Abstract
Severe factor V (FV) deficiency is associated with mild to severe bleeding diathesis, but many patients with FV levels lower than 1% bleed less than anticipated. We used calibrated automated thrombography to screen patients with severe FV deficiency for protective procoagulant defects. Thrombin generation in FV-deficient plasma was only measurable at high tissue factor concentrations. Upon reconstitution of FV-deficient plasma with purified FV, thrombin generation increased steeply with FV concentration, reaching a plateau at approximately 10% FV. FV-deficient plasma reconstituted with 100% FV generated severalfold more thrombin than normal plasma, especially at low tissue factor concentrations (1.36 pM) or in the presence of activated protein C, suggesting reduced tissue factor pathway inhibitor (TFPI) levels in FV-deficient plasma. Plasma TFPI antigen and activity levels were indeed lower (P < .001) in FV-deficient patients (n = 11; 4.0 ± 1.0 ng/mL free TFPI) than in controls (n = 20; 11.5 ± 4.8 ng/mL), while persons with partial FV deficiency had inter-mediate levels (n = 16; 7.9 ± 2.5 ng/mL). FV immunodepletion experiments in normal plasma and surface plasmon resonance analysis provided evidence for the existence of a FV/TFPI complex, possibly affecting TFPI stability/clearance in vivo. Low TFPI levels decreased the FV requirement for minimal thrombin generation in FV-deficient plasma to less than 1% and might therefore protect FV-deficient patients from severe bleeding.
Introduction
Coagulation factor V (FV) is a large multidomain glycoprotein structurally and functionally homologous to factor VIII (FVIII).1 After biosynthesis in the liver, FV is released in the bloodstream, where it is found in both plasma (80%; concentration of 21-25 nM) and platelets (20%). The activated form of FV (FVa) acts as an essential cofactor of activated factor X (FXa) in prothrombin (PT) activation, thereby enhancing thrombin formation by several orders of magnitude.2
The generation of thrombin is physiologically down-regulated by several anticoagulant mechanisms, including the protein C pathway3 and the tissue factor pathway inhibitor (TFPI) system.4 Activated protein C (APC) is a vitamin K–dependent serine protease which, in concert with its nonenzymatic cofactor protein S, inactivates FVa and FVIIIa by limited proteolysis. A poor anticoagulant response of plasma to exogenous APC (APC resistance5) is the most common risk factor for venous thrombosis. Conversely, TFPI is a Kunitz-type protease inhibitor that binds and inhibits both FXa and the tissue factor (TF)/FVIIa complex in a 2-step reaction,6 the first step being stimulated by protein S.7,8 TFPI is synthesized primarily by the vascular endothelium, and most of it (approximately 80%) is associated with the endothelial surface as a full-length protein, the form that most effectively inhibits FXa.9 Another 2% of all TFPI is stored in platelets.10,11 The remainder circulates in plasma at a concentration of 2.0 to 2.5 nM, of which approximately 80% is C-terminally truncated and bound to lipoproteins, while 5% to 20% is present as a free protein (both full-length and truncated forms).12 Low levels of plasma TFPI, particularly free TFPI, have been associated with an increased risk of venous thrombosis.13–16
Severe FV deficiency (Owren parahemophilia; OMIM 227400) is a rare bleeding disorder with an estimated prevalence of 1:106. It is inherited as an autosomal recessive trait, and several intragenic mutations impairing FV gene (F5) expression have been described (reviewed by Asselta et al17). Whereas most heterozygous carriers are asymptomatic, homozygotes and compound heterozygotes present with a wide spectrum of symptoms,18,19 ranging from mucosal bleeding and postoperative/postpartum hemorrhages to life-threatening intracranial hematomas. Although severe bleeding manifestations are usually confined to people with FV levels less than 1%, there seems to be no clear-cut relationship between plasma FV levels and bleeding phenotype, and some FV-deficient patients have apparently remained completely asymptomatic for several years despite their undetectable plasma FV levels.20,21
The overall moderate bleeding diathesis associated with severe FV deficiency in humans contrasts sharply with the invariable embryonic/perinatal lethality of the FV knockout mouse model.22 In addition, compared with patients with severe hemophilia, who frequently develop joint and muscle bleedings,23 many patients with severe FV deficiency show a milder clinical course.18 Even admitting that the FV level required for minimal hemostasis is 1% or less,24 the general viability of FV deficiency in humans suggests the existence of widespread compensating mechanisms that protect FV-deficient patients from excessive bleeding. In contrast to severe hemophilia25 and other bleeding disorders,26–28 where coinherited thrombophilic mutations (F5 Leiden, F2 G20210A mutation) or low levels of the natural anticoagulants have been shown to mitigate the bleeding manifestations, no similar protective mechanisms have ever been reported for severe FV deficiency.
In this study, we have used in vitro thrombin generation assays to investigate the overall coagulation function in 11 patients with severe FV deficiency, and to screen for possible procoagulant defects that may contribute to improve their clinical phenotype.
Methods
Study population
Experiments were conducted in plasma from 11 subjects (10 unrelated) with congenital severe FV deficiency: 8 were patients referred to Padua Academic Hospital from district hospitals in northeastern Italy and 3 were blood donors from George King Bio-Medical (Overland Park, KS). Patient characteristics are reported in Table 1. No DNA and only limited information could be obtained for the George King donors.
Table 1.
Demographic and clinical characteristics of patients with severe FV deficiency
| Patient | Sex | Age, y | FV level, % | Coinherited thrombophilic defects | Clinical phenotype* |
|---|---|---|---|---|---|
| PD I | F | 64 | < 0.5 | Moderate/severe (5) | |
| PD II | F | 44 | < 0.5 | 170% FVIII | Severe (7) |
| PD III | F | 35 | 0.6 | 131% PT (no F2 G20210A) | Asymptomatic (0†) |
| PD IV | F | 27 | < 0.5 | Mild (2) | |
| PD V | F | 52 | < 0.5 | Severe (10†) | |
| PD VI | M | 28 | < 0.5 | Mild (1†) | |
| PD VII | F | 62 | 4.8 | Mild (1) | |
| PD VII-A | F | 46 | 6.2 | Mild (1) | |
| GK 502 | F | 57 | < 0.5 | Mild | |
| GK 505 | F | 56 | < 0.5 | 53% PC | Unknown |
| GK 506 | M | 65 | < 0.5 | Unknown |
PT indicates prothrombin; and PC, protein C.
Numbers in parentheses represent the bleeding score calculated according to Rodeghiero et al.29
Prophylaxis (with plasma and/or antifibrinolytic agents) often given during risk situations after the diagnosis of severe FV deficiency was made.
Patients with severe FV deficiency were compared with 16 people with partial FV deficiency (9 men and 7 women; FV level 42.9% ± 9.9%) and to 20 healthy controls (8 men and 12 women; FV level 87.0% ± 17.8%) recruited at Padua Academic Hospital among relatives of FV-deficient and FV Leiden pseudohomozygous30 patients and among healthy hospital personnel. Subjects with partial FV deficiency were all asymptomatic, except one who had experienced epistaxis and gum bleeding during childhood. None of the subjects under study was on oral contraceptives or hormone replacement therapy at the time of blood sampling.
As an additional control group, 15 unrelated (male) patients with hemophilia A (FVIII levels, < 1%-23%; mean, 3.9%) and bleeding symptoms ranging from mild to severe were included in the study. Of these, 13 were patients at Padua Academic Hospital and 2 were blood donors from George King Biomedical.
