Abstract
The major fungal pathogen of humans, Aspergillus fumigatus, lacks a defined sexual cycle, although the presence of genes encoding putative mating type idiomorphs and regulators of Aspergillus sexual development heightens the potential for cryptic sexuality in this deuteromycete. To test the functionality of these genetic determinants, we transferred the alpha box-encoding mat1-1 idiomorph from an A. fumigatus isolate to the homothallic fertile species Aspergillus nidulans. Abundant formation of fruiting bodies (cleistothecia) containing viable ascospores establishes functionality of this mating type gene product in the transgenic strain. Using a similar approach, we also established that the conserved transcriptional regulator from A. fumigatus, the nsdD gene product, can act as a functional, positively acting factor for A. nidulans cleistothecium development; moreover, high-level expression of NsdD in the endogenous host A. fumigatus profoundly alters hyphal development by triggering the formation of coiled hyphae. Our findings demonstrate that the presumably asexual pathogen A. fumigatus encodes functional regulators of mating and sexual development, thereby potentiating the case for cryptic sexuality in this fungal pathogen.
In recent decades, pathogenic fungi, such as Aspergillus spp., have gained significance as severe threats to human health, particularly in the immunocompromised individual. Recent genomic advances (10, 12, 27) have provided the opportunity to undertake comparative studies of pathogenic and nonpathogenic aspergilli. In an evolutionary context, restricted access to sexuality may have driven acquisition of pathogenicity in A. fumigatus (13, 18, 26). The existence of cryptic sexuality in this species has been proposed by Paoletti and coworkers, based on the near-1:1 distribution of both mating type idiomorphs among a population of worldwide isolates (29). Moreover, phylogenetic analyses support the occurrence of genetic recombination within the Aspergillus fumigatus population (32). As deduced from in silico analyses, the genome of the A. fumigatus sequence reference strain Af293 contains many putative gene products that are orthologous to recognized determinants of fruiting body formation in the fertile species Aspergillus nidulans (10, 27). Among these is the GATA-type transcription factor NsdD, which has been characterized as a crucial regulator of sexual development (14). NsdD acts positively on early steps of sexual reproduction in A. nidulans without affecting asexual conidiation, as demonstrated by analysis of deletion mutants and overexpression studies (16).
Sexual propagation in fungi is widespread and comprises distinct developmental steps to eventually form recombinant progeny from meiotic spores. To support genetic variability, two nuclei must fuse in a process termed karyogamy. Karyogamy follows fusion of haploid ascomycete cells, resulting in diploid formation. Diploid cells eventually undergo meiosis to produce ascospore-containing asci. These may be formed within specialized structures, the fruiting bodies, that differ in shape among the ascomycetous phylum (2). In order to establish nuclear identity, mating type systems have evolved (5, 7). In heterothallic ascomycetes, sexual reproduction is governed by a unique region of the chromosome known as the mating type (mat) locus, which exists in two idiomorphic forms. In dothideomycetes, one locus encodes a transcription factor with an α-box DNA binding domain whereas the other encodes a transcription factor with a high-mobility-group (HMG) domain. By convention (44), these are termed MAT1-1 and MAT1-2, respectively, for some ascomycetes, among them Aspergillus. Deuteromycetes (syn. fungi imperfecti), on the other hand, display an asexual life-style; however, some of them may have maintained ways of recombining genes even though sexual reproduction has not been observed.
Aspergillus species may be homothallic or heterothallic (8). From genome sequence analyses of the homothallic model ascomycete, A. nidulans, genes encoding α-box and HMG-domain proteins (matB and matA, respectively) were both found to be present and necessary for sexual reproduction (30). Homothallism does not therefore circumvent the need for the mating type proteins. The genome sequence of the apparently asexual deuteromycete A. fumigatus has revealed that the fungus is potentially heterothallic (9, 29, 31, 45), and functionality of its mat1-2 gene product, an HMG protein, has been demonstrated (34).
To further the working hypothesis that A. fumigatus harbors cryptic sexuality, we tested the functionality of conserved key factors of Aspergillus mating and sexual development. First, the A. fumigatus mating type mat1-1 open reading frame (ORF) obtained from a clinical isolate was assessed for its capacity to support sexual development in the ascomycetous host A. nidulans. Likewise, an A. fumigatus orthologue of the positive regulator of sexual development, NsdD, was found to support cleistothecium formation in A. nidulans upon overexpression and to rescue NsdD deficiency as judged by complementation studies. Thus, the presumed asexual pathogen A. fumigatus has retained functional regulators of mating and sexual development.
MATERIALS AND METHODS
Strains, media, and growth conditions.
For general cloning procedures, the bacterial strain Escherichia coli DH5α [F− φ80dlacZΔM15 Δ(lacZYA-argF)U169 recA1 endA1 hsdR17(rK− mK+) supE44 λ− thi-1 gyrA96 relA1] (47) was used, which was cultivated in LB (1% Bacto-tryptone, 0.5% yeast extract, 1% NaCl, pH 7.5) medium. Fungal strains used in this study are listed in Table 1. The A. fumigatus isolate D141 was typed to be a mat1-1 strain according to the method of Paoletti et al. (30) by a multiplex PCR approach using the three priming oligonucleotides AFM1, AFM2, and AFM3. Growth of Aspergillus strains was carried out at 37°C on minimal medium prepared according to the method of Käfer (20). Antibiotic concentrations were 100 μg/ml for ampicillin and 0.1 μg/ml for pyrithiamine; supplements for nutritional auxotrophies were added according to an online resource (http://www.gla.ac.uk/ibls/molgen/aspergillus/supplement.html).
TABLE 1.
