Abstract
The infectious cycle of human cytomegalovirus (HCMV) is intricately linked to the host's cell cycle. Viral gene expression can be initiated only in G0/G1 phase. Once expressed, the immediate-early gene product IE2 prevents cellular DNA synthesis, arresting infected cells with a G1 DNA content. This function is required for efficient viral replication in vitro. A prerequisite for addressing its in vivo relevance is the characterization of cell cycle-regulatory activities of CMV species for which animal models have been established. Here, we show that murine CMV (MCMV), like HCMV, has a strong antiproliferative capacity and arrests cells in G1. Unexpectedly, and in contrast to HCMV, MCMV can also block cells that have passed through S phase by arresting them in G2. Moreover, MCMV can also replicate in G2 cells. This is made possible by the cell cycle-independent expression of MCMV immediate-early genes. Transfection experiments show that of several MCMV candidate genes, only immediate-early gene 3 (ie3), the homologue of HCMV IE2, exhibits cell cycle arrest activity. Accordingly, an MCMV ie3 deletion mutant has lost the ability to arrest cells in either G1 or G2. Thus, despite interspecies variations in the cell cycle dependence of viral gene expression, the central theme of HCMV IE2-induced cell cycle arrest is conserved in the murine counterpart, raising the possibility of studying its physiological relevance at the level of the whole organism.
Cytomegaloviruses (CMVs), the prototypical members of the betaherpesvirus subfamily, are ubiquitous, species-specific pathogens that typically establish an asymptomatic, lifelong infection in the immunocompetent host. In the immunocompromised host, CMV can reactivate and cause severe or even life-threatening disease. Consequently, human CMV (HCMV) infections are particularly hazardous in neonates as well as transplant recipients and AIDS patients.
During their evolution, CMVs have evolved many ways to functionally interfere with their host's biology in order to ensure their replication and survival (43). In this respect, the analysis of cell cycle regulation by HCMV has been a constant focus of interest over recent years and by now has been studied to some extent mainly in lytically infected fibroblasts (2, 12, 16, 29, 55). These studies revealed a complex interplay between HCMV and the cell cycle state of the host cell. HCMV can infect cells throughout the cell cycle, but for unknown reasons viral de novo gene expression is initiated only in G0 and early to mid-G1 phases (17, 51). In later cell cycle phases, viral entry takes place, but viral gene expression is delayed until cells have divided and reentered the following G1 phase (17, 51). There, or indeed also in G0 cells, HCMV stimulates proproliferative cellular pathways by activating the S-phase-promoting cyclin E-Cdk2-Rb-E2F pathway (6, 9, 18, 21, 24, 26, 28, 41, 49, 56, 64, 67, 69) and the M-phase-promoting cyclin B1-Cdk1 complex (26, 52, 61, 65, 67). However, once infected cells reach the G1/S transition, the viral IE2 protein inhibits further cell cycle progression by specifically blocking cellular DNA synthesis (8, 15, 34, 38, 46, 48, 57, 66). Together, these viral activities lead to a profoundly deregulated cell cycle state of HCMV-infected cells, where the initiation of cellular DNA replication is selectively inhibited (4, 68) but other features of S-phase-cells, such as an active nucleotide metabolism and the expression of replication factors, are induced. Even morphological changes typical of mitotic cells can be observed in infected cells (22, 23). These virally induced alterations are important for HCMV physiology since the IE2-mediated block of cellular DNA synthesis as well as the activation of cyclin-dependent kinases and critical enzymes for nucleotide metabolism are prerequisites for efficient virus replication in vitro (7, 48, 53, 54).
The physiological importance of HCMV-mediated cell cycle regulation at the level of the whole organism is unknown and, for obvious reasons, cannot be addressed experimentally. Thus, it would be desirable to use animal models of HCMV pathogenesis to address this issue. Such model systems do exist and typically employ murine CMV (MCMV) (33, 50). However, there is a surprising lack of information as to whether MCMV regulates cell cycle progression. Therefore, we have initiated an analysis of cell cycle-regulatory effects imposed by MCMV. Unexpectedly, this virus causes early (G1) and late (G2) cell cycle blocks. Consistent with this, viral immediate-early gene expression occurs in a cell cycle-independent manner. We further find that the viral ie3 protein is necessary and sufficient for mediating the observed cell cycle-regulatory effects of MCMV.
MATERIALS AND METHODS
Viruses and cells.
The Smith strain of MCMV (provided by Hartmut Hengel, Düsseldorf) was grown in primary murine embryonic fibroblasts (MEF). The MCMV recombinants MW97.01 (62), MCMVdie1 (19), and MCMVdie3 and MCMVrev (1) were propagated by passage on NIH 3T3-Bam25 cells (1). Both the recombinant viruses and the complementing cell line were provided by Ana Angulo, Barcelona. The HCMV laboratory strain AD169 (ATCC) was grown on human embryonic lung (HEL) fibroblasts. NIH 3T3 cells (ATCC CRL-1658), NIH 3T3-Bam25 cells and primary MEF were maintained in Dulbecco's modified Eagle's medium supplemented with 5% newborn calf serum, 5% fetal calf serum, 5 mM glutamine, 100 U/ml penicillin, and 100 μg/ml streptomycin. HEL fibroblasts were maintained in Eagle minimum essential medium supplemented with Earle's balanced salt solution, 25 mM HEPES, 1 mM sodium pyruvate, 2 mM glutamine, nonessential amino acids, 50 μg/ml gentamicin, 5% fetal calf serum, and 5% newborn calf serum. Virus were titers were determined on 24-well plates by standard plaque assays. A multiplicity of infection (MOI) of 5 PFU per cell was used for infection experiments. Where indicated, ganciclovir was used at a final concentration of 50 μM.
Plasmids and transfections.
Full-length cDNAs of MCMV immediate-early genes ie1, ie2, and ie3 were PCR amplified from a cDNA preparation of MCMV-infected cells, thereby introducing flanking restriction sites suitable for cloning into the eukaryotic expression vector pSG5-3HA, which contains an in-frame triple-hemagglutinin (3HA) epitope tag (66). The correctness of the resulting plasmids pSG5-3HA-ie1, pSG5-3HA-ie2, and pSG5-3HA-ie3 was confirmed by sequencing. The HCMV IE2 expression plasmid pSG5-3HA-IE2 was designed correspondingly as described before (66). The plasmid pMACS Kk.II expressing a truncated H-2Kk surface marker was obtained from Miltenyi Biotec (Bergisch Gladbach). All plasmids were purified by CsCl ethidium bromide equilibrium centrifugation before being used for transfection. The pSG5-3HA-derived effector plasmids and the pMACS Kk.II marker plasmid were used in a 4:1 molar ratio to transfect proliferating NIH 3T3 cells. An optimized calcium phosphate coprecipitation method was used for transfection (14). Cells were washed and supplied with fresh medium at 16 h and harvested at 48 h posttransfection. Where indicated, nocodazole at a final concentration of 50 ng/ml was added to the cells 24 h before harvesting.
Immunoblot analysis.
After harvest, cell lysates were prepared by sonicating cells in 50 mM Tris-Cl (pH 6.8)-2% sodium dodecyl sulfate-10% glycerol-1 mM dithiothreitol-2 μg/ml aprotinin-10 μg/ml leupeptin-1 μM pepstatin-0.1 mM Pefabloc. Lysates were clarified by centrifugation at 17,500 × g, and then the protein concentration was determined using the Bio-Rad DC protein assay kit. The lysates were adjusted to equal protein concentration, supplemented with 100 mM dithiothreitol and bromphenol blue, and heated to 95°C for 5 min. Sodium dodecyl sulfate-polyacrylamide gel electrophoresis and immunoblotting were performed essentially as described previously (67). The following primary antibodies were used: rat anti-HA (clone 3F10) from Roche (Mannheim) and mouse anti-GAPDH (anti-glyceraldehyde-3-phosphate dehydrogenase) (mAbcam 9484) from Abcam (Cambridge, United Kingdom).
