Abstract
Background
Fabry disease is a rare X linked lysosomal storage disorder resulting from deficiency of α‐galactosidase A activity. Although the severity of clinical features in male patients is well described, only recently have studies reported the high prevalence of disabling clinical features in heterozygous females.
Aims
This study sets out to examine the clinical features and natural history of Fabry disease in further detail in a large group of female patients.
Methods
Data were obtained from 303 females enrolled in the Fabry Outcome Survey. Pain was assessed using the Brief Pain Inventory, and health related quality of life (HRQoL) was assessed using the European Quality of Life Questionnaire. A modified version of the Mainz Severity Score Index was also applied. Data on left ventricular mass (LVM) index, mean ventricular wall thickness, and glomerular filtration rate (GFR) were used to assess cardiac and renal involvement.
Results
The most commonly reported clinical features in females were neurological (77%) and cardiac (59%). A history of renal involvement was recorded in 40% of cases. Neurological features were the earliest to develop (mean age: 16 years), whereas cardiac (mean age: 33.5 years) and renal (mean age: 37.3 years) features developed later. LVM index increased exponentially with age. In addition, age was negatively correlated with estimated GFR and HRQoL.
Conclusions
Females with Fabry disease report important age related clinical features and clinical investigation demonstrates evidence of disease progression. This study highlights the importance of careful and longitudinal assessment of female heterozygote patients with Fabry disease.
Keywords: Fabry disease, Fabry Outcome Survey, heterozygotes
Fabry disease (OMIM 301500) is a rare X linked lysosomal storage disorder resulting from deficiency of α‐galactosidase A activity.1 The estimated incidence of Fabry disease is 1 in 117 000 male live births,2 though estimates vary from 1 in 40 000 to over 1 in 400 000. Fabry disease is a multi‐system disorder in which storage of the principal substrate globotriaosylceramide occurs in a wide variety of tissues including vascular endothelium, renal glomeruli and tubules, dorsal root ganglia, cardiac myocytes, conducting tissue and valves, cornea, and skin. Clinical features include vascular skin lesions (angiokeratoma), proteinuria, progressive renal impairment, painful paraesthesiae of the hands and feet (acroparaesthesiae), cardiac hypertrophy, conduction abnormalities, corneal dystrophy, hypohidrosis, and impaired temperature regulation. In affected males, life expectancy is approximately 50 years.3
Although textbook descriptions of Fabry disease refer to X linked recessive inheritance and only a mild clinical phenotype in a minority of female “carriers”, recent research has emphasised the prevalence of disabling clinical features in most heterozygous females.4,5,6,7,8 In a recent study of female “carriers” aged 12–65 years, clinical manifestations were observed in all 20 females studied.7 Acroparaesthesiae were reported in 18 females and were usually the first symptom to occur, with a mean age of onset of 10 years (range: 4–23 years). Clinical examination also revealed signs of renal and cardiac involvement in more than half of this group.
Enzyme replacement therapy (ERT) for Fabry disease was licensed in Europe in 2001 under the provisions of the orphan drug legislation; however, evidence of safety and efficacy was provided by the results of clinical trials conducted almost entirely in male patients.9,10 Data from one open label uncontrolled study in 15 heterozygous female patients showed evidence of improvement in health related quality of life (HRQoL) and a reduction in left ventricular mass (LVM) following treatment with agalsidase alfa.11 Published information on the effects of ERT on signs and symptoms in females is otherwise limited. Although ERT has been licensed for use in female patients, the paucity of evidence of clinical benefit in women has led to difficulties in securing this treatment in some European countries.
The Fabry Outcome Survey (FOS) is a European outcomes database for all patients with Fabry disease who are receiving or are candidates for ERT with agalsidase alfa (Replagal, TKT Europe, Danderyd, Sweden). In recent years asymptomatic female heterozygotes who are not immediate candidates for ERT have also been recruited to the database. The description of the database and the baseline demographic data within it have been published recently.12 Of the 366 patients registered at the time of that report, 165 were female and 61 of these were index cases. The mean age of onset of symptoms in these index cases was 22.6 years; however, on average patients were not diagnosed until 16.3 years following the onset of symptoms. Neuropathic pain was reported in 64% of all registered female patients, with dermatological features in 50%, proteinuria in 33%, cardiac features in 65%, left ventricular hypertrophy in 28%, cerebrovascular events in 27%, auditory symptoms in 47%, gastrointestinal symptoms in 50%, fatigue in 28%, and ocular signs (mainly corneal dystrophy) in 53%. Mean age at death in 24 affected female relatives of patients enrolled in FOS was 55.4 years.