Fungal strains
| Name | Descriptiona | Reference |
|---|---|---|
| FGSC A4 | Aspergillus nidulans Glasgow wild type | Fungal Genetics Stock Center |
| KHH52 | A. nidulans nsdDΔ deletion strain | 16 |
| TNO2A3 | A. nidulans nkuAΔ deletion strain; pyrG89 pyroA4 | 25 |
| AnS22 | Complementation of KHH52 by forced expression of AfnsdD via pSK393 | This study |
| AnS23 | Complementation of KHH52 by forced expression of AnnsdD via pSK395 | This study |
| AnS24 | Overexpression of AfnsdD in FGSC A4 via pSK393 | This study |
| AnS25 | Overexpression of AnnsdD in FGSC A4 via pSK395 | This study |
| AnS30 | A. nidulans expressing A. fumigatus MAT1-1 AfpyrG | This study |
| AnS31 | A. nidulans expressing A. fumigatus MAT1-1Δα-box allele; AfpyrG | This study |
| D141 | Aspergillus fumigatus wild-type strain (syn. NRRL 6585), clinical isolate [mat1-1] | 41 |
| Af293 | A. fumigatus genome sequence reference strain, clinical isolate [mat1-2] | 28 |
| AfS41 | pabaA::loxP; riboB::loxP derivative of D141 | This study |
| AfS45 | pabaA::loxP; pyroA::loxP derivative of Af293 | This study |
| AfS53 | Overexpression of AfnsdD via pSK393 in AfS41 | This study |
| AfS54 | Overexpression of AnnsdD via pSK395 in AfS41 | This study |
| AfS55 | Overexpression of AfnsdD via pSK393 in AfS45 | This study |
| AfS56 | Overexpression of AnnsdD via pSK395 in AfS45 | This study |
| AfS60 | Overexpression of AfnsdDA455P allele via pSK446 in AfS41 | This study |
AfnsdD, A. fumigatus nsdD; AnnsdD, A. nidulans nsdD. Other genes are shown similarly.
Transformation procedures.
Calcium-manganese-treated E. coli cells were used for transformation (17); Aspergillus recipients were transformed by polyethylene glycol-mediated fusion of protoplasts as described previously (33).
Manipulation of nucleic acids and plasmid constructions.
Standard protocols of recombinant DNA technology were carried out (36). Phusion high-fidelity DNA polymerase was used in PCRs (35), and essential cloning steps were verified by sequencing at the Göttingen Genomics Laboratory (G2L; www.g2l.bio.uni-goettingen.de) or GATC Biotech. Sequence analyses were carried out using the Lasergene biocomputing software package from DNAStar and the CLC Free Workbench 4 program. Fungal genomic DNA was prepared according to the method of Kolar et al. (21), and Southern analyses were carried out essentially as described previously (40). Total RNA samples were isolated using the TRIzol reagent of Invitrogen, followed by Northern hybridization according to the protocols cited by Brown and Mackey (3). Random primed labeling (11) was performed using the Stratagene Prime-It II Kit in the presence of [α-32P]dATP. Autoradiographs were produced by exposing washed membranes to Kodak X-oMAT films. cDNAs were amplified from total RNA preparations using the RevertAid H Minus First Strand cDNA synthesis kit from Fermentas. Plasmids constructed during the course of this study are listed and briefly described in Table 2; Table 3 lists the sequences of utilized oligonucleotides. The replacement cassette for the A. nidulans matB locus was generated using a fusion PCR approach (42), using amplicons covering the 5′ and 3′ region (Sv491/Sv492 and Sv497/Sv498, respectively), the A. fumigatus mat1-1 coding sequence (Sv493/Sv494), and the A. fumigatus pyrG locus (Sv495/Sv496) as a complementing marker gene; the Sv511/Sv512 pair of nested primers was used for amplification of the complete replacement module, which was inserted into the EcoRV site of the pJET1 plasmid (Fermentas) to yield pSK432. From this construct, a derivative replacement cassette with the mat1-1 gene carrying a deletion of the α-box was amplified by whole-vector PCR with primers Sv515 and Sv516 to result in pSK433. The expression cassette of pSK390 was generated by fusing the A. nidulans niiA/niaD intergenic region to the 3′ region of the A. fumigatus his2A gene to promote integration into this site of the recipient's genome. For forced nsdD expressions, cDNA amplicons generated with primer pair Sv135/Sv136 or Sv207/Sv406 to cover the coding sequences of the A. fumigatus or A. nidulans gene, respectively, were inserted into the PmeI site of pSK390 to yield the integrative vectors pSK393 and pSK395. The nsdDA455P expression plasmid pSK446 was constructed via fusion PCR using the oligonucleotides Sv549 and Sv550 together with the primer pair Sv135/Sv136. The resulting amplicon, which was inserted into PmeI of pSK390, carries a G-to-C substitution in the A. fumigatus nsdD ORF at position 1363, which changes an alanine residue at position 455 in the NsdD primary structure to a proline.
TABLE 2.
Plasmid constructs
| Name | Descriptiona | Reference |
|---|---|---|
| pBluescript II KS | General cloning plasmid (bla, MCS) | Stratagene |
| pJET1 | Positive selection vector, pUC19 derivative (bla, MCS in eco47IR) | Fermentas |
| pSK432 | 5′matB::mat1-1::AfpyrG::3′matB gene replacement cassette | This study |
| pSK433 | 5′matB::mat1-1Δα-box::AfpyrG::3′matB gene replacement cassette | This study |
| pSK390 | Aspergillus expression vector (AnpniiA/pniaD-PmeI-Afhis2At; ptrA) | This study |
| pSK393 | Construct for forced expression of A. fumigatus nsdD (pniaD::AfnsdD::his2At; ptrA) | This study |
| pSK395 | Construct for forced expression of A. nidulans nsdD (pniaD::AnnsdD::his2At; ptrA) | This study |
| pSK446 | Construct for forced expression of A. fumigatus nsdDA455P loss-of-function allele | This study |
AfpyrG, A. fumigatus pyrG; AnpniiA/pniaD, A. nidulans niiA/niaD promoter; other genes are shown similarly. MCS, multiple cloning site.
TABLE 3.