Proliferation assay and flow cytometry.
Cells were plated at a density of 1 × 104 cells/cm2, infected with MCMV the day after, harvested at the indicated time points, and counted in a hemacytometer. To measure cellular DNA content, cells were first suspended in 1 volume 0.25 M sucrose-40 mM sodium citrate (pH 7.4), and then 4 volumes of phosphate-buffered saline (PBS)-0.5% NP-40-0.5 mM EDTA-25 μg/ml propidium iodide-250 μg/ml RNase A were added. The cell suspension was incubated for 10 min at 25°C and analyzed by flow cytometry using the CellQuest and Modfit software packages (Becton Dickinson). To measure both DNA content and protein expression, cells were first permeabilized by incubation in 75% ethanol for at least 12 h at 0°C. Afterwards, cells were stained with fluorescently labeled antibodies and propidium iodide as previously described (64). The mouse monoclonal antibody CroMA101, provided by Stipan Jonjic (Rijeka, Croatia), was used to detect the MCMV ie1 protein. A rat monoclonal antibody was used for detection of MCMV ie2 and a rat antiserum for detection of MCMV ie3. Both were provided by Yoshihiro Tsutsui (Hamamatsu, Japan). For detection of HCMV IE1/IE2 the antibody clone E13 (Argene) was used. The monoclonal antibody 6G3 (Cell Signaling) was used to measure histone H3 phosphorylation at serine 10. An Alexa 488-conjugated goat anti-mouse antibody (Molecular Probes) served as the secondary antibody. To discriminate transfected from nontransfected cells, a fluorescein isothiocyanate-labeled anti-H-2Kk antibody (Myltenyi Biotec) was used. All experiments were performed at least three times, and only representative results were included in the figures.
Cell cycle synchronization.
To synchronize HEL fibroblasts in G0, they were kept in serum-free medium for 3 days. For restimulation, this medium was replaced by fresh medium containing 10% serum. For synchronization in G1, cells were grown to confluence and left contact inhibited for 1 to 2 days. To allow for reentry into the cell cycle, cells were replated at lower density. Where indicated, the replated cells were further synchronized at the G1/S transition by treatment with 1 mM hydroxyurea (HU). To relieve the HU block, cells were washed several times with normal growth medium.
Analysis of chromatin condensation.
After harvest, cells were first subjected to hypotonic treatment with 0.075 M KCl for 10 min at 37°C. Cells were then fixed in 3:1 (vol/vol) methanol-acetic acid for several hours. Afterwards, cells were dropped onto microscope slides and stained with phosphate-buffered (pH 6.8) Giemsa's azur-eosin-methylene blue solution (Merck) following the manufacturer's instructions. Phase-contrast images of the cells were obtained by using an Axiovert S100 inverted microscope (Zeiss).
UV irradiation.
To purify MCMV particles, infectious cell culture supernatant was centrifuged for 30 min at 25,000 rpm in an SW40Ti rotor (Beckman). The virus pellet was resuspended in PBS and transferred in 1-ml quantities to petri dishes of 6 cm in diameter. The suspension was evenly distributed over the dish and irradiated with the indicated doses of UV light using an UV-Stratalinker 2400 (Stratagene). In addition, PBS without virus was irradiated and used for mock infections. To determine whether virion activities were still preserved after UV treatment, an NF-κB electrophoretic mobility shift assay was performed as previously described (44).
RESULTS
Inhibition of cell proliferation by MCMV.
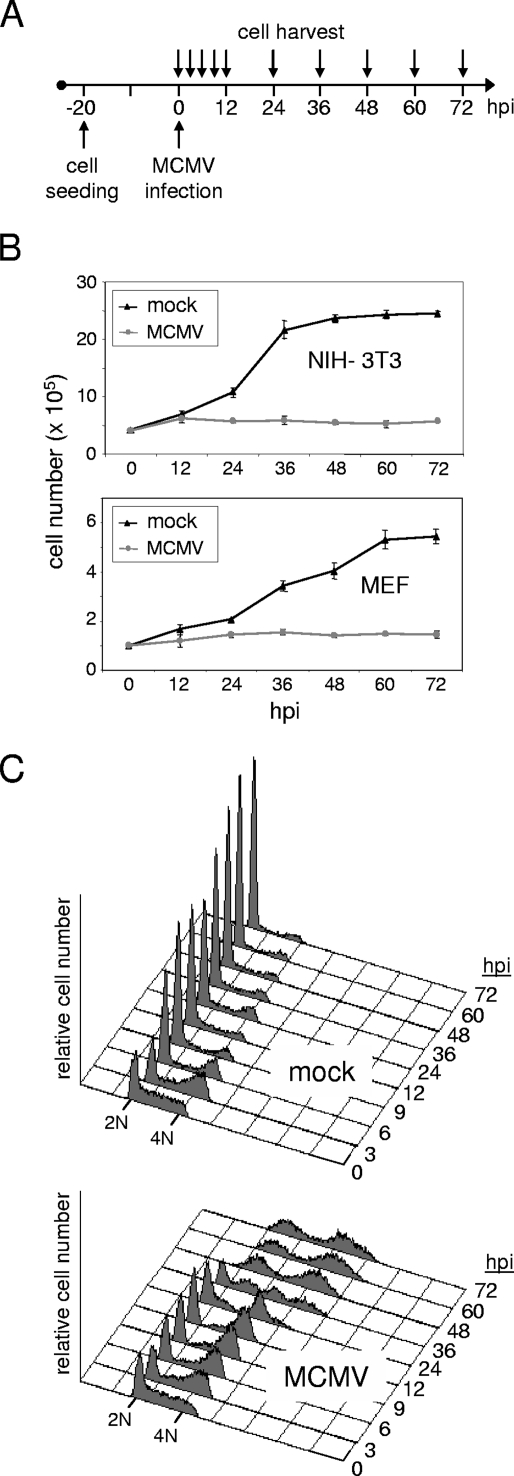
We first set out to analyze the general influence of MCMV on cell proliferation. To this end, we infected murine fibroblast cell cultures matched in cell numbers with MCMV or a mock control according to the schematic in Fig. 1A. In addition to an immortalized cell line (NIH 3T3), we included primary MEF in the analysis to control for potential cell type-specific effects and for effects that might be pertinent to immortalized cells. As can be seen from Fig. 1B, cells were seeded at a density that allowed primary cell cultures a linear proliferation rate over almost 3 days, whereas the immortalized 3T3 cells grew faster, reaching a growth plateau after 48 h. In contrast to mock-infected cultures, MCMV-infected cultures did not increase in cell number to any appreciable extent (Fig. 1B). As monitored by light microscopy, the stability in cell number appeared to be the consequence of a lack of cellular proliferation rather than a loss of cells due to cell death (data not shown). Also, the fact that both cellular systems tested behaved in a similar manner shows that the reduced proliferation rate is not a cell type-specific effect following CMV infection. Given the estimated doubling time of cells and the finding that the antiproliferative effect could be observed as early as 8 to 12 h postinfection (hpi), these results indicate that cells infected with MCMV cease to proliferate in an early phase of infection.
FIG. 1.
