Abstract
Background
It has been reported that the activating mutation, E133K, in the angiogenic factor VG5Q (formally named AGGF1) causes Klippel‐Trenaunay Syndrome (KTS), a rare vascular disease associated with asymmetric overgrowth. This proposal followed from the observation that five out of 130 KTS patients were constitutionally heterozygous for VG5Q, E133K.
Objective
To explore the possibility that VG5Q, and specifically E133K, is implicated in other mosaic overgrowth syndromes.
Results
24 patients were analysed for this sequence change.
One patient was constitutionally heterozygous for E133K. Analysis of both parents revealed that the patient's mother, who was healthy, also carried E133K. An analysis of 275 healthy controls showed that 3.3% (9/275) of the population were carriers of E133K.
Conclusions
The findings bring into question the assertion that VG5Q, E133K is a mutation and that it causes KTS.
Keywords: Klippel‐Trenaunay syndrome, VG5Q, asymmetric overgrowth
Klippel‐Trenaunay syndrome (KTS) (MIM 149000; http://www.ncbi.nlm.nih.gov/entrez/dispomim.cgi?id = 149000) is one of a number of rare asymmetric growth disorders. KTS is characterised by the presence of vascular malformations of the capillary, venous, and lymphatic systems and overgrowth of the affected limb, usually the lower limbs.1 Currently a molecular diagnosis for KTS is not available. Recently the vascular growth factor VG5Q was implicated in the aetiology of KTS,2 with the observation that five of 130 KTS patients (4%) were constitutionally heterozygous for the sequence change E133K.
To explore the possibility that VG5Q, and specifically E133K, is implicated in other mosaic overgrowth syndromes we analysed 24 patients with asymmetric overgrowth, including 11 individuals with Proteus syndrome (PS) (MIM 176920; http://www.ncbi.nlm.nih.gov/entrez/dispomim.cgi?id = 176920),3 which phenotypically overlaps with KTS. Nineteen were ascertained from the dermatology clinic of Great Ormond Street Children's Hospital, London, three from Greenwood Genetics Center, South Carolina, and two from Montreal Children's Hospital, Montreal. A series of 275 healthy, unrelated spouses of cancer patients served as population controls. Samples were obtained with informed consent and local ethics review board approval in accordance with the tenets of the Helsinki Declaration.
Fibroblasts were cultured from skin biopsies obtained from normal tissue and from regions of overgrowth. Genomic DNA was extracted from either cultured cells or affected tissue using the Qiagen QIAamp DNA mini kit (Qiagen Inc, Valencia, California, USA). Venous blood samples were obtained from the controls, and genomic DNA extracted using a standard sucrose lysis protocol. Exon 3 of VG5Q was amplified using published primer sequences.2 Patient DNA was analysed by direct sequencing. Controls were initially analysed by denaturing high performance liquid chromatography (DHPLC; WAVE system, Transgenomics, Santa Clara, California, USA). Any samples showing migration defects were sequenced.
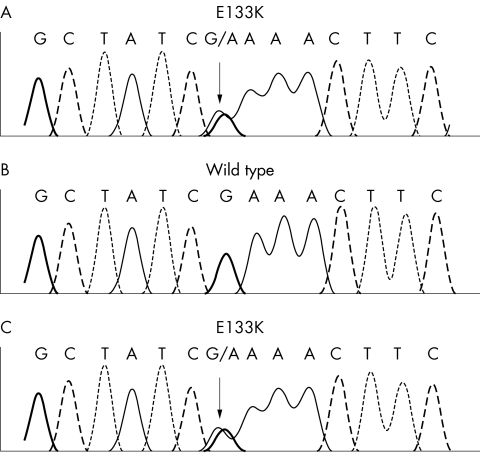
One patient was identified as being constitutionally heterozygous for E133K (fig 1A). The manifestations observed in this patient were overgrowth of the right leg, macrodactyly of third and fourth left toes and second and third right toes (severe enough to warrant surgery), macrocephaly, an overgrown right ear, right hemimegalencephaly, right visceromegaly, overgrowth of the left maxilla and mandible, and lipomas. KTS was excluded because no vascular malformations were present. The remaining 23 cases did not possess this variant.
Figure 1 Sequence chromatograms of VG5Q, codon 133. (A) DNA from patient 15 (heterozygous for E133K mutation). (B) DNA from patient's father (homozygous wild type E133 codon). (C) DNA from patient's mother (heterozygous for E133K mutation). Arrows indicate sequence changes.
To determine whether the sequence change arose de novo, we sequenced constitutional DNA from both parents. One parent, who was clinically normal, also harboured the E133K mutation (fig 1C). To determine the frequency of this variant in the general population, we analysed 275 unrelated, healthy, white, population controls for the E133K sequence change. Nine (3.3%) of the controls carried the E133K sequence change, indicating that this variant has reached polymorphic frequency in population controls (p value for difference between overgrowth cases and controls, 0.56).
In the study of Wang and colleagues,2 5/130 KTS cases and 0/200 age, sex, and ethnicity matched controls carried the E133K variant. Therefore, our identification of nine carriers among 275 healthy individuals was unexpected. This difference may be a result of population stratification in that our controls were of European descent. Nevertheless, this observation together with the presence of the variant in one healthy parent of the single affected carrier we identified throws into doubt the validity of the specific observation that the E133K variant in VG5Q is associated with KTS. It seems plausible to suggest that E133K is a relatively common polymorphism rather than a rare gain of function mutation, especially given the rarity of KTS and the fact that we found it with a similar frequency in our healthy controls as Tian et al in their cases.2 The more general question of whether any other alterations in VG5Q predispose to KTS remains unanswered.
Acknowledgements
We would like to thank all the patients and families for participating in this study, Anna Martinez, and V Der Kaloustian for patient recruitment/collection, and Athena Matakidou and Hayley Spendlove for technical assistance.
Abbreviations
KTS - Klippel‐Trenaunay syndrome
Footnotes
Conflicts of interests: none declared
References
- 1.Cohen M M. Klippel‐Trenaunay syndrome. Am J Med Genet 200093171–175. [DOI] [PubMed] [Google Scholar]
- 2.Tian X L, Kadaba R, You S A, Liu M, Timur A A, Yang L, Chen Q, Szafranski P, Rao S, Wu L, Housman D E, DiCorleto P E, Driscoll D J, Borrow J, Wang Q. Identification of an angiogenic factor that when mutated causes susceptibility to Klippel‐Trenaunay syndrome. Nature 2004427640–645. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Biesecker L G, Happle R, Mulliken J B, Weksberg R, Graham J M, Viljoen D L, Cohen M M. Proteus syndrome: diagnostic criteria, differential diagnosis, and patient evaluation. Am J Med Genet 199984389–395. [DOI] [PubMed] [Google Scholar]