Abstract
Indole-3-carbinol (I3C) conjugates are phytochemicals expressed in brassica vegetables and have been associated with the anticancer activities of vegetable consumption. I3C and its metabolite bis(3′-indolyl)methane (DIM) induce overlapping and unique responses in multiple cancer cell lines and tumors, and these include growth inhibition, apoptosis and antiangiogenic activities. The mechanisms of these responses are complex and dependent on cell context. I3C and/or DIM activate or inactivate multiple nuclear receptors, induce endoplasmic reticulum stress, decrease mitochondrial membrane potential, and modulate multiple signaling pathways including kinases. DIM has been used as a template to synthesize a series of 1,1-bis(3′indolyl)-1-(substituted aromatic)methanes (i.e. C-DIMs) which are also cytotoxic to cancer cells and tumors. Some of the effects of C-DIMs resemble those reported for DIM analogs; however, structure–activity studies with the aromatic ring has resulted in generation of highly unique receptor agonists. For example, p-trifluoromethylphenyl, p-t-butylphenyl and p-biphenyl analogs activate peroxisome proliferator-activated receptor γ (PPARγ), and p-methoxyphenyl and p-phenyl compounds activate nerve growth factor-induced-Bα (NGFI-Bα, Nur77) orphan nuclear receptor. The effects of C-DIMs on PPARγ and Nur77 coupled with their receptor-independent activities has resulted in the development of a novel group of multi-targeted anticancer drugs with excellent potential for clinical treatment of cancer.
Keywords: I3C, DIMs, Anticarcinogenesis, Nur77, PPAR
1. Introduction
The development of cancer is a complex process that is initiated by some form of DNA damage which ultimately can lead to tumor formation. The conversion of a damaged cell into a tumor requires a series of steps associated with tumor promotion and progression and, for many cancers, the initiation–promotion–progression pathways are accompanied by activating specific protooncogenes and inactivating mutations of tumor suppressor genes [1,2]. Not surprisingly, many of these critical genes are involved in DNA repair and maintenance of DNA integrity, regulation of cell proliferation, and maintenance of cell survival and cell death pathways. Inherited genetic factors play an important role and can explain enhanced cancer susceptibility in only 5–15% of most cancers, whereas “environmental/lifestyle” or dietary factors are major determinants in cancer formation. With few exceptions, the precise contributions of environmental, lifestyle, dietary or other risk factors for cancer are not well-defined, although, it is generally considered that diets high in vegetables, fruit and fish products and low in red meats are generally protective. Nevertheless, the precise contributions of various food products as protective against or as risk factors for development of specific cancers is not well-defined.
2. Cruciferous vegetables and cancer
Epidemiology studies on various population groups have been used to investigate the association of various food-types with development of specific cancers, and results of these studies are highly variable and sometimes conflicting. Large prospective studies in North America and Europe have examined the association between consumption of different foods and cancer incidence, and this approach provides important insights on cancer chemoprevention. Examination of participants in the Nurses’ Health and Health Professionals follow-up studies (US cohort) showed that high fruit and vegetable consumption was associated with a decreased risk for cardiovascular disease [relative risk (RR) = 0.88] but had no effect on cancer (RR = 1.0) [3]. Similar results were observed for two large prospective studies on breast cancer [4,5]; however, in the US cohort, it was reported that higher dietary intakes of fruits and vegetables were associated with lower risks of lung cancer in men, but not in women [6]. Several other prospective studies have demonstrated a protective effect of high intakes of fruit and/or vegetables or specific sub-types of vegetable consumption for urothelial cancer, non-Hodgkin’s lymphoma, colorectal cancer, pancreatic cancer, bladder cancer, and renal cell carcinoma [7–12]. Moreover, in many of these studies, the vegetable sub-type frequently associated with decreased cancer risk was cruciferous vegetables including broccoli, cauliflower and Brussel’s sprouts. For example, in the bladder cancer study, total fruit and vegetable intake was not significantly associated with decreased bladder cancer; however, intake of cruciferous vegetables was inversely associated with risk with a relative risk of 0.49 in the highest consumption group [11]. Although prostate cancer has been inversely associated with high consumption of cruciferous vegetables in case-control studies [13,14], results of a prospective study did not confirm the protective effects [15]. The predictive nature of prospective and case-control epidemiology studies depends on several factors including recall of dietary intakes which has several problems. Nevertheless, there is evidence that dietary consumption of cruciferous vegetables may provide protection from some chronic diseases and specific cancers.
2.1. Anticancer activities of cruciferous vegetables and indole-3-carbinol (I3C)
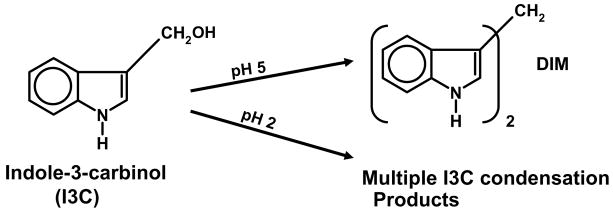
Natural products from vegetable sources have been widely used in traditional medicine as potions for treatment of a variety of human ailments, and phytochemicals and their derivatives are major sources and building blocks for development of new drugs [16–18]. Cruciferous vegetables contain a wide spectrum of active phytochemicals; however, most studies have focused on isothiocyanates and indole-3-carbinol (I3C) as the major chemopreventive and chemotherapeutic phytochemicals associated with the anticancer activities of cruciferous vegetables [19–23]. Glucobrassican, a major component of cruciferous vegetables, is a 3-indolylmethyl glucosinolate which is readily hydrolyzed in the acidic conditions of the gut to give I3C. This compound is highly unstable in acidic conditions (pH 2–4) and undergoes a series of condensation reactions to give a broad spectrum of products [23–25]. At higher pHs (pH 5–7), such as those encountered under cell culture conditions, I3C is primarily converted into the dimeric product bis(3-indolyl)methane (DIM) [26] (Fig. 1). Thus, many of the responses observed for I3C in vivo are due to a complex mixture of acid-catalyzed indole derivatives, whereas in cell culture, the effects may be due to a combination of I3C/DIM.
Fig. 1.
I3C undergoes acid-catalyzed rearrangement into multiple condensation products including DIM.
Early in vivo reports demonstrated that I3C inhibited carcinogen-induced mammary tumor growth, and subsequent studies in other animal models have confirmed the antitumorigenic activity of I3C [27–37]. In some animal models, the effects of I3C have been related to its activity as an inducer of phase I (CYPs) and phase II (glutathione S-transferase) drug-metabolizing enzymes. This may be particularly relevant for hormone-dependent cancers where I3C induces changes in the 2-hydroxylation (increases) and 16α-hydroxylation (increases) of 17β-estradiol [24,28,32,38–41].
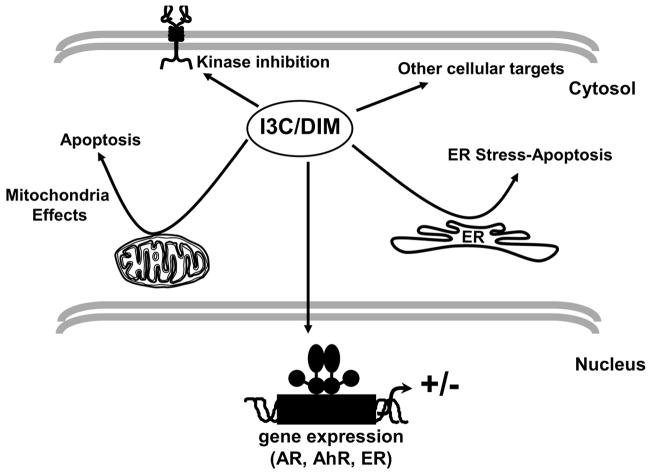
The mechanism of action of I3C has been extensively investigated in cancer cell culture models, and a recent review article has summarized these responses [41]. Since the effects are probably due to a combination of I3C and DIM, it is difficult to dissect out those responses that are due to I3C alone. It is also important to note that direct comparisons between studies are difficult since only selected responses are observed, and there may be important cell context-dependent differences. Fig. 2 summarizes major pathways associated with the effects of I3C on cancer cells, namely inhibition of cell proliferation and cell cycle progression and induction of cell death or decreased cell survival. These effects undoubtedly contribute to the cytotoxicity of I3C in most cancer cell lines over concentrations that range from the low μM to 500 μM. For example, several studies show that I3C modulates expression of several genes and proteins that lead to inhibition of cell growth, and these include decreased cyclin D1, cyclin-dependent kinases (cdks) 2 and 6, phospho-retinoblastoma protein, and increased expression of several cyclin-dependent kinase inhibitors [42–49]. Many of these responses are highly dependent on cell context, suggesting that the underlying mechanisms of growth inhibition are different. For example, treatment of MCF-7 breast cancer cells with I3C blocks G1 to S phase progression, and this is associated with conversion of the active 90 kDa cdk2 complex to an inactive higher molecular weight (200 kDa) complex [44]. Moreover, in this cell line, I3C did not affect expression of p21 or p27 cdk inhibitors, and the effects on cdk2 were not observed for DIM. Interestingly, a previous study by the same group showed that I3C decreased cdk6, and this response was linked to decreased Sp1 binding to the cdk6 promoter in MCF-7 cells [45,47].