Thus it is clear that heterozygous females suffer from clinically important symptoms and signs of Fabry disease. However, the rate of progression of serious complications in females, in particular renal and cardiac dysfunction and structural abnormalities, is not known. Nevertheless, such information is necessary in order to judge the effectiveness of ERT in this chronic disorder. The aim of the present study was to examine in further detail the clinical features and natural history of Fabry disease, including the results of key clinical investigations, in a large group of female patients using data available from FOS.
Methods
Patients
The FOS database has been approved by the ethics review boards of all participating centres, and all patients have given written informed consent. At the time of this analysis (August 2004), FOS contained data on 608 patients, of whom 303 (50%) were female. Of the female patients, 141 (47%) were receiving ERT with agalsidase alfa at a dose of 0.2 mg/kg body weight intravenously every 2 weeks. Patients had received treatment for a mean (±SD) duration of 12.7±12.1 months.
Data collection
Upon enrolment into FOS, each patient's medical history is documented by a physician or nurse specialist. Uniform case reports documenting demographic details, family history, mode of diagnosis, clinical features, treatment, and outcomes are submitted electronically to the central FOS database. Pain is assessed using the Brief Pain Inventory (BPI), which has been validated for most European countries.13,14,15 HRQoL is assessed using the European Quality of Life Questionnaire (EQ‐5D).16,17 Overall disease severity is assessed using a minor modification of the Mainz Severity Score Index (MSSI)18 to make use of the format of the data as entered in FOS (FOS‐MSSI). This involves the modification of several parameters, which are recorded in ordinal format in the original MSSI index, for the binary format of data in FOS. Datasets are not complete for all registrants; where numbers of registrants are quoted below, n is the number for whom the relevant data are complete.
Input of data on cardiac and renal function is optional in FOS. Echocardiographic data are reported in a uniform manner. LVM is calculated using the equations:
LVM (g) = 0.8[1.04(IVSd+LVd+PWd)3−LVd3]+0.6
LVM (g/m2.7) = LV mass/height2.7
MWT (cm) = (IVSd+PWd)/2
where IVS is interventricular septum thickness at end diastole, LVd is left ventricular internal diameter in end diastole, PW is posterior wall thickness at end diastole, and MWT is mean wall thickness.
Echocardiographic data are reported by a consultant cardiologist in each participating centre. Glomerular function is assessed using an estimated glomerular filtration rate (GFR) derived from the serum creatinine concentration (measured locally) and sex using the short version of the Modification of Diet in Renal Disease (MDRD) formula: GFR (ml/min/1.73 m2) = ([186.3×serum creatinine (mg/dl)−1.154]×[age (years)−0.203])×1.212 (if of African origin)×0.742 (if female).
Serum creatinine concentrations were adjusted to account for differences in methodology used in individual laboratories and changes in methodology over time. Results from enzymatic assays for serum creatinine were modified to bring them into line with results from the classic Jaffé method.
α‐Galactosidase A activity was estimated in plasma or leukocytes by the participating centres according to their own methodology and reported as absent, reduced, or normal according to local reference ranges.
Statistical analysis
Numbers are reported as means (±SD). For correlations, Spearman's correlation coefficient for ranked data was used. Regression analysis was used to describe the effect of age on disease parameters.
Results
Patient demographics and clinical features
Data are available from 303 female patients enrolled in FOS. Mean age at entry into FOS was 38.2±18.0 years. Diagnosis was made at a mean age of 31.8±16.8 years (n = 268), while the mean age at onset of symptoms was 19.7±15.4 years (n = 187). Patients were recruited from 65 centres in 11 European countries. The 303 females came from 148 families.
The frequency and age at onset of disease specific clinical features, as reported by the patient using the checklist of predefined clinical features developed for FOS, are shown in table 1. The most common features were neurological and cardiac, which were reported in 77% and 59% of patients, respectively. Neurological features were also the earliest to develop, beginning at an average age of 16.0 years, whereas cardiac features began somewhat later, at an average age of 33.5 years. Of note, self‐reported indicators of renal involvement were observed in 40% of patients and these began at an average age of 37.3 years. Other signs and symptoms involving the eye and gastrointestinal system were also common, being observed in 50% of the females for whom these data were available.
