Abstract
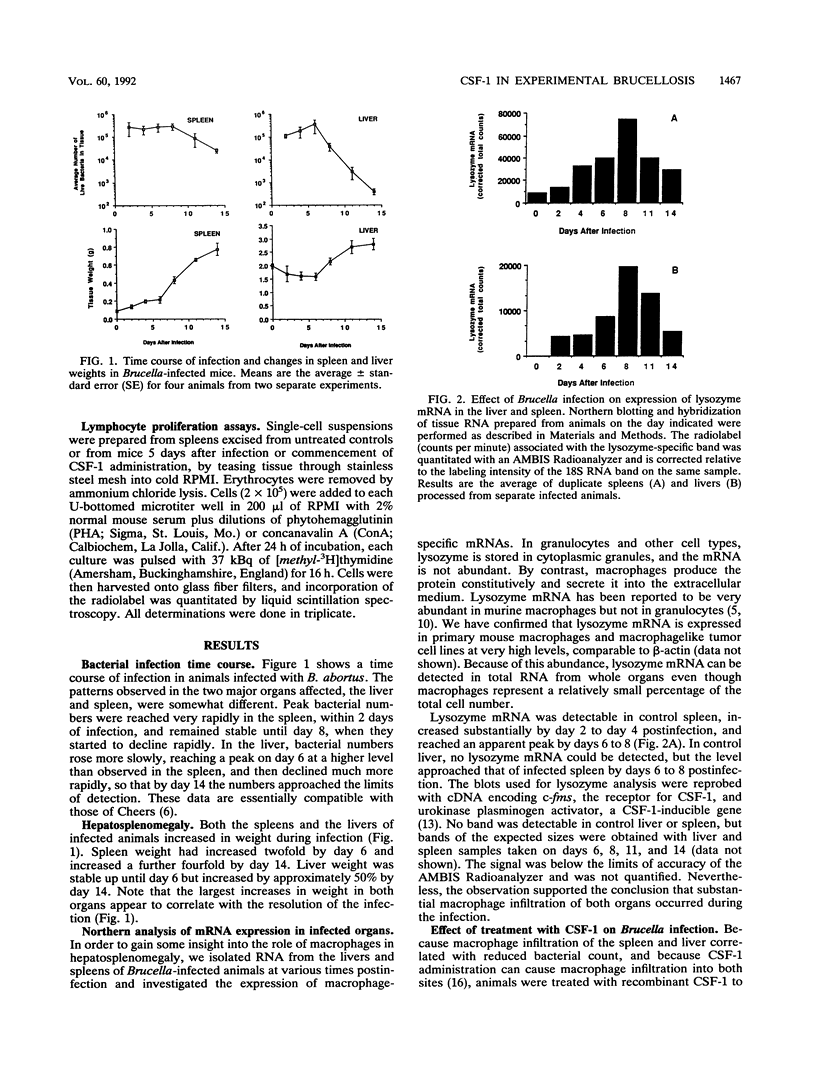
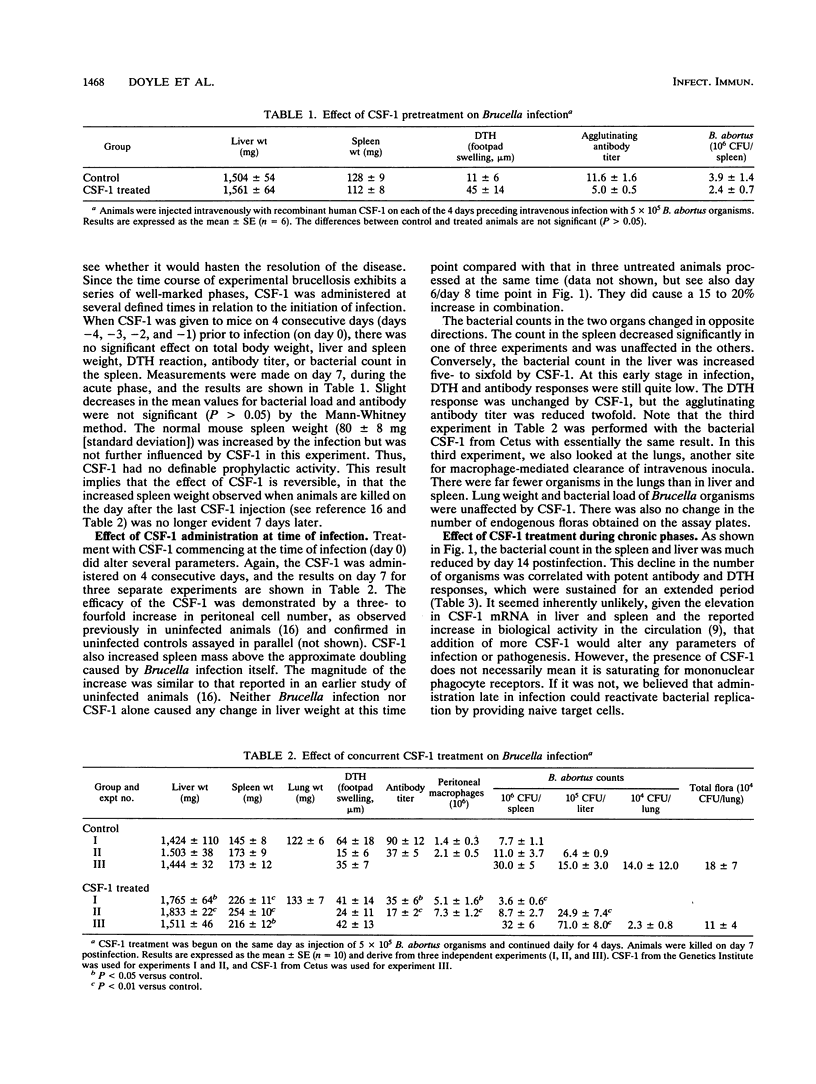
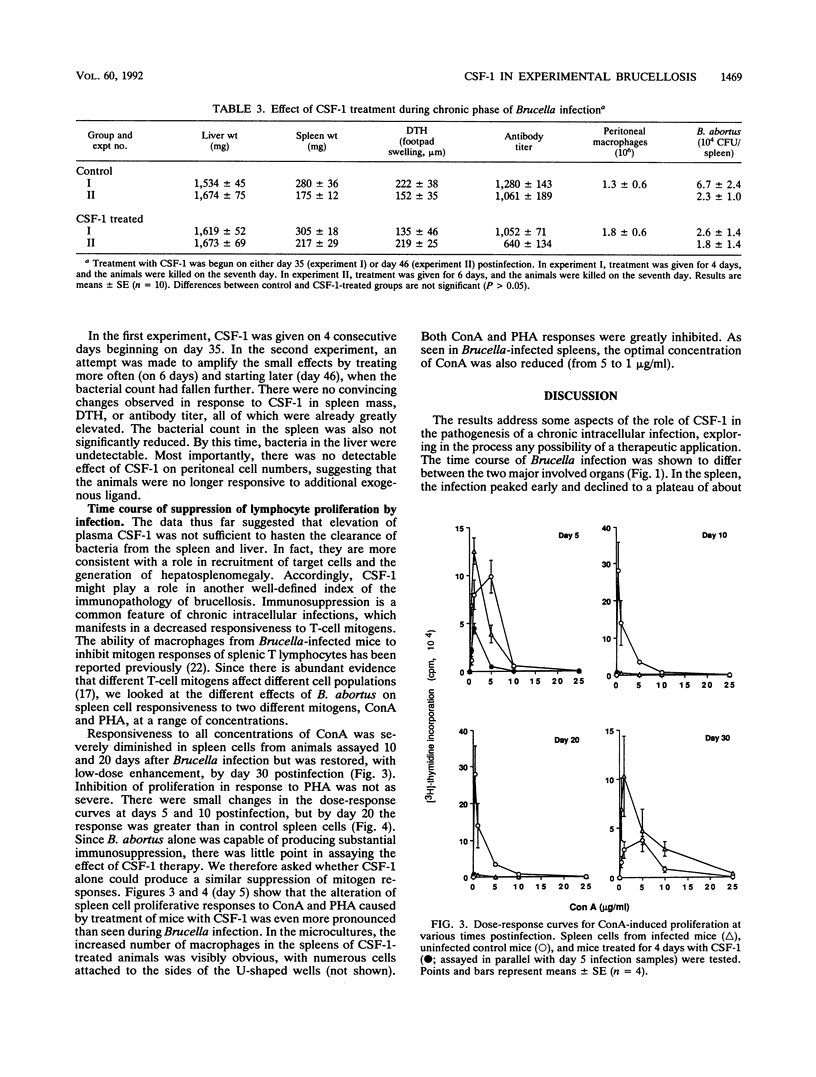
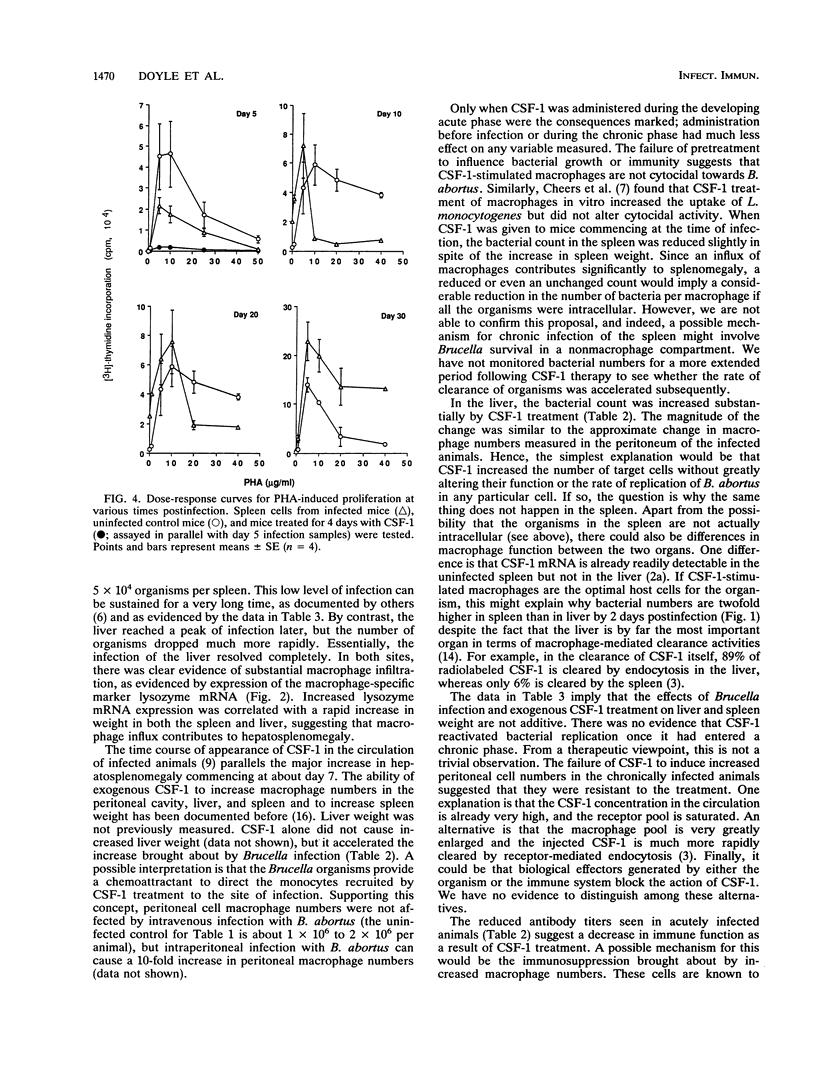
Brucella abortus injected into CBA mice replicated primarily in the spleen and liver, reaching a peak bacterial count in both organs about 7 days postinfection. The organism was eliminated from the liver but declined to a chronic phase in the spleen. The infection caused hepatosplenomegaly. An influx of macrophages into the two organs was monitored by quantitative Northern (RNA blot) analysis of the macrophage-specific marker lysozyme mRNA. Lysozyme mRNA was detectable in spleen and increased three- to fourfold during infection. In liver, lysozyme mRNA was initially undetectable, but at about the peak of infection it reached a level comparable to that in the spleen. Macrophage colony-stimulating factor 1 (CSF-1) has been reported to be elevated in the circulation of animals infected with B. abortus and is known to stimulate monocytopoiesis. To investigate the role of CSF-1 in pathogenesis, we studied the effect of further increasing the CSF-1 concentration by administration of recombinant human CSF-1. Since the infection is characterized by several distinct phases, recombinant human CSF-1 was administered at defined times relative to these phases. Pronounced effects were observed only when CSF-1 administration was begun during the developing acute phase. The consequences were decreased bacterial numbers in the spleen but an increase in the liver, reduced antibody generation, and increased hepatosplenomegaly. A feature of many chronic intracellular infections is immunosuppression. B. abortus caused a substantial diminution of responsiveness of spleen cells to T-cell mitogens, particularly concanavalin A. This action was mimicked by CSF-1 treatment of the animals prior to spleen cell isolation. The results suggest that CSF-1 plays a role in macrophage recruitment in brucellosis and that recruited macrophages contribute to the immunopathology and immunosuppression.