The study was carried out in accordance with the Declaration of Helsinki, and all subjects gave informed consent to participation. George King Bio-Medical donors routinely sign an informed consent statement at the moment of blood collection. The study was approved by the ethical commission of Padua Academic Hospital.
Blood collection and plasma preparation
Venous blood was drawn by venipuncture in 3.8% sodium citrate (wt/vol) and centrifuged at 2000g for 15 minutes. Platelet-poor plasma was aliquoted, snap-frozen, and stored at −80°C until use; buffy coats were stored at −20°C for later DNA isolation.
Plasma purchased from George King Bio-Medical was collected from individual donors by plasmapheresis in 3.8% sodium citrate, divided into aliquots, and immediately frozen at −80°C.
Commercial factor-depleted plasmas
FV-depleted plasmas were purchased from Affinity Biologicals (Ancaster, ON), Dade-Behring (Marburg, Germany), Diagen (Thame, United Kingdom), Diagnostica Stago (Asnières sur Seine, France), and Organon-Teknika (Durham, NC). PT-, FVII-, FX-, protein C–, and antithrombin-depleted plasmas were from Affinity Biologicals.
Measurement of coagulation factor levels in plasma
A complete thrombophilia screening, including plasma levels of antithrombin, protein C, and protein S as well as PT and FVIII, was performed in all patients with severe FV deficiency. Antithrombin levels were measured with the Coamatic Antithrombin kit (Chromogenix, Mölndal, Sweden). For the quantification of protein C levels, protein C was activated in 1:20-diluted plasma with 0.05 U/mL Protac (Kordia Life Sciences, Leiden, The Netherlands) for 1 hour at 37°C, and the amidolytic activity of APC toward the chromogenic substrate S2366 was measured spectrophotometrically. Total and free protein S were determined by enzyme-linked immunosorbent assay (ELISA) as described.31 PT levels were measured with a chromogenic assay after complete activation of PT with Ecarin (Kordia Life Sciences).31 FVIII levels were measured by a one-stage activated partial thromboplastin time (aPTT)–based clotting assay in FVIII-deficient plasma.
FV and FX activity levels were determined with prothrombinase-based assays using in-house purified proteins, essentially as described.32 Briefly, FV or FX was activated in highly diluted plasma with 2 nM thrombin (Innovative Research, Southfield, MI) or 0.27 μg/mL RVV-X (Kabi Diagnostica/Chromogenix), respectively, for 10 minutes at 37°C. The FVa or FXa concentration was subsequently quantified via a prothrombinase-based assay under the following conditions: limiting amounts of FVa (< 25 pM), 5 nM FXa, 1 μM PT, 40 μM DOPS/DOPC (10/90 mol/mol) phospholipid vesicles, and 2.6 mM CaCl2 for the FVa assay; and limiting amounts of FXa (< 25 pM), 5 nM FVa, 40 μM DOPS/DOPC (10/90 mol/mol) phospholipid vesicles, 1 μM PT, and 3 mM CaCl2 for the FXa assay (final concentrations). The minimum FV concentration that could be reliably detected with this assay in plasma was 0.5%. To attain maximal precision, the FV levels of the different FV-deficient patients were measured 2 times in duplicate using independent plasma dilutions.
Plasma levels of total and free TFPI antigen were measured with commercial ELISA kits (Asserachrom; Diagnostica Stago). Due to the poor sensitivity of the free TFPI ELISA in the low-level range, a more sensitive homemade full-length TFPI ELISA (C. F. A. Maurissen, J.R., and T. M. Hackeng, manuscript in preparation) was also performed in some cases. TFPI activity was determined with a thrombin generation–based assay (see “Thrombin generation assays”).
A normal plasma pool prepared with plasma from 85 healthy donors not using any medication was used as a reference in all measurements. All factor levels were expressed as percentage of normal plasma, except total and free TFPI antigen levels, which were compared with a standard provided with the respective ELISA kit and expressed in nanograms per milliliter.
Thrombin generation assays
Thrombin generation was measured in platelet-poor plasma with the calibrated automated thrombogram (CAT) method.33 Briefly, coagulation was initiated with varying concentrations of recombinant TF (Innovin; Dade-Behring), 30 μM DOPS/DOPC/DOPE (20/60/20 mol/mol/mol) phospholipid vesicles, and 16 mM added CaCl2, and thrombin activity was monitored continuously via a low-affinity fluorogenic substrate (I-1140; BACHEM, Bubendorf, Switzerland). A thrombin calibrator (Thrombinoscope, Maastricht, The Netherlands) was used to correct each curve for inner-filter effects and substrate consumption. Fluorescence was read in a Fluoroskan Ascent reader (Thermo Labsystems, Helsinki, Finland), and thrombin generation curves were calculated with the Thrombinoscope software or as previously described27 (Figure 1). Apart from the experiments presented in Figure 1, all thrombin generation curves were run in duplicate. The height of the thrombin peak or the area under the curve (endogenous thrombin potential [ETP]) was taken as a measure of the amount of thrombin formed. The lag time of the thrombin generation curve, which is a measure of the plasma clotting time, was also analyzed in some experiments.
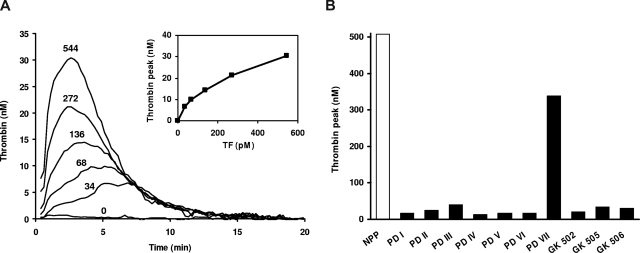
Figure 1.
Thrombin generation in FV-deficient plasma. (A) TF-titration of thrombin generation in FV-deficient plasma. Thrombin generation was determined in plasma from patient GK 506 at 0, 34, 68, 136, 272, and 544 pM TF. Inset shows peak height of thrombin generation as a function of TF concentration. (B) Peak height of thrombin generation evoked by 544 pM TF in normal pooled plasma (NPP) and in plasma from patients with severe FV deficiency.
To obtain reliable thrombin generation curves, FV-deficient plasma was supplemented with purified FV to various final concentrations (see “Results”), and FVIII-deficient plasma was spiked with 1% normal pooled plasma. FV was purified from normal pooled plasma as described.32 For reconstitution purposes, 23 nM FV was considered equal to 100% of the normal plasma concentration. In some experiments, FV-deficient plasma was supplemented with recombinant full-length TFPI (kind gift from Dr Lindhout, Maastricht University, Maastricht, The Netherlands). The amount of added TFPI was chosen to normalize thrombin generation in FV-deficient plasma reconstituted with 100% FV. To prevent contact activation, all thrombin generation curves were measured in the presence of 30 μg/mL corn trypsin inhibitor (CTI; Hematologic Technologies, Essex Junction, VT).
To determine plasma APC resistance, thrombin generation was triggered with 13.6 pM TF in the absence and presence of 12 nM purified human APC (Innovative Research). The APC concentration was chosen to reduce the ETP in normal plasma to approximately 10% of the ETP without APC.