Fig. 2.
I3C and DIM modulate multiple responses and genes in cancer cell lines through several pathways.
I3C also causes G1 cell cycle arrest in prostate cancer cells; however, the specific targets of I3C exhibit both similar and overlapping effects in LNCaP and PC3 prostate cancer cells compared to MCF-7 breast cancer cells. In androgen-responsive LNCaP prostate cancer cells, treatment with I3C decreased expression of cdk6, decreased cdk2- dependent enzymatic activity, and induced the cdk inhibitors p16, p21 and p27 [42]. The effects of I3C in androgen-insensitive PC3 cells were similar to those observed in LNCaP cells, and expression of both p21 and p27 were increased and cdk6 protein and cdk6-dependent activity were decreased [48]. Growth inhibitory responses in breast and prostate cancer cells required concentrations of I3C as high as 100 μM, whereas the characteristic I3C-induced G1 arrest and decreased proliferation of human immortalized keratinocyte HaCaT cells was only observed at concentrations between 200 and 500 μM [43]. Moreover, in this cell line, the cdk inhibitor p15 was induced but expression of p21, p27, p19 and cdk6 was unchanged at all concentrations of I3C [43].
The cytotoxicity of I3C in cancer cell lines is also associated with the induction of cell death and the modulation of multiple genes and proteins associated with this response [48,50–61]. Rahman and coworkers reported that I3C induced apoptosis and caspase-dependent PARP cleavage in ER-negative MDA-MB-453 breast cancer cells, and this was accompanied by caspase-3 activation, mitochondrial uptake of bax, decreased bcl-2, and an overall increase of bax/bcl-2 ratios. In contrast, another report showed that induction of apoptosis in MCF-7 cells was bax- and p53-dependent [56]. These results suggest that I3C may activate the intrinsic mitochondrial pathway; however, the proapoptotic effects of I3C in cancer cell lines need further investigation.
The mechanisms of I3C-induced cytotoxicity in cancer cells may be related, in part, to a host of other responses that can effect cell proliferation and cell death (Fig. 2). For example, I3C inhibits both androgenic responses in prostate and estrogenic responses in cervical and breast cancer cells [61–67], and induction of CYP-dependent estradiol metabolism also decreases the mitogenic effects of this hormone [28,32,38–41]. In addition, I3C modulates expression or activation of many other genes including p53 (increase), interferon γ, NFκB (decrease), non-steroidal anti-inflammatory drug-activated gene-1 (NAG-1, increase), breast cancer susceptibility gene (BRCA1, increase), and the phosphatidylinositol-3-kinase (PI3-K, decrease) survival pathways [59,60,66–73]. I3C is also an aryl hydrocarbon receptor (AhR) agonist which can activate growth inhibitory pathways in breast and other cancer cell lines [74,75]. A recent paper showed that I3C induces BRCA1 and BRCA2 in breast (MCF-7 and T47D) and prostate (LNCaP and DU145) cancer cells [67]. Using RNA interference studies, they showed that I3C-mediated antiestrogenic/antiandrogenic activity, and cytotoxicity was due to BRCA1 (and/or BRCA2). Moreover, the results suggest that induction of BRCA1 and BRCA2 by I3C is due to activation of ER stress and these results, coupled with studies on DIM and synthetic DIM analogs, may be one of the important underlying mechanisms of action of I3C (see below).
2.2. Anticarcinogenic activity of DIM
DIM is the major dimeric condensation product of I3C (Fig. 1) and, although this compound is photolabile, it is relatively stable in cell culture media. In MCF-7 cells treated with radiolabeled I3C, a significant amount of the radiolabel is found as DIM in the nuclear fraction [26]. Like I3C, DIM is cytotoxic to cancer cells and inhibits growth of multiple tumor types [32,50,53,55,72,76], and these responses are accompanied by activation of growth inhibitory and proapoptotic pathways [32,50,53,55,72,73,75–95] (Fig. 2). DIM induces some of the same responses observed for I3C, and this is consistent with the conversion of I3C into DIM in cell culture media [26]. However, the mechanisms of action of DIM are more well-defined than those for I3C, and DIM induces several unique pathways that are not observed for I3C. In this section of the review, a more detailed examination of some tumor type-specific effects and mechanisms of action of DIM will be discussed and, where possible, compared to those observed for I3C.
DIM inhibits breast cancer cell and tumor growth and the mechanisms of action are complex and cell context-dependent (Fig. 2). Studies in this laboratory have shown that DIM inhibits carcinogen- induced rat mammary tumor growth at a dose of 5 mg/kg/d and, in MCF-7 cells, DIM acts as a selective AhR modulator (SAhRM) and activates inhibitory AhR-ERα crosstalk [96]. This mechanism has been extensively investigated in breast cancer cells for many other AhR agonists and undoubtedly contributes to the activity of DIM in breast cancer cells [97,98]. The importance of the AhR in MCF-7 cells has been investigated by RNA interference, and knockdown of this receptor increased the percentage of cells progressing through G0/G1 to S phase, indicating a constitutive (ligand-independent) role for this receptor as an inhibitor of MCF-7 cell proliferation [99]. However, there are also reports that DIM is estrogenic and activates genomic and non-genomic ER-dependent pathways in breast cancer cell lines and rainbow trout [100,101]. This seemingly contradicts the antiestrogenic effects of DIM through activation of inhibitory AhR-ERα crosstalk in breast cancer cells. However, recent studies have demonstrated that many AhR agonists, including 2,3,7,8-tetrachlorodibenzo- p-dioxin (TCDD) and DIM, directly activate ERα; however, these effects are decreased by competitive AhR binding and do not necessarily block inhibitory AhR-ERα crosstalk [102–104]. Despite the evidence that activation of the AhR by DIM plays a role in blocking estrogen-dependent breast cancer and tumor growth, there is evidence that DIM also activates AhR-independent growth inhibitory/proapoptotic breast cancer cells [76,78–88]. However, there are some inconsistencies in some of the proposed mechanisms of growth inhibition by DIM and I3C in breast cancer cells. For example, DIM induces p21 expression in MCF-7 and MDA-MB-231 cells, and this is associated with enhanced Sp1 binding to the p21 promoter [85], whereas I3C does not activate p21 through enhanced Sp1 binding [45]. Another report shows that DIM induces p21 in MCF-7 but not MDA-MB-231 cells and does not affect Sp1, Sp3 or Sp4 protein expression [81]. These differences between I3C and DIM and between studies have not been resolved and may be due to multiple factors including the type of serum used and the cell passage number since Sp protein expression can also change with passage number [105].
DIM also modulates kinase activities and other proapoptotic and growth inhibitory genes and proteins in breast cancer cells, and there is also evidence that DIM decreases mitochondrial membrane potential (MMP) and induces ER stress in breast cancer cells [80,81,86]. Both of these responses may be important underlying mechanisms of action for DIM in breast and other cancer cell lines. For example, in MCF-7 cells, DIM induces several markers of ER stress including CHOP and JNK phosphorylation [80], and studies in pancreatic cancer cells show that induction of these responses by DIM leads to constitutive activation of death receptor 5 (DR5) and the extrinsic apoptosis pathway [82]. Based on these results, it is possible that many of the proapoptotic and growth inhibitory effects of DIM may be due to direct targeting of mitochondria and the ER, and it is possible that perturbation of these organelles may be related.