Table 1 Frequency and age at onset of specific signs and symptoms of Fabry disease in females (n = 248).
| Frequency | Age at onset | |||
|---|---|---|---|---|
| Proportion of population (%) | Number of patients | Mean±SD | Number of patients | |
| Cerebrovascular | 25 | 61 | 38.8±17.5 | 35 |
| Stroke | 7 | 18 | 49.9±14.5 | 15 |
| TIA | 7 | 17 | 42.9±14.6 | 13 |
| Neurological | 77 | 192 | 16.0±14.0 | 99 |
| Pain attacks | 57 | 142 | 16.6±15.0 | 98 |
| Chronic pain | 32 | 80 | 20.7±14.5 | 55 |
| Cardiac | 59 | 147 | 33.5±18.1 | 66 |
| Chest pain | 15 | 37 | 41.5±16.3 | 16 |
| Palpitations | 20 | 50 | 41.6±15.3 | 30 |
| LV hypertrophy | 26 | 65 | 50.4±11.3 | 35 |
| Renal/urinary | 40 | 100 | 37.3±16.6 | 61 |
| Proteinuria | 35 | 86 | 38.8±18.2 | 59 |
| Dialysis | 1 | 40.0 | 1 | |
| Transplants | 1 | 2 | 40.1±1.2 | 2 |
| Gastrointestinal | 50 | 124 | 21.4±16.6 | 65 |
| Diarrhoea | 19 | 46 | 19.0±15.8 | 23 |
| Constipation | 15 | 37 | 23.3±18.5 | 22 |
| Auditory | 48 | 119 | 29.8±18.7 | 71 |
| Tinnitus | 29 | 71 | 27.9±15.9 | 46 |
| Vertigo | 32 | 80 | 34.1±17.9 | 58 |
| Sudden deafness | 4 | 9 | 36.5±20.0 | 8 |
| Ophthalmological | 50 | 123 | 31.1±18.9 | 76 |
| Cornea verticillata | 40 | 99 | 32.3±20.2 | 72 |
| Tortuous vessels | 6 | 14 | 34.4±23.6 | 8 |
| Posterior subcapsular cataract | 2 | 6 | 61.3±8.7 | 3 |
| Dermatological | 44 | 108 | 29.0±16.1 | 60 |
| Angiokeratomas | 40 | 100 | 28.6±15.5 | 59 |
| Telangiectasia | 8 | 19 | 29.3±17.9 | 10 |
LV, left ventricular; TIA, transient ischaemic attack.
When the signs and symptoms were analysed in more detail, the predominant symptoms reported were acute attacks of pain (classed as neurological), affecting 57% of patients. The next most frequent features were angiokeratoma and cornea verticillata. A high proportion of patients (35%) were also reported to have proteinuria. Of the cardiac manifestations reported by the patient, left ventricular hypertrophy was declared in 26% of the female population studied, and palpitations were reported by 20%.
Clinical investigations
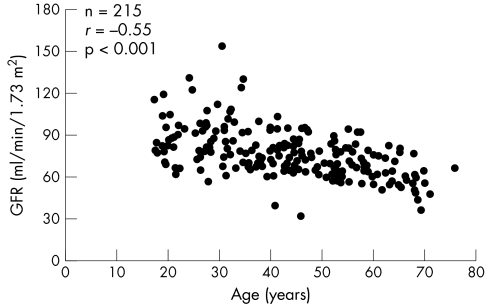
Echocardiographic assessment of LVM prior to ERT was available in 140 patients. LVM was measured according to Devereux19 and indexed to actual body height to the power of 2.7. Mean LVM index was 50.2±26.1 g/m2.7 at a mean age of 38.9±17.7 years. Hypertrophic cardiomyopathy was considered to be present if the LVM index exceeded 50 g/m2.7. Mean ventricular wall thickness in female patients enrolled in FOS was 11.0±3.4 mm. Mean GFR in 215 female patients in FOS prior to ERT was 76.8±17.3 ml/min/1.73 m2 at a mean age of 43.3±14.5 years, excluding those below 18 years of age. Classification of renal function according to the Kidney Disease Outcomes Quality Initiative guidelines showed that among females in FOS, 13% have stage III (moderate), 66% have stage II (mild), and 21% have stage I (normal) renal function.20
Disease severity and progression in untreated female patients
The LVM index (fig 1) increased exponentially with age (r = 0.73, p<0.001, n = 140) and mean ventricular wall thickness increased linearly with age (r = 0.8, p<0.001, n = 154). Age was also negatively correlated with estimated GFR (assessed using the MDRD equation), with a decline in GFR of 0.64 ml/min per year (fig 2; r = −0.55, p<0.001, n = 215). In addition, age was negatively correlated with HRQoL; however, there was no correlation when adjusted for the expected normal decline in quality of life with age. No correlations with age were found for the BPI dimensions.