By analogy with the diluted prothrombin time assay, which measures the ability of TFPI to prolong the plasma clotting time at a low TF concentration,34 TFPI activity was quantified as the ability of TFPI to reduce the peak height of thrombin generation. Briefly, plasma was incubated with (and without) 16 μg/mL of a monoclonal antibody against human TFPI (TFPI-6; Sanquin Reagents, Amsterdam, The Netherlands) for 15 minutes at 37°C, after which thrombin generation was initiated with 1.36 pM TF. The outcome of the test was expressed as an anti-TFPI ratio, defined as the ratio of the peak heights of the thrombin generation curves determined in the presence and absence of antibody. The higher the anti-TFPI ratio, the more functional TFPI is present in plasma. Normal plasma yields an anti-TFPI ratio of approximately 4.0.
DNA analysis
Genomic DNA was isolated from peripheral blood leukocytes by “salting out.” Carriership of the F5 Leiden and F2 G20210A mutations was determined by polymerase chain reaction (PCR)–mediated amplification and restriction analysis, as described.31 TFPI gene (TFPI) mutation screening was performed by PCR-mediated amplification and direct sequencing of all exons and splicing junctions, including approximately 500 bp of the promoter region. Primer sequences and amplification/sequencing conditions are available on request.
Plasma FV immunodepletion
For immunodepletion experiments, blood was drawn from healthy donors both in 3.2% sodium citrate (wt/vol) and in a mixture of recombinant hirudin (3.5 μM; Kordia Life Sciences), recombinant tick anticoagulant protein (TAP; 0.5 nM; Corvas International, San Diego, CA), and CTI (50 μg/mL). Platelet-poor plasma was prepared by centrifugation at 2000g for 15 minutes. Pooled citrated or hirudin/TAP/CTI-anticoagulated plasma (625 μL) was pretreated with 250 μL (drained volume) protein A sepharose beads (rProtein A Sepharose Fast Flow; GE Healthcare, Uppsala, Sweden) for 1 hour at room temperature to bind all endogenous immunoglobulins (preclearance). After removing the beads by centrifugation at 3000g for 2 minutes, precleared plasma was incubated with protein A sepharose beads bearing the anti-human FV monoclonal antibody 3B1 (kind gift from Prof B. N. Bouma, Utrecht University Hospital, Utrecht, The Netherlands) or no antibody (negative control) at room temperature under rotation. From these mixtures, 50-μL aliquots were taken at regular intervals and assayed for FV35 and full-length TFPI antigen by ELISA.
Surface plasmon resonance analysis
Surface plasmon resonance (SPR) measurements were performed on a Biacore T100 (GE Healthcare). Three flow cells of a CM5 chip were coated with approximately 1500, 3400, and 7000 resonance units (RUs) of recombinant TFPI, respectively. The fourth cell was not coated and served as a reference cell. The chip was perfused with running buffer (25 mM HEPES, 175 mM NaCl, 0.005% Tween, and 5 mM CaCl2 [pH 7.4]) until a stable baseline was obtained. Then, plasma-purified FV (2.6-100 nM in running buffer) was injected for 240 seconds and binding to immobilized TFPI was recorded. Since spontaneous dissociation was rather slow, the chip was stripped with regeneration buffer (25 mM HEPES, 1 M NaCl, and 0.005% Tween [pH 7.4]) for 150 seconds. All experiments were carried out at 25°C with a flow rate of 20 μL/min. Binding was expressed in RUs after correction for the signal obtained in the reference cell.
Statistical analysis
All data are expressed as means plus or minus standard deviation. Due to the small number of people per group, factor levels and thrombin generation parameters were compared among groups using the nonparametric Mann-Whitney-Wilcoxon 2-sample test (U). The effect of age and sex on coagulation factor levels and thrombin generation parameters was assessed by (multiple) linear regression analysis. Statistical analyses were performed with SPSS 14.0 (SPSS, Chicago, IL).
Results
Characteristics of FV-deficient patients
The demographic and clinical characteristics of the 11 patients with congenital severe FV deficiency included in this study are presented in Table 1. Patients PD VII and PD VII-A were sisters, whereas all others were unrelated. Most patients had undetectable FV levels, except for PD III (0.6%), PD VII (4.8%), and PD VII-A (6.2%). Although all patients were bleeders, only patient PD V had experienced life-threatening events (hemorrhagic shock after tonsillectomy and severe hemoperitoneum following rupture of an ovarian cyst).
FV gene mutation screening was performed in all patients whose DNA was available for study. All were found to be homozygous or compound heterozygous for F5 mutations that severely impair gene expression36,37 (P.S. et al, manuscript in preparation). None of the patients was a carrier of the F5 Leiden or F2 G20210A mutations, and the levels of antithrombin, protein C, and protein S, as well as PT, FVIII, and FX, were in the normal range, unless otherwise stated in Table 1.
Thrombin generation in FV-deficient plasma
The coagulation phenotype of the different FV-deficient plasmas was characterized by thrombin generation measurements. When coagulation was initiated with 13.6 pM TF, a concentration that elicits maximal thrombin generation in normal plasma, very little thrombin, if any, was formed in most FV-deficient plasmas (data not shown), except for that of patient PD VII (patient PD VII-A was not tested). The thrombin generation curve obtained in this plasma was comparable with that of normal plasma (although slightly later in time), indicating that approximately 5% FV is sufficient for maximal thrombin generation at 13.6 pM TF.
When the TF concentration was increased, thrombin formation became apparent also in the plasmas with undetectable FV levels and progressively increased with the TF concentration (Figure 1A). At 544 pM TF, all FV-deficient patients showed some degree of thrombin generation, with peak heights ranging between 13.9 nM (PD IV) and 340 nM (PD VII) compared with approximately 500 nM in normal plasma (Figure 1B).
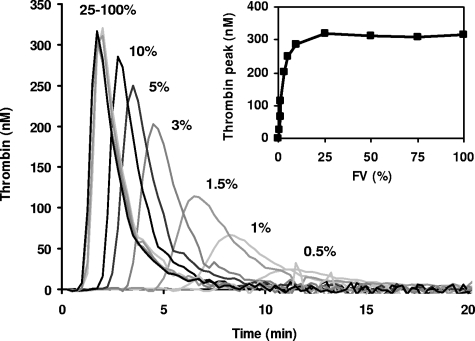
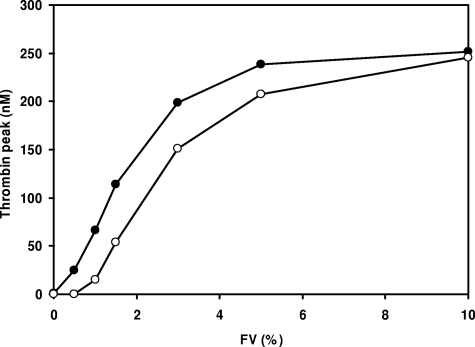
To determine the effect of FV level on thrombin generation, FV-deficient plasma was reconstituted with increasing amounts of purified FV ranging from 0% to 100% of the normal plasma concentration, and thrombin generation was measured at 13.6 pM TF (Figure 2). Although at this TF concentration no thrombin was formed in the absence of exogenous FV, thrombin generation increased steeply (and showed gradually shorter lag times) at increasing FV concentrations, reaching a plateau at approximately 10% FV. Thrombin generation was already detectable at 0.5% FV and attained half-maximal ETP and peak height at 1% and 2% FV, respectively.
Figure 2.
