Abstract
The alc gene of bacteriophage T4 was originally defined on the basis of mutations which allow late protein synthesis directed by T4 DNA containing cytosine rather than hydroxymethylcytosine. The question remained whether the normal alc gene product (gpalc) also blocks the transcription of early genes from cytosine-containing DNA. Complementation experiments were performed between hydroxymethylcytosine-containing phage which direct gpalc synthesis but carry mutations in a given gene(s) and cytosine-containing phage carrying that gene(s). The required protein would then have to be directed by the cytosine-containing DNA: it is looked for directly on polyacrylamide gels or through its physiological effects or both. For all early proteins examined in this way, no synthesis was observed when 95 to 100% of the hydroxymethylcytosine was substituted by cytosine in the infecting DNA, whereas there was significant synthesis with 75% substitution or less. The results indicate that gpalc is carried in with the infecting DNA or is made very early to block transcription of all cytosine-containing DNA.
Full text
PDF


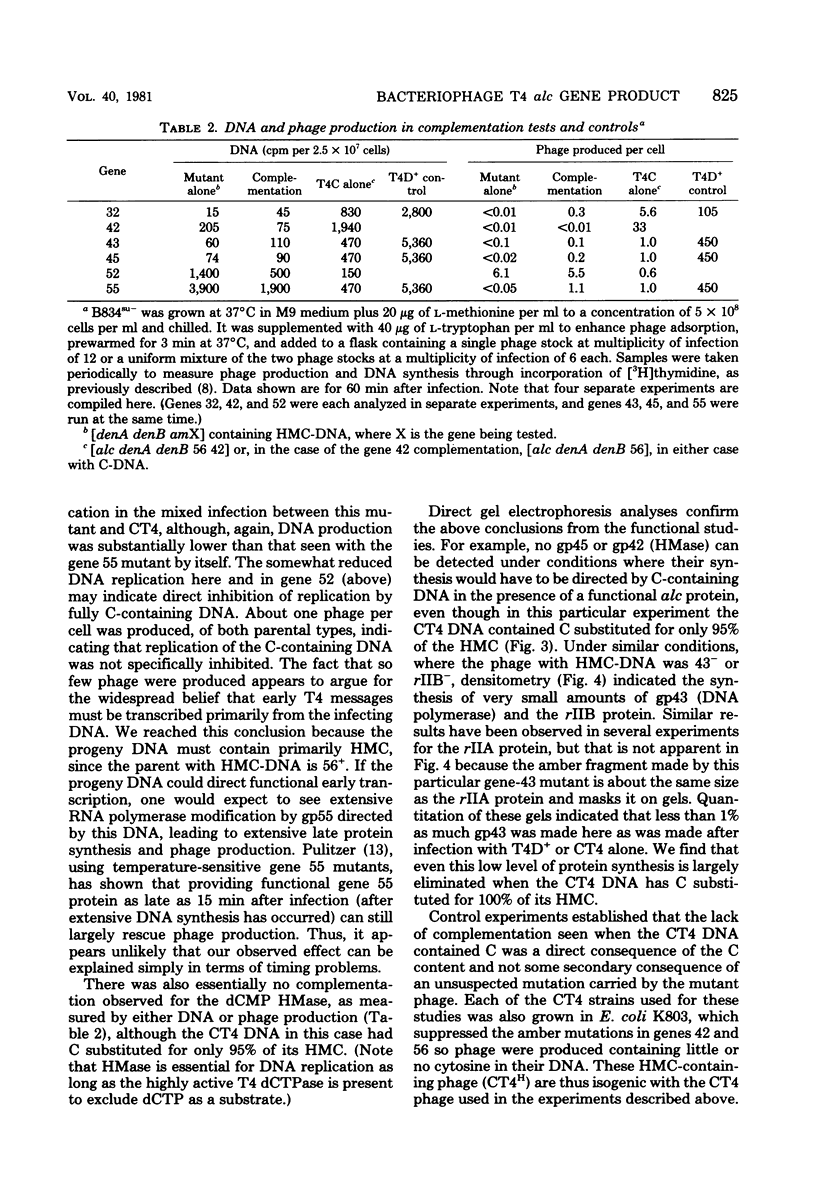
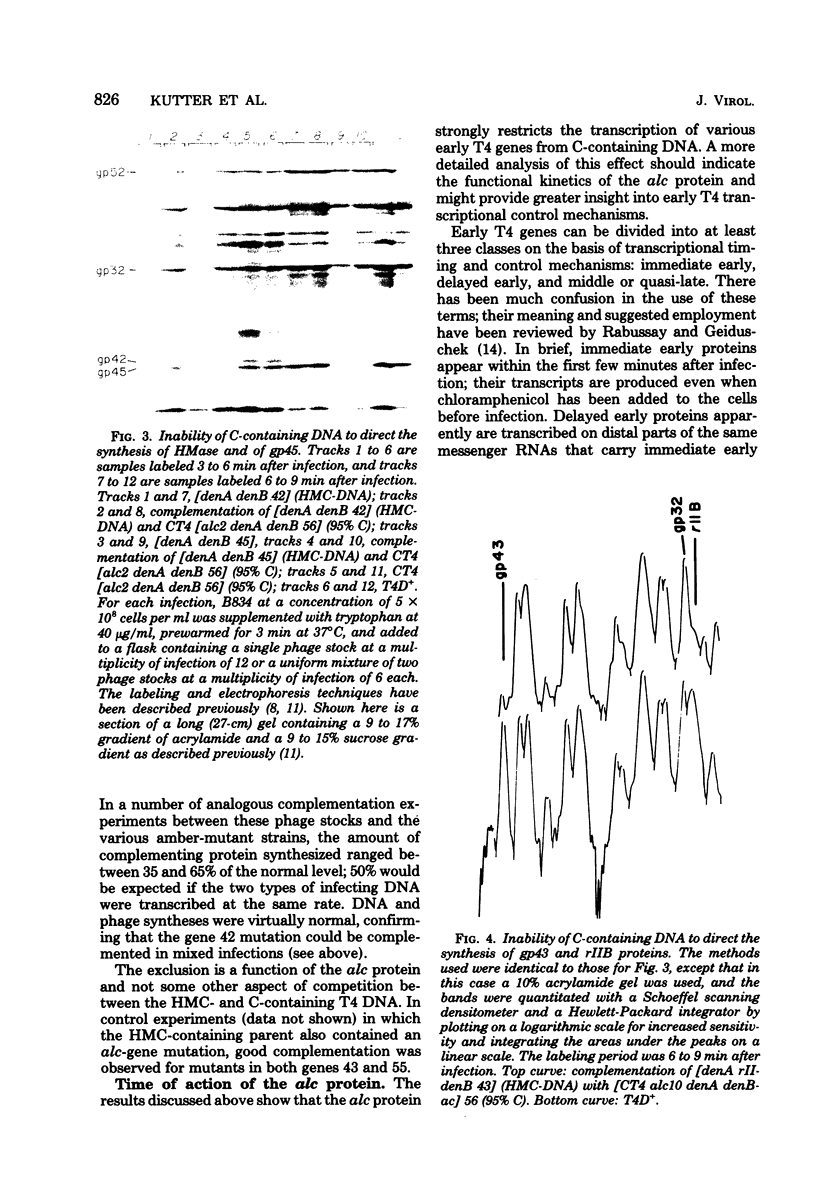



Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Bolle A., Epstein R. H., Salser W., Geiduschek E. P. Transcription during bacteriophage T4 development: synthesis and relative stability of early and late RNA. J Mol Biol. 1968 Feb 14;31(3):325–348. doi: 10.1016/0022-2836(68)90413-0. [DOI] [PubMed] [Google Scholar]
- Brody E. N., Diggelmann H., Geiduschek E. P. Transcription of the bacteriophage T4 template. Obligate synthesis of T4 prereplicative RNA in vitro. Biochemistry. 1970 Mar 17;9(6):1289–1299. doi: 10.1021/bi00808a001. [DOI] [PubMed] [Google Scholar]
- Homyk T., Jr, Weil J. Deletion analysis of two nonessential regions of the T4 genome. Virology. 1974 Oct;61(2):505–523. doi: 10.1016/0042-6822(74)90286-4. [DOI] [PubMed] [Google Scholar]