Oligonucleotides
| Name | Sequence |
|---|---|
| AFM1 | 5′-CCT TGA CGC GAT GGG GTG G-3′ |
| AFM2 | 5′-CGC TCC TCA TCA GAA CAA CTC G-3′ |
| AFM3 | 5′-CGG AAA TCT GAT GTC GCC ACG-3′ |
| Sv135 | 5′-ATG GGG TCA TTA GAG GCA ACA CAT AG-3′ |
| Sv136 | 5′-CTA TCG GGT ACC GGG AGG CGT GGG ATC-3′ |
| Sv207 | 5′-ATG GGA TCA CTA GAG GCT GGA C-3′ |
| Sv406 | 5′-TTA ATG ACT CCT CGG TGA CAC C-3′ |
| Sv491 | 5′-TTC GAG GTC CTC ACT CTT AAA GC-3′ |
| Sv492 | 5′-GAT TGC AGC TTC CAT GAC GGC TGG GGG ATT GGC GTT CTA TCT TGG-3′ |
| Sv493 | 5′-AAT CCC CCA GCC GTC ATG GAA GCT GCA ATC TCT CCC CTC GAG CGT GC-3′ |
| Sv494 | 5′-AAG AGG GTG AAG AGC TCA GAC GTT GAT GTA TTG ATC AAT GTC-3′ |
| Sv495 | 5′-TAC ATC AAC GTC TGA GCT CTT CAC CCT CTT CGC GGG TCT GAA ATA CC-3′ |
| Sv496 | 5′-CTG GAA CCA ACC GAG ACC TGT CTG AGA GGA GGC ACT GAT GCG-3′ |
| Sv497 | 5′-CTC CTC TCA GAC AGG TCT CGG TTG GTT CCA GGC AAC TCA TGA TC-3′ |
| Sv498 | 5′-AGT ACT ACG GAG TAG TGC CGG TGG-3′ |
| Sv511 | 5′-ATG AAA GAG ATA TAC AAC AAG G-3′ |
| Sv512 | 5′-ATC TCA CAA GAC CAA TGT ACC-3′ |
| Sv515 | 5′-GAC GAC CAT GAA AGC GAG G-3′ |
| Sv516 | 5′-TTT CTT TCC TTC CTG GGT GC-3′ |
| Sv549 | 5′-ATT GTG CAA TCC CTG TGG CCT ACA CTA TGC-3′ |
| Sv550 | 5′-TAG GCC ACA GGG ATT GCA CAA TGT CCG AGC-3′ |
Light and electron microscopy.
Aspergillus colonies and fruiting bodies were inspected using a Nikon SMZ800 binocular microscope combined with a Coolpix 4500 digital camera. Fungal hyphae were examined with an Olympus SZX12 binocular or a Zeiss Axiolab microscope; photographs were taken with a Kappa digital camera using the Image Base software package or with a Xillix Microimager digital camera in combination with the Improvision Openlab software. For scanning electron microscopy, samples from strains grown on culture medium-coated glass slides were essentially prepared according to the procedure described by Sohn and Yoon (39).
RESULTS
The A. fumigatus mating type gene mat1-1 codes for a functional orthologue of the A. nidulans α-box counterpart.
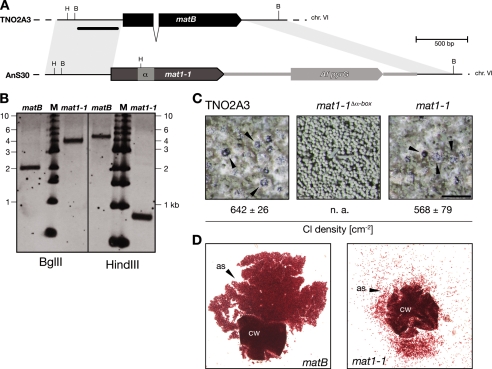
In contrast to the A. fumigatus genome sequence reference strain, Af293, which encodes the mat1-2 idiomorph, the clinical isolate D141 (41) was identified as a mat1-1 strain encoding an α-box domain protein (data not shown). Recent studies suggest the existence of a cryptic sexual cycle of A. fumigatus, which would require functional DNA binding proteins expressed from the respective mating type loci. To put this to the test, the native matB ORF of the homothallic species A. nidulans was swapped for its A. fumigatus orthologue by a one-step procedure (Fig. 1A). After transformation of the NHEJ-deficient recipient TNO2A3 (25) with a suitable gene replacement cassette, several isolates were recovered for which correct gene replacement could be validated by Southern hybridization analyses (Fig. 1B). One representative (AnS30) was chosen for phenotypic characterization. After 8 days of incubation under conditions favorable for sexual differentiation, its ability to form fruiting bodies at densities similar to the ones displayed by the recipient strain and with cleistothecial dimensions comparable to those of its progenitor strain was observed (Fig. 1C). As estimated from crushed cleistothecia, fruiting bodies of either origin contained red-pigmented ascospores (Fig. 1D). However, the number of ascospores was reduced in fruiting bodies derived from the mat1-1 transgenic strain (data not shown), and analysis of cleistothecial contents revealed the presence of ascogenous hyphae and ascocarp cells, suggestive of slower maturation. Continued incubation for an additional 4 days eventually resulted in mature fruiting bodies accompanied by formation of fertile ascospores, which displayed similar viabilities to those harvested from the progenitor strain (data not shown). When the endogenous matB gene of TNO2A3 was replaced by an allele of A. fumigatus mat1-1 in which the α-box had been deleted, cleistothecium formation was abolished in all (n = 8) validated transformants (Fig. 1C), which conclusively demonstrates the functionality of the A. fumigatus mat1-1-encoded DNA-binding protein for cleistothecial development.
FIG. 1.
The A. fumigatus mat1-1 ORF supports sexual development in A. nidulans. (A) Schematic presentation of matB/mat1-1 ORF replacement procedure showing the native matB locus from A. nidulans and the genome architecture after replacement with the A. fumigatus α-box protein-encoding mat1-1 ORF from isolate D141. Homologous regions are depicted by rhombic gray areas; BglII (B) and HindIII (H) recognition sites are indicated. The black bar represents the hybridizing probe in Southern analyses (B) of recipient strain TNO2A3 and one representative carrying the A. fumigatus mat1-1 gene after digestion of chromosomal DNA with BglII or HindIII; M, DNA molecular weight marker. (C) Colony appearance and cleistothecium (Cl) formation of strains AnS30 (mat1-1) and AnS31 (mat1-1Δα-box), expressing a functioning and malfunctioning allele of the A. fumigatus mat1-1 ORF, respectively: mature fruiting bodies (arrowheads) were formed only when functional MatB or Mat1-1 proteins were expressed from the endogenous matB locus in A. nidulans, and as estimated from cleistothecial densities, the A. fumigatus mat1-1 ORF supported fruiting body formation to an extent similar to that for its A. nidulans orthologue (scale bar, 500 μm; n. a., not applicable; Cl densities were determined from five different 0.5-cm2 areas of two independently inoculated culture plates). (D) Crushed cleistothecia from 8-day-old fruiting bodies of strain TNO2A3 (left) or AnS30 (right): ascosporogenesis of the mat1-1 strain appeared delayed, as indicated by the presence of unripe asci and ascogenous hyphae (cw, cleistothecial wall; as, ascospores). Yet after prolonged incubation, equal amounts of fertile ascospores were formed by the two strains.