FV titration of thrombin generation in FV-deficient plasma. Plasma from patient GK 502 was reconstituted with 0 to 23 nM purified FV (0%, 0.5%, 1%, 1.5%, 3%, 5%, 10%, 25%, 50%, 75%, and 100% of the normal plasma concentration), and thrombin generation was determined at 13.6 pM TF. Thrombin generation curves obtained at 25%, 50%, 75%, and 100% FV are perfectly superimposable. Inset shows peak height of thrombin generation as a function of FV concentration.
Comparison between FV-deficient plasma reconstituted with 100% FV and normal plasma
To screen FV-deficient plasma for the presence of procoagulant defects, thrombin generation determined under different experimental conditions was compared between FV-deficient plasma reconstituted with 100% purified FV (to eliminate the effect of low FV levels) and normal plasma. Since all reconstituted FV-deficient plasmas behaved essentially the same in these experiments, only a representative example is shown in Figure 3.
Figure 3.
Thrombin generation in FV-deficient plasma reconstituted with 100% FV and in normal plasma. Thrombin generation was measured in FV-deficient plasma (pooled plasma from patients GK 502, GK 505, and GK 506) reconstituted with 100% FV (●) and in normal plasma (○) after triggering coagulation with (A) 1.36 pM TF, (B) 13.6 pM TF, and (C) 13.6 pM TF in the presence of 12 nM APC.
When the TF concentration was varied between 0.34 and 13.6 pM, much more thrombin was formed in reconstituted FV-deficient plasma than in normal plasma at the lowest TF concentrations, but this difference gradually disappeared as the TF concentration was increased. Thus, while the thrombin peak obtained at 1.36 pM was 4 times higher in reconstituted FV-deficient plasma than in normal plasma (183.6 nM vs 44.6 nM; Figure 3A), the thrombin generation curves obtained at 13.6 pM TF were virtually superimposable (Figure 3B). However, when APC (12 nM) was included in the reaction mixture at 13.6 pM TF, thrombin generation was again much higher in reconstituted FV-deficient plasma (peak height, 166.3 nM; ETP, 515.8 nM × min) than in normal plasma (peak height, 21.3 nM; ETP, 62.3 nM × min), revealing a pronounced APC resistance in (reconstituted) FV-deficient plasma (Figure 3C). Similarly, the lag time of thrombin generation was shorter in reconstituted FV-deficient plasma than in normal plasma, both at 1.36 pM TF (2.79 minutes vs 3.67 minutes; Figure 3A) and at 13.6 pM TF in the presence of APC (1.75 minutes vs 2.25 minutes; Figure 3C). Since TFPI is the major determinant of the lag time and peak height of thrombin generation at low TF concentrations and of APC resistance measured with the ETP-based test,38,39 these findings suggested that TFPI levels might be decreased in FV-deficient plasma.
Plasma TFPI levels in FV-deficient patients
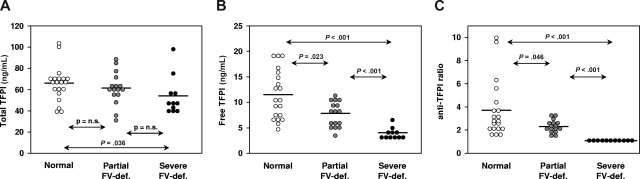
Plasma levels of total and free TFPI antigen were determined in all FV-deficient patients and in 20 healthy controls. As an additional control, 16 people with partial FV deficiency (mean FV levels, 42.9%) were also included. Total TFPI levels (Figure 4A) decreased slightly from healthy controls (66.0 ± 16.5 ng/mL) to those with partial FV deficiency (61.4 ± 15.3 ng/mL; P = nonsignificant) to patients with severe FV deficiency (53.8 ± 17.9 ng/mL; P = .036 vs healthy controls). A similar trend, but more pronounced, was observed for free TFPI levels (Figure 4B), which were higher in healthy controls (11.5 ± 4.8 ng/mL) than in those with partial FV deficiency (7.9 ± 2.5 ng/mL; P = .023) than in patients with severe FV deficiency (4.0 ± 1.0 ng/mL; P < .001 vs healthy controls). Correction for age and sex, which are well-known determinants of TFPI levels,13,40 did not alter these results. Full-length TFPI levels, as determined with an in-house ELISA, were also markedly reduced in FV-deficient plasma (23.9% ± 17.4% of normal pooled plasma).
Figure 4.
Plasma TFPI antigen and activity levels in groups of people with different FV levels. Total and free TFPI antigen levels were measured with commercial ELISAs, whereas TFPI activity levels were determined by a thrombin generation–based assay and expressed as an anti-TFPI ratio, as described in “Thrombin generation assays.” (A) Total TFPI antigen levels. (B) Free TFPI antigen levels. (C) TFPI activity levels. The horizontal bars represent the means of the respective groups. Probabilities of the Mann-Whitney Wilcoxon U test are indicated.
TFPI activity was determined by a thrombin generation–based assay in which coagulation was initiated with 1.36 pM TF in the absence and presence of a neutralizing anti-TFPI antibody. To correct for a possible effect of FV on thrombin generation, the FV level was normalized in all FV-deficient plasmas by adding purified FV up to 100%. TFPI activity, expressed as anti-TFPI ratio (thrombin peak with antibody/thrombin peak without antibody), was 3.70 plus or minus 2.41 in healthy controls, 2.29 plus or minus 0.48 in those with partial FV deficiency (P = .046), and 1.18 plus or minus 0.08 in patients with severe FV deficiency (P < .001 vs healthy controls), indicating that also the activity of TFPI is markedly reduced in FV-deficient plasma (Figure 4C).
Thrombin generation curves obtained in the absence and presence of anti-TFPI antibody were also analyzed separately. In the absence of antibody, plasma free TFPI levels showed a strong direct correlation with the lag time of thrombin generation (r = 0.582; P < .001) and an inverse correlation with the peak height (r = −0.725; P < .001), in line with the notion that free TFPI is a major determinant of these parameters at low TF concentration.39 The lag time was 3.91 plus or minus 1.73 minutes in healthy controls, 2.90 plus or minus 0.44 minutes in those with partial FV deficiency (P = .020), and 2.47 plus or minus 0.29 minutes in patients with severe FV deficiency (P = .001 vs healthy controls). The peak height was 61.4 plus or minus 34.6 nM in healthy controls, 81.2 plus or minus 27.1 nM in those with partial FV deficiency (P = .043), and 156.5 plus or minus 20.7 nM in patients with severe FV deficiency (P < .001 vs healthy controls). Conversely, the lag time and peak height of thrombin generation obtained in the presence of antibody were virtually independent of the free TFPI level, confirming complete neutralization of plasma TFPI by the added antibody.
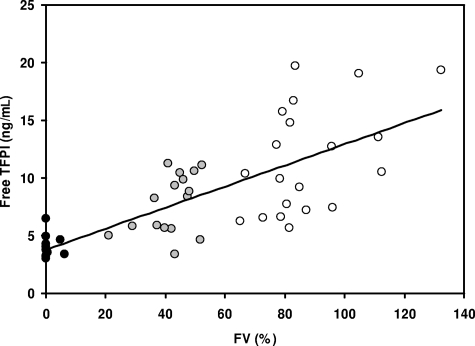
In the population as a whole, FV levels were strongly correlated with the plasma levels of total TFPI (r = 0.372; P = .010), free TFPI (r = 0.739; P < .001; Figure 5), and TFPI activity (r = 0.666; P < .001). Even in the small group of healthy controls, and after correction for age and sex, the FV level correlated with the levels of total TFPI (P = .057) and free TFPI (P = .008), as well as with the anti-TFPI ratio (P = .015).