Figure 1 Left ventricular mass (LVM) index according to age in 140 untreated female patients with Fabry disease enrolled in FOS.
Figure 2 Glomerular filtration rate (GFR) according to age in 215 untreated female patients with Fabry disease enrolled in FOS.
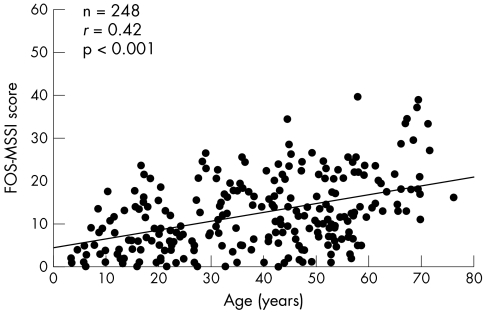
The FOS‐MSSI score correlated positively with age (fig 3; r = 0.42, p<0.001, n = 248) and the number of affected organ systems (r = 0.82, p<0.001, n = 248). The FOS‐MSSI correlated inversely with HRQoL, as assessed using the EQ‐5D utility score (r = −0.42, p<0.001, n = 130), a measure of the deviance of the EQ‐5D scores from age and gender matched UK reference data21 (r = −0.35, p<0.001, n = 130), and the visual analogue scale of the EQ‐5D (r = −0.47, p<0.001, n = 110).
Figure 3 Positive correlation between the FOS‐MSSI score and age in 248 untreated female patients enrolled in FOS.
Proteinuria
No effect of age on GFR was seen in a subgroup of 38 female patients with proteinuria (protein >300 mg/24 h). The mean GFR was 74.5±14.6 ml/min/1.73 m2, at ages ranging from 17 to 68 years.
Longitudinal data
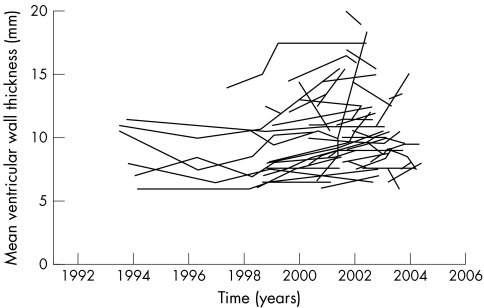
Longitudinal data are available for many parameters in FOS in individual untreated patients; however, more data are needed before they can be subjected to a thorough statistical analysis. The longitudinal data available in FOS on mean ventricular wall thickness are shown as an example (fig 4).
Figure 4 Longitudinal data on mean ventricular wall thickness in untreated female patients with Fabry disease enrolled in FOS.
Mortality
Mortality data are available for relatives of some of the patients enrolled in FOS. The mean age at death in 53 female relatives (assumed by the registrant to be heterozygous for a Fabry disease mutation) was 49.8±12.3 years; median: 49.0) (fig 5).
Figure 5 Age at death of 53 female relatives of patients enrolled in FOS.
Enzyme activity
Data on plasma or leukocyte enzyme activity were available in 213 patients. Cross‐tabulation with disease severity, as determined by the FOS‐MSSI, demonstrated that among 83 patients with normal enzyme activity, 31% had a score above 20 (moderate disease severity), whereas among 97 patients with reduced enzyme activity, 18% had a score above 20. No women carrying the D313Y amino acid substitution, associated with pseudodeficient activity in plasma, were included in this analysis.