A. fumigatus expresses a functional NsdD orthologue.
Having demonstrated that the deuteromycete A. fumigatus expresses mating type gene products that are functional in an ascomycetous host, we extended our analysis to another regulator of Aspergillus sexual development. From a BLAST search at the CADRE database (www.cadre-genomes.org.uk) (24), an A. fumigatus locus (Afu3g13870) with the capacity to encode an A. nidulans NsdD transcription factor orthologue was identified. Alignment of the translation product sequences illustrates large regions of identity, particularly among the carboxy-terminal residues (Fig. 2A), where the conserved DNA-binding motif resides. This conserved feature is a GATA zinc finger domain of the IVb type (43), characterized by a C-X2-C-X18-C-X2-C sequence, which is known to be required for functionality of A. nidulans NsdD in triggering sexual development (16). The zinc finger domain is identically present in the A. fumigatus orthologue, which hints at a conserved cellular role among the NsdD factors from A. fumigatus and A. nidulans.
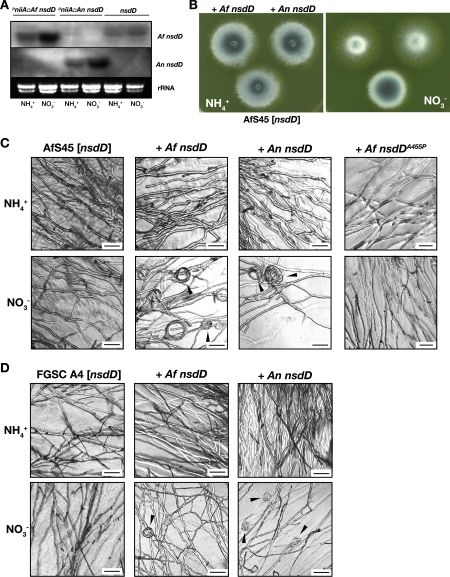
FIG. 2.
The NsdD orthologue of A. fumigatus is functional in A. nidulans. (A) Pairwise alignment of NsdD proteins from A. fumigatus and A. nidulans. The alignment was created based on accession number sequences Afu3g13870 (A. fumigatus) and AAB16914 (A. nidulans); identical residues are shown in black and nonconserved ones in gray, and the GATA zinc finger domain is boxed with the Ala455 residue of A. fumigatus NsdD highlighted. (B) Colony appearance of A. nidulans FGSC A4 derivatives expressing A. fumigatus (Af) or A. nidulans (An) nsdD from the nitrate-inducible niiA promoter under repressing and inducing conditions: overexpression of either nsdD gene results in repression of asexual sporulation and formation of numerous nests and fruiting bodies. (C) Complementation of deletant KHH52: the recipient strain is unable to form cleistothecia but develops mature fruiting bodies (arrows) that harbor fertile ascospores when either nsdD gene is expressed. Scale bar, 300 μm (upper panels) or 50 μm (lower panel).
We next tested functionality of the A. fumigatus NsdD protein by cross-complementation studies. To transfer the nsdD coding sequence from A. fumigatus to the heterologous host A. nidulans, a suitable expression vector was constructed by inserting a cDNA fragment into the plasmid pSK390. The A. fumigatus nsdD cDNA could be amplified from mycelial as well as sporulating samples, which indicates that the nsdD gene is indeed expressed in this asexual fungus. To assess whether forced expression of the A. fumigatus nsdD gene is able to trigger sexual development in a wild-type genetic background, the expression construct pSK393 was transformed into the A. nidulans strain FGSC A4. Overexpression of either native or A. fumigatus NsdD resulted in formation of numerous cleistothecia (Fig. 2B). In stark contrast, untransformed FGSC A4 formed only a few fruiting bodies under these conditions of illumination and aeration, which favor asexual sporulation and the formation of conidiophores. Transforming the expression constructs into the nsdD deletion recipient KHH52 revealed complementation capacities of the A. fumigatus nsdD coding sequence. The deletion recipient is characterized by retarded hyphal growth, earlier conidiophore formation, and most strikingly, the absence of any cellular structures of sexual development, such as Hülle cells or cleistothecia. In contrast, the majority (6 out of 8 for pSK393 and 11 out of 14 for pSK395) of the resulting transformants formed fruiting bodies on inducing medium (Fig. 2C). As a positive control, KHH52 was transformed with the expression construct harboring the A. nidulans nsdD coding sequence to display profound sexual development. Cleistothecia harvested from either transformant contained numerous ascospores that were able to germinate when transferred to culture medium. We therefore conclude that the A. fumigatus NsdD factor is able to replace its A. nidulans counterpart in a functional manner.
Forced expression of NsdD results in circular growing hyphae.
Given the pronounced phenotype of nsdD overexpression in fertile A. nidulans, we were interested in whether forced expression of this transcriptional activator in the asexual species A. fumigatus would result in any related phenotype. The expression constructs for the A. fumigatus and A. nidulans transcription factors were transformed into A. fumigatus strain AfS41, which is a descendant of the wild-type isolate D141, or strain AfS45, which stems from the reference strain Af293. To confirm proper and inducible transcription of each nsdD coding sequence, RNA preparations from selected transformants were subjected to Northern analysis (Fig. 3A). As expected, strong hybridization signals were obtained for samples from nitrate-containing cultures, whereas from medium with ammonium as the sole source of nitrogen, weaker expression of each transcript was evident. Transcription of the endogenous nsdD gene appeared to be repressed by forced expression of the A. nidulans gene, which supports previous data suggestive of autoregulation of NsdD expression (15). Validated transformants were inspected on inducing medium to find two distinct irregularities: the sporulation zone of colonies from transgenic A. fumigatus strains that expressed the nsdD genes at high levels was reduced (Fig. 3B), and moreover, transformants of both genetic backgrounds reproducibly formed coiled hyphal structures in the growth zone of mycelial extension (Fig. 3C). Whereas the majority of hyphal tip segments appeared normal and regularly shaped, some of them developed a curved and intertwining assembly, resulting in curls of approximately 20 μm in diameter. To exclude that these structures are due to expression of a foreign gene per se, a presumptive loss-of-function allele of the A. fumigatus nsdD gene encoding a proline instead of an alanine residue at position 455, which maps to the GATA zinc finger domain (37, 46), was expressed. The corresponding construct was unable to complement the nsdD deletion in strain KHH52 (data not shown) and did not result in hyphal coil formation in A. fumigatus (Fig. 3C).
FIG. 3.