Figure 5.
Correlation between FV and free TFPI levels in plasma. Plasma FV and free TFPI levels were determined with a prothrombinase-based assay and a commercial ELISA, respectively, in 11 patients with severe FV deficiency (●), 16 people with partial FV deficiency ( ), and 20 healthy controls (○).
), and 20 healthy controls (○).
Effect of TFPI levels on thrombin generation in FV-deficient plasma
Since low plasma levels of TFPI are associated with a hypercoagulable state, they might be beneficial in (severe) FV deficiency. To quantify this possible protective effect, thrombin generation in FV-deficient plasma reconstituted with increasing amounts of FV (0.5%-10% of normal plasma) was determined in the absence and presence of 3.84 ng/mL exogenous TFPI (Figure 6). At the lowest FV concentrations, considerably more thrombin was formed in the absence than in the presence of added TFPI, but this difference gradually decreased at higher FV concentrations and eventually disappeared at 10% FV. In particular, in plasma containing 0.5% FV there was appreciable thrombin generation in the absence of TFPI, but no thrombin generation at all in the presence of TFPI.
Figure 6.
FV-titration of thrombin generation in FV-deficient plasma with and without added TFPI. Thrombin generation was triggered with 13.6 pM TF in FV-deficient plasma from patient GK 502 reconstituted with increasing amounts of purified FV in the absence (●) and presence (○) of 3.84 ng/mL added TFPI.
Analysis of the TFPI gene in FV-deficient patients
To account for the observed low plasma levels of TFPI, the whole coding region and the proximal promoter of the TFPI gene were sequenced in 6 patients with severe FV deficiency (all unrelated). Apart from known polymorphisms, no mutations were found.
TFPI levels in factor-depleted plasmas
TFPI antigen levels were also measured in commercial plasmas depleted of FV or other factors (PT, FVII, FX, protein C, and antithrombin, respectively). All FV-depleted plasmas (n = 5) showed markedly reduced levels of both free (4.3 ± 0.6 ng/mL) and full-length (26.3% ± 12.5%) TFPI, whereas other depleted plasmas had normal TFPI levels (10.4 ± 2.2 ng/mL free TFPI and 121.8% ± 33.2% full-length TFPI).
Interaction between FV and TFPI
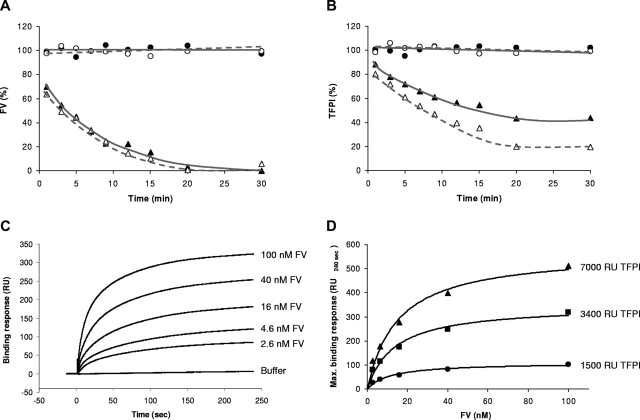
To account for the low TFPI levels in FV-depleted plasma, we investigated the effect of FV immunodepletion on TFPI levels in normal plasma. For this purpose, we used both citrated plasma, in which free calcium is low, and hirudin/TAP/CTI-anticoagulated plasma, which retains normal levels of ionized calcium. Upon the addition of protein A sepharose–coupled anti-FV antibodies, FV disappeared with the same kinetics from both plasmas and became unmeasurable after approximately 20 minutes (Figure 7A). Interestingly, full-length TFPI also disappeared from plasma at a similar rate, suggesting that it binds to FV in plasma. Within 20 minutes from anti-FV addition, the full-length TFPI concentration had dropped to 43% and 20% of its initial value in citrated and hirudin/TAP/CTI-anticoagulated plasma, respectively, and remained constant afterward (Figure 7B). In the absence of anti-FV antibodies, no appreciable decrease of FV (Figure 7A) or TFPI (Figure 7B) levels was observed for 1 hour.
Figure 7.
FV-TFPI interaction. (A,B) Plasma FV immunodepletion. Pooled plasma from 2 healthy donors was incubated with protein A sepharose beads bearing an anti-FV monoclonal antibody (treated plasma; triangles) or no antibody (control plasma; circles). At regular intervals, subsamples were taken and assayed for FV and full-length TFPI antigen as described in “Plasma FV immunodepletion.” FV levels (A) and TFPI levels (B) were expressed as percentage of the average control plasma level. The experiment was conducted at low (citrated plasma; closed symbols, solid lines) and normal (hirudin/TAP/CTI-anticoagulated plasma; open symbols, dashed lines) levels of free calcium ions. (C,D) Measurement of FV-TFPI interaction by SPR. A CM5 chip with immobilized TFPI was perfused with increasing concentrations of FV as described in “Surface plasmon resonance analysis.” (C) Sensograms recorded at 3400 RU of immobilized TFPI with the indicated FV concentrations. (D) Dose-response curves showing the equilibrium binding response (RU240 seconds) as a function of the FV concentration at 1500, 3400, and 7000 RU of immobilized TFPI.
Physical interaction between FV and TFPI was further verified by SPR measurements. FV bound to immobilized TFPI in a specific and concentration-dependent manner (Figure 7C). For all 3 TFPI-containing flow cells, the equilibrium binding response versus FV concentration plot could be fitted with a hyperbolic function (Figure 7D). The FV concentration at which half-maximal binding to TFPI was observed was 13.5 plus or minus 1.7 nM.
Plasma TFPI levels in patients with hemophilia A
To verify whether the reduction of TFPI levels is specific for FV deficiency, 15 patients with hemophilia A were also investigated and compared with the male healthy controls (Table 2). Although both total and free TFPI antigen levels as well as TFPI activity levels determined via the thrombin generation–based assay were all lower in patients with hemophilia A than in healthy controls, only the difference in free TFPI levels reached statistical significance (P = .001). Correction for the age difference between patients with hemophilia and controls did not alter this result.
Table 2.
TFPI levels in patients with hemophilia A and healthy controls
| Group | n | Age, y | FVIII:C, % | Total TFPI, ng/mL | Free TFPI, ng/mL | TFPI activity, anti-TFPI ratio |
|---|---|---|---|---|---|---|
| Patients with hemophilia | 15 | 27.8 ± 19.2 | 3.9 (range, < 1-23) | 65.4 ± 13.8 | 9.8 ± 2.4* | 3.51 ± 1.40 |
| Male controls | 8 | 45.9 ± 18.9 | 79.0 ± 32.0 | 72.6 ± 14.7 | 15.3 ± 3.3* | 5.22 ± 2.92 |
Data are expressed as mean plus or minus SD.
P = .001.