MCMV inhibits proliferation by arresting cells at two points in the cell division cycle. (A) Experimental setup. Proliferating mouse fibroblasts were infected with MCMV (MOI = 5) the day after plating. (B and C) Infected and mock-infected control cells were harvested at the indicated time points and analyzed for cell number (B) and cell cycle distribution (C). Time points between 0 and 12 hpi are applicable only to the analysis of cell cycle distribution. (B) Shown are growth curves where the absolute number of cells per dish is given for each time point of harvest. Mean values and standard deviations represent the results of four independent experiments. (C) DNA histograms of 3T3 cells, in which the relative DNA content is plotted against cell number. A 2N DNA content is indicative of G0/G1 cells and a 4N DNA content of cells in either G2 or mitosis.
MCMV arrests cells with G1 and G2/M DNA contents.
In order to examine whether MCMV-infected cells were blocked at a specific phase of the cell division cycle, we compared their DNA content with that of mock-infected cells by flow cytometry. In principle, we were following the same time course as shown above (Fig. 1A), but this time we included more time points within the first 12 h of infection. The analysis was initiated by splitting a near-confluent culture 20 h prior to infection. This led to a partly synchronized culture containing high numbers of S-phase cells at the time of infection (Fig. 1C). Three hours later, the majority of cells had acquired a G2/M DNA content regardless of whether they were MCMV infected or not. However, while mock-infected cells underwent cell division between 3 and 9 hpi, as indicated by the sharp increase of the G1 peak and decrease of the G2/M peak, MCMV-infected cells continued to accumulate with a G2/M DNA content. This happened at the expense of the S-phase population, which was progressively depleted, whereas the fraction of cells with a G1 DNA content slightly increased from 3 to 6 hpi and then remained constant until 24 hpi. The most likely explanation for these findings is that MCMV interfered with normal cell cycle progression at two points, i.e., before the onset and after the completion of cellular DNA synthesis. These findings are consistent with the loss of proliferative capacity of MCMV-infected cells shown in Fig. 1B. Furthermore, the observed arrest late in the cell cycle, just before control cells started to divide, also explained why the proliferation defect of MCMV-infected cells was observed so early after infection.
MCMV prevents mitotic entry.
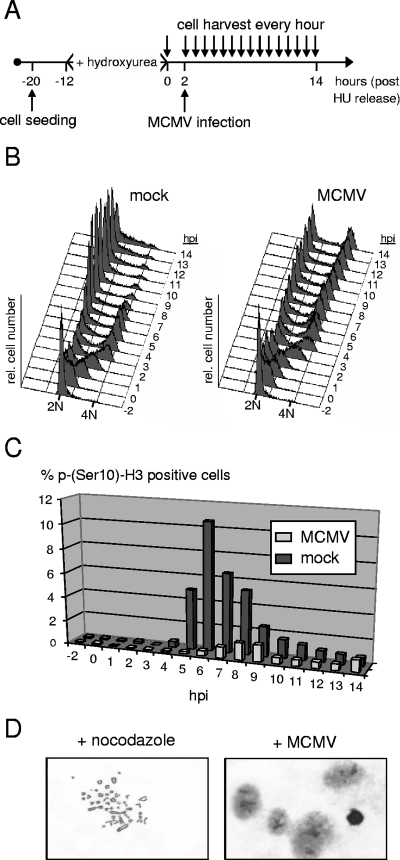
The block that was induced by MCMV late in the cell cycle was unexpected, since it is without precedent in HCMV infection. Whether it occurred in G2 phase or mitosis was not possible to judge from the above analysis because these two cell cycle phases cannot be differentiated on the basis of cellular DNA content. To address this point directly, we analyzed MCMV-arrested cells for the presence of a widely used mitotic marker, the phosphorylation of histone H3 at residue serine 10. This specific histone modification is involved in the genome-wide condensation of cellular chromatin early in mitosis. In principle, we used the same experimental setup as in Fig. 1 but further increased the level of cell cycle synchronization by arresting cells at the G1/S transition with HU (Fig. 2A). Two hours after release from the HU block, when most cells (about 65%) had entered early S phase (Fig. 2B), cultures were infected with MCMV. This procedure allowed an enriched fraction of control cells to simultaneously traverse through the relatively short period of time (typically 30 to 60 min) that is required for mitosis, enabling us to better monitor the kinetics of mitotic entry and exit. The proportion of mock-infected cells staining positive for H3 serine 10 phosphorylation sharply increased at the 5-hpi time point, reached a maximum of 11% around 6 hpi, and faded away after 8 hpi (Fig. 2C). This is in accordance with the DNA content analysis performed in parallel using an aliquot of this culture (Fig. 2B), suggesting that most control cells had completed mitosis within a time window of 4 h. In contrast, only a small minority of MCMV-infected cells entered and passed through mitosis (Fig. 2C), consistent with the small increase of the G1 population shown in Fig. 1C. Instead, the vast majority of cells were arrested with a 4N DNA content apparently prior to entering mitosis. This finding was further confirmed by looking at the state of chromatin condensation in a more direct way, using microscopic imaging of specifically stained cellular chromatin. Unlike control NIH 3T3 cells, which were kept under nocodazole treatment and hence were arrested in prometaphase, MCMV-arrested cells displayed only a diffuse chromatin staining (Fig. 2D). Thus, the MCMV-induced late cell cycle block clearly occurs before cells enter mitosis.
FIG. 2.
The late cell cycle block of MCMV-infected cells occurs after completion of S phase but prior to the onset of mitosis. (A) Experimental setup. Density-arrested NIH 3T3 fibroblasts were first replated at lower density and then treated with HU for synchronization at G1/S. Two hours after removal of HU, cells were MCMV or mock infected. (B) During the following 14 h, cell cycle progression was monitored at constant intervals of 1 h by flow cytometry. (C) In addition, the proportion of mitotic cells was determined for each sample by analyzing phosphorylation of histone H3 at serine 10. (D) To analyze chromatin condensation in a more direct way, cells arrested by MCMV with a G2 DNA content were stained with Giemsa's solution and examined by light microscopy. Nocodazole-treated cells were used as a positive control. Representative results are shown.
MCMV DNA replication is equally efficient in cells arrested with a G1 or G2 DNA content.
As can be seen from Fig. 1C a further dramatic change in the cell cycle profiles of MCMV-infected cells became apparent between 24 and 48 hpi. During this time window, MCMV-infected cells were shifted from a normal 2N or 4N to a ca. 50% higher DNA content. This was reminiscent of HCMV-infected cells, which when arrested with a G1 DNA content in the early phase of infection, start to accumulate newly synthesized DNA during later stages of the infectious cycle due to the considerable output of the viral replication machinery (38). To address the question whether in the case of MCMV-infected cells the increase in DNA content was also a consequence of viral DNA synthesis rather than, e.g., due to overreplication of the cellular genome, we infected cells in the presence or absence of the viral DNA polymerase inhibitor ganciclovir. Again, between 24 and 48 hpi the aforementioned changes were observed in infected cells (Fig. 3, upper panel). An overall increase of DNA content obscured the normal cell cycle profile, resulting in two remaining peaks, one positioned between a 2N and 4N DNA content and the second one beyond the 4N DNA content of normal G2 cells. In the presence of ganciclovir, these changes were entirely absent (Fig. 3, lower panel). This is consistent with the view that the newly synthesized DNA is likely to be of viral origin, since in MCMV-infected cells ganciclovir is only inefficiently phosphorylated (63), which in turn suggests a significantly higher affinity of this nucleotide analog for the repression of viral over cellular DNA synthesis. Therefore, and in analogy to HCMV, it appears that it is MCMV DNA replication that leads to the observed increase in the DNA content of MCMV-infected cells. Thus, remarkably, and in contrast to HCMV, MCMV is able not only to block cell cycle progression in G1 and G2 but also to replicate its genome in both cell cycle compartments with similar efficiency.
