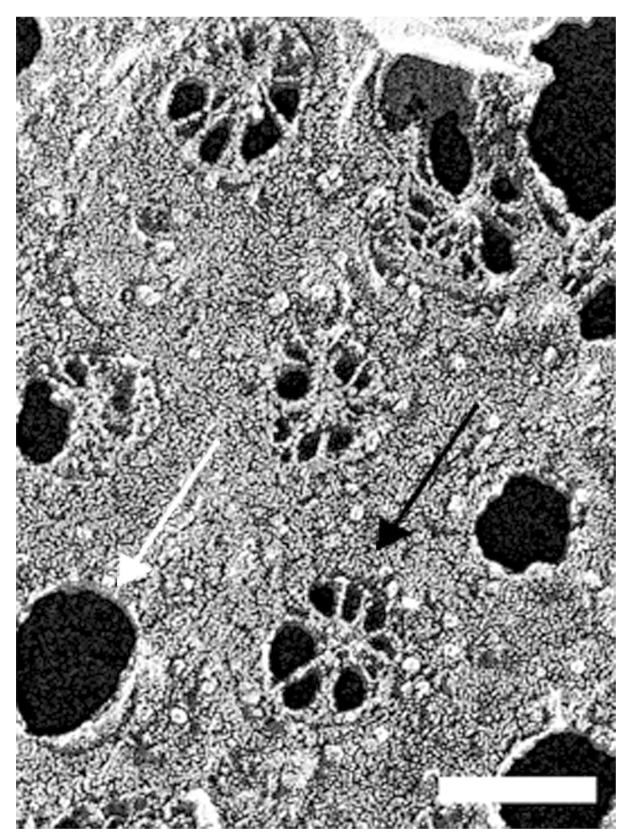
Figure 2 shows the choriocapillaris and the outermost layers of BrM, the basal lamina of the choriocapillaris, and the OCL. These were all very similar in structure in the macular (Fig. 2A) and peripheral regions (Fig. 2B). The small holes seen in the choriocapillaris are artifacts that likely occur at locations of fenestrations. Occasionally, we saw intact fenestra as shown in Figure 3. There was no significant accumulation of LLPs in either the macula or periphery of the basal lamina of the choriocapillaris or the OCL of these older eyes, consistent with our other studies.6,7
FIGURE 2.
Replica showing choriocapillaris (CC), basal lamina (BL), and the outer collagenous layer (OCL) of macular (A) and peripheral (B) BrM; 63-year-old eye. Bars are 500 nm.
FIGURE 3.
Surface of the endothelium of the choriocapillaris showing several fenestrations (dark arrow) and presumed artifactual pores (white arrow) that formed during tissue preparation for QFDE; 64-year-old eye, macular region. Bar is 100 nm.
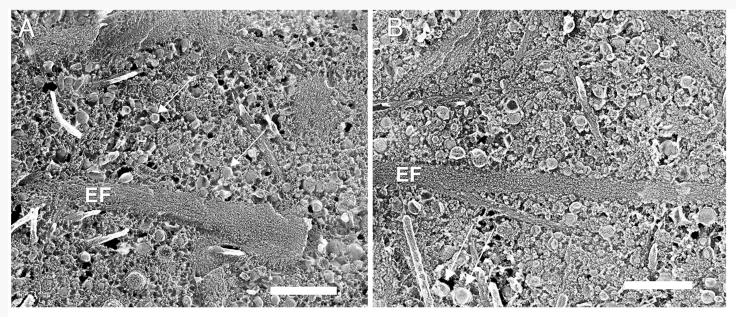
We have recently shown that the age-related accumulation of LLPs in macular BrM occurs earliest in or near the elastic layer.7,12 In eyes greater than 60 years of age, this layer is always filled with LLP and other debris.7 For the current study, in both macular and peripheral regions, this layer was also densely packed with elastic fibers (EF), LLPs, and other small extracellular debris in these older eyes (Fig. 4). Similar results were seen in our OTAP images. In a 64-year-old eye, many LLPs were seen to be associated with the elastic layer in both macular and peripheral regions (Fig. 5). As we have previously reported,7 LLPs were sometimes associated with elastin and collagen fibrils by fine filaments, both in macular and peripheral regions. However, LLPs were also found without direct contact to either collagen or elastin fibrils (Fig. 4).
FIGURE 4.