Forced expression of nsdD in the deuteromycete A. fumigatus results in the formation of coiled hyphal structures. (A) Validation of forced nsdD overexpression in the A. fumigatus host strain AfS45 by Northern analysis. Autoradiographs derived from Northern analysis of RNA samples obtained under repressing (NH4+) or inducing (NO3−) conditions to demonstrate nsdD steady-state transcript levels expressed from the endogenous locus or the niiA promoter, respectively, are shown. Ethidium bromide-stained rRNA serves as a loading control. (B) Colony appearance of strains expressing the nsdD gene from A. fumigatus (Af) or A. nidulans (An). Expression constructs were introduced in the recipient strain AfS45, and confirmed transformants were inoculated on ammonium- or nitrate-containing medium. Reduced sporulation and altered hyphal extension at the colony periphery are characteristic for the transgenic strains expressing either nsdD gene at high levels. (C) Hyphal morphology resulting from forced expression of nsdD in A. fumigatus. The formation of coiled hyphal structures (arrows) resulting from curling tip extension is induced specifically by high nsdD expression, whereas expression of an nsdD loss-of-function allele does not support coil formation. (D) Coil formation in A. nidulans upon overexpression of nsdD. Strain FGSC A4 transformed with either construct for forced expression of the nsdD gene from A. fumigatus (AnS24) or A. nidulans (AnS25) displays fortified hyphal coil formation (arrowheads) on inducing medium. Scale bar, 50 μm.
To examine whether A. nidulans expressing any nsdD gene at high levels would form similar structures, strains AnS24 and AnS25 were inspected microscopically. In comparison to their progenitor, which formed hyphal coils very rarely, both strains displayed coiled hyphal tips at significantly higher frequencies on culture medium inducing expression of the nsdD gene (Fig. 3D). Accordingly, formation of these coils could be attributed specifically to the forced expression of either of the two NsdD transcriptional activators in two Aspergillus species, which further supports a conserved cellular function of this regulatory protein. By microscopic inspection, we were unable to track any further developmental fate of these hyphal coils or to correlate their appearance to cleistothecium formation.
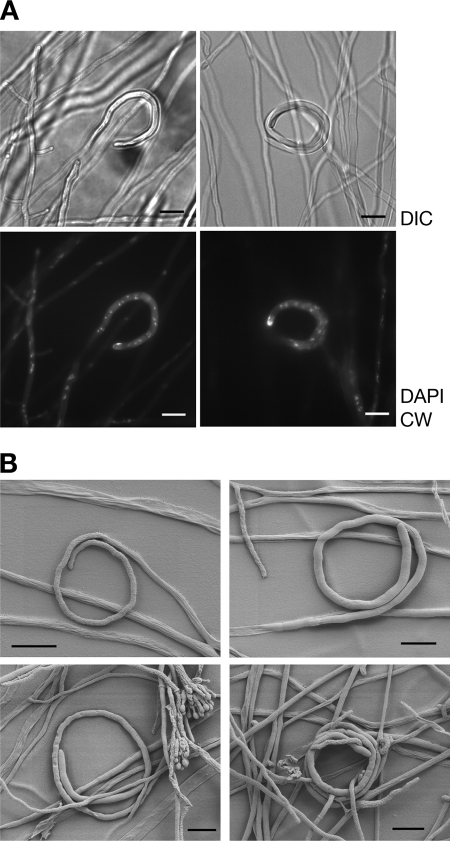
In order to characterize the A. fumigatus hyphal coils further, microscopic inspection after nuclear and cell wall staining was carried out, revealing normal nuclear distribution within these hyphal segments and typical profound apical cell wall synthesis (Fig. 4A). In this respect, the curled hyphae do not appear distinct from vegetative growing ones, which is also evident from scanning electron microscopy images (Fig. 4B). The fate of these structures within the vegetative mycelium could not be pursued since subsequent intertwining of growing hyphae superposed by asexual sporulation structures hampered microscopic inspection.
FIG. 4.
Ultrastructure of hyphal coils resulting from nsdD overexpression in A. fumigatus. Images are representative of A. fumigatus background (mat1-1 or mat1-2) and source of the nsdD gene (A. fumigatus or A. nidulans). (A) Differential interference contrast (DIC) and fluorescence microscopy images of strain AfS54 reveal nuclear distribution and cell wall composition of the induced coils by 4′,6′-diamidino-2-phenylindole (DAPI) staining and calcoflour white (CW) incorporation, respectively; no obvious differences from vegetative, straight-line hyphae are evident. (B) Representative images of AfS54 from scanning electron microscopy to illustrate size and structure of the nsdD-induced morphological alterations. Scale bar, 10 μm.
DISCUSSION
The data presented in this study demonstrate that the presumably asexual human pathogen A. fumigatus encodes functional determinants of sexual development, as determined by functional complementation in A. nidulans, a homothallic species where sexual development is closely linked to mating, since strains with any of the two mating types deleted are infertile and do not form ascospores (30). In agreement with this is the noncomplementation phenotype observed for the A. fumigatus α-box-deleted mat1-1 allele, which expresses a nonfunctional mating type factor and therefore is unable to substitute for its A. nidulans orthologue. The intact A. fumigatus mat1-1 ORF, however, supports the A. nidulans sexual cycle accompanied by Hülle cell formation, cleistothecium development, and ascosporogenesis. This finding complements a recent study (34) in which the A. nidulans matA ORF was swapped for its A. fumigatus counterpart: Mat1-2 supported cleistothecial differentiation in the transgenic strain, which produced viable ascospores. According to this study, however, substitution by the A. fumigatus mat1-2 ORF resulted in the delayed formation of hypertrophic fruiting bodies; we did not find this to be the case in our mat1-1 complementation experiments, which hints at subtle differences in the functional conservation of each mating type factor. In summary, we can now conclude that both mating type idiomorphs of A. fumigatus encode products capable of supporting sexual differentiation and fruiting body formation (although to differing degrees) in a fertile, homothallic species of Aspergillus.
This finding is an extension of prior studies, in which the idiomorphs had been identified and demonstrated to be expressed in A. fumigatus (29). Moreover, based on a near-1:1 ratio of distribution for both mating types within a worldwide collection of A. fumigatus isolates, the existence of a sexual cycle has been proposed. Our study strongly supports this hypothesis by demonstrating that two key factors of A. nidulans sexual development appear to be functionally conserved. Of course, the possibility exists that the tested gene products, Mat1-1 and NsdD, have been assigned to alternative biological roles in A. fumigatus. Characterization of A. fumigatus mutant strains may shed light on this prospect, and the generation of corresponding A. fumigatus deletants is a next logical step.