Discussion
This study, involving the largest cohort of females heterozygous for Fabry disease, confirms the findings of MacDermot et al4 and Whybra et al7 that the majority of females who are heterozygous for disease causing mutations in the α‐galactosidase A gene report clinical features of Fabry disease. Indeed, in the present study severe manifestations of disease were common, with 77% of women reporting neurological involvement, 59% cardiac involvement, and 40% renal involvement. Although the onset of symptoms was not as early as is typically observed in males, the majority of signs and symptoms assessed had developed by the third and fourth decades of life; however, there was considerable variation between individuals. This study demonstrates the considerable delay between onset of symptoms and diagnosis in this group of women. In an earlier analysis of the FOS database, Mehta and co‐workers12 reported a similar prevalence of symptoms and a wide range of disease expression in female patients. One possible explanation for this variation is skewed X chromosome inactivation.22 Early in embryonic development one of the two X chromosomes in each somatic cell becomes inactivated, which results in patchy and variable expression of the defective gene. This may impact upon the degree of disease expression observed. Two groups have shown an association between patterns of X inactivation in peripheral blood leukocytes and clinical severity of Fabry disease.23,24 In a recent review, Dobyns et al suggest that most X linked conditions are neither dominant nor recessive.25 In view of the high prevalence of symptoms in female patients, we suggest that the X linked inheritance of Fabry disease should no longer be regarded as recessive and that the term “carrier” should be avoided, if possible, as it may give the false impression to a patient or relative, ignorant of the terminology of medical genetics, that females are asymptomatic or only mildly affected.
It is worth commenting on the fact that the severity of disease was not related to plasma or leukocyte activity of α‐galactosidase A. This serves to reinforce the point that the measurement of α‐galactosidase A activity in plasma or leukocytes is of no diagnostic or prognostic value in females and should not be used to determine treatment options. The enzyme deficiency is intracellular and occurs only in cells where the X chromosome carrying the wild type allele is inactivated. Activity of the enzyme in plasma or leukocytes may not reflect the situation within the lysosome of relevant cell types in Fabry disease.
It is also noteworthy that proteinuria was not associated with a more rapid decline in GFR in these women; in fact, the reverse was the case. This finding should prompt further study into the prognostic relevance of proteinuria in Fabry disease.
One potential source of bias of data in FOS relates to the possibility that the more severely affected female patients, who attend their physicians on a more frequent basis and who are either on ERT or are immediate candidates for this treatment, are more likely to be registered in the database. However, a secular trend apparent in FOS is that as the relative number of women registered in FOS has approached the number of male patients and as more mildly affected and asymptomatic women have been recruited, the average reported severity of the condition has lessened. The prevalence of features of Fabry disease in women enrolled in FOS should not be interpreted as being the same as the prevalence in all females heterozygous for disease causing mutations. Nevertheless, this is a rare condition, and we report on 303 women, a number equal to that of males registered in the database. The prevalence of disease manifestations in this study is similar to the prevalence of features as reported by MacDermot and co‐workers,4 who in their study, attempted to trace all known obligate heterozygous females in the UK. One way to examine potential bias would be to compare disease severity in index cases and individuals diagnosed through pedigree analysis. FOS permits only an approximate estimate of index cases according to whether the diagnosis was made by a clinician or through an affected family member. Using this method, we found no significant difference between the number of affected organ systems between index cases and those identified by pedigree.
Assessment of the severity of disease revealed that cardiac and renal involvement increased progressively with age. Using the relationship between age and disease parameters as a tool to examine the natural history of a disease has potential disadvantages. Survivor bias (based on the early death of more severely affected patients) may dilute the apparent rate of progression of the disease and perhaps even give rise to a false impression of improvement over time.26 Selection bias, as a result of the recruitment of young, mildly affected females through pedigree analysis, and older severely affected index cases, may also give a false impression of disease progression. We examined our data for such bias (data not shown), and although older women were more likely to be index cases, their disease severity did not differ significantly from the severity of those identified by pedigree analysis. Despite these disadvantages, this approach allows the early reporting of results from large numbers of individuals, which is important in rare diseases such as Fabry disease.
It has previously been established that the MSSI is a useful tool for assessing the severity of Fabry disease. This disease specific severity score has the advantage of summarising the diverse aspects of disease expression and of concentrating on those aspects of health most relevant to Fabry disease. In the present study, a modification of this index, the FOS‐MSSI, was used and results showed that the score obtained correlated positively with age and the number of affected organ systems. The score was also found to correlate inversely with HRQoL, indicating that increasingly severe disease is associated with a poorer quality of life. This suggests that the modified score is a suitable index for assessing the severity of Fabry disease in women enrolled in FOS.