Replica of the elastic layer in macular (A) and peripheral (B) BrM of a 63-year-old eye. Arrows point to LLPs. Elastic fiber (EF). Bars are 500 nm.
FIGURE 5.
Negative contrast OTAP images of macular (A) and peripheral (B) BrM from a 64-year-old donor. Basal lamina of the RPE (BL), inner collagenous layer (ICL), elastic fiber (EF), outer collagenous layer (OCL), endothelial cell (E), red blood cell (RBC). The small white dots are collagen fibrils cut in cross section. (A) Arrow shows four LLPs in elastic layer. (B) Arrows show two LLPs, one of which was only partially preserved by the OTAP. Bars are 500 nm.
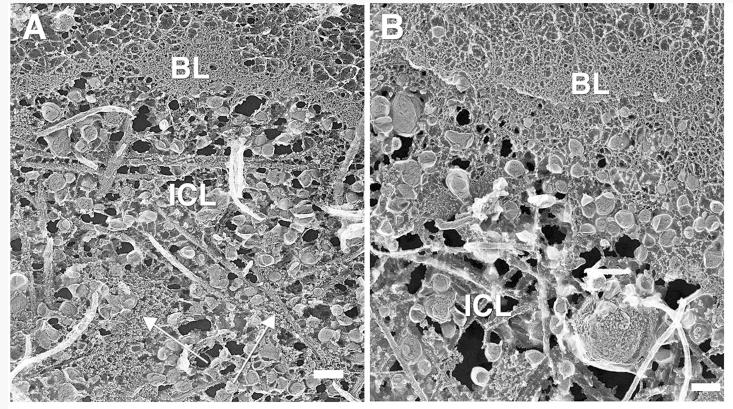
With aging, there was a progressive filling of the ICL with debris, particularly LLPs and small granules.6,7 In the 64-year-old eye, accumulations of LLPs were seen in the ICL of both the macula (Fig. 6A) and periphery (Fig. 6B). Also seen in the macular region was what might be early accumulation of the small granules in the ICL, although these appeared mostly clumped in this eye. We have recently reported that these small granules accumulate in BrM concurrently with the age-related accumulation of LLPs.7
FIGURE 6.
Replicas of the ICL and basal lamina (BL) of the RPE from the macula (A) and periphery (B) of BrM; 64-year-old eye. Arrows in (A) point to clumps of small granules. Bars are 200 nm.
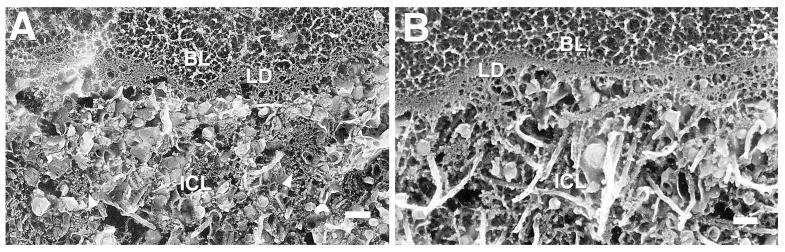
In eyes more than 70 years of age, the collagen fibrils are not as apparent in the ICL as this region now contained many LLPs and small granules. In the macular ICL (Fig. 7A), many LLPs and numerous small granules were seen among a few collagen fibrils. There were more collagen fibrils and fewer LLPs and small granules in the peripheral ICL (Fig. 7B), suggesting that the process here might not be as advanced as in the macular region. The structure of individual LLPs and small granules were similar in macula and periphery. OTAP images showed more lipid accumulation in the macular ICL than in the peripheral ICL of this eye (Fig. 8).
FIGURE 7.
Replica of the ICL layer in the macular (A) and peripheral (B) regions of a 76-year-old eye. Arrowheads in (A) point at a few of the many small granules. Many LLPs and a few collagen fibrils are evident. Lamina densa (LD). Bars are 200 nm.
FIGURE 8.