FIG. 3.
MCMV replicates both in cells arrested with a G1 DNA content and in cells arrested with a G2 DNA content. Asynchronously proliferating NIH 3T3 cells were MCMV infected (+MCMV) as described in the legend to Fig. 1. Where indicated, ganciclovir (+GCV) was added immediately after infection. Cells were harvested at the indicated time points, and their DNA content was analyzed by flow cytometry.
MCMV immediate-early gene expression is cell cycle independent.
The finding that both G1 and G2 cells fully support viral DNA synthesis implies also that the preceding steps of the viral replicative cycle are fully in place and not restricted to G1 cells as in HCMV-infected cells. To directly test the prediction of cell cycle-independent MCMV immediate-early gene expression, we analyzed MCMV-infected cells by flow cytometry for the expression of the major immediate-early genes ie1, ie2, and ie3 as a function of cell cycle position. We did the analysis at 4 hpi, when, according to our above findings, the normal cell cycle distribution of infected cells is still undisturbed by MCMV (Fig. 1). At this time the level of ie1 expression is known to exceed by far that of ie2 and ie3, which are expressed at only low (ie3) to moderate (ie2) abundance (30). This was reflected by the different intensities of antibody staining in our analysis (Fig. 4, far right panels). Importantly, though, all three immediate-early proteins showed an entirely cell cycle-independent expression pattern. Thus, MCMV lacks the G1 dependence of HCMV immediate-early gene expression, explaining its ability to block progression and to replicate late in the cell cycle. In addition, the fact that S-phase cells readily support viral gene expression but are not arrested by the virus before reaching G2 underscores the specificity of the MCMV-induced cell cycle arrest in G1 and G2.
FIG. 4.
MCMV lacks the strict cell cycle dependence of HCMV major immediate-early gene expression. Proliferating 3T3 fibroblasts were infected with MCMV or a mock control. Cells were harvested at 4 hpi and double stained with propidium iodide and fluorescently labeled antibodies against the indicated MCMV immediate-early proteins. Cells were analyzed by flow cytometry for cellular DNA content and viral gene expression. Data are depicted as dot plots and histogram overlays.
The differences in cell cycle regulation between MCMV and HCMV are virus but not host cell specific.
As outlined above, the cell cycle-independent expression of MCMV immediate-early genes was unexpected and was required to be addressed in more detail. Therefore, we set out to directly compare the two viruses in cross-species infection assays. We took advantage of the fact that human fibroblasts can be efficiently infected with MCMV and are fully permissive for its immediate-early gene expression program (27, 31, 35, 58). Similarly, murine fibroblasts have been reported to allow murine immediate-early gene expression after infection with HCMV (35). In the case of HCMV-infected human fibroblasts, we readily observed the well-described phenomenon of G1-dependent IE1/IE2 gene expression (Fig. 5A, upper panels), and a very similar observation was made with HCMV-infected murine fibroblasts (Fig. 5A, lower panels). In contrast, virtually all MCMV-infected human cells stained positive for ie1 expression irrespective of their cell cycle position (Fig. 5A, middle panels). This clearly indicates the significance of virus over host determinants in the establishment of cell cycle-independent (MCMV) or -dependent (HCMV) viral gene expression programs.
FIG. 5.
Conservation of MCMV cell cycle characteristics in human fibroblasts. (A) Asynchronously proliferating HEL fibroblasts and 3T3 cells were infected with MCMV or HCMV as indicated. Cells were harvested at 4 hpi and analyzed for DNA content and viral immediate-early gene expression as detailed in the legend to Fig. 4. (B and C) HEL fibroblasts were serum starved for 3 days and then restimulated by serum readdition (0 h). Restimulated cells were infected by HCMV or MCMV or left noninfected. Infections were carried out at the beginning of serum stimulation or at 18 h after restimulation, when cells were just entering S phase. Cells were harvested at 2-h intervals between 20 and 36 h and analyzed for cell cycle distribution by flow cytometry.
This further prompted us to ask whether the cell cycle-independent initiation of MCMV gene expression in human fibroblasts also translates into an early and late cell cycle block like in murine cells. To test this, we synchronized cells by serum starvation and then infected serum-restimulated cells at either the G0/G1 or G1/S transition (Fig. 5B). As expected, HCMV was very efficient in blocking S-phase entry but largely unable to block cell cycle progression in S, G2, and M phases (Fig. 5C, middle panels). Remarkably, MCMV was able to prevent the onset of cellular DNA synthesis just as efficiently as HCMV, and in addition, MCMV also led to a slowdown of S-phase progression that gave way to a marked G2 arrest at later times of infection (right panels). Of note, MCMV-infected cells showed no signs of viral DNA replication, as demonstrated by the absence of the increased DNA content that becomes visible after 32 hpi in HCMV-infected G0/G1 cells (upper middle panel). This is consistent with a previous study showing that viral DNA synthesis is inefficient in MCMV-infected human fibroblasts (27, 58). Taken together, the cross-species infection experiments provide clear evidence that the late cell cycle block of MCMV is a virus-related and not a host-specific phenomenon that correlates well with the S/G2 expression of MCMV immediate-early genes in infected cells.
MCMV cell cycle arrest depends on de novo expression of viral genes.
To clarify whether the cell cycle-independent initiation of viral gene expression rather than, for instance, preexisting virion proteins would be necessary for the cell cycle arrest capacity of MCMV, we next selectively prevented de novo synthesis of viral gene products by UV irradiation of viral particles. In order to minimize undesired side effects of UV treatment, we first performed a dose-response experiment and defined a UV dose (1,000 J/m2) that was sufficient to fully suppress immediate early gene expression (Fig. 6A) but left virion activities such as the induction of the NF-κB signaling pathway intact (Fig. 6B). The same dose of UV light completely abolished both the G1 and G2 arrest activities of MCMV (Fig. 6C). Indeed, irradiated virions displayed no cell cycle activities at all, as DNA profiles of cells infected with UV-irradiated MCMV were indistinguishable from those of mock-infected cells. These data strongly suggest that the onset of viral gene expression is a prerequisite for both the early and late cell cycle blocks in MCMV-infected cells.
FIG. 6.
De novo expression of viral genes is needed for the MCMV-mediated G1 and G2 cell cycle arrest. (A and B) Purified MCMV particles were irradiated with increasing doses of UV light and used to infect NIH 3T3 cells. Cells were harvested at 24 hpi to analyze the number of MCMV ie1-expressing cells by flow cytometry (A) or at 1 hpi to measure the extent of virus-induced NF-κB activation by electrophoretic mobility shift assay (B). (C and D) NIH 3T3 cells were released from the density arrest and infected in G1 phase (4 h after replating) (C) or early S phase (15 h after replating) (D) as indicated. MCMVUV, MCMV irradiated with 1,000 J/m2 UV-C light. Cell cycle progression was monitored at constant intervals by flow cytometry analysis of cellular DNA content as indicated.
MCMV ie3 arrests cells with a G1 DNA content.