- Horvitz H. R. Polypeptide bound to the host RNA polymerase is specified by T4 control gene 33. Nat New Biol. 1973 Aug 1;244(135):137–140. doi: 10.1038/newbio244137a0. [DOI] [PubMed] [Google Scholar]
- Hosoda J., Levinthal C. Protein synthesis by Escherichia coli infected with bacteriophage T4D. Virology. 1968 Apr;34(4):709–727. doi: 10.1016/0042-6822(68)90092-5. [DOI] [PubMed] [Google Scholar]
- Koerner J. F., Snustad D. P. Shutoff of host macromolecular synthesis after T-even bacteriophage infection. Microbiol Rev. 1979 Jun;43(2):199–223. doi: 10.1128/mr.43.2.199-223.1979. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kutter E. M., Wiberg J. S. Degradation of cytosin-containing bacterial and bacteriophage DNA after infection of Escherichia coli B with bacteriophage T4D wild type and with mutants defective in genes 46, 47 and 56. J Mol Biol. 1968 Dec;38(3):395–411. doi: 10.1016/0022-2836(68)90394-x. [DOI] [PubMed] [Google Scholar]
- Kutter E., Beug A., Sluss R., Jensen L., Bradley D. The production of undegraded cytosine-containing DNA by bacteriophage T4 in the absence of dCTPase and endonucleases II and IV, and its effects on T4-directed protein synthesis. J Mol Biol. 1975 Dec 25;99(4):591–607. doi: 10.1016/s0022-2836(75)80174-4. [DOI] [PubMed] [Google Scholar]
- Mattson T., Richardson J., Goodin D. Mutant of bacteriophage T4D affecting expression of many early genes. Nature. 1974 Jul 5;250(461):48–50. doi: 10.1038/250048a0. [DOI] [PubMed] [Google Scholar]
- Morton D., Kutter E. M., Guttman B. S. Synthesis of T4 DNA and bacteriophage in the absence of dCMP hydroxymethylase. J Virol. 1978 Oct;28(1):262–269. doi: 10.1128/jvi.28.1.262-269.1978. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pearson R. E., Snyder L. Shutoff of lambda gene expression by bacteriophage T4: role of the T4 alc gene. J Virol. 1980 Jul;35(1):194–202. doi: 10.1128/jvi.35.1.194-202.1980. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pulitzer J. F. Function of T4 gene 55. I. Characterization of temperature-sensitive mutations in the "maturation" gene 55. J Mol Biol. 1970 Apr 28;49(2):473–488. doi: 10.1016/0022-2836(70)90258-5. [DOI] [PubMed] [Google Scholar]