The NsdD-associated phenotype of hyphal coil formation in the fertile species A. nidulans and the deuteromycete A. fumigatus remains vague thus far, although some parallels with early-stage cleistothecial formation in A. nidulans can be drawn, since cleistothecial development is characterized by the formation of circular growing hyphae that surround the ascogenous system (39). Ascocarp formation in several Aspergillus species is initiated by an unbranched hyphal coil, the ascogonium, which may represent a precursor of the spherical cleistothecial wall (1). Moreover, hyphal coils are similar to structures observed in related species, such as Aspergillus ruber, for which the ascogonial coil has been described as a fertilization structure (4, 6). In other fungal genera, the formation of tightly coiled hyphae has been attributed to sexual development and cleistothecium formation, exemplified by the pathogenic fungus Histoplasma capsulatum (22). We did not observe further development of the A. nidulans coils into primordial structures, and even after prolonged incubation of the nsdD-overexpressing A. fumigatus strains, cleistothecium-like structures were not formed. The presence of both mating types is likely to be a strict necessity, but preliminary data from crossing experiments indicate that NsdD overexpression in an A. fumigatus mat1-1/mat1-2 heterokaryon is not sufficient to drive further development (our unpublished results). Balanced regulation of mating type expression could be a prerequisite for the progression of a sexual cycle (38), or specific environmental conditions might have to be present that would trigger sexual differentiation.
A. fumigatus reproduces by dispersal of clonal conidia; however, evidence for recent unlimited sexual recombination has been provided (29, 32). It is an open question to what extent either reproductive mode, sexual or clonal, directs A. fumigatus evolution. The apparent lack of sexuality of A. fumigatus is not based on the absence of functional mating type gene products; other reasons, such as inappropriate expression or the absence of up- or downstream components, are likely to account for this. Genome data mining has revealed the presence of a variety of A. fumigatus genes that have been characterized as being required for sexual development of A. nidulans (12), and, as exemplified for nsdD, their products might have retained functionality. From comparative analyses among closely related aspergilli, it is evident these genes are under purifying selection irrespective of the sexual nature of their host (10). Accordingly, their cellular roles in A. fumigatus remain to be scrutinized, which will yield further insight into cryptic sexuality of this human pathogen. This not only will enhance molecular biology for this fungus but might also reveal any impact of restricted sexuality on fungal pathogenicity: limiting genetic recombination and accordingly ensuring genome stability to a certain extent could support maintenance of virulence traits (18). In this respect, A. fumigatus behaves in line with at least two other fungal pathogens, the basidiomycete Cryptococcus neoformans and the commensal Candida albicans, which have maintained capacities for genetic recombination sexually via monokaryotic fruiting (23) or mitotically by parasexuality (19), respectively. For A. fumigatus, any sexual cycle awaits discovery; nevertheless, the data presented in this study are in favor of a cryptic sexuality in this saprophytic pathogen.
Acknowledgments
We thank all former and present members of the departments for continuous support and inspiring discussions, especially Gerhard Braus and Joachim Morschhäuser and his group for help and advice. Michaela Dümig is thanked for brilliant technical assistance, and we are indebted to Andreas Reimer from the Department of Geobiology, Center for Geosciences at the University of Göttingen, for assistance and for providing the scanning electron microscopy images. Kap-Hoon Han is thanked for the generous gift of strain KHH52 and Elaine Bignell for proofreading the manuscript.
Financial aid was received from the German Research Foundation, the Free State of Bavaria, and the University of Würzburg.
Footnotes
Published ahead of print on 29 August 2008.
REFERENCES
- 1.Benjamin, C. R. 1955. Ascocarps of Aspergillus and Penicillium. Mycologia 47669-687. [Google Scholar]
- 2.Braus, G. H., S. Krappmann, and S. E. Eckert. 2002. Sexual development in ascomycetes—fruit body formation of Aspergillus nidulans, p. 215-244. In H. D. Osiewacz (ed.), Molecular biology of fungal development. Marcel Dekker, Inc., New York, NY.
- 3.Brown, T., and K. Mackey. 1997. Analysis of RNA by Northern and slot blot hybridization, p. 4.9.1-4.9.16. In F. M. Ausubel, R. Brendt, R. E. Kingston, et al. (ed.), Current protocols in molecular biology. John Wiley & Sons Inc., Hoboken, NJ. [DOI] [PubMed]
- 4.Bruggeman, J. 2003. Thesis. Wageningen University, Wageningen, The Netherlands.
- 5.Casselton, L. A. 2002. Mate recognition in fungi. Heredity 88142-147. [DOI] [PubMed] [Google Scholar]
- 6.Champe, S. P., and L. D. Simon. 1992. Cellular differentiation and tissue formation in the fungus Aspergillus nidulans, p. 63-91. In E. F. Rossomando and S. Alexander (ed.), Morphogenesis: an analysis of the development of biological form. Dekker, New York, NY.
- 7.Coppin, E., R. Debuchy, S. Arnaise, and M. Picard. 1997. Mating types and sexual development in filamentous ascomycetes. Microbiol. Mol. Biol. Rev. 61411-428. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Dyer, P. S. 2007. Sexual reproduction and significance of MAT in the aspergilli, p. 123-142. In J. Heitman, J. Kronstad, J. Taylor, and L. Casselton (ed.), Sex in fungi: molecular determination and evolutionary implications. ASM Press, Washington, DC.