The finding of an MCMV-mediated cell cycle arrest that occurs in the early phase of infection and relies on de novo viral gene expression led us to examine whether, as in HCMV, one or more of the MCMV major immediate-early gene products have cell cycle arrest activity. We used a transfection-based approach similar to that we have previously employed to identify the cell cycle arrest function of HCMV IE2 (66). Expression of ie1, ie2, and ie3 in proliferating 3T3 cells was detected by immunoblot analysis using an antibody recognizing the HA tag fused to the N termini of these MCMV proteins (Fig. 7A). Upon expression of ie1 and ie2, we failed to detect any abnormality in cell cycle progression (Fig. 7B, upper panels). In contrast, ie3 transfection led to a reproducible increase in the number of cells with G1 DNA content (67% of all cells, compared to 48% in the empty vector control). This was in a range comparable to that for HCMV IE2, which is known to arrest mouse fibroblasts (66) and therefore served as a positive control. To test whether the ie3-induced changes were caused by a block of the G1/S transition or by altered cell cycle kinetics such as, for example, an acceleration of S/G2, we performed a “mitotic trap” experiment. This is based on nocodazole-dependent inhibition of microtubule formation and leads to trapping of cycling cells in prometaphase. As expected, ie1- and ie2- as well as control-transfected cells accumulated in the presence of nocodazole with a 4N DNA content (Fig. 7B, lower panels). In contrast, only a minority of ie3-transfected cells acquired a 4N DNA content, suggesting that ie3, at least at the level of DNA replication, inhibits most cells from passing through the cell cycle and therefore prevents these cells from encountering the nocodazole block. Although in this setting HCMV IE2 showed an even stronger effect, the results indicated that ie3, like its human homologue, has a clear antiproliferative capacity.
FIG. 7.
MCMV ie3 has cell cycle arrest activity. HA-tagged versions of the indicated CMV genes or an empty vector control were transfected together with the KK surface marker (Miltenyi Biotec) into proliferating NIH 3T3 cells. (A) Cells were controlled for expression of the transfected genes by immunoblot analysis. An anti-HA antibody was used for detection of the viral proteins and an anti-GAPDH antibody to control for equal loading. (B) The cells were further analyzed for cell cycle distribution of the KK positive fraction by flow cytometry. To assay for G1 arrest activity, cycling cells were blocked in prometaphase by nocodazole treatment (+ noco, lower panel). Cell cycle distributions are given as percent G1/percent S/percent G2 + M.
MCMV cell cycle arrest depends on ie3.
Having shown that ie3 is sufficient to block cell cycle progression when expressed by itself outside the viral context, we next asked whether ie3 is necessary for inducing the cell cycle arrest mediated by MCMV. To this end, we made use of existing MCMV deletion mutants lacking major parts of ie1 (exon 4) or ie3 (exon 5) (1, 19). In agreement with the data from our transfection experiment (see above), the lack of ie1 had no influence on the cell cycle arrest function of MCMV, as the ie1 deletion mutant (MCMVdie1) blocked cells in G1 and G2 with the same efficiency as parental virus (Fig. 8). In contrast, cultures infected with the ie3 deletion mutant (MCMVdie3) behaved like mock-infected cells, showing a normal progression through the cell division cycle (Fig. 8). This suggested that ie3 expression is required by MCMV for both the early and late cell cycle arrest. To prove that the loss of cell cycle arrest function was indeed a consequence of ie3 deletion, we further included in our analysis a revertant virus in which the ie3 open reading frame was restored. This revertant (MCMVrev) displayed the full cell cycle arrest capacity of wild-type MCMV (Fig. 8), demonstrating the specificity of our finding. Hence, ie3 not only is sufficient to induce a cell cycle arrest when expressed outside the context of the viral genome but also is a necessary prerequisite for the prominent cell cycle block in MCMV-infected cells.
FIG. 8.
ie3 is essential for the MCMV-mediated cell cycle arrest. NIH 3T3 cells were released from density arrest and infected in G1 phase (4 h after replating) (A) or early to mid-S phase (16 h after replating) (B) with the indicated MCMV recombinants (MCMV, parental virus; MCMVdie1, ie1 deletion mutant; MCMVdie3, ie3 deletion mutant; MCMVrev, ie3 revertant virus). Cell cycle progression was monitored at constant intervals by flow cytometry of cellular DNA content as indicated.
DISCUSSION
This is the first comprehensive analysis of interactions between MCMV and the cell cycle. We found that MCMV uses one of its major immediate-early gene products, ie3, to induce an inhibition of cellular DNA synthesis and of cell division. Thus, a major strategy applied by HCMV to gain control over the host's cell cycle is conserved in its murine relative. We also revealed a clear difference between the two viruses in that MCMV lacks the strict G1 dependence of HCMV immediate-early gene expression. Instead, MCMV is able to start up its replicative cycle regardless of the current cell cycle position of the host cell. As a result, this virus can efficiently arrest the cell cycle at two points: in G1 (possibly at the G1/S transition) and G2, depending on the time of infection. In contrast, HCMV blocks cell cycle progression predominantly at the G1/S transition, and only a minor subpopulation of G2-arrested cells can be found after HCMV infection of proliferating cells (17, 38). Such differences between human and murine CMVs have to be taken into consideration whenever one is trying to employ an MCMV-based animal model for studying the relevance of CMV-mediated cell cycle alterations in vivo.
Our finding that MCMV arrests fibroblasts with either a G1 or a G2 DNA content is in general agreement with an earlier study showing that MCMV infection inhibits proliferation and cellular DNA synthesis of fetal neuronal stem cells (32). However, it stands in contrast to a recent study using time lapse video microscopy to demonstrate undisturbed cell divisions in MCMV-infected MEF (40). There are several possible explanations for this discrepancy. First, those authors used a genetically modified virus in which the ie1/ie3 N terminus was fused to green fluorescent protein (GFP). It is conceivable that the relatively large GFP domain affects the ie3 cell cycle arrest function in a negative way. Second, their analysis was obviously performed at a low MOI because, judged by the number of GFP-positive cells, the infection rate was only 10 to 20%. Since our experiments were carried out at high MOI (5 to 10 PFU per cell), leading to productive infection of nearly every cell, we cannot rule out a reduced cell cycle arrest capacity at low MOI. Third, cells were analyzed at a single-cell level in that, study and only a small number (two out of seven) of infected cells presented were undergoing cell division during the first 20 h of infection. This complicates a direct comparison to the flow cytometry-based data presented here, providing cell cycle profiles of whole-cell populations.
The observation of a ganciclovir-sensitive increase of DNA content in both G1- and G2-arrested cells suggests that both cell cycle compartments are supportive for viral DNA synthesis. It might appear paradoxical to find that a virus known to depend on the host's nucleotide metabolism (20, 37) replicates its genome outside S phase. However, it is well documented that MCMV is able, even under conditions of growth factor deprivation, to upregulate a number of enzymes involved in the synthesis of nucleoside triphosphate precursors (13, 20, 36). The expression of those genes normally is controlled in an E2F-Rb-dependent manner and therefore closely associated with S-phase progression and proliferation. This suggests that MCMV, similarly to HCMV, has evolved mechanisms to uncouple S-phase-specific gene expression from cellular DNA replication.
Of crucial importance for understanding the differences in cell cycle regulation between MCMV and HCMV is the finding of a cell cycle-independent start of immediate-early gene expression in MCMV-infected cells. Because the molecular basis for the repression of HCMV immediate-early gene expression in S/G2 phase is unknown, one can only speculate about the reason for the higher permissiveness of S/G2 cells for MCMV. One important distinction can be made from the results of our cross-species infection experiments. They show that the cell cycle-independent expression of MCMV ie1 takes place even in MCMV-infected human cells and, vice versa, that HCMV major immediate-early expression is cell cycle dependent in murine fibroblasts. This proves that the different behaviors of MCMV and HCMV are not a mere consequence of the different biologies of human and mouse cells but rather goes back to intrinsic properties of the viruses. Possibly, it has to do with the slightly distinct mechanisms that the two viruses have evolved to establish a nuclear microenvironment favorable for the initiation of immediate-early gene expression at ND10 domains (40, 59). Alternatively, differences in the promoter/enhancer structure or other cis-acting regulatory elements residing in the major immediate-early gene loci may be responsible (25). Work in progress in our laboratory addresses this issue. On a more general note, one can expect two possible scenarios: either MCMV has evolved an activity that overcomes an otherwise hostile environment in S/G2 or HCMV has evolved a mechanism to avoid initiation of viral gene transcription in these cell cycle phases.