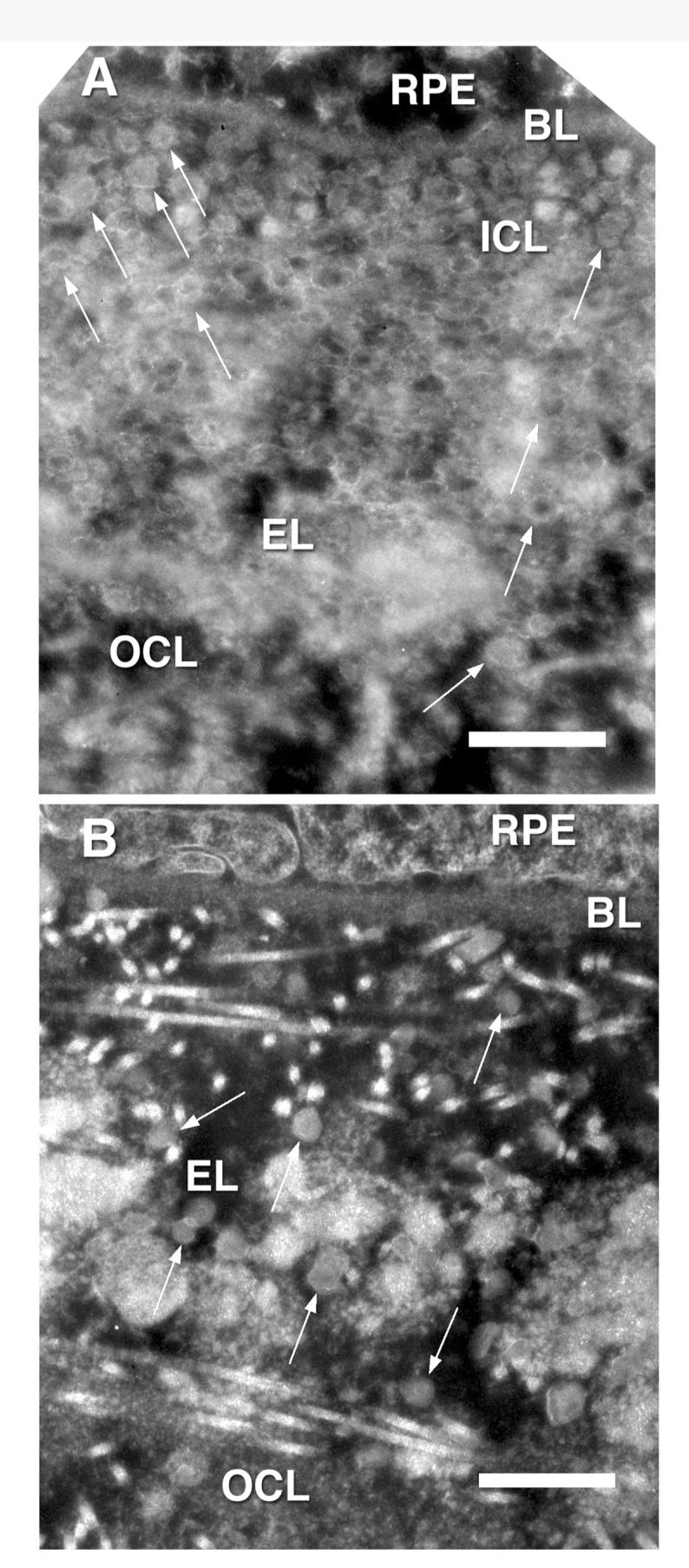
Negative contrast OTAP images of the macular (A) and peripheral (B) regions of BrM of a 76-year-old eye. Arrows point at LLPs that in OTAP images are bright on their boundaries and usually somewhat darker in their core. Rod-like structures and small bright dots in (B) are collagen fibrils. Basal lamina of the RPE (BL), elastic fiber (EF). Bars are 500 nm.
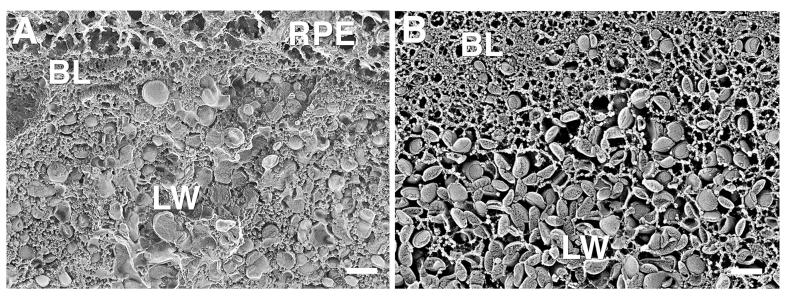
In some eyes more than 70 years of age, and most eyes over age 80, a new layer was seen in macular BrM that we previously called the “lipid wall”6 (Fig. 9). This layer, nearly filled with LLPs and devoid of collagen, was found between the ICL and the basal lamina of the RPE. The latter was usually free of LLPs, but in eyes in which a lipid wall had formed, the LLPs sometimes appeared to be crowded into this structure (Fig. 9A). As shown in Figure 10, the lipid wall sometimes also contained many small granules.