The cytotoxicity of DIM has also been observed in many other cancer cell lines; however, there has been an extensive focus on both androgen- sensitive and androgen-insensitive prostate cancer [89–93]. DIM binds directly to the AR [106] and is an AR antagonist in AR-positive LNCaP prostate cancer cells. However, it is evident from studies in AR-positive and AR-negative prostate cancer cells that DIM induces growth inhibitory and proapoptotic responses that are AR-independent. Savino and coworkers investigated the role of DIM on ER stress and intracellular calcium levels in mediating the cytotoxicity of DIM in AR-negative DU145 prostate and C33A cervical cancer cell lines and found cell context- dependent differences [92]. There is also evidence that DIM induces apoptosis through the mitochondrial pathway in AR-negative PC3 cells [93], demonstrating that in prostate cancer cells, both the ER and mitochondria can also be targets for DIM (Fig. 2). Future studies in different cancer cell lines are required to confirm the role of the ER and mitochondria or their interactions on the activity of DIM and also identify other critical cellular sites responsible for the remarkable anticancer activity of DIM.
2.3. Synthetic analogs of DIM as anticancer agents
2.3.1. Ring substituted DIMs
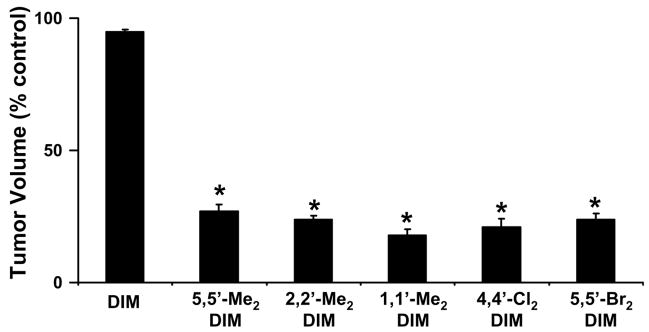
Initial studies in our laboratory focused on the AhR agonist activity of DIM in breast cancer cells and activation of inhibitory AhR-ER crosstalk in which DIM inhibits estrogen-induced responses in breast cancer cells and mammary tumors [77]. Our results characterized the antiestrogenic activity of DIM and the inhibition of carcinogen-induced mammary tumor growth in female Sprague–Dawley rats at a dose of 5 mg/kg/d [77]. A series of symmetrical ring-substituted DIM analogs were readily synthesized by the condensation of commercially available substituted indoles with formaldehyde. These compounds were initially screened in the carcinogen- induced rat mammary tumor model at a dose of 1 mg/kg/d to identify compounds that were at least 5 times more active than DIM [107,108]. Fig. 3 illustrates the anticarcinogenic activity of several active ring-substituted DIM analogs that inhibited tumor growth at a dose of 1 mg/kg/d, whereas in this same model, DIM was not active at this dose. Subsequent in vivo studies at lower doses have not been carried out; however, it was evident that the anticarcinogenic activity of some ring-substituted DIMs was >5-fold higher than DIM [77]. 5,5′-DibromoDIM has been used as a prototype in two studies comparing the activity of the ring-substituted compound with DIM. Both 5,5′-dibromoDIM and DIM inhibited pancreatic cancer cell survival and activated ER stress pathways which enhanced DR5 and the extrinsic apoptotic pathway [81,82].
Fig. 3.
Comparative effects of DIM and ring-substituted DIMs as inhibitors of carcinogen-induced rat mammary tumor growth in female Sprague–Dawley rats. Animals were treated with 1.0 mg/kg/d DIM or ring-substituted DIMs, and corn oil (100%) served as a vehicle control [77,107,108]. 5,5′-Dimethyl- DIM (5,5′-Me2), 2,2′-dimethylDIM (2,2′-Me2), 1,1′-dimethylDIM (1,1′-Me2), 4,4-dichloroDIM (4,4′-Cl2) and 5,5′-dibromoDIM (5,5′-Br2) all significantly (p < 0.05) decreased tumor weights and volumes (*).
In this study, 5,5′-dibromoDIM was clearly more potent that DIM (⩾2-fold); however, both compounds induced the same ER stress-dependent responses in pancreatic cancer cells. In contrast, in ER-negative MDA-MB-231 and ER-positive MCF-7 breast cancer cells, the anticarcinogenic activity of 5,5′-dibromoDIM was not only >2-fold higher than DIM but there was also evidence for differences in their mechanisms of action [81]. DIM significantly induced p21 expression in MCF-7 cells; 5,5′-dibromoDIM decreased p21 in the same cell line, and neither compound affected levels of p21 protein in MDA-MB-231 cells. Similar differences were observed for the mitochondriotoxic effects of these compounds where 5,5′-dibromoDIM significantly decreased MMP in MCF-7 and MDAMB- 231 cells, whereas minimal effects were observed for DIM. These data, coupled with results of ongoing studies, demonstrate differences in the potency and mechanism of action between DIM and ring-substituted DIMs. Moreover, there are also similar differences among ring-substituted DIMs which are dependent on the substituent and its position on indole ring.
2.3.2. PPARγ-active C-DIMs
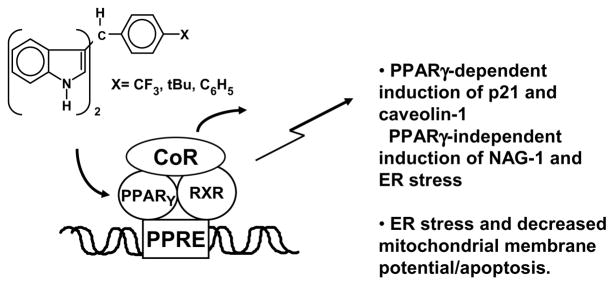
A second class of DIM derivatives was developed by condensing indole or substituted indoles with substituted benzaldehyde derivatives to give 1,1-bis(3′-indolyl)-1-(p-substituted phenyl)methanes (C-DIMs) [81,82,109–122]. These compounds are triarylmethane derivatives which differ from DIM and ring-substituted DIMs which are diarylmethanes. These compounds did not bind or activate the AhR, ER or AR; however, initial studies showed that some C-DIMs also inhibited carcinogen- induced rat mammary tumor growth and growth of various cancer cell lines [109–118]. Initial studies surveyed the activation of several orphan nuclear receptors by a series of C-DIMs containing various p-substituted phenyl groups, and the results showed that some analogs activated peroxisome proliferator-activated receptor γ (PPARγ) in breast cancer cells [109]. Subsequent studies showed that one or more of the three most active compounds, namely the p-trifluoromethyl (DIM-C-pPhCF3), p-t- butyl (DIM-C-pPhtBu), and p-phenyl (DIM-CpPhC6H5) analogs (Fig. 4) also activated PPARγ in colon, pancreatic, prostate, bladder, breast, endometrial and kidney cancer cell lines [109–113,121]. The PPARγ-active C-DIMs exhibit highly tissue-specific receptor-dependent activation of responses and genes. PPARγ-active C-DIMs induced differentiation in 3T3-L1 adipocyte cells characterized by an increase in lipid droplets and Oil red-O staining [109]. In addition, these compounds induced p21 gene expression in Panc28 pancreatic cancer cells and caveolin-1 in colon and bladder cancer cells that was PPARγ-dependent [110,111,113]. However, for most other responses, the C-DIM-induced proapoptotic and growth inhibitory effects were PPARγ independent. A direct comparison of the effects of DIM, 5,5′-dibromoDIM and DIM-C-pPhtBu was carried out in Panc1 and Panc28 pancreatic cancers, and all three compounds activated ER stress pathways leading to induction of CHOP and DR5 and the extrinsic apoptosis pathway [82]. PPARγ-active C-DIMs activated ER stress in other cancer cell lines [82,117,122], but not in breast cancer cells [81]. In contrast, DIM activated ER stress pathways in breast cancer cells. This demonstrates that DIM and C-DIMs exhibit cell context-dependent differences in their activation of ER stress in cancer cell lines.
Fig. 4.
PPARγ-active C-DIMs and their effects on cancer cells and tumors [109–118].