- 9.Dyer, P. S., M. Paoletti, and D. B. Archer. 2003. Genomics reveals sexual secrets of Aspergillus. Microbiology 1492301-2303. [DOI] [PubMed] [Google Scholar]
- 10.Fedorova, N. D., N. Khaldi, V. S. Joardar, R. Maiti, P. Amedeo, M. J. Anderson, J. Crabtree, J. C. Silva, J. H. Badger, A. Albarraq, S. Angiuoli, H. Bussey, P. Bowyer, P. J. Cotty, P. S. Dyer, A. Egan, K. Galens, C. M. Fraser-Liggett, B. J. Haas, J. M. Inman, R. Kent, S. Lemieux, I. Malavazi, J. Orvis, T. Roemer, C. M. Ronning, J. P. Sundaram, G. Sutton, G. Turner, J. C. Venter, O. R. White, B. R. Whitty, P. Youngman, K. H. Wolfe, G. H. Goldman, J. R. Wortman, B. Jiang, D. W. Denning, and W. C. Nierman. 2008. Genomic islands in the pathogenic filamentous fungus Aspergillus fumigatus. PLoS Genet. 4e1000046. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Feinberg, A. P., and B. Vogelstein. 1983. A technique for radiolabeling DNA restriction endonuclease fragments to high specific activity. Anal. Biochem. 1326-13. [DOI] [PubMed] [Google Scholar]
- 12.Galagan, J. E., S. E. Calvo, C. Cuomo, L. J. Ma, J. R. Wortman, S. Batzoglou, S. I. Lee, M. Basturkmen, C. C. Spevak, J. Clutterbuck, V. Kapitonov, J. Jurka, C. Scazzocchio, M. Farman, J. Butler, S. Purcell, S. Harris, G. H. Braus, O. Draht, S. Busch, C. D'Enfert, C. Bouchier, G. H. Goldman, D. Bell-Pedersen, S. Griffiths-Jones, J. H. Doonan, J. Yu, K. Vienken, A. Pain, M. Freitag, E. U. Selker, D. B. Archer, M. A. Penalva, B. R. Oakley, M. Momany, T. Tanaka, T. Kumagai, K. Asai, M. Machida, W. C. Nierman, D. W. Denning, M. Caddick, M. Hynes, M. Paoletti, R. Fischer, B. Miller, P. Dyer, M. S. Sachs, S. A. Osmani, and B. W. Birren. 2005. Sequencing of Aspergillus nidulans and comparative analysis with A. fumigatus and A. oryzae. Nature 4381105-1115. [DOI] [PubMed] [Google Scholar]
- 13.Geiser, D. M. 12 June 2008, posting date. Sexual structures in Aspergillus: morphology, importance and genomics. Med. Mycol. doi: 10.1080/13693780802139859. [DOI] [PubMed]
- 14.Han, K.-H., Y. J. Han, and D. M. Han. 1995. Cloning of nsdD gene cDNA in Aspergillus nidulans. Bull. Life Sci. Biotechnol. 339-42. [Google Scholar]
- 15.Han, K. H., K.-Y. Han, M.-S. Kim, D.-B. Lee, J.-H. Kim, S.-K. Chae, K.-S. Chae, and D.-M. Han. 2003. Regulation of nsdD expression in Aspergillus nidulans. J. Microbiol. 41259-261. [Google Scholar]
- 16.Han, K. H., K. Y. Han, J. H. Yu, K. S. Chae, K. Y. Jahng, and D. M. Han. 2001. The nsdD gene encodes a putative GATA-type transcription factor necessary for sexual development of Aspergillus nidulans. Mol. Microbiol. 41299-309. [DOI] [PubMed] [Google Scholar]
- 17.Hanahan, D., J. Jessee, and F. R. Bloom. 1991. Plasmid transformation of Escherichia coli and other bacteria. Methods Enzymol. 20463-113. [DOI] [PubMed] [Google Scholar]
- 18.Heitman, J. 2006. Sexual reproduction and the evolution of microbial pathogens. Curr. Biol. 16R711-R725. [DOI] [PubMed] [Google Scholar]
- 19.Johnson, A. 2003. The biology of mating in Candida albicans. Nat. Rev. Microbiol. 1106-116. [DOI] [PubMed] [Google Scholar]
- 20.Käfer, E. 1965. Origins of translocations in Aspergillus nidulans. Genetics 52217-232. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Kolar, M., P. J. Punt, C. A. van den Hondel, and H. Schwab. 1988. Transformation of Penicillium chrysogenum using dominant selection markers and expression of an Escherichia coli lacZ fusion gene. Gene 62127-134. [DOI] [PubMed] [Google Scholar]
- 22.Kwon-Chung, K. J. 1972. Sexual stage of Histoplasma capsulatum. Science 175326. [DOI] [PubMed] [Google Scholar]
- 23.Lin, X., C. M. Hull, and J. Heitman. 2005. Sexual reproduction between partners of the same mating type in Cryptococcus neoformans. Nature 4341017-1021. [DOI] [PubMed] [Google Scholar]
- 24.Mabey, J. E., M. J. Anderson, P. F. Giles, C. J. Miller, T. K. Attwood, N. W. Paton, E. Bornberg-Bauer, G. D. Robson, S. G. Oliver, and D. W. Denning. 2004. CADRE: the Central Aspergillus Data REpository. Nucleic Acids Res. 32D401-D405. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Nayak, T., E. Szewczyk, C. E. Oakley, A. Osmani, L. Ukil, S. L. Murray, M. J. Hynes, S. A. Osmani, and B. R. Oakley. 2006. A versatile and efficient gene-targeting system for Aspergillus nidulans. Genetics 1721557-1566. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Nielsen, K., and J. Heitman. 2007. Sex and virulence of human pathogenic fungi. Adv. Genet. 57143-173. [DOI] [PubMed] [Google Scholar]
- 27.Nierman, W. C., A. Pain, M. J. Anderson, J. R. Wortman, H. S. Kim, J. Arroyo, M. Berriman, K. Abe, D. B. Archer, C. Bermejo, J. Bennett, P. Bowyer, D. Chen, M. Collins, R. Coulsen, R. Davies, P. S. Dyer, M. Farman, N. Fedorova, N. Fedorova, T. V. Feldblyum, R. Fischer, N. Fosker, A. Fraser, J. L. Garcia, M. J. Garcia, A. Goble, G. H. Goldman, K. Gomi, S. Griffith-Jones, R. Gwilliam, B. Haas, H. Haas, D. Harris, H. Horiuchi, J. Huang, S. Humphray, J. Jimenez, N. Keller, H. Khouri, K. Kitamoto, T. Kobayashi, S. Konzack, R. Kulkarni, T. Kumagai, A. Lafon, J. P. Latge, W. Li, A. Lord, C. Lu, W. H. Majoros, G. S. May, B. L. Miller, Y. Mohamoud, M. Molina, M. Monod, I. Mouyna, S. Mulligan, L. Murphy, S. O'Neil, I. Paulsen, M. A. Penalva, M. Pertea, C. Price, B. L. Pritchard, M. A. Quail, E. Rabbinowitsch, N. Rawlins, M. A. Rajandream, U. Reichard, H. Renauld, G. D. Robson, S. Rodriguez de Cordoba, J. M. Rodriguez-Pena, C. M. Ronning, S. Rutter, S. L. Salzberg, M. Sanchez, J. C. Sanchez-Ferrero, D. Saunders, K. Seeger, R. Squares, S. Squares, M. Takeuchi, F. Tekaia, G. Turner, C. R. Vazquez de Aldana, J. Weidman, O. White, J. Woodward, J. H. Yu, C. Fraser, J. E. Galagan, K. Asai, M. Machida, N. Hall, B. Barrell, and D. W. Denning. 2005. Genomic sequence of the pathogenic and allergenic filamentous fungus Aspergillus fumigatus. Nature 4381151-1156. [DOI] [PubMed] [Google Scholar]