FIGURE 9.
Two distinct appearances of lipid wall (LW) are seen in this 78-year-old eye. The macula (A) shows many LLPs and small granules, and the periphery (B) shows mainly LLPs in the lipid wall. Bars are 200 nm.
FIGURE 10.
Detailed view of lipid wall layer in macula of 78-year-old eye. Many small granules surround the LLPs. Bar is 100 nm.
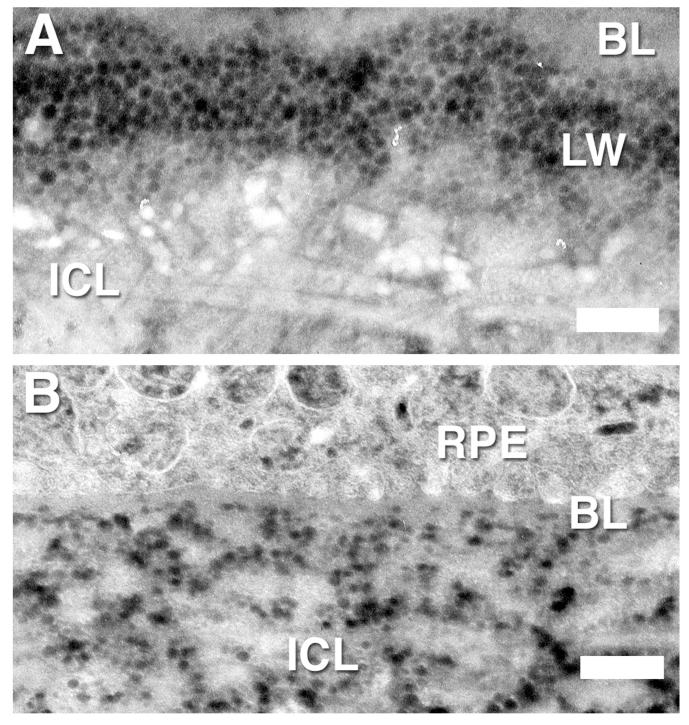
Of note in this study, we saw the lipid wall more often in the macular region of these older eyes, but we did see it also in the peripheral region (Fig. 9B). In some eyes, the lipid wall was further developed in the macular region than in the peripheral (Fig. 11), but in other eyes, they looked remarkably similar. The thin section results were, for the most part, consistent with our QFDE findings and also showed formation of the lipid walls (Fig. 11A). However, in some eyes, the lipid wall appeared in some sections but not in others (compare Figs. 9B and 11B).
FIGURE 11.
Negative-contrast thin-section images of the region between inner collagenous layer (ICL) and basal lamina (BL) of RPE in the macula (A) and periphery (B) of BrM of a 78-year-old eye. Lipid wall (LW). Bar is 500 nm.
DISCUSSION
In this study, we examined BrM of older eyes, comparing macular and peripheral regions. Previous work by our group6,7,12 has shown significant age-related accumulation of LLPs and other debris in macular BrM. This accumulation began in or near the elastic layer that progressively led to an accumulation in the ICL, finally resulting in the formation of a “lipid wall” in some older eyes between the ICL and the basal lamina of the RPE.6,7 Along with LLPs, we saw an accumulation in BrM of small granules, roughly 10 nm in diameter.7 We do not know the identity of these particles. However, they look similar to particles seen in the basal lamina, particularly the lamina densa (Figs. 7 and 9B).13,14
In the current study, we compared the debris accumulation of older BrM in the macular region with that seen in the peripheral region. We found that peripheral BrM looked similar to macular BrM in these older eyes. Both regions showed significant accumulation of extracellular material, particularly LLPs, in the elastic layer and the ICL. There was a tendency to see a greater number of these particles in the macular region compared with the periphery, but the differences were not dramatic. Good agreement was seen between those images prepared by QFDE and those prepared using OTAP.
Age-related changes were observed in the ICL of both the macular and peripheral tissue. We did not notice age-related changes in the elastic layer and OCL, as changes in those tissues occur in eyes younger than the eyes examined in the current study.10 Nonetheless, our findings are consistent with those we have previously reported. In the older eyes examined for the current study, in both the macular and peripheral regions, the elastic layer was filled with elastin fibers, LLPs, and other debris with little open space apparent.