- Ratner D. The interaction bacterial and phage proteins with immobilized Escherichia coli RNA polymerase. J Mol Biol. 1974 Sep 15;88(2):373–383. doi: 10.1016/0022-2836(74)90488-4. [DOI] [PubMed] [Google Scholar]
- Rohrer H., Zillig W., Mailhammer R. ADP-ribosylation of DNA-dependent RNA polymerase of Escherichia coli by an NAD+: protein ADP-ribosyltransferase from bacteriophage T4. Eur J Biochem. 1975 Dec 1;60(1):227–238. doi: 10.1111/j.1432-1033.1975.tb20995.x. [DOI] [PubMed] [Google Scholar]
- Sadowski P. D., Hurwitz J. Enzymatic breakage of deoxyribonucleic acid. I. Purification and properties of endonuclease II from T4 phage-infected Escherichia coli. J Biol Chem. 1969 Nov 25;244(22):6182–6191. [PubMed] [Google Scholar]
- Sadowski P. D., Hurwitz J. Enzymatic breakage of deoxyribonucleic acid. II. Purification and properties of endonuclease IV from T4 phage-infected Escherichia coli. J Biol Chem. 1969 Nov 25;244(22):6192–6198. [PubMed] [Google Scholar]
- Silverstein J. L., Goldberg E. B. T4 DNA injection. II. Protection of entering DNA from host exonuclease V. Virology. 1976 Jul 1;72(1):212–223. doi: 10.1016/0042-6822(76)90324-x. [DOI] [PubMed] [Google Scholar]
- Sirotkin K., Wei J., Snyder L. T4 Bacteriophage-coded RNA polymerase subunit blocks host transcription and unfolds the host chromosome. Nature. 1977 Jan 6;265(5589):28–32. doi: 10.1038/265028a0. [DOI] [PubMed] [Google Scholar]
- Snustad D. P., Tigges M. A., Parson K. A., Bursch C. J., Caron F. M., Koerner J. F., Tutas D. J. Identification and preliminary characterization of a mutant defective in the bacteriophage T4-induced unfolding of the Escherichia coli nucleoid. J Virol. 1976 Feb;17(2):622–641. doi: 10.1128/jvi.17.2.622-641.1976. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Snyder L., Gold L., Kutter E. A gene of bacteriophage T4 whose product prevents true late transcription on cytosine-containing T4 DNA. Proc Natl Acad Sci U S A. 1976 Sep;73(9):3098–3102. doi: 10.1073/pnas.73.9.3098. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stevens A. Deoxyribonucleic acid dependent ribonucleic acid polymerases from two T4 phage-infected systems. Biochemistry. 1974 Jan 29;13(3):493–503. doi: 10.1021/bi00700a015. [DOI] [PubMed] [Google Scholar]
- Stevens A. New small polypeptides associated with DNA-dependent RNA polymerase of Escherichia coli after infection with bacteriophage T4. Proc Natl Acad Sci U S A. 1972 Mar;69(3):603–607. doi: 10.1073/pnas.69.3.603. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Szybalski W. Initiation and patterns of transcription during phage development. Proc Can Cancer Conf. 1969;8:183–215. [PubMed] [Google Scholar]
- Tigges M. A., Bursch C. J., Snustad D. P. Slow switchover from host RNA synthesis to bacteriophage RNA synthesis after infection of Escherichia coli with a T4 mutant defective in the bacteriophage T4-induced unfolding of the host nucleoid. J Virol. 1977 Dec;24(3):775–785. doi: 10.1128/jvi.24.3.775-785.1977. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tomich P. K., Chiu C. S., Wovcha M. G., Greenberg G. R. Evidence for a complex regulating the in vivo activities of early enzymes induced by bacteriophage T4. J Biol Chem. 1974 Dec 10;249(23):7613–7622. [PubMed] [Google Scholar]
- Travers A. A. Positive control of transcription by a bacteriophage sigma factor. Nature. 1970 Mar 14;225(5237):1009–1012. doi: 10.1038/2251009a0. [DOI] [PubMed] [Google Scholar]
- Wilson G. G., Tanyashin V. I., Murray N. E. Molecular cloning of fragments of bacteriophage T4 DNA. Mol Gen Genet. 1977 Nov 14;156(2):203–214. doi: 10.1007/BF00283493. [DOI] [PubMed] [Google Scholar]
- Wood W. B. Host specificity of DNA produced by Escherichia coli: bacterial mutations affecting the restriction and modification of DNA. J Mol Biol. 1966 Mar;16(1):118–133. doi: 10.1016/s0022-2836(66)80267-x. [DOI] [PubMed] [Google Scholar]