PPARγ-active C-DIMs also induce other proapoptotic responses that are both receptor- and ER stress-independent. For example, in SW480 colon cancer cells, DIM-C-pPhCF3 and DIM-C-pPhC6H5 do not induce ER stress and, in both SW480 and HCT116 cells, the C-DIM compounds induce expression of NAG-1 and activating transcription factor 3 (ATF3) which are proapoptotic genes and proteins [115,116]. DIM also induces NAG-1 and ATF3 in HCT116 cells; however, the mechanism of this response has not been reported. Induction of NAG-1 in HCT116 cells by PPARγ-active C-DIMs is dependent on PI3-K-dependent activation of early growth response-1 (Egr-1) gene which in turn activates NAG-1 through interactions with a proximal Egr-1 element in the NAG-1 promoter [115]. In contrast, induction of NAG-1 by DIM-C- pPhCF3 in LNCaP cells is MAPK-dependent[118] and suggests that induction of some proapoptotic genes such as NAG-1 are dependent on activation of kinase pathways by C-DIMs. However, the mechanisms of kinase activation have not been determined.
Both C-DIMs and DIM induce apoptosis in prostate cancer cells and, in LNCaP cells, DIM is an antiandrogen and blocks hormone-induced responses [118]. In contrast, C-DIM-dependent modifications of androgen responsiveness in LNCaP cells is completely different from the antiandrogenic activity of DIM. PPARγ-active C-DIMs decrease AR mRNA and protein levels and AR-promoter activity and these responses are PPARγ-independent. The effects on AR mRNA are not affected by cycloheximide, suggesting that C-DIM decreases AR mRNA stability or decreases promoter activity in a transcription-independent manner. C-DIMs also decrease prostate specific antigen (PSA) protein and mRNA and promoter activity; however, cycloheximide inhibits the effects of C-DIMs on PSA mRNA and this suggests induction of an inhibitory trans-acting factor. These C-DIM-induced effects on AR clearly distinguish between these compounds and DIM in androgen-sensitive prostate cancer cells. Current studies on C-DIMs are focused on determining their cell context- dependent mechanisms of action and on development of clinical applications for cancer chemotherapy.
2.3.3. Nur77-active D-CIMs
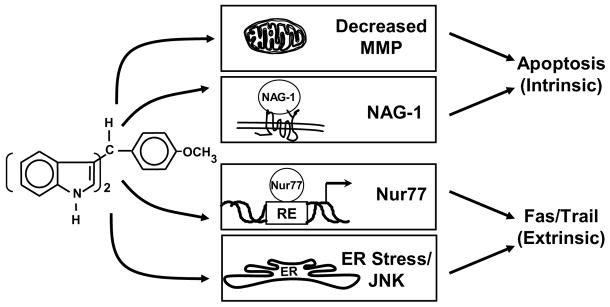
Another sub-class of C-DIMs containing para-methoxy (DIM-C-pPhOCH3) or hydrogen (DIMC-Ph) groups (Fig. 5) activate the orphan nuclear receptor nerve growth factor-induced Bα (NGFI-Bα) [125,126] often referred to as Nur77 [4]. The NGFI-B subfamily contains three members (Nur77, Nurr1 and Nor1) which were initially identified as immediate early genes induced by nerve growth factor in neuronal PC-12 cells [123].
Fig. 5.
Nur77-active D-DIMs and their effects on cancer cells and tumors [119,120].
Nur77 plays an important role in thymocyte-negative selection and in TCR-mediated apoptosis in thymocytes [124,125], and overexpression of Nur77 in transgenic mice resulted in high levels of apoptosis in thymocytes [126,127]. Several studies suggest that Nur77 plays a role in cell death pathways activated by apoptosis-inducing agents [128–134]. Li and coworkers [128] reported that treatment of LNCaP prostate cancer cells with several apoptosis-inducing agents, such as retinoids, TPA and TNF-α, resulted in induction of Nur77 gene expression. Surprisingly, induction of apoptosis and cytochrome c release from the mitochondria was independent of the DBD of Nur77. Treatment with leptomycin B (a blocker of nuclear export) inhibited induction of Nur77-dependent apoptosis. Using a series of wild-type and deletion GFP-Nur77 constructs, it was reported that induction of apoptosis was accompanied by translocation of Nur77 from the nucleus to the mitochondria. Moreover, Nur77 specifically interacted with Bcl-2 to form a proapoptotic complex in HEK293T and HCT116 cells [129]. Nur77 translocation from the nucleus has been observed in several cell lines after treatment with apoptosis inducers [128–130]. However, a study in colon cancer cells reported that butyrate-induced apoptosis was associated with nuclear to cytoplasmic translocation of Nur77 which was not accompanied by subsequent mitochondrial interactions [133]. Results with C-DIM compounds showed that DIM-C-pPhOCH3 and DIM-C-Ph activated nuclear Nur77 in pancreatic and colon cancer cells lines [119,120]. It was also reported that these same compounds activated a construct (pNuRE) containing a Nur responsive element (NuRE), and also activated a Gal4-Nur77(EF) construct containing the ligand binding domain of Nur77. We also demonstrated that these Nur77 agonists induced growth inhibition and apoptosis in pancreatic and colon cancer cells [119,120], and this was associated with induction of tumor necrosis factor-related apoptosis-inducing ligand (TRAIL) mRNA and protein. Moreover, these responses were partially reversed either by the Nur77 antagonist DIM-C- pPhOH or by transfecting small inhibitory RNA for Nur77 (iNur77). Microarray studies show that DIM-C-pPhOCH3 activates several proapoptotic genes in colon, pancreatic, prostate and bladder cancer cells through both Nur77-dependent and Nur77-independent pathways, and these are currently being investigated.
It is clear from studies on C-DIM compounds that DIM is an excellent scaffold from which new chemotherapeutic agents can be derived. A recent paper reported the synthesis and antitumorigenic activities of a series of substituted benzenesulfonyl or benzoyl I3C derivatives in which the indole nitrogen was derivatized [135]. Like I3C, these compounds were antitumorigenic and proapoptotic; however, they were up to 100 times more potent than I3C and were stable under acidic conditions. These I3C derivatives and the C-DIMs demonstrate that the chemoprotective and chemotherapeutic properties of I3C and DIM can be retained and enhanced in their synthetic analogs which are a promising class of anticancer drugs that activate multiple pathways in cancer cells and tumors but not in non-tumor tissues or organs.
Acknowledgments
The financial assistance of the National Institutes of Health (CA124998, CA108718 and CA112337) and the Texas Agricultural Experiment Station is gratefully acknowledged.