Discussion
Despite the pivotal role of FV in PT activation and the absence of alternative pathways to generate thrombin, many patients with FV levels lower than 1% experience only moderate bleeding and have a milder clinical course than patients with severe hemophilia.18 Also in comparison with FV knockout mice, which die in utero or shortly after birth of massive hemorrhage,22 most patients with severe FV deficiency show a relatively mild bleeding diathesis. This is generally attributed to the fact that FV gene mutations found in patients are compatible with some residual (though often undetectable) FV expression, which may be sufficient to prevent serious bleeding.18,19 As a matter of fact, several in vivo, in vitro, and in silico data,20,24,41,42 as well as the thrombin generation experiments presented in Figures 1 and 2, support the concept that tiny amounts of FV are sufficient for minimal hemostasis.
In the present study, we have explored the additional possibility that patients with severe FV deficiency may be protected from life-threatening bleeding by a concomitant procoagulant defect. By performing a standard thrombophilic screening, low levels of protein C or high levels of PT or FVIII were found in 3 patients (Table 1). However, upon reconstitution with purified FV up to the normal plasma concentration, all FV-deficient plasmas generated considerably more thrombin than normal plasma, both at low TF without APC (Figure 3A) and at higher TF in the presence of APC (Figure 3C). This turned out to be due to markedly reduced TFPI levels in FV-deficient plasma. All measured TFPI-related parameters, including total and free antigen levels as well as activity levels determined with a thrombin generation–based functional assay, were significantly reduced in patients with severe FV deficiency compared with controls, while those with partial FV deficiency showed intermediate levels (Figure 4). The value of 4.0 ng/mL for the mean free TFPI levels of patients with severe FV deficiency (corresponding to approximately 35% of the levels of healthy controls) is even likely to be an overestimate, because (1) in our hands this was approximately the lower limit of the commercial ELISA used for free TFPI quantification; (2) full-length TFPI levels as determined with a homemade ELISA were even lower; and (3) in the thrombin generation–based TFPI activity assay (which reflects free TFPI levels), addition of an anti-TFPI antibody hardly affected the peak height of thrombin generation in FV-deficient patients (mean anti-TFPI ratio, 1.18).
Since plasma levels of free TFPI are a major determinant of ETP-based (and, to a lesser extent, aPTT-based) APC resistance,38,39 the low free TFPI levels may contribute to the APC resistance phenotype observed in FV-deficient plasma,30,43 which had been previously entirely attributed to the missing/reduced APC-cofactor activity of FV.
Since plasma TFPI represents only a small fraction of all intravascular TFPI and is structurally and functionally heterogeneous,12 its pathophysiologic significance is uncertain. However, low plasma levels of (free) TFPI have been shown to increase the risk of venous thrombosis13–16 and to functionally compensate for the low levels of the procoagulant factors in newborns.44 Therefore, they might also enhance thrombin generation and mitigate bleeding symptoms in patients with severe FV deficiency. As a matter of fact, when FV-deficient plasma was reconstituted with increasing amounts of purified FV, considerably more thrombin was generated in the absence than in the presence of added TFPI (Figure 6), especially at the lowest FV concentrations (< 2%). These results indicate that plasma TFPI deficiency lowers the FV level required for minimal thrombin generation in vitro. Whether this mechanism is also physiologically relevant remains to be established. In this respect, it should be emphasized that our thrombin generation experiments performed in platelet-poor plasma may not be entirely representative of the in vivo situation, where platelet FV also contributes to thrombin generation. Although platelets from patients with congenital severe FV deficiency contain hardly any FV,20,45 preliminary thrombin generation experiments in platelet-rich plasma from FV-deficient patients suggest that platelets may play a pivotal role in maintaining the hemostatic balance in patients with severe FV deficiency.
The origin of the (partial) TFPI deficiency in plasma from FV-deficient patients is still under investigation. A genetic cause appears unlikely, as no mutation was identified in the coding region and proximal promoter of the TFPI gene in any of 6 FV-deficient patients. On the other hand, our experiments indicate that FV and TFPI can bind to each other and that a large fraction of full-length TFPI is actually complexed with FV in normal plasma (Figure 7). Although the physiologic significance of this complex is presently unknown, FV-bound TFPI might be protected from plasma proteases and/or from receptor-mediated clearance, and therefore be more stable in vivo. This would account for the low levels of free/full-length TFPI in FV-deficient plasma and for the strong correlation between the levels of FV and TFPI observed in this (Figure 5) and other studies.13,46 Moreover, the finding of a FV/TFPI complex in plasma provides a rationale for the low TFPI levels in FV-immunodepleted plasma.
Finally, since FVIII is structurally and functionally homologous to FV, we reasoned that patients with hemophilia A might also have low plasma levels of TFPI as compared with sex-matched healthy controls. This indeed appeared to be the case, at least for free TFPI, although the reduction of TFPI levels was less consistent and far less pronounced than in severe FV deficiency (Table 2). While the evidence that naturally occurring low TFPI levels could ameliorate the clinical course of patients with severe hemophilia is rather scanty,47 newborns with hemophilia tend to be protected from bleeding by their low TFPI levels.48 Moreover, TFPI inhibitors known as nonanticoagulant sulfated polysaccharides (NASPs) have been reported to effectively improve hemostasis in animal models of hemophilia A and B.49,50
In conclusion, we have shown that congenital FV deficiency is associated with reduced plasma levels of free/full-length TFPI. Partial TFPI deficiency might be a common feature of congenital FV deficiency and possibly help to counteract the severe bleeding tendency associated with this disorder. However, further research is required to fully appreciate the causes and physiologic significance of low plasma levels of TFPI in FV-deficient patients.
Acknowledgments
The authors thank Dr E. Zanon from the Hemophilia Center of Padua University Hospital, Dr V. De Angelis and Dr P. Pradella from the Transfusion Medicine Department of Trieste University Hospital, Dr G. Tagariello from the Regional Blood Disease Center of Castelfranco Veneto Hospital, and Dr G. Barillari from the Institute of Immuno-Hematology and Transfusion Medicine of Udine General and University Hospital (Italy) for their help in recruiting the FV-deficient and hemophilic patients. Dr R. Hartmann from Maastricht University (The Netherlands) is gratefully acknowledged for his expert assistance in the SPR measurements. Dr T. Lindhout from Maastricht University (The Netherlands) and Prof B. N. Bouma from Utrecht University Hospital (The Netherlands) kindly provided recombinant full-length TFPI and monoclonal antibody 3B1, respectively.
This study was supported by a VIDI grant (no. 917-76-312 to E.C.) from the Dutch Organization for Scientific Research (NWO; Amsterdam, The Netherlands).
Footnotes
The publication costs of this article were defrayed in part by page charge payment. Therefore, and solely to indicate this fact, this article is hereby marked “advertisement” in accordance with 18 USC section 1734.
Authorship
Contribution: J.R., E.C., and C.D. designed the study; P.S. and L.S. selected and enrolled patients; C.D., C.R., S.G., and E.C. performed experiments; C.D., E.C., J.R., and P.S. analyzed data; C.D. and E.C. wrote the manuscript; and P.S. and J.R. performed critical review of the manuscript.
Conflict-of-interest disclosure: The authors declare no competing financial interests.
Correspondence: Elisabetta Castoldi, Department of Biochemistry, Maastricht University, PO Box 616, 6200 MD Maastricht, The Netherlands; e-mail: e.castoldi@bioch.unimaas.nl.