Full text
PDF



Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Abd A. H., Savage N. W., Halliday W. J., Hume D. A. The role of macrophages in experimental arthritis induced by Streptococcus agalactiae sonicate: actions of macrophage colony-stimulating factor (CSF-1) and other macrophage-modulating agents. Lymphokine Cytokine Res. 1991 Apr;10(1-2):43–50. [PubMed] [Google Scholar]
- Araya L. N., Elzer P. H., Rowe G. E., Enright F. M., Winter A. J. Temporal development of protective cell-mediated and humoral immunity in BALB/c mice infected with Brucella abortus. J Immunol. 1989 Nov 15;143(10):3330–3337. [PubMed] [Google Scholar]
- Bartocci A., Mastrogiannis D. S., Migliorati G., Stockert R. J., Wolkoff A. W., Stanley E. R. Macrophages specifically regulate the concentration of their own growth factor in the circulation. Proc Natl Acad Sci U S A. 1987 Sep;84(17):6179–6183. doi: 10.1073/pnas.84.17.6179. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Berman D. T., Wilson B. L., Moreno E., Angus R. D., Jones L. M. Characterization of Brucella abortus soluble antigen employed in immunoassay. J Clin Microbiol. 1980 Apr;11(4):355–362. doi: 10.1128/jcm.11.4.355-362.1980. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bonifer C., Vidal M., Grosveld F., Sippel A. E. Tissue specific and position independent expression of the complete gene domain for chicken lysozyme in transgenic mice. EMBO J. 1990 Sep;9(9):2843–2848. doi: 10.1002/j.1460-2075.1990.tb07473.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cheers C., Hill M., Haigh A. M., Stanley E. R. Stimulation of macrophage phagocytic but not bactericidal activity by colony-stimulating factor 1. Infect Immun. 1989 May;57(5):1512–1516. doi: 10.1128/iai.57.5.1512-1516.1989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cheers C. Pathogenesis and cellular immunity in experimental murine brucellosis. Dev Biol Stand. 1984;56:237–246. [PubMed] [Google Scholar]
- Cheers C., Stanley E. R. Macrophage production during murine listeriosis: colony-stimulating factor 1 (CSF-1) and CSF-1-binding cells in genetically resistant and susceptible mice. Infect Immun. 1988 Nov;56(11):2972–2978. doi: 10.1128/iai.56.11.2972-2978.1988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cheers C., Young A. M. Serum colony stimulating activity and colony forming cells in murine brucellosis: relationship to immunopathology. Microb Pathog. 1987 Sep;3(3):185–194. doi: 10.1016/0882-4010(87)90095-7. [DOI] [PubMed] [Google Scholar]
- Chung L. P., Keshav S., Gordon S. Cloning the human lysozyme cDNA: inverted Alu repeat in the mRNA and in situ hybridization for macrophages and Paneth cells. Proc Natl Acad Sci U S A. 1988 Sep;85(17):6227–6231. doi: 10.1073/pnas.85.17.6227. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Goodwin J. S., Ceuppens J. Regulation of the immune response by prostaglandins. J Clin Immunol. 1983 Oct;3(4):295–315. doi: 10.1007/BF00915791. [DOI] [PubMed] [Google Scholar]
- Hume D. A., Donahue R. E., Fidler I. J. The therapeutic effect of human recombinant macrophage colony stimulating factor (CSF-1) in experimental murine metastatic melanoma. Lymphokine Res. 1989 Spring;8(1):69–77. [PubMed] [Google Scholar]