References
- 1.Hanahan D, Weinberg RA. The hallmarks of cancer. Cell. 2000;100:57–70. doi: 10.1016/s0092-8674(00)81683-9. [DOI] [PubMed] [Google Scholar]
- 2.Knudson AG., Jr Mutation and cancer: statistical study of retinoblastoma. Proc Natl Acad Sci USA. 1971;68:820–823. doi: 10.1073/pnas.68.4.820. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Hung HC, Joshipura KJ, Jiang R, Hu FB, Hunter D, Smith-Warner SA, Colditz GA, Rosner B, Spiegelman D, Willett WC. Fruit and vegetable intake and risk of major chronic disease. J Natl Cancer Inst. 2004;96:1577–1584. doi: 10.1093/jnci/djh296. [DOI] [PubMed] [Google Scholar]
- 4.Smith-Warner SA, Spiegelman D, Yaun SS, Adami HO, Beeson WL, Van den Brandt PA, Folsom AR, Fraser GE, Freudenheim JL, Goldbohm RA, Graham S, Miller AB, Potter JD, Rohan TE, Speizer FE, Toniolo P, Willett WC, Wolk A, Zeleniuch-Jacquotte A, Hunter DJ. Intake of fruits and vegetables and risk of breast cancer: a pooled analysis of cohort studies. JAMA. 2001;285:769–776. doi: 10.1001/jama.285.6.769. [DOI] [PubMed] [Google Scholar]
- 5.van Gils CH, Peeters PH, Bueno-de-Mesquita HB, Boshuizen HC, Lahmann PH, Clavel-Chapelon F, Thiebaut A, Kesse E, Sieri S, Palli D, Tumino R, Panico S, Vineis P, Gonzalez CA, Ardanaz E, Sanchez MJ, Amiano P, Navarro C, Quiros JR, Key TJ, Allen N, Khaw KT, Bingham SA, Psaltopoulou T, Koliva M, Trichopoulou A, Nagel G, Linseisen J, Boeing H, Berglund G, Wirfalt E, Hallmans G, Lenner P, Overvad K, Tjonneland A, Olsen A, Lund E, Engeset D, Alsaker E, Norat T, Kaaks R, Slimani N, Riboli E. Consumption of vegetables and fruits and risk of breast cancer. JAMA. 2005;293:183–193. doi: 10.1001/jama.293.2.183. [DOI] [PubMed] [Google Scholar]
- 6.Feskanich D, Ziegler RG, Michaud DS, Giovannucci EL, Speizer FE, Willett WC, Colditz GA. Prospective study of fruit and vegetable consumption and risk of lung cancer among men and women. J Natl Cancer Inst. 2000;92:1812–1823. doi: 10.1093/jnci/92.22.1812. [DOI] [PubMed] [Google Scholar]
- 7.Zeegers MP, Goldbohm RA, Van den Brandt PA. Consumption of vegetables and fruits and urothelial cancer incidence: a prospective study. Cancer Epidemiol Biomarkers Prev. 2001;10:1121–1128. [PubMed] [Google Scholar]
- 8.Zhang SM, Hunter DJ, Rosner BA, Giovannucci EL, Colditz GA, Speizer FE, Willett WC. Intakes of fruits, vegetables, and related nutrients and the risk of non-Hodgkin’s lymphoma among women. Cancer Epidemiol Biomarkers Prev. 2000;9:477–485. [PubMed] [Google Scholar]
- 9.Terry P, Giovannucci E, Michels KB, Bergkvist L, Hansen H, Holmberg L, Wolk A. Fruit, vegetables, dietary fiber, and risk of colorectal cancer. J Natl Cancer Inst. 2001;93:525–533. doi: 10.1093/jnci/93.7.525. [DOI] [PubMed] [Google Scholar]
- 10.Larsson SC, Hakansson N, Naslund I, Bergkvist L, Wolk A. Fruit and vegetable consumption in relation to pancreatic cancer risk: a prospective study. Cancer Epidemiol Biomarkers Prev. 2006;15:301–305. doi: 10.1158/1055-9965.EPI-05-0696. [DOI] [PubMed] [Google Scholar]
- 11.Michaud DS, Spiegelman D, Clinton SK, Rimm EB, Willett WC, Giovannucci EL. Fruit and vegetable intake and incidence of bladder cancer in a male prospective cohort. J Natl Cancer Inst. 1999;91:605–613. doi: 10.1093/jnci/91.7.605. [DOI] [PubMed] [Google Scholar]
- 12.Rashidkhani B, Lindblad P, Wolk A. Fruits, vegetables and risk of renal cell carcinoma: a prospective study of Swedish women. Int J Cancer. 2005;113:451–455. doi: 10.1002/ijc.20577. [DOI] [PubMed] [Google Scholar]
- 13.Kristal AR, Lampe JW. Brassica vegetables and prostate cancer risk: a review of the epidemiological evidence. Nutr Cancer. 2002;42:1–9. doi: 10.1207/S15327914NC421_1. [DOI] [PubMed] [Google Scholar]
- 14.Murillo G, Mehta RG. Cruciferous vegetables and cancer prevention. Nutr Cancer. 2001;41:17–28. doi: 10.1080/01635581.2001.9680607. [DOI] [PubMed] [Google Scholar]
- 15.Giovannucci E, Rimm EB, Liu Y, Stampfer MJ, Willett WC. A prospective study of cruciferous vegetables and prostate cancer. Cancer Epidemiol Biomarkers Prev. 2003;12:1403–1409. [PubMed] [Google Scholar]
- 16.Newman DJ, Cragg GM, Snader KM. Natural products as sources of new drugs over the period 1981–2002. J Nat Prod. 2003;66:1022–1037. doi: 10.1021/np030096l. [DOI] [PubMed] [Google Scholar]
- 17.Butler MS. The role of natural product chemistry in drug discovery. J Nat Prod. 2004;67:2141–2153. doi: 10.1021/np040106y. [DOI] [PubMed] [Google Scholar]
- 18.Koehn FE, Carter GT. The evolving role of natural products in drug discovery. Nat Rev Drug Discov. 2005;4:206–220. doi: 10.1038/nrd1657. [DOI] [PubMed] [Google Scholar]
- 19.Broadbent TA, Broadbent HS. The chemistry and pharmacology of indole-3-carbinol (indole-3-methanol) and 3-(methoxymethyl)indole [Part II] Curr Med Chem. 1998;5:469–491. [PubMed] [Google Scholar]
- 20.Preobrazhenskaya MN, Bukhman VM, Korolev AM, Efimov SA. Ascorbigen and other indole-derived compounds from Brassica vegetables and their analogs as anticarcinogenic and immunomodulating agents. Pharmacol Therap. 1993;60:301–313. doi: 10.1016/0163-7258(93)90012-3. [DOI] [PubMed] [Google Scholar]
- 21.Safe S, Chen I, Porter W. Dietary indoles with antiestrogenic activity in common vegetables and their implications. In: Pavlik E, editor. Estrogens, Progestins and their Antagonists. Birkhouser; Boston: 1996. pp. 73–97. [Google Scholar]
- 22.Verhoeven DT, Verhagen H, Goldbohm RA, Van den Brandt PA, van PG. A review of mechanisms underlying anticarcinogenicity by brassica vegetables. Chem Biol Interact. 1997;103:79–129. doi: 10.1016/s0009-2797(96)03745-3. [DOI] [PubMed] [Google Scholar]
- 23.Aggarwal BB, Ichikawa H. Molecular targets and anticancer potential of indole-3-carbinol and its derivatives. Cell Cycle. 2005;4:1201–1215. doi: 10.4161/cc.4.9.1993. [DOI] [PubMed] [Google Scholar]
- 24.Bjeldanes LF, Kim JY, Grose KR, Bartholomew JC, Bradfield CA. Aromatic hydrocarbon responsiveness-receptor agonists generated from indole-3-carbinol in vitro and in vivo – comparisons with 2,3,7,8-tetrachlorodibenzo-p-dioxin. Proc Natl Acad Sci USA. 1991;88:9543–9547. doi: 10.1073/pnas.88.21.9543. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Grose KR, Bjeldanes LF. Oligomerization of indole-3-carbinol in aqueous acid. Chem Res Toxicol. 1992;5:188– 193. doi: 10.1021/tx00026a007. [DOI] [PubMed] [Google Scholar]
- 26.Anderton MJ, Manson MM, Verschoyle RD, Gescher A, Lamb JH, Farmer PB, Steward WP, Williams ML. Pharmacokinetics and tissue disposition of indole-3-carbinol and its acid condensation products after oral administration to mice. Clin Cancer Res. 2004;10:5233–5241. doi: 10.1158/1078-0432.CCR-04-0163. [DOI] [PubMed] [Google Scholar]
- 27.Wattenberg LW, Loub WD. Inhibition of polycyclic hydrocarbon-induced neoplasia by naturally occurring indoles. Cancer Res. 1978;38:1410–1413. [PubMed] [Google Scholar]
- 28.Bradlow HL, Michnovicz JJ, Telang NT, Osborne MP. Effects of dietary indole-3-carbinol on estradiol metabolism and spontaneous mammary tumors in mice. Carcinogenesis. 1991;12:1571–1574. doi: 10.1093/carcin/12.9.1571. [DOI] [PubMed] [Google Scholar]