Although longitudinal data are limited, the data available from FOS are consistent with the hypothesis that Fabry disease in women is progressive. Interestingly, data available on the female relations of women enrolled in FOS show that the mean age at death was approximately 50 years.
In conclusion, females with Fabry disease show evidence of deterioration of HRQoL, renal function, and cardiac size with advancing age. FOS is a useful tool to evaluate the phenotype and natural history of this orphan disorder, and the information gathered will serve as a baseline against which to evaluate the effects of enzyme replacement or other new treatments. Change in the rate of decline of quality of life parameters, severity score indices, renal function, and left ventricular mass should be considered as measures of outcome of enzyme replacement therapy. The progressive nature of the condition mandates thorough and repeated clinical assessment of affected female heterozygotes.
Acknowledgements
The authors very much appreciate the support of all patients enrolled in FOS and are very much obliged to all those who entered data into the database.
Abbreviations
BPI - Brief Pain Inventory
ERT - enzyme replacement therapy
EQ‐5D - European Quality of Life Questionnaire
FOS - the Fabry Outcome Survey
GFR - glomerular filtration rate
HRQoL - health related quality of life
LVM - left ventricular mass
MDRD - Modification of Diet in Renal Disease
MSSI - Mainz Severity Score Index
Footnotes
Sponsorship: The FOS database is under the independent control of the FOS European board. Data collection and analysis in FOS are supported by TKT Europe, Danderyd, Sweden. The sponsor had no role in the interpretation of data or writing of this report.
Competing interests: P B Deegan, F Baehner, and D A Hughes have received honoraria and travel grants from TKT Europe. M‐Á Barba Romero has received honoraria and travel grants from TKT Europe and travel grants from Genzyme. M Beck has received honoraria, travel grants, and research grants from TKT Europe and Genzyme.
References
- 1.Desnick R J, Ioannou Y A, Eng C M. α‐Galactosidase A deficiency: Fabry disease. In: Scriver CR, Beaudet AL, Sly WS, Valle D, eds. The metabolic and molecular basis of inherited disease. 8th ed. New York: McGraw‐Hill, 20013733–3774.
- 2.Meikle P J, Hopwood J J, Clague A E, Carey W F. Prevalence of lysosomal storage disorders. JAMA 1999281249–254. [DOI] [PubMed] [Google Scholar]
- 3.MacDermot K D, Holmes A, Miners A H. Anderson‐Fabry disease: clinical manifestations and impact of disease in a cohort of 98 hemizygous males. J Med Genet 200138750–760. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.MacDermot K D, Holmes A, Miners A H. Anderson‐Fabry disease: clinical manifestations and impact of disease in a cohort of 60 obligate carrier females. J Med Genet 200138769–775. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Kampmann C, Baehner F, Whybra C, Martin C, Wiethoff C M, Ries M, Gal A, Beck M. Cardiac manifestations of Anderson‐Fabry disease in heterozygous females. J Am Coll Cardiol 2002401668–1674. [DOI] [PubMed] [Google Scholar]
- 6.Guffon N. Clinical presentation in female patients with Fabry disease. J Med Genet 200340e38. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Whybra C, Kampmann C, Willers I, Davies J, Winchester B, Kriegsmann J, Bruhl K, Gal A, Bunge S, Beck M. Anderson‐Fabry disease: clinical manifestations of disease in female heterozygotes. J Inherit Metab Dis 200124715–724. [DOI] [PubMed] [Google Scholar]
- 8.MacDermot K D, Holmes A, Miners A H. Natural history of Fabry disease in affected males and obligate carrier females. J Inherit Metab Dis 200124(Suppl 2)13–14. [DOI] [PubMed] [Google Scholar]
- 9.Eng C M, Guffon N, Wilcox W R, Germain D P, Lee P, Waldek S, Caplan L, Linthorst G E, Desnick R J. Safety and efficacy of recombinant human alpha‐galactosidase A‐replacement therapy in Fabry's disease. N Engl J Med 20013459–16. [DOI] [PubMed] [Google Scholar]