The fact that MCMV is able to establish a late cell cycle block not only in murine cells but also in human cells demonstrates that the MCMV-induced G2 arrest is a virus-specific, not a host-specific, phenomenon. Most likely, the expression of MCMV immediate-early genes is, as in murine cells, responsible for this arrest. An extrapolation from this finding is that HCMV, if able to express its immediate-early genes in S phase, might also elicit a G2 arrest. This assumption is supported by observations of Fortunato et al., who showed that the minor fraction of immediate-early gene-positive S-phase cells arising after infection of cells the near G1/S transition traverse S phase and finally accumulate in G2 (17). This interpretation of our data is summarized in the comparative model of CMV-mediated cell cycle arrest that envisages the cell cycle arrest functions of HCMV and MCMV as a consequence of their cell cycle-dependent expression of IE2 and ie3, respectively (Fig. 9).
FIG. 9.
Comparative model of CMV-mediated cell cycle arrest functions. This model summarizes our current knowledge of the interdependencies between CMV immediate-early gene expression and the cell cycle position of the host cell. Both HCMV and MCMV can infect cells throughout the cell cycle. The expression of the cell cycle regulator IE2 is restricted to G0/G1 cells during HCMV infection, which causes these cells to run into the well-described cell cycle block at the G1/S transition. In contrast, the cell cycle-independent expression of ie3 can result in early (G1/S) and late (G2) cell cycle blocks. The exact position of the early ie3-mediated cell cycle block is unknown, but in analogy to IE2 it appears likely that it occurs at a similar position as in HCMV-infected cells. This model implies that HCMV might also be able to block cells at a later time point of the cell cycle if immediate-early genes were expressed in those cell cycle phases.
The cellular mediators of the CMV-induced G2 arrest in human and mouse cells are still unknown. Recent studies discovering the ability of more and more herpesviruses, including HCMV, to activate the DNA damage checkpoint via the action of their immediate-early gene products (5, 11, 39, 57, 60) could offer an attractive explanation. Because this checkpoint typically arrests the cell cycle at two points, i.e., G1 and G2, such a single mechanism would satisfy all the criteria we established so far from our experimental results.
Our finding that ie3 has an autonomous cell cycle arrest function and is essential for induction of the G1 and G2 arrest in MCMV-infected cells adds a further activity to this positional, structural, and functional homologue of HCMV IE2 (1, 10, 42). The fact that we observed only a G1 arrest and no G2 arrest in ie3-transfected cells is not necessarily in disagreement with an essential role of ie3 in the establishment of both cell cycle blocks. Either ie3 expression alone is not sufficient for inducing a G2 arrest and needs the contribution of other, yet-undefined viral factors or, more likely from our viewpoint, this observation might be due to the limitations of our ie3 expression system. The nuclear uptake of transiently transfected plasmids is allowed to occur only during mitosis, when the barrier of the nuclear membrane disintegrates (47). Therefore, expression from the ie3 expression plasmid starts in G1, and hence, cells expressing significant levels of ie3 are likely to be arrested before they enter or even traverse S phase to reach later cell cycle phases. Future investigations are required to show which of the possible scenarios will apply. Regarding the experimental setups that have been used so far to study the effects of HCMV IE2 on cell cycle progression, one cannot exclude that IE2 has a yet-overlooked G2 arrest activity. Even studies employing viral transduction methods to examine IE2-mediated cell cycle effects have used serum-starved cells as starting material and made no use of the possibility of transducing cycling cells or cells synchronized in S phase (34, 45, 46).
A recent study described an HCMV mutant that has lost the ability to arrest cells with a G1 DNA content due to a single amino acid exchange within the IE2 C terminus (48). Parts of the IE2 C terminus, including the critical arginine at position 548, are conserved in ie3 (3). This opens up the intriguing possibility of introducing the same mutation in the MCMV genome and, provided that this has similar consequences as for HCMV, to use such a mutant to study the relevance of the CMV cell cycle arrest function in vivo.
Acknowledgments
We are grateful to Ana Angulo and Hartmut Hengel for the generous supply of cells and viruses; to Wolfram Brune, Stipan Jonjic, and Yoshihiro Tsutsui for providing antibodies; and to Martin Messerle for the gift of various plasmids. We thank Iris Gruska and Christina Montag for their help with electrophoretic mobility shift assays and Nadezhda Glezeva and Ralf Uecker for their excellent technical assistance.
This work was supported by grants HA 1575/2-1 and WI 2043/2-2 from the Deutsche Forschungs-gemeinschaft (DFG).
Footnotes
Published ahead of print on 30 July 2008.
REFERENCES
- 1.Angulo, A., P. Ghazal, and M. Messerle. 2000. The major immediate-early gene ie3 of mouse cytomegalovirus is essential for viral growth. J. Virol. 7411129-11136. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Bain, M., and J. Sinclair. 2007. The S phase of the cell cycle and its perturbation by human cytomegalovirus. Rev. Med. Virol. 17423-434. [DOI] [PubMed] [Google Scholar]
- 3.Barrasa, M. I., N. Harel, Y. Yu, and J. C. Alwine. 2003. Strain variations in single amino acids of the 86-kilodalton human cytomegalovirus major immediate-early protein (IE2) affect its functional and biochemical properties: implications of dynamic protein conformation. J. Virol. 774760-4772. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Biswas, N., V. Sanchez, and D. H. Spector. 2003. Human cytomegalovirus infection leads to accumulation of geminin and inhibition of the licensing of cellular DNA replication. J. Virol. 772369-2376. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Boutell, C., and R. D. Everett. 2004. Herpes simplex virus type 1 infection induces the stabilization of p53 in a USP7- and ATM-independent manner. J. Virol. 788068-8077. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Bresnahan, W. A., T. Albrecht, and E. A. Thompson. 1998. The cyclin E promoter is activated by human cytomegalovirus 86-kDa immediate early protein. J. Biol. Chem. 27322075-22082. [DOI] [PubMed] [Google Scholar]
- 7.Bresnahan, W. A., I. Boldogh, P. Chi, E. A. Thompson, and T. Albrecht. 1997. Inhibition of cellular Cdk2 activity blocks human cytomegalovirus replication. Virology 231239-247. [DOI] [PubMed] [Google Scholar]
- 8.Bresnahan, W. A., I. Boldogh, E. A. Thompson, and T. Albrecht. 1996. Human cytomegalovirus inhibits cellular DNA synthesis and arrests productively infected cells in late G1. Virology 224150-160. [DOI] [PubMed] [Google Scholar]
- 9.Bresnahan, W. A., E. A. Thompson, and T. Albrecht. 1997. Human cytomegalovirus infection results in altered Cdk2 subcellular localization. J. Gen. Virol. 781993-1997. [DOI] [PubMed] [Google Scholar]
- 10.Busche, A., A. Angulo, P. Kay-Jackson, P. Ghazal, and M. Messerle. 2008. Phenotypes of major immediate-early gene mutants of mouse cytomegalovirus. Med. Microbiol. Immunol. 197233-240. [DOI] [PubMed] [Google Scholar]
- 11.Castillo, J. P., F. M. Frame, H. A. Rogoff, M. T. Pickering, A. D. Yurochko, and T. F. Kowalik. 2005. Human cytomegalovirus IE1-72 activates ataxia telangiectasia mutated kinase and a p53/p21-mediated growth arrest response. J. Virol. 7911467-11475. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Castillo, J. P., and T. F. Kowalik. 2004. HCMV infection: modulating the cell cycle and cell death. Int. Rev. Immunol. 23113-139. [DOI] [PubMed] [Google Scholar]