- 29.Morse MA, LaGreca SD, Amin SG, Chung FL. Effects of indole-3-carbinol on lung tumorigenesis and DNA methylation induced by 4-(methylnitrosamino)-1-(3-pyridl)-1-butanone (NNK) and on the metabolism and disposition of NNK in A/J mice. Cancer Res. 1990;50:1613–1617. [PubMed] [Google Scholar]
- 30.Tanaka T, Kojima T, Morishita Y, Mori H. Inhibitory effects of the natural products indole-3-carbinol and sinigrin during initiation and promotion phases of 4-nitroquinoline 1-oxide-induced rat tongue carcinogenesis. Jpn J Cancer Res. 1992;83:835–842. doi: 10.1111/j.1349-7006.1992.tb01988.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Tanaka T, Mori Y, Morishita Y, Hara A, Ohno T, Kojima T, Mori H. Inhibitory effect of sinigrin and indole-3-carbinol in diethylnitrosamine-induced hepatocarcinogenesis in male ACI/N rats. Carcinogenesis. 1990;11:1403– 1406. doi: 10.1093/carcin/11.8.1403. [DOI] [PubMed] [Google Scholar]
- 32.Grubbs CJ, Steele VE, Casebolt T, Juliana MM, Eto I, Whitaker LM, Dragnev KH, Kello GJ, Lubet RL. Chemoprevention of chemically induced mammary carcinogenesis by indole-3-carbinol. Anticancer Res. 1995;15:709–716. [PubMed] [Google Scholar]
- 33.Kim DJ, Lee KK, Han BS, Ahn B, Bae JH, Jang JJ. Biphasic modifying effect on indole-3-carbinol on diethylnitrosamine- induced preneoplastic glutathione S-transferase placental form-positive liver cell foci in Sprague–Dawley rats. Jpn J Cancer Res. 1994;85:578–583. doi: 10.1111/j.1349-7006.1994.tb02399.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Kojima T, Tanaka T, Mori H. Chemoprevention of spontaneous endometrial cancer in female Donryu rats by dietary indol-3-carbinol. Cancer Res. 1994;54:1446–1449. [PubMed] [Google Scholar]
- 35.Jin L, Qi M, Chen DZ, Anderson A, Yang GY, Arbeit JM, Auborn KJ. Indole-3-carbinol prevents cervical cancer in human papilloma virus type 16 (HPV16) transgenic mice. Cancer Res. 1999;59:3991–3997. [PubMed] [Google Scholar]
- 36.Oganesian A, Hendricks JD, Williams DE. Long term dietary indole-3-carbinol inhibits diethylnitrosamine-initiated hepatocarcinogenesis in the infant mouse model. Cancer Lett. 1997;118:87–94. doi: 10.1016/s0304-3835(97)00235-8. [DOI] [PubMed] [Google Scholar]
- 37.Blum CA, Xu M, Orner GA, Fong AT, Bailey GS, Stoner GD, Horio DT, Dashwood RH. beta-Catenin mutation in rat colon tumors initiated by 1,2-dimethylhydrazine and 2-amino-3-methylimidazo[4,5-f]quinoline, and the effect of post-initiation treatment with chlorophyllin and indole-3-carbinol. Carcinogenesis. 2001;22:315–320. doi: 10.1093/carcin/22.2.315. [DOI] [PubMed] [Google Scholar]
- 38.Michnovicz JJ, Bradlow HL. Induction of estradiol metabolism by dietary indole-3-carbinol in humans. J Natl Cancer Inst. 1990;82:947–949. doi: 10.1093/jnci/82.11.947. [DOI] [PubMed] [Google Scholar]
- 39.Wortelboer HM, De Kruif CA, Van Iersel AJ, Falke HE, Noordhoek J, Blaauboer BJ. Acid reaction products of indole-3-carbinol and their effects on cytochrome P450 and phase II enzymes in rat and monkey hepatocytes. Biochem Pharmacol. 1992;43:1439–1447. doi: 10.1016/0006-2952(92)90200-3. [DOI] [PubMed] [Google Scholar]
- 40.Niwa T, Swaneck G, Bradlow HL. Alterations in estradiol metabolism in MCF-7 cells induced by treatment with indole-3-carbinol and related compounds. Steroids. 1994;59:523–527. doi: 10.1016/0039-128x(94)90070-1. [DOI] [PubMed] [Google Scholar]
- 41.Jellinck PH, Michnovicz JJ, Bradlow HL. Influence of indole-3-carbinol on the hepatic microsomal formation of catechol estrogens. Steroids. 1991;56:446–450. doi: 10.1016/0039-128x(91)90034-s. [DOI] [PubMed] [Google Scholar]
- 42.Zhang J, Hsu JC, Kinseth MA, Bjeldanes LF, Firestone GL. Indole-3-carbinol induces a G1 cell cycle arrest and inhibits prostate-specific antigen production in human LNCaP prostate carcinoma cells. Cancer. 2003;98:2511– 2520. doi: 10.1002/cncr.11844. [DOI] [PubMed] [Google Scholar]
- 43.Matsuzaki Y, Koyama M, Hitomi T, Kawanaka M, Sakai T. Indole-3-carbinol activates the cyclin-dependent kinase inhibitor p15(INK4b) gene. FEBS Lett. 2004;576:137–140. doi: 10.1016/j.febslet.2004.09.002. [DOI] [PubMed] [Google Scholar]
- 44.Garcia HH, Brar GA, Nguyen DH, Bjeldanes LF, Firestone GL. Indole-3-carbinol (I3C) inhibits cyclin-dependent kinase-2 function in human breast cancer cells by regulating the size distribution, associated cyclin E forms, and subcellular localization of the CDK2 protein complex. J Biol Chem. 2005;280:8756–8764. doi: 10.1074/jbc.M407957200. [DOI] [PubMed] [Google Scholar]
- 45.Cram EJ, Liu BD, Bjeldanes LF, Firestone GL. Indole-3-carbinol inhibits CDK6 expression in human MCF-7 breast cancer cells by disrupting Sp1 transcription factor interactions with a composite element in the CDK6 gene promoter. J Biol Chem. 2001;276:22332– 22340. doi: 10.1074/jbc.M010539200. [DOI] [PubMed] [Google Scholar]
- 46.Rahman KM, Sarkar FH, Banerjee S, Wang Z, Liao DJ, Hong X, Sarkar NH. Therapeutic intervention of experimental breast cancer bone metastasis by indole-3-carbinol in SCID-human mouse model. Mol Cancer Ther. 2006;5:2747–2756. doi: 10.1158/1535-7163.MCT-06-0221. [DOI] [PubMed] [Google Scholar]
- 47.Cover CM, Hsieh SJ, Tran SH, Hallden G, Kim GS, Bjeldanes LF, Firestone GL. Indole-3-carbinol inhibits the expression of cyclin-dependent kinase-6 and induces a G1 cell cycle arrest of human breast cancer cells independent of estrogen receptor signaling. J Biol Chem. 1998;273:3838–3847. doi: 10.1074/jbc.273.7.3838. [DOI] [PubMed] [Google Scholar]
- 48.Chinni SR, Li Y, Upadhyay S, Koppolu PK, Sarkar FH. Indole-3-carbinol (I3C) induced cell growth inhibition, G1 cell cycle arrest and apoptosis in prostate cancer cells. Oncogene. 2001;20:2927–2936. doi: 10.1038/sj.onc.1204365. [DOI] [PubMed] [Google Scholar]
- 49.Frydoonfar HR, McGrath DR, Spigelman AD. Inhibition of proliferation of a colon cancer cell line by indole-3-carbinol. Colorectal Dis. 2002;4:205–207. doi: 10.1046/j.1463-1318.2002.00346.x. [DOI] [PubMed] [Google Scholar]
- 50.Rahman KM, Aranha O, Sarkar FH. Indole-3-carbinol (I3C) induces apoptosis in tumorigenic but not in non-tumorigenic breast epithelial cells. Nutr Cancer. 2003;45:101–112. doi: 10.1207/S15327914NC4501_12. [DOI] [PubMed] [Google Scholar]
- 51.Howells LM, Gallacher-Horley B, Houghton CE, Manson MM, Hudson EA. Indole-3-carbinol inhibits protein kinase B/Akt and induces apoptosis in the human breast tumor cell line MDA MB468 but not in the non-tumorigenic HBL100 line. Mol Cancer Ther. 2002;1:1161–1172. [PubMed] [Google Scholar]
- 52.Zheng Q, Hirose Y, Yoshimi N, Murakami A, Koshimizu K, Ohigashi H, Sakata K, Matsumoto Y, Sayama Y, Mori H. Further investigation of the modifying effect of various chemopreventive agents on apoptosis and cell proliferation in human colon cancer cells. J Cancer Res Clin Oncol. 2002;128:539–546. doi: 10.1007/s00432-002-0373-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Nachshon-Kedmi M, Yannai S, Haj A, Fares FA. Indole-3-carbinol and 3,3′-diindolylmethane induce apoptosis in human prostate cancer cells. Food Chem Toxicol. 2003;41:745–752. doi: 10.1016/s0278-6915(03)00004-8. [DOI] [PubMed] [Google Scholar]