Some differences between macular and peripheral regions were apparent in the ICL layer. Although both of these tissues showed an age-related accumulation of LLPs and small granules and an apparent decrease in collagen fibril density, this process appeared more advanced in the macular region. However, such differences were not dramatic, and it appeared that the same process was taking place in both regions.
Our results were generally consistent with those of Holz et al.4 showing that all lipid classes accumulated roughly twice as rapidly in macular BrM compared with the periphery.4 Within the constraints of small tissue samples, our results are not consistent with those of Curcio et al.,5 who reported that cholesterol accumulates sevenfold more rapidly in macular BrM than in the periphery. It remains possible that LLPs accumulating in the macula are cholesterol-rich relative to those accumulating in the periphery of BrM, or that the cholesterol accumulating with age in BrM is not contained exclusively within the LLPs. It will be informative to envision the topography of BrM lipid deposition with methods allowing larger samples and more precisely defined locations.15
Our studies have focused on older eyes that did not have overt indications of ARM. Thus, any changes in retinal function that may be caused by the LLP accumulation in BrM might be better characterized as age-related changes. We saw such changes in both the macular and peripheral aspects of the BrM. Age-related changes in both macular and peripheral function have been reported in a number of studies.16–19
It was somewhat surprising that we saw the formation of lipid walls in both macular and peripheral regions of BrM given that age-related maculopathy is thought to be primarily a macular disease. However, a recent study comparing proteome changes in macular and peripheral retina concluded that AMD also impacts the peripheral retina.20 Geographic atrophy and drusen have been reported in the periphery of some AMD patients.21 Still, while the lipid walls that we have described are found in locations where basal linear deposits arise (between the basal lamina of the RPE and the ICL), AMD is associated with formation of basal laminar deposits in the periphery,22 not basal linear deposits.
It is not known whether the lipid walls seen in this study are precursors to pathologic structures such as the basal deposits found in eyes with age-related maculopathy. Our study suggests that perhaps retinal functional deficits might also be found in the periphery of eyes with age-related maculopathy, although perhaps at a later stage of the disease than in the macular region.
ACKNOWLEDGMENTS
This study was supported by The American Health Assistance Foundation and by NIH grants EY014662 and EY06109.
REFERENCES
- [1].Lewis H, Straatsma BR, Foos RY. Chorioretinal juncture. Multiple extramacular drusen. Ophthalmology. 1986;93:1098–1112. doi: 10.1016/s0161-6420(86)33615-7. [DOI] [PubMed] [Google Scholar]
- [2].Malek G, Cousins S. Is our current clinical classification of AMD up to the job? Br J Ophthalmol. 2006;90:1080–1081. doi: 10.1136/bjo.2006.098715. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [3].Sheraidah G, Steinmetz R, Maguire J, Pauleikhoff D, Marshall J, Bird AC. Correlation between lipids extracted from Bruch’s membrane and age. Ophthalmology. 1993;100:47–51. doi: 10.1016/s0161-6420(13)31712-6. [DOI] [PubMed] [Google Scholar]
- [4].Holz FG, Sheraidah G, Pauleikhoff D, Bird AC. Analysis of lipid deposits extracted from human macular and peripheral Bruch’s membrane. Arch Ophthalmol. 1994;112:402–406. doi: 10.1001/archopht.1994.01090150132035. [DOI] [PubMed] [Google Scholar]
- [5].Curcio CA, Millican CL, Bailey T, Kruth HS. Accumulation of cholesterol with age in human Bruch’s membrane. Invest Ophthalmol Vis Sci. 2001;42:265–274. [PubMed] [Google Scholar]