- 13.Cavallo, R., D. Lembo, G. Gribaudo, and S. Landolfo. 2001. Murine cytomegalovirus infection induces cellular folylpolyglutamate synthetase activity in quiescent cells. Intervirology 44224-226. [DOI] [PubMed] [Google Scholar]
- 14.Chen, C., and H. Okayama. 1987. High-efficiency transformation of mammalian cells by plasmid DNA. Mol. Cell. Biol. 72745-2752. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Dittmer, D., and E. S. Mocarski. 1997. Human cytomegalovirus infection inhibits G1/S transition. J. Virol. 711629-1634. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Fortunato, E. A., A. K. McElroy, I. Sanchez, and D. H. Spector. 2000. Exploitation of cellular signaling and regulatory pathways by human cytomegalovirus. Trends Microbiol. 8111-119. [DOI] [PubMed] [Google Scholar]
- 17.Fortunato, E. A., V. Sanchez, J. Y. Yen, and D. H. Spector. 2002. Infection of cells with human cytomegalovirus during S phase results in a blockade to immediate-early gene expression that can be overcome by inhibition of the proteasome. J. Virol. 765369-5379. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Fortunato, E. A., M. H. Sommer, K. Yoder, and D. H. Spector. 1997. Identification of domains within the human cytomegalovirus major immediate-early 86-kilodalton protein and the retinoblastoma protein required for physical and functional interaction with each other. J. Virol. 718176-8185. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Ghazal, P., A. E. Visser, M. Gustems, R. Garcia, E. M. Borst, K. Sullivan, M. Messerle, and A. Angulo. 2005. Elimination of ie1 significantly attenuates murine cytomegalovirus virulence but does not alter replicative capacity in cell culture. J. Virol. 797182-7194. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Gribaudo, G., L. Riera, D. Lembo, M. De Andrea, M. Gariglio, T. L. Rudge, L. F. Johnson, and S. Landolfo. 2000. Murine cytomegalovirus stimulates cellular thymidylate synthase gene expression in quiescent cells and requires the enzyme for replication. J. Virol. 744979-4987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Hagemeier, C., R. Caswell, G. Hayhurst, J. Sinclair, and T. Kouzarides. 1994. Functional interaction between the HCMV IE2 transactivator and the retinoblastoma protein. EMBO J. 132897-2903. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Hertel, L., S. Chou, and E. S. Mocarski. 2007. Viral and cell cycle-regulated kinases in cytomegalovirus-induced pseudomitosis and replication. PLoS Pathog. 3e6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Hertel, L., and E. S. Mocarski. 2004. Global analysis of host cell gene expression late during cytomegalovirus infection reveals extensive dysregulation of cell cycle gene expression and induction of pseudomitosis independent of US28 function. J. Virol. 7811988-12011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Hume, A. J., J. S. Finkel, J. P. Kamil, D. M. Coen, M. R. Culbertson, and R. F. Kalejta. 2008. Phosphorylation of retinoblastoma protein by viral protein with cyclin-dependent kinase function. Science 320797-799. [DOI] [PubMed] [Google Scholar]
- 25.Isomura, H., and M. F. Stinski. 2003. The human cytomegalovirus major immediate-early enhancer determines the efficiency of immediate-early gene transcription and viral replication in permissive cells at low multiplicity of infection. J. Virol. 773602-3614. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Jault, F. M., J. M. Jault, F. Ruchti, E. A. Fortunato, C. Clark, J. Corbeil, D. D. Richman, and D. H. Spector. 1995. Cytomegalovirus infection induces high levels of cyclins, phosphorylated Rb, and p53, leading to cell cycle arrest. J. Virol. 696697-6704. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Jurak, I., and W. Brune. 2006. Induction of apoptosis limits cytomegalovirus cross-species infection. EMBO J. 252634-2642. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Kalejta, R. F., J. T. Bechtel, and T. Shenk. 2003. Human cytomegalovirus pp71 stimulates cell cycle progression by inducing the proteasome-dependent degradation of the retinoblastoma family of tumor suppressors. Mol. Cell. Biol. 231885-1895. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Kalejta, R. F., and T. Shenk. 2002. Manipulation of the cell cycle by human cytomegalovirus. Front Biosci. 7d295-d306. [DOI] [PubMed] [Google Scholar]
- 30.Keil, G. M., A. Ebeling-Keil, and U. H. Koszinowski. 1987. Immediate-early genes of murine cytomegalovirus: location, transcripts, and translation products. J. Virol. 61526-533. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Kim, K. S., and R. I. Carp. 1972. Abortive infection of human diploid cells by murine cytomegalovirus. Infect. Immun. 6793-797. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Kosugi, I., Y. Shinmura, H. Kawasaki, Y. Arai, R. Y. Li, S. Baba, and Y. Tsutsui. 2000. Cytomegalovirus infection of the central nervous system stem cells from mouse embryo: a model for developmental brain disorders induced by cytomegalovirus. Lab. Investig. 801373-1383. [DOI] [PubMed] [Google Scholar]
- 33.Krmpotic, A., I. Bubic, B. Polic, P. Lucin, and S. Jonjic. 2003. Pathogenesis of murine cytomegalovirus infection. Microbes Infect. 51263-1277. [DOI] [PubMed] [Google Scholar]
- 34.Kronschnabl, M., M. Marschall, and T. Stamminger. 2002. Efficient and tightly regulated expression systems for the human cytomegalovirus major transactivator protein IE2p86 in permissive cells. Virus Res. 8389-102. [DOI] [PubMed] [Google Scholar]
- 35.Lafemina, R. L., and G. S. Hayward. 1988. Differences in cell-type-specific blocks to immediate early gene expression and DNA replication of human, simian and murine cytomegalovirus. J. Gen. Virol. 69355-374. [DOI] [PubMed] [Google Scholar]
- 36.Lembo, D., A. Angeretti, M. Gariglio, and S. Landolfo. 1998. Murine cytomegalovirus induces expression and enzyme activity of cellular dihydrofolate reductase in quiescent cells. J. Gen. Virol. 792803-2807. [DOI] [PubMed] [Google Scholar]
- 37.Lembo, D., G. Gribaudo, L. Riera, A. Mondo, R. Cavallo, A. Angeretti, and S. Landolfo. 2000. The thymidylate synthase inhibitor ZD1694 potently inhibits murine and human cytomegalovirus replication in quiescent fibroblasts. Antiviral Res. 47111-120. [DOI] [PubMed] [Google Scholar]
- 38.Lu, M., and T. Shenk. 1996. Human cytomegalovirus infection inhibits cell cycle progression at multiple points, including the transition from G1 to S. J. Virol. 708850-8857. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Luo, M. H., K. Rosenke, K. Czornak, and E. A. Fortunato. 2007. Human cytomegalovirus disrupts both ataxia telangiectasia mutated protein (ATM)- and ATM-Rad3-related kinase-mediated DNA damage responses during lytic infection. J. Virol. 811934-1950. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Maul, G. G., and D. Negorev. 2008. Differences between mouse and human cytomegalovirus interactions with their respective hosts at immediate early times of the replication cycle. Med. Microbiol. Immunol. 197241-249. [DOI] [PubMed] [Google Scholar]
- 41.McElroy, A. K., R. S. Dwarakanath, and D. H. Spector. 2000. Dysregulation of cyclin E gene expression in human cytomegalovirus-infected cells requires viral early gene expression and is associated with changes in the Rb-related protein p130. J. Virol. 744192-4206. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Messerle, M., B. Buhler, G. M. Keil, and U. H. Koszinowski. 1992. Structural organization, expression, and functional characterization of the murine cytomegalovirus immediate-early gene 3. J. Virol. 6627-36. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Mocarski, E. S., T. Shenk, and R. F. Pass. 2007. Cytomegaloviruses, p. 2701-2772. In D. M. Knipe, P. M. Howley, D. E. Griffin, R. A. Lamb, M. A. Martin, B. Roizman, and S. E. Straus (ed.), Fields virology, 5th ed. Lippincott Williams & Wilkins, Philadelphia, PA.