- 28.Pain, A., J. Woodward, M. A. Quail, M. J. Anderson, R. Clark, M. Collins, N. Fosker, A. Fraser, D. Harris, N. Larke, L. Murphy, S. Humphray, S. O'Neil, M. Pertea, C. Price, E. Rabbinowitsch, M. A. Rajandream, S. Salzberg, D. Saunders, K. Seeger, S. Sharp, T. Warren, D. W. Denning, B. Barrell, and N. Hall. 2004. Insight into the genome of Aspergillus fumigatus: analysis of a 922 kb region encompassing the nitrate assimilation gene cluster. Fungal Genet. Biol. 41443-453. [DOI] [PubMed] [Google Scholar]
- 29.Paoletti, M., C. Rydholm, E. U. Schwier, M. J. Anderson, G. Szakacs, F. Lutzoni, J. P. Debeaupuis, J.-P. Latgé, D. W. Denning, and P. S. Dyer. 2005. Evidence for sexuality in the opportunistic fungal pathogen Aspergillus fumigatus. Curr. Biol. 151242-1248. [DOI] [PubMed] [Google Scholar]
- 30.Paoletti, M., F. A. Seymour, M. J. Alcocer, N. Kaur, A. M. Calvo, D. B. Archer, and P. S. Dyer. 2007. Mating type and the genetic basis of self-fertility in the model fungus Aspergillus nidulans. Curr. Biol. 171384-1389. [DOI] [PubMed] [Google Scholar]
- 31.Pöggeler, S. 2002. Genomic evidence for mating abilities in the asexual pathogen Aspergillus fumigatus. Curr. Genet. 42153-160. [DOI] [PubMed] [Google Scholar]
- 32.Pringle, A., D. M. Baker, J. L. Platt, J. P. Wares, J.-P. Latgé, and J. W. Taylor. 2005. Cryptic speciation in the cosmopolitan and clonal human pathogenic fungus Aspergillus fumigatus. Evolution 591886-1899. [PubMed] [Google Scholar]
- 33.Punt, P. J., and C. A. van den Hondel. 1992. Transformation of filamentous fungi based on hygromycin B and phleomycin resistance markers. Methods Enzymol. 216447-457. [DOI] [PubMed] [Google Scholar]
- 34.Pyrzak, W., K. Y. Miller, and B. L. Miller. 2008. The mating type protein Mat1-2 from asexual Aspergillus fumigatus drives sexual reproduction in fertile Aspergillus nidulans. Eukaryot. Cell 71029-1040. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Saiki, R. K., S. Scharf, F. Faloona, K. B. Mullis, G. T. Horn, H. A. Erlich, and N. Arnheim. 1985. Enzymatic amplification of beta-globin genomic sequences and restriction site analysis for diagnosis of sickle cell anemia. Science 2301350-1354. [DOI] [PubMed] [Google Scholar]
- 36.Sambrook, J., E. F. Fritsch, and T. Maniatis. 1989. Molecular cloning: a laboratory manual. Cold Spring Harbor Laboratory Press, Cold Spring Harbor, NY.
- 37.Scazzocchio, C. 2000. The fungal GATA factors. Curr. Opin. Microbiol. 3126-131. [DOI] [PubMed] [Google Scholar]
- 38.Scazzocchio, C. 2006. Aspergillus genomes: secret sex and the secrets of sex. Trends Genet. 22521-525. [DOI] [PubMed] [Google Scholar]
- 39.Sohn, K. T., and K. S. Yoon. 2002. Ultrastructural study on the cleistothecium development in Aspergillus nidulans. Mycobiology 30117-127. [Google Scholar]
- 40.Southern, E. 2006. Southern blotting. Nat. Protoc. 1518-525. [DOI] [PubMed] [Google Scholar]
- 41.Staib, F., S. K. Mishra, C. Rajendran, R. Voigt, J. Steffen, K. H. Neumann, C. A. Hartmann, and G. Heins. 1980. A notable Aspergillus from a mortal aspergilloma of the lung. New aspects of the epidemiology, serodiagnosis and taxonomy of Aspergillus fumigatus. Zentralbl. Bakteriol. A 247530-536. [PubMed] [Google Scholar]
- 42.Szewczyk, E., T. Nayak, C. E. Oakley, H. Edgerton, Y. Xiong, N. Taheri-Talesh, S. A. Osmani, and B. R. Oakley. 2006. Fusion PCR and gene targeting in Aspergillus nidulans. Nat. Protoc. 13111-3120. [DOI] [PubMed] [Google Scholar]
- 43.Teakle, G. R., and P. M. Gilmartin. 1998. Two forms of type IV zinc-finger motif and their kingdom-specific distribution between the flora, fauna and fungi. Trends Biochem. Sci. 23100-102. [DOI] [PubMed] [Google Scholar]
- 44.Turgeon, B. G., and O. C. Yoder. 2000. Proposed nomenclature for mating type genes of filamentous ascomycetes. Fungal Genet. Biol. 311-5. [DOI] [PubMed] [Google Scholar]
- 45.Varga, J. 2003. Mating type gene homologues in Aspergillus fumigatus. Microbiology 149816-819. [DOI] [PubMed] [Google Scholar]
- 46.Wilson, R. A., and H. N. Arst, Jr. 1998. Mutational analysis of AREA, a transcriptional activator mediating nitrogen metabolite repression in Aspergillus nidulans and a member of the “streetwise” GATA family of transcription factors. Microbiol. Mol. Biol. Rev. 62586-596. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Woodcock, D. M., P. J. Crowther, J. Doherty, S. Jefferson, E. DeCruz, M. Noyer-Weidner, S. S. Smith, M. Z. Michael, and M. W. Graham. 1989. Quantitative evaluation of Escherichia coli host strains for tolerance to cytosine methylation in plasmid and phage recombinants. Nucleic Acids Res. 173469-3478. [DOI] [PMC free article] [PubMed] [Google Scholar]