- [6].Ruberti JW, Curcio CA, Millican L, Menco BPM, Huang J-D, Johnson M. Quick-freeze/deep-etch visualization of age-related lipid accumulation in Bruch’s membrane. Invest Ophthalmol Vis Sci. 2003;44:1753–1759. doi: 10.1167/iovs.02-0496. [DOI] [PubMed] [Google Scholar]
- [7].Huang J-D, Presley JB, Chimento MF, Curcio CA, Johnson M. Age-related changes in human macular Bruch’s membrane as seen by quick-freeze/deep-etch. Exp Eye Res. 2007;85:202–218. doi: 10.1016/j.exer.2007.03.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [8].Marshall J, Hussain AA, Moore DJ, Patmore AL. Aging and Bruch’s membrane. In: Marmor MF, Wolfensberger TJ, editors. The Retinal Pigment Epithelium: Function and Disease. Oxford University Press; New York: 1998. pp. 669–692. [Google Scholar]
- [9].Ethier C, Johnson M, Ruberti J. Ocular biomechanics and biotransport. Annu Rev Biomed Eng. 2004;6:249–273. doi: 10.1146/annurev.bioeng.6.040803.140055. [DOI] [PubMed] [Google Scholar]
- [10].Haimovici R, Gantz DL, Rumelt S, Freddo TF, Small DM. The lipid composition of drusen, Bruch’s membrane, and sclera by hot stage polarizing light microscopy. Invest Ophthalmol Vis Sci. 2001;42:1592–1599. [PubMed] [Google Scholar]
- [11].Guyton JR, Klemp KF. Ultrastructural discrimination of lipid droplets and vesicles in atherosclerosis: value of osmium-thiocarbohydrazide-osmium and tannic acid-paraphenylenediamine techniques. J Histochem Cytochem. 1988;36:1319–1328. doi: 10.1177/36.10.2458408. [DOI] [PubMed] [Google Scholar]
- [12].Curcio CA, Presley JB, Millican CL, Medeiros NE. Basal deposits and drusen in eyes with age-related maculopathy: evidence for solid lipid particles. Exp Eye Res. 2005;80:761–775. doi: 10.1016/j.exer.2004.09.017. [DOI] [PubMed] [Google Scholar]
- [13].Kubosawa J, Kondo Y. Quick-freeze, deep-etch studies of renal basement membranes. Microsc Res Tech. 1994;28:2–12. doi: 10.1002/jemt.1070280103. [DOI] [PubMed] [Google Scholar]
- [14].Kubosawa H, Kondo Y. Ultrastructural organization of the glomerular basement membrane as revealed by a deep-etch replica method. Cell Tissue Res. 1985;242:33–39. doi: 10.1007/BF00225560. [DOI] [PubMed] [Google Scholar]
- [15].Rudolf M, Li C-M, Chimento M, Curcio C.Topography of esterified cholesterol deposition in human Bruch membrane (BrM) wholemounts ARVO 2007Investigative Ophthalmology & Visual Science; Ft. Lauderdale, FL: e-abstract 2184. [Google Scholar]
- [16].Sunness JS, Massof RW, Johnson MA, Finkelstein D, Fine SL. Peripheral retinal function in age-related macular degeneration. Arch Ophthalmol. 1985;103:811–816. doi: 10.1001/archopht.1985.01050060071029. [DOI] [PubMed] [Google Scholar]
- [17].Casson EJ, Johnson CA, Nelson-Quigg JM. Temporal modulation perimetry: the effects of aging and eccentricity on sensitivity in normals. Invest Ophthalmol Vis Sci. 1993;34:3096–3102. [PubMed] [Google Scholar]
- [18].Holopigian K, Seiple W, Greenstein V, Kim D, Carr RE. Relative effects of aging and age-related macular degeneration on peripheral visual function. Optom Vision Sci. 1997;74:152–159. doi: 10.1097/00006324-199703000-00026. [DOI] [PubMed] [Google Scholar]
- [19].Tam WK, Chan H, Brown B, Leung KW, Woo V, Yap M. Aging and mfERG topography. Eye. 2006;20:18–24. doi: 10.1038/sj.eye.6701777. [DOI] [PubMed] [Google Scholar]
- [20].Ethen CM, Reilly C, Feng X, Olsen TW, Ferrington DA. The proteome of central and peripheral retina with progression of age-related macular degeneration. Invest Ophthalmol Vis Sci. 2006;47:2280–2290. doi: 10.1167/iovs.05-1395. [DOI] [PubMed] [Google Scholar]
- [21].Postel EA, Agarwal A, Schmidt S, et al. Comparing age-related macular degeneration phenotype in probands from singleton and multiplex families. Am J Ophthalmol. 2005;139:820–825. doi: 10.1016/j.ajo.2004.12.029. [DOI] [PubMed] [Google Scholar]
- [22].van der Schaft TL, de Bruijn WC, Mooy CM, de Jong PTVM. Basal laminar deposit in the aging peripheral human retina. Graefes Arch Clin Exp Ophthalmol. 1993;231:470–475. doi: 10.1007/BF02044234. [DOI] [PubMed] [Google Scholar]