References
- 1.Segers K, Dahlbäck B, Nicolaes GA. Coagulation factor V and thrombophilia: background and mechanisms. Thromb Haemost. 2007;98:530–542. [PubMed] [Google Scholar]
- 2.Rosing J, Tans G, Govers-Riemslag JW, Zwaal RF, Hemker HC. The role of phospholipids and factor Va in the prothrombinase complex. J Biol Chem. 1980;255:274–283. [PubMed] [Google Scholar]
- 3.Dahlbäck B, Villoutreix BO. The anticoagulant protein C pathway. FEBS Lett. 2005;579:3310–3316. doi: 10.1016/j.febslet.2005.03.001. [DOI] [PubMed] [Google Scholar]
- 4.Crawley JT, Lane DA. The haemostatic role of tissue factor pathway inhibitor. Arterioscler Thromb Vasc Biol. 2008;28:233–242. doi: 10.1161/ATVBAHA.107.141606. [DOI] [PubMed] [Google Scholar]
- 5.Dahlbäck B, Carlsson M, Svensson PJ. Familial thrombophilia due to a previously unrecognized mechanism characterized by poor anticoagulant response to activated protein C: prediction of a cofactor to activated protein C. Proc Natl Acad Sci U S A. 1993;90:1004–1008. doi: 10.1073/pnas.90.3.1004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Broze GJ, Jr, Girard TJ, Novotny WF. Regulation of coagulation by a multivalent Kunitz-type inhibitor. Biochemistry. 1990;29:7539–7546. doi: 10.1021/bi00485a001. [DOI] [PubMed] [Google Scholar]
- 7.Hackeng TM, Seré KM, Tans G, Rosing J. Protein S stimulates inhibition of the tissue factor pathway by tissue factor pathway inhibitor. Proc Natl Acad Sci U S A. 2006;103:3106–3111. doi: 10.1073/pnas.0504240103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Ndonwi M, Broze G., Jr Protein S enhances the tissue factor pathway inhibitor inhibition of factor Xa but not its inhibition of factor VIIa-tissue factor. J Thromb Haemost. 2008;6:1044–1046. doi: 10.1111/j.1538-7836.2008.02980.x. [DOI] [PubMed] [Google Scholar]
- 9.Wesselschmidt R, Likert K, Girard T, Wun TC, Broze GJ., Jr Tissue factor pathway inhibitor: the carboxy-terminus is required for optimal inhibition of factor Xa. Blood. 1992;79:2004–2010. [PubMed] [Google Scholar]
- 10.Novotny WF, Girard TJ, Miletich JP, Broze GJ., Jr Platelets secrete a coagulation inhibitor functionally and antigenically similar to the lipoprotein associated coagulation inhibitor. Blood. 1988;72:2020–2025. [PubMed] [Google Scholar]
- 11.Maroney SA, Haberichter SL, Friese P, et al. Active tissue factor pathway inhibitor is expressed on the surface of coated platelets. Blood. 2007;109:1931–1937. doi: 10.1182/blood-2006-07-037283. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Broze GJ, Jr, Lange GW, Duffin KL, MacPhail L. Heterogeneity of plasma tissue factor pathway inhibitor. Blood Coagul Fibrinolysis. 1994;5:551–559. [PubMed] [Google Scholar]
- 13.Dahm A, Van Hylckama Vlieg A, Bendz B, Rosendaal F, Bertina RM, Sandset PM. Low levels of tissue factor pathway inhibitor (TFPI) increase the risk of venous thrombosis. Blood. 2003;101:4387–4392. doi: 10.1182/blood-2002-10-3188. [DOI] [PubMed] [Google Scholar]
- 14.Ariëns RA, Alberio G, Moia M, Mannucci PM. Low levels of heparin-releasable tissue factor path-way inhibitor in young patients with thrombosis. Thromb Haemost. 1999;81:203–207. [PubMed] [Google Scholar]
- 15.Amini-Nekoo A, Futers TS, Moia M, Mannucci PM, Grant PJ, Ariëns RA. Analysis of the tissue factor pathway inhibitor gene and antigen levels in relation to venous thrombosis. Br J Haematol. 2001;113:537–543. doi: 10.1046/j.1365-2141.2001.02752.x. [DOI] [PubMed] [Google Scholar]
- 16.Hoke M, Kyrle PA, Minar E, et al. Tissue factor pathway inhibitor and the risk of recurrent venous thromboembolism. Thromb Haemost. 2005;94:787–790. doi: 10.1160/TH05-06-0412. [DOI] [PubMed] [Google Scholar]
- 17.Asselta R, Tenchini ML, Duga S. Inherited defects of coagulation factor V: the hemorrhagic side. J Thromb Haemost. 2006;4:26–34. doi: 10.1111/j.1538-7836.2005.01590.x. [DOI] [PubMed] [Google Scholar]
- 18.Lak M, Sharifian R, Peyvandi F, Mannucci PM. Symptoms of inherited factor V deficiency in 35 Iranian patients. Br J Haematol. 1998;103:1067–1069. doi: 10.1046/j.1365-2141.1998.01077.x. [DOI] [PubMed] [Google Scholar]
- 19.Acharya SS, Coughlin A, Dimichele DM. Rare Bleeding Disorder Registry: deficiencies of factors II, V, VII, X, XIII, fibrinogen and dysfibrinogenemias. J Thromb Haemost. 2004;2:248–256. doi: 10.1111/j.1538-7836.2003.t01-1-00553.x. [DOI] [PubMed] [Google Scholar]
- 20.Guasch JF, Cannegieter S, Reitsma PH, van't Veer-Korthof ET, Bertina RM. Severe coagulation factor V deficiency caused by a 4 bp deletion in the factor V gene. Br J Haematol. 1998;101:32–39. doi: 10.1046/j.1365-2141.1998.00664.x. [DOI] [PubMed] [Google Scholar]
- 21.Castoldi E, Lunghi B, Mingozzi F, et al. A missense mutation (Y1702C) in the coagulation factor V gene is a frequent cause of factor V deficiency in the Italian population. Haematologica. 2001;86:629–633. [PubMed] [Google Scholar]
- 22.Cui J, O'Shea KS, Purkayastha A, Saunders TL, Ginsburg D. Fatal haemorrhage and incomplete block to embryogenesis in mice lacking coagulation factor V. Nature. 1996;384:66–68. doi: 10.1038/384066a0. [DOI] [PubMed] [Google Scholar]
- 23.Bolton-Maggs PH, Pasi KJ. Haemophilias A and B. Lancet. 2003;361:1801–1809. doi: 10.1016/S0140-6736(03)13405-8. [DOI] [PubMed] [Google Scholar]
- 24.Mann KG. How much factor V is enough? Thromb Haemost. 2000;83:3–4. [PubMed] [Google Scholar]
- 25.van Dijk K, van der Bom JG, Fischer K, Grobbee DE, van den Berg HM. Do prothrombotic factors influence clinical phenotype of severe haemophilia? A review of the literature. Thromb Haemost. 2004;92:305–310. doi: 10.1160/TH04-02-0112. [DOI] [PubMed] [Google Scholar]
- 26.Franchini M, Veneri D, Poli G, Manzato F, Salvagno GL, Lippi G. High prevalence of inherited prothrombotic risk factors in 134 consecutive patients with von Willebrand disease. Am J Hematol. 2006;81:465–467. doi: 10.1002/ajh.20623. [DOI] [PubMed] [Google Scholar]