- Hume D. A., Pavli P., Donahue R. E., Fidler I. J. The effect of human recombinant macrophage colony-stimulating factor (CSF-1) on the murine mononuclear phagocyte system in vivo. J Immunol. 1988 Nov 15;141(10):3405–3409. [PubMed] [Google Scholar]
- Ladner M. B., Martin G. A., Noble J. A., Wittman V. P., Warren M. K., McGrogan M., Stanley E. R. cDNA cloning and expression of murine macrophage colony-stimulating factor from L929 cells. Proc Natl Acad Sci U S A. 1988 Sep;85(18):6706–6710. doi: 10.1073/pnas.85.18.6706. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Montaraz J. A., Winter A. J., Hunter D. M., Sowa B. A., Wu A. M., Adams L. G. Protection against Brucella abortus in mice with O-polysaccharide-specific monoclonal antibodies. Infect Immun. 1986 Mar;51(3):961–963. doi: 10.1128/iai.51.3.961-963.1986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Plommet M. Brucellosis and immunity: humoral and cellular components in mice. Ann Inst Pasteur Microbiol. 1987 Jan-Feb;138(1):105–110. doi: 10.1016/0769-2609(87)90086-x. [DOI] [PubMed] [Google Scholar]
- Ralph P., Warren M. K., Nakoinz I., Lee M. T., Brindley L., Sampson-Johannes A., Kawasaki E. S., Ladner M. B., Strickler J. E., Boosman A. Biological properties and molecular biology of the human macrophage growth factor, CSF-1. Immunobiology. 1986 Sep;172(3-5):194–204. doi: 10.1016/S0171-2985(86)80099-7. [DOI] [PubMed] [Google Scholar]
- Riglar C., Cheers C. Macrophage activation during experimental murine brucellosis. II. Inhibition of in vitro lymphocyte proliferation by brucella-activated macrophages. Cell Immunol. 1980 Jan;49(1):154–167. doi: 10.1016/0008-8749(80)90065-9. [DOI] [PubMed] [Google Scholar]
- Stanley E. R. Action of the colony-stimulating factor, CSF-1. Ciba Found Symp. 1986;118:29–41. doi: 10.1002/9780470720998.ch3. [DOI] [PubMed] [Google Scholar]
- Strassmann G., Bertolini D. R., Kerby S. B., Fong M. Regulation of murine mononuclear phagocyte inflammatory products by macrophage colony-stimulating factor. Lack of IL-1 and prostaglandin E2 production and generation of a specific IL-1 inhibitor. J Immunol. 1991 Aug 15;147(4):1279–1285. [PubMed] [Google Scholar]
- Wing E. J., Magee D. M., Pearson A. C., Waheed A., Shadduck R. K. Peritoneal macrophages exposed to purified macrophage colony-stimulating factor (M-CSF) suppress mitogen- and antigen-stimulated lymphocyte proliferation. J Immunol. 1986 Nov 1;137(9):2768–2773. [PubMed] [Google Scholar]
- Wong G. G., Temple P. A., Leary A. C., Witek-Giannotti J. S., Yang Y. C., Ciarletta A. B., Chung M., Murtha P., Kriz R., Kaufman R. J. Human CSF-1: molecular cloning and expression of 4-kb cDNA encoding the human urinary protein. Science. 1987 Mar 20;235(4795):1504–1508. doi: 10.1126/science.3493529. [DOI] [PubMed] [Google Scholar]
- Young D. A., Lowe L. D., Clark S. C. Comparison of the effects of IL-3, granulocyte-macrophage colony-stimulating factor, and macrophage colony-stimulating factor in supporting monocyte differentiation in culture. Analysis of macrophage antibody-dependent cellular cytotoxicity. J Immunol. 1990 Jul 15;145(2):607–615. [PubMed] [Google Scholar]