- 54.Bonnesen C, Eggleston IM, Hayes JD. Dietary indoles and isothiocyanates that are generated from cruciferous vegetables can both stimulate apoptosis and confer protection against DNA damage in human colon cell lines. Cancer Res. 2001;61:6120–6130. [PubMed] [Google Scholar]
- 55.Chen DZ, Qi M, Auborn KJ, Carter TH. Indole-3-carbinol and diindolylmethane induce apoptosis of human cervical cancer cells and in murine HPV16-transgenic preneoplastic cervical epithelium. J Nutr. 2001;131:3294–3302. doi: 10.1093/jn/131.12.3294. [DOI] [PubMed] [Google Scholar]
- 56.Ge X, Fares FA, Yannai S. Induction of apoptosis in MCF-7 cells by indole-3-carbinol is independent of p53 and bax. Anticancer Res. 1999;19:3199–3203. [PubMed] [Google Scholar]
- 57.Rahman KM, Aranha O, Glazyrin A, Chinni SR, Sarkar FH. Translocation of Bax to mitochondria induces apoptotic cell death in indole-3-carbinol (I3C) treated breast cancer cells. Oncogene. 2000;19:5764–5771. doi: 10.1038/sj.onc.1203959. [DOI] [PubMed] [Google Scholar]
- 58.Jeon KI, Rih JK, Kim HJ, Lee YJ, Cho CH, Goldberg ID, Rosen EM, Bae I. Pretreatment of indole-3-carbinol augments TRAIL-induced apoptosis in a prostate cancer cell line, LNCaP. FEBS Lett. 2003;544:246–251. doi: 10.1016/s0014-5793(03)00473-3. [DOI] [PubMed] [Google Scholar]
- 59.Chinni SR, Sarkar FH. Akt inactivation is a key event in indole-3-carbinol-induced apoptosis in PC-3 cells. Clin Cancer Res. 2002;8:1228–1236. [PubMed] [Google Scholar]
- 60.Takada Y, Andree M, Aggarwal BB. Indole-3-carbinol suppresses NF-kappaB and IkappaBalpha kinase activation, causing inhibition of expression of NF-kappaB-regulated antiapoptotic and metastatic gene products and enhancement of apoptosis in myeloid and leukemia cells. Blood. 2005;106:641–649. doi: 10.1182/blood-2004-12-4589. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Ashok BT, Chen Y, Liu X, Bradlow HL, Mittelman A, Tiwari RK. Abrogation of estrogen-mediated cellular and biochemical effects by indole-3-carbinol. Nutr Cancer. 2001;41:180–187. doi: 10.1080/01635581.2001.9680630. [DOI] [PubMed] [Google Scholar]
- 62.Ashok BT, Chen YG, Liu X, Garikapaty VP, Seplowitz R, Tschorn J, Roy K, Mittelman A, Tiwari RK. Multiple molecular targets of indole-3-carbinol, a chemopreventive anti-estrogen in breast cancer. Eur J Cancer Prev. 2002;11(Suppl 2):S86–S93. [PubMed] [Google Scholar]
- 63.Yuan F, Chen DZ, Liu K, Sepkovic DW, Bradlow HL, Auborn K. Anti-estrogenic activities of indole-3-carbinol in cervical cells: implication for prevention of cervical cancer. Anticancer Res. 1999;19:1673–1680. [PubMed] [Google Scholar]
- 64.Telang NT, Katdare M, Bradlow HL, Osborne MP, Fishman J. Inhibition of proliferation and modulation of estradiol metabolism: novel mechanisms for breast cancer prevention by the phytochemical indole-3-carbinol. Proc Soc Exp Biol Med. 1997;216:246–252. doi: 10.3181/00379727-216-44174. [DOI] [PubMed] [Google Scholar]
- 65.Meng Q, Yuan F, Goldberg ID, Rosen EM, Auborn K, Fan S. Indole-3-carbinol is a negative regulator of estrogen receptor-alpha signaling in human tumor cells. J Nutr. 2000;130:2927–2931. doi: 10.1093/jn/130.12.2927. [DOI] [PubMed] [Google Scholar]
- 66.Meng Q, Qi M, Chen DZ, Yuan R, Goldberg ID, Rosen EM, Auborn K, Fan S. Suppression of breast cancer invasion and migration by indole-3-carbinol: associated with up-regulation of BRCA1 and E-cadherin/catenin complexes. J Mol Med. 2000;78:155–165. doi: 10.1007/s001090000088. [DOI] [PubMed] [Google Scholar]
- 67.Fan S, Meng Q, Auborn K, Carter T, Rosen EM. BRCA1 and BRCA2 as molecular targets for phytochemicals indole-3-carbinol and genistein in breast and prostate cancer cells. Br J Cancer. 2006;94:407–426. doi: 10.1038/sj.bjc.6602935. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Sundar SN, Kerekatte V, Equinozio CN, Doan VB, Bjeldanes LF, Firestone GL. Indole-3-carbinol selectively uncouples expression and activity of estrogen receptor subtypes in human breast cancer cells. Mol Endocrinol. 2006;20:3070–3082. doi: 10.1210/me.2005-0263. [DOI] [PubMed] [Google Scholar]
- 69.Hsu JC, Zhang J, Dev A, Wing A, Bjeldanes LF, Firestone GL. Indole-3-carbinol inhibition of androgen receptor expression and downregulation of androgen responsiveness in human prostate cancer cells. Carcinogenesis. 2005;26:1896–1904. doi: 10.1093/carcin/bgi155. [DOI] [PubMed] [Google Scholar]
- 70.Hsu JC, Dev A, Wing A, Brew CT, Bjeldanes LF, Firestone GL. Indole-3-carbinol mediated cell cycle arrest of LNCaP human prostate cancer cells requires the induced production of activated p53 tumor suppressor protein. Biochem Pharmacol. 2006;72:1714–1723. doi: 10.1016/j.bcp.2006.08.012. [DOI] [PubMed] [Google Scholar]
- 71.Chatterji U, Riby JE, Taniguchi T, Bjeldanes EL, Bjeldanes LF, Firestone GL. Indole-3-carbinol stimulates transcription of the interferon gamma receptor 1 gene and augments interferon responsiveness in human breast cancer cells. Carcinogenesis. 2004;25:1119–1128. doi: 10.1093/carcin/bgh121. [DOI] [PubMed] [Google Scholar]
- 72.Lee SH, Kim JS, Yamaguchi K, Eling TE, Baek SJ. Indole-3-carbinol and 3,3′-diindolylmethane induce expression of NAG-1 in a p53-independent manner. Biochem Biophys Res Commun. 2005;328:63–69. doi: 10.1016/j.bbrc.2004.12.138. [DOI] [PubMed] [Google Scholar]
- 73.Rahman KM, Li Y, Sarkar FH. Inactivation of akt and NF-kappaB play important roles during indole-3-carbinol-induced apoptosis in breast cancer cells. Nutr Cancer. 2004;48:84–94. doi: 10.1207/s15327914nc4801_12. [DOI] [PubMed] [Google Scholar]
- 74.Traber PG, Chianale J, Florence R, Kim K, Wojcik E, Gumucio JJ. Expression of cytochrome P450b and P450e genes in small intestinal mucosa of rats following treatment with phenobarbital, polyhalogenated biphenyls, and organochlorine pesticides. J Biol Chem. 1988;263:9449–9455. [PubMed] [Google Scholar]
- 75.Chen I, Safe S, Bjeldanes L. Indole-3-carbinol and diindolylmethane as aryl hydrocarbon (Ah) receptor agonists and antagonists in T47D human breast cancer cells. Biochem Pharmacol. 1996;51:1069–1076. doi: 10.1016/0006-2952(96)00060-3. [DOI] [PubMed] [Google Scholar]
- 76.Chang X, Tou JC, Hong C, Kim HA, Riby JE, Firestone GL, Bjeldanes LF. 3,3′-Diindolylmethane inhibits angiogenesis and the growth of transplantable human breast carcinoma in athymic mice. Carcinogenesis. 2005;26:771–778. doi: 10.1093/carcin/bgi018. [DOI] [PubMed] [Google Scholar]
- 77.Chen I, McDougal A, Wang F, Safe S. Aryl hydrocarbon receptor-mediated antiestrogenic and antitumorigenic activity of diindolylmethane. Carcinogenesis. 1998;19:1631–1639. doi: 10.1093/carcin/19.9.1631. [DOI] [PubMed] [Google Scholar]