- 10.Schiffmann R, Kopp J B, Austin H A, 3rd, Sabnis S, Moore D F, Weibel T, Balow J E, Brady R O. nzyme replacement therapy in Fabry disease: a randomized controlled trial. JAMA 20012852743–2749. [DOI] [PubMed] [Google Scholar]
- 11.Baehner F, Kampmann C, Whybra C, Miebach E, Wiethoff C M, Beck M. Enzyme replacement therapy in heterozygous females with Fabry disease: results of a phase IIIB study. J Inherit Metab Dis 200326617–627. [DOI] [PubMed] [Google Scholar]
- 12.Mehta A, Ricci R, Widmer U, Dehout F, Garcia de Lorenzo A, Kampmann C, Linhart A, Sunder‐Plassmann G, Ries M, Beck M. Fabry disease defined: baseline clinical manifestations of 366 patients in the Fabry Outcome Survey. Eur J Clin Invest 200434236–242. [DOI] [PubMed] [Google Scholar]
- 13.Tan G, Jensen M P, Thornby J I, Shanti B F. Validation of the Brief Pain Inventory for chronic nonmalignant pain. J Pain 20045133–137. [DOI] [PubMed] [Google Scholar]
- 14.Cleeland C S. Pain assessment: the advantages of using pain scales in lysosomal storage diseases. Acta Paediatr Suppl 20029143–47. [DOI] [PubMed] [Google Scholar]
- 15.Cleeland C S, Ryan K M. Pain assessment: global use of the Brief Pain Inventory. Ann Acad Med Singapore 199423129–138. [PubMed] [Google Scholar]
- 16.Miners A H, Holmes A, Sherr L, Jenkinson C, MacDermot K D. Assessment of health‐related quality‐of‐life in males with Anderson Fabry Disease before therapeutic intervention. Qual Life Res 200211127–133. [DOI] [PubMed] [Google Scholar]
- 17.Rabin R, de Charro F. EQ‐5D: a measure of health status from the EuroQol Group. Ann Med 200133337–343. [DOI] [PubMed] [Google Scholar]
- 18.Whybra C, Kampmann C, Krummenauer F, Ries M, Mengel E, Miebach E, Baehner F, Kim K, Bajbouj M, Schwarting A, Gal A, Beck M. The Mainz Severity Score Index: a new instrument for quantifying the Anderson‐Fabry disease phenotype, and the response of patients to enzyme replacement therapy. Clin Genet 200465299–307. [DOI] [PubMed] [Google Scholar]
- 19.Devereux R B, Alonso D R, Lutas E M, Gottlieb G J, Campo E, Sachs I, Reichek N. Echocardiographic assessment of left ventricular hypertrophy: comparison to necropsy findings. Am J Cardiol 198657450–458. [DOI] [PubMed] [Google Scholar]
- 20.Levey A S, Coresh J, Balk E, Kausz A T, Levin A, Steffes M W, Hogg R J, Perrone R D, Lau J, Eknoyan G. National Kidney Foundation practice guidelines for chronic kidney disease: evaluation, classification, and stratification. Ann Intern Med 2003139137–147. [DOI] [PubMed] [Google Scholar]
- 21.Kind P, Hardman G, Macran S.UK population norms for EQ‐5D. Trans vol ed. York: Centre for Health Economics, 1999172
- 22.Lyon M F. X‐chromosome inactivation. Curr Biol 19999R235–R237. [DOI] [PubMed] [Google Scholar]
- 23.Dobrovolny R, Dvorakova L, Ledvinova J, Magage S, Bultas J, Lubanda J C, Elleder M, Karetova D, Pavlikova M, Hrebicek M. Relationship between X‐inactivation and clinical involvement in Fabry heterozygotes. Eleven novel mutations in the alpha‐galactosidase A gene in the Czech and Slovak population. J Mol Med 200583(8)647–654. [DOI] [PubMed] [Google Scholar]
- 24.Morrone A, Cavicchi C, Bardelli T, Antuzzi D, Parini R, Di Rocco M, Feriozzi S, Gabrielli O, Barone R, Pistone G, Spisni C, Ricci R, Zammarchi E. Fabry disease: molecular studies in Italian patients and X inactivation analysis in manifesting carriers. J Med Genet 200340(8)e103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Dobyns W B, Filauro A, Tomson B N, Chan A S, Ho A W, Ting N T, Oosterwijk J C, Ober C. Inheritance of most X‐linked traits is not dominant or recessive, just X‐linked. Am J Med Genet A 2004129(2)136–143. [DOI] [PubMed] [Google Scholar]
- 26.Lilienfeld A M. Practical limitations of epidemiologic methods. Environ Health Perspect 1983523–8. [DOI] [PMC free article] [PubMed] [Google Scholar]