- 44.Montag, C., J. Wagner, I. Gruska, and C. Hagemeier. 2006. Human cytomegalovirus blocks tumor necrosis factor alpha- and interleukin-1β-mediated NF-κB signaling. J. Virol. 8011686-11698. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Murphy, E. A., D. N. Streblow, J. A. Nelson, and M. F. Stinski. 2000. The human cytomegalovirus IE86 protein can block cell cycle progression after inducing transition into the S phase of permissive cells. J. Virol. 747108-7118. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Noris, E., C. Zannetti, A. Demurtas, J. Sinclair, M. De Andrea, M. Gariglio, and S. Landolfo. 2002. Cell cycle arrest by human cytomegalovirus 86-kDa IE2 protein resembles premature senescence. J. Virol. 7612135-12148. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Pellegrin, P., A. Fernandez, N. J. Lamb, and R. Bennes. 2002. Macromolecular uptake is a spontaneous event during mitosis in cultured fibroblasts: implications for vector-dependent plasmid transfection. Mol. Biol. Cell 13570-578. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Petrik, D. T., K. P. Schmitt, and M. F. Stinski. 2006. Inhibition of cellular DNA synthesis by the human cytomegalovirus IE86 protein is necessary for efficient virus replication. J. Virol. 803872-3883. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Prichard, M. N., E. Sztul, S. L. Daily, A. L. Perry, S. L. Frederick, R. B. Gill, C. B. Hartline, D. N. Streblow, S. M. Varnum, R. D. Smith, and E. R. Kern. 2008. Human cytomegalovirus UL97 kinase activity is required for the hyperphosphorylation of retinoblastoma protein and inhibits the formation of nuclear aggresomes. J. Virol. 825054-5067. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Reddehase, M. J., J. Podlech, and N. K. Grzimek. 2002. Mouse models of cytomegalovirus latency: overview. J. Clin. Virol. 25(Suppl. 2)S23-S36. [DOI] [PubMed] [Google Scholar]
- 51.Salvant, B. S., E. A. Fortunato, and D. H. Spector. 1998. Cell cycle dysregulation by human cytomegalovirus: influence of the cell cycle phase at the time of infection and effects on cyclin transcription. J. Virol. 723729-3741. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Sanchez, V., A. K. McElroy, and D. H. Spector. 2003. Mechanisms governing maintenance of Cdk1/cyclin B1 kinase activity in cells infected with human cytomegalovirus. J. Virol. 7713214-13224. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Sanchez, V., A. K. McElroy, J. Yen, S. Tamrakar, C. L. Clark, R. A. Schwartz, and D. H. Spector. 2004. Cyclin-dependent kinase activity is required at early times for accurate processing and accumulation of the human cytomegalovirus UL122-123 and UL37 immediate-early transcripts and at later times for virus production. J. Virol. 7811219-11232. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Sanchez, V., and D. H. Spector. 2006. Cyclin-dependent kinase activity is required for efficient expression and posttranslational modification of human cytomegalovirus proteins and for production of extracellular particles. J. Virol. 805886-5896. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Sanchez, V., and D. H. Spector. 2006. Exploitation of host cell cycle regulatory pathways by HCMV, p. 205-230. In M. J. Reddehase (ed.), Cytomegaloviruses. Caister Academic Press, Norfolk, United Kingdom.
- 56.Song, Y. J., and M. F. Stinski. 2002. Effect of the human cytomegalovirus IE86 protein on expression of E2F-responsive genes: a DNA microarray analysis. Proc. Natl. Acad. Sci. USA 992836-2841. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Song, Y. J., and M. F. Stinski. 2005. Inhibition of cell division by the human cytomegalovirus IE86 protein: role of the p53 pathway or cyclin-dependent kinase 1/cyclin B1. J. Virol. 792597-2603. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Tang, Q., and G. G. Maul. 2006. Mouse cytomegalovirus crosses the species barrier with help from a few human cytomegalovirus proteins. J. Virol. 807510-7521. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Tang, Q., and G. G. Maul. 2003. Mouse cytomegalovirus immediate-early protein 1 binds with host cell repressors to relieve suppressive effects on viral transcription and replication during lytic infection. J. Virol. 771357-1367. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Tarakanova, V. L., V. Leung-Pineda, S. Hwang, C. W. Yang, K. Matatall, M. Basson, R. Sun, H. Piwnica-Worms, B. P. Sleckman, and H. W. t. Virgin. 2007. Gamma-herpesvirus kinase actively initiates a DNA damage response by inducing phosphorylation of H2AX to foster viral replication. Cell Host Microbe 1275-286. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Tran, K., J. A. Mahr, J. Choi, J. G. Teodoro, M. R. Green, and D. H. Spector. 2008. Accumulation of substrates of the anaphase-promoting complex (APC) during human cytomegalovirus infection is associated with the phosphorylation of Cdh1 and the dissociation and relocalization of APC subunits. J. Virol. 82529-537. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Wagner, M., S. Jonjic, U. H. Koszinowski, and M. Messerle. 1999. Systematic excision of vector sequences from the BAC-cloned herpesvirus genome during virus reconstitution. J. Virol. 737056-7060. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Wagner, M., D. Michel, P. Schaarschmidt, B. Vaida, S. Jonjic, M. Messerle, T. Mertens, and U. Koszinowski. 2000. Comparison between human cytomegalovirus pUL97 and murine cytomegalovirus (MCMV) pM97 expressed by MCMV and vaccinia virus: pM97 does not confer ganciclovir sensitivity. J. Virol. 7410729-10736. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Wiebusch, L., J. Asmar, R. Uecker, and C. Hagemeier. 2003. Human cytomegalovirus immediate-early protein 2 (IE2)-mediated activation of cyclin E is cell-cycle-independent and forces S-phase entry in IE2-arrested cells. J. Gen. Virol. 8451-60. [DOI] [PubMed] [Google Scholar]
- 65.Wiebusch, L., M. Bach, R. Uecker, and C. Hagemeier. 2005. Human cytomegalovirus inactivates the G0/G1-APC/C ubiquitin ligase by Cdh1 dissociation. Cell Cycle. 41435-1439. [DOI] [PubMed] [Google Scholar]
- 66.Wiebusch, L., and C. Hagemeier. 1999. Human cytomegalovirus 86-kilodalton IE2 protein blocks cell cycle progression in G1. J. Virol. 739274-9283. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Wiebusch, L., and C. Hagemeier. 2001. The human cytomegalovirus immediate early 2 protein dissociates cellular DNA synthesis from cyclin-dependent kinase activation. EMBO J. 201086-1098. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Wiebusch, L., R. Uecker, and C. Hagemeier. 2003. Human cytomegalovirus prevents replication licensing by inhibiting MCM loading onto chromatin. EMBO Rep. 442-46. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Zhang, Z., S. M. Huong, X. Wang, D. Y. Huang, and E. S. Huang. 2003. Interactions between human cytomegalovirus IE1-72 and cellular p107: functional domains and mechanisms of up-regulation of cyclin E/cdk2 kinase activity. J. Virol. 7712660-12670. [DOI] [PMC free article] [PubMed] [Google Scholar]