- 27.Castoldi E, Govers-Riemslag JW, Pinotti M, et al. Coinheritance of Factor V (FV) Leiden enhances thrombin formation and is associated with a mild bleeding phenotype in patients homozygous for the FVII 9726+5G>A (FVII Lazio) mutation. Blood. 2003;102:4014–4020. doi: 10.1182/blood-2003-04-1199. [DOI] [PubMed] [Google Scholar]
- 28.Strey RF, Siegemund A, Siegemund T, et al. Influence of factor V HR2 on thrombin generation and clinical manifestation in rare bleeding disorders. Pathophysiol Haemost Thromb. 2005;34:279–283. doi: 10.1159/000093108. [DOI] [PubMed] [Google Scholar]
- 29.Rodeghiero F, Tosetto A, Castaman G. How to estimate bleeding risk in mild bleeding disorders. J Thromb Haemost. 2007;5:157–166. doi: 10.1111/j.1538-7836.2007.02520.x. [DOI] [PubMed] [Google Scholar]
- 30.Brugge JM, Simioni P, Bernardi F, et al. Expression of the normal factor V allele modulates the APC resistance phenotype in heterozygous carriers of the factor V Leiden mutation. J Thromb Haemost. 2005;3:2695–2702. doi: 10.1111/j.1538-7836.2005.01634.x. [DOI] [PubMed] [Google Scholar]
- 31.Castoldi E, Simioni P, Tormene D, et al. Differential effects of high prothrombin levels on thrombin generation depending on the cause of the hyperprothrombinemia. J Thromb Haemost. 2007;5:971–979. doi: 10.1111/j.1538-7836.2007.02448.x. [DOI] [PubMed] [Google Scholar]
- 32.Nicolaes GA, Tans G, Thomassen MC, et al. Peptide bond cleavages and loss of functional activ-ity during inactivation of factor Va and factor VaR506Q by activated protein C. J Biol Chem. 1995;270:21158–21166. doi: 10.1074/jbc.270.36.21158. [DOI] [PubMed] [Google Scholar]
- 33.Hemker HC, Giesen P, Al Dieri R, et al. The calibrated automated thrombogram (CAT): a universal routine test for hyper- and hypocoagulability. Pathophysiol Haemost Thromb. 2002;32:249–253. doi: 10.1159/000073575. [DOI] [PubMed] [Google Scholar]
- 34.Dahm AE, Andersen TO, Rosendaal F, Sandset PM. A novel anticoagulant activity assay of tissue factor pathway inhibitor I (TFPI). J Thromb Haemost. 2005;3:651–658. doi: 10.1111/j.1538-7836.2005.01237.x. [DOI] [PubMed] [Google Scholar]
- 35.Segers K, Dahlbäck B, Bock PE, Tans G, Rosing J, Nicolaes GA. The role of thrombin exosites I and II in the activation of human coagulation factor V. J Biol Chem. 2007;282:33915–33924. doi: 10.1074/jbc.M701123200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Olds R, Simioni P, Thompson E, Morgan T, Girolami A, Lane DA. Factor V Gly2112Asp, a C2-domain variant, is associated with severe deficiency and a bleeding tendency [abstract]. J Thromb Haemost. 2003:P1206. [Google Scholar]
- 37.Tognin G, Rossetto V, Barillari G, et al. Clinical and molecular characterization of three Italian patients with severe factor V deficiency and bleeding disorder (‘parahaemophilia’): report of a novel homozygous mutation (FV: Asp524His) [abstract]. J Thromb Haemost. 2005:P0250. [Google Scholar]
- 38.de Visser MC, van Hylckama Vlieg A, Tans G, et al. Determinants of the APTT- and ETP-based APC sensitivity tests. J Thromb Haemost. 2005;3:1488–1494. doi: 10.1111/j.1538-7836.2005.01430.x. [DOI] [PubMed] [Google Scholar]
- 39.Dielis AW, Castoldi E, Spronk HM, et al. Coagulation factors and the protein C system as determinants of thrombin generation in a normal population. J Thromb Haemost. 2008;6:125–131. doi: 10.1111/j.1538-7836.2007.02824.x. [DOI] [PubMed] [Google Scholar]
- 40.Ariëns RA, Coppola R, Potenza I, Mannucci PM. The increase with age of the components of the tissue factor coagulation pathway is gender-dependent. Blood Coagul Fibrinolysis. 1995;6:433–437. doi: 10.1097/00001721-199507000-00010. [DOI] [PubMed] [Google Scholar]
- 41.Yang TL, Cui J, Taylor JM, Yang A, Gruber SB, Ginsburg D. Rescue of fatal neonatal hemorrhage in factor V deficient mice by low level transgene expression. Thromb Haemost. 2000;83:70–77. [PubMed] [Google Scholar]
- 42.Al Dieri R, Peyvandi F, Santagostino E, et al. The thrombogram in rare inherited coagulation disorders: its relation to clinical bleeding. Thromb Haemost. 2002;88:576–582. [PubMed] [Google Scholar]
- 43.Simioni P, Girolami A. Homozygous factor V-deficient patients show resistance to activated protein C whereas heterozygotes do not. Blood Coagul Fibrinolysis. 1994;5:825–827. doi: 10.1097/00001721-199410000-00021. [DOI] [PubMed] [Google Scholar]
- 44.Cvirn G, Gallistl S, Leschnik B, Muntean W. Low tissue factor pathway inhibitor (TFPI) together with low antithrombin allows sufficient thrombin generation in neonates. J Thromb Haemost. 2003;1:263–268. doi: 10.1046/j.1538-7836.2003.00081.x. [DOI] [PubMed] [Google Scholar]
- 45.Ajzner EE, Balogh I, Szabo T, Marosi A, Haramura G, Muszbek L. Severe coagulation factor V deficiency caused by 2 novel frameshift mutations: 2952delT in exon 13 and 5493insG in exon 16 of factor 5 gene. Blood. 2002;99:702–705. doi: 10.1182/blood.v99.2.702. [DOI] [PubMed] [Google Scholar]
- 46.Vossen CY, Callas PW, Hasstedt SJ, Long GL, Rosendaal FR, Bovill EG. A genetic basis for the interrelation of coagulation factors. J Thromb Haemost. 2007;5:1930–1935. doi: 10.1111/j.1538-7836.2007.02678.x. [DOI] [PubMed] [Google Scholar]
- 47.Shetty S, Vora S, Kulkarni B, et al. Contribution of natural anticoagulant and fibrinolytic factors in modulating the clinical severity of haemophilia patients. Br J Haematol. 2007;138:541–544. doi: 10.1111/j.1365-2141.2007.06693.x. [DOI] [PubMed] [Google Scholar]
- 48.Fritsch P, Cvirn G, Cimenti C, et al. Thrombin generation in factor VIII-depleted neonatal plasma: nearly normal because of physiologically low antithrombin and tissue factor pathway inhibitor. J Thromb Haemost. 2006;4:1071–1077. doi: 10.1111/j.1538-7836.2006.01947.x. [DOI] [PubMed] [Google Scholar]
- 49.Liu T, Scallan CD, Broze GJ, Jr, Patarroyo-White S, Pierce GF, Johnson KW. Improved coagulation in bleeding disorders by non-anticoagulant sulfated polysaccharides (NASP). Thromb Haemost. 2006;95:68–76. [PubMed] [Google Scholar]
- 50.Prasad S, Lillicrap D, Labelle A, et al. Efficacy and safety of a new-class of hemostatic drug candidate, AV513, in hemophilia A dogs. Blood. 2008;111:672–679. doi: 10.1182/blood-2007-07-098913. [DOI] [PubMed] [Google Scholar]