- 78.Rahman KW, Sarkar FH. Inhibition of nuclear translocation of nuclear factor-κB contributes to 3,3′-diindolylmethane-induced apoptosis in breast cancer cells. Cancer Res. 2005;65:364–371. [PubMed] [Google Scholar]
- 79.Riby JE, Chang GH, Firestone GL, Bjeldanes LF. Ligand-independent activation of estrogen receptor function by 3,3′-diindolylmethane in human breast cancer cells. Biochem Pharmacol. 2000;60:167–177. doi: 10.1016/s0006-2952(00)00307-5. [DOI] [PubMed] [Google Scholar]
- 80.Sun S, Han J, Ralph WM, Jr, Chandrasekaran A, Liu K, Auborn KJ, Carter TH. Endoplasmic reticulum stress as a correlate of cytotoxicity in human tumor cells exposed to diindolylmethane in vitro. Cell Stress Chaperones. 2004;9:76–87. doi: 10.1379/CSC-2R.1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Vanderlaag K, Samudio I, Burghardt R, Barhoumi R, Safe S. Inhibition of breast cancer cell growth and induction of cell death by 1,1-bis(3′-indolyl)methane (DIM) and 5,5′-dibromoDIM. Cancer Lett. 2005;236:198–212. doi: 10.1016/j.canlet.2005.05.036. [DOI] [PubMed] [Google Scholar]
- 82.Abdelrahim M, Newman K, Vanderlaag K, Samudio I, Safe S. 3,3′-Diindolylmethane (DIM) and derivatives induce apoptosis in pancreatic cancer cells through endoplasmic reticulum stress-dependent upregulation of DR5. Carcinogenesis. 2006;27:717–728. doi: 10.1093/carcin/bgi270. [DOI] [PubMed] [Google Scholar]
- 83.Leong H, Firestone GL, Bjeldanes LF. Cytostatic effects of 3,3′-diindolylmethane in human endometrial cancer cells result from an estrogen receptor-mediated increase in transforming growth factor-alpha expression. Carcinogenesis. 2001;22:1809–1817. doi: 10.1093/carcin/22.11.1809. [DOI] [PubMed] [Google Scholar]
- 84.Hong C, Firestone GL, Bjeldanes LF. Bcl-2 family-mediated apoptotic effects of 3,3′-diindolylmethane (DIM) in human breast cancer cells. Biochem Pharmacol. 2002;63:1085–1097. doi: 10.1016/s0006-2952(02)00856-0. [DOI] [PubMed] [Google Scholar]
- 85.Hong C, Kim HA, Firestone GL, Bjeldanes LF. 3,3′-Diindolylmethane (DIM) induces a G1 cell cycle arrest in human breast cancer cells that is accompanied by Sp1- mediated activation of p21WAF1/CIP1 expression. Carcinogenesis. 2002;23:1297–1305. doi: 10.1093/carcin/23.8.1297. [DOI] [PubMed] [Google Scholar]
- 86.Gong Y, Sohn H, Xue L, Firestone GL, Bjeldanes LF. 3,3′-Diindolylmethane is a novel mitochondrial H+-ATP synthase inhibitor that can induce p21Cip1/Waf1 expression by induction of oxidative stress in human breast cancer cells. Cancer Res. 2006;66:4880–4887. doi: 10.1158/0008-5472.CAN-05-4162. [DOI] [PubMed] [Google Scholar]
- 87.Xue L, Firestone GL, Bjeldanes LF. DIM stimulates IFNγ gene expression in human breast cancer cells via the specific activation of JNK and p38 pathways. Oncogene. 2005;24:2343–2353. doi: 10.1038/sj.onc.1208434. [DOI] [PubMed] [Google Scholar]
- 88.Rahman KW, Li Y, Wang Z, Sarkar SH, Sarkar FH. Gene expression profiling revealed survivin as a target of 3,3′-diindolylmethane-induced cell growth inhibition and apoptosis in breast cancer cells. Cancer Res. 2006;66:4952–4960. doi: 10.1158/0008-5472.CAN-05-3918. [DOI] [PubMed] [Google Scholar]
- 89.Garikapaty VP, Ashok BT, Tadi K, Mittelman A, Tiwari RK. 3,3′-Diindolylmethane downregulates prosurvival pathway in hormone independent prostate cancer. Biochem Biophys Res Commun. 2006;340:718–725. doi: 10.1016/j.bbrc.2005.12.059. [DOI] [PubMed] [Google Scholar]
- 90.Nachshon-Kedmi M, Fares FA, Yannai S. Therapeutic activity of 3,3′-diindolylmethane on prostate cancer in an in vivo model. Prostate. 2004;61:153–160. doi: 10.1002/pros.20092. [DOI] [PubMed] [Google Scholar]
- 91.Kong D, Li Y, Wang Z, Banerjee S, Sarkar FH. Inhibition of angiogenesis and invasion by 3,3′-diindolylmethane is mediated by the NF-κB downstream target genes MMP-9 and uPA that regulated bioavailability of vascular endothelial growth factor in prostate cancer. Cancer Res. 2007;67:3310–3319. doi: 10.1158/0008-5472.CAN-06-4277. [DOI] [PubMed] [Google Scholar]
- 92.Savino JA, III, Evans JF, Rabinowitz D, Auborn KJ, Carter TH. Multiple, disparate roles for calcium signaling in apoptosis of human prostate and cervical cancer cells exposed to diindolylmethane. Mol Cancer Ther. 2006;5:556–563. doi: 10.1158/1535-7163.MCT-05-0355. [DOI] [PubMed] [Google Scholar]
- 93.Nachshon-Kedmi M, Yannai S, Fares FA. Induction of apoptosis in human prostate cancer cell line, PC3, by 3,3′-diindolylmethane through the mitochondrial pathway. Br J Cancer. 2004;91:1358–1363. doi: 10.1038/sj.bjc.6602145. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Kim EJ, Park SY, Shin HK, Kwon DY, Surh YJ, Park JH. Activation of caspase-8 contributes to 3,3′-diindolylmethane-induced apoptosis in colon cancer cells. J Nutr. 2007;137:31–36. doi: 10.1093/jn/137.1.31. [DOI] [PubMed] [Google Scholar]
- 95.Gong Y, Firestone GL, Bjeldanes LF. 3,3′-Diindolylmethane is a novel topoisomerase IIα catalytic inhibitor that induces S-phase retardation and mitotic delay in human hepatoma HepG2 cells. Mol Pharmacol. 2006;69:1320– 1327. doi: 10.1124/mol.105.018978. [DOI] [PubMed] [Google Scholar]
- 96.Safe S, Wormke M. Inhibitory aryl hydrocarbon–estrogen receptor α cross-talk and mechanisms of action. Chem Res Toxicol. 2003;16:807–816. doi: 10.1021/tx034036r. [DOI] [PubMed] [Google Scholar]
- 97.McDougal A, Wormke M, Calvin J, Safe S. Tamoxifen-induced antitumorigenic/antiestrogenic action synergized by a selective Ah receptor modulator. Cancer Res. 2001;61:3901–3907. [PubMed] [Google Scholar]
- 98.Safe S, McDougal A, Gupta MS, Ramamoorthy K. Selective Ah receptor modulators (SAhRMs): progress towards development of a new class of inhibitors of breast cancer growth. J Women’s Cancer. 2001;3:37–45. [Google Scholar]
- 99.Abdelrahim M, Smith R, III, Safe S. Aryl hydrocarbon receptor gene silencing with small inhibitory RNA differentially modulates Ah-responsiveness in MCF-7 and HepG2 cancer cells. Mol Pharmacol. 2003;63:1373–1381. doi: 10.1124/mol.63.6.1373. [DOI] [PubMed] [Google Scholar]
- 100.Shilling AD, Carlson DB, Katchamart S, Williams DE. 3,3′-Diindolylmethane, a major condensation product of indole-3-carbinol, is a potent estrogen in the rainbow trout. Toxicol Appl Pharmacol. 2001;170:191–200. doi: 10.1006/taap.2000.9100. [DOI] [PubMed] [Google Scholar]
- 101.Leong H, Riby JE, Firestone GL, Bjeldanes LF. Potent ligand-independent estrogen receptor activation by 3,3′-diindolylmethane is mediated by cross talk between the protein kinase A and mitogen-activated protein kinase signaling pathways. Mol Endocrinol. 2004;18:291–302. doi: 10.1210/me.2003-0196. [DOI] [PubMed] [Google Scholar]
- 102.Pearce ST, Liu H, Radhakrishnan I, Abdelrahim M, Safe S, Jordan VC. Interaction of the aryl hydrocarbon receptor ligand 6-methyl-1,3,8-trichlorodibenzofuran (MCDF) with estrogen receptor α. Cancer Res. 2004;64:2889–2897. doi: 10.1158/0008-5472.can-03-1770. [DOI] [PubMed] [Google Scholar]
- 103.Abdelrahim M, Ariazi E, Kim K, Khan S, Barhoumi R, Burghardt R, Liu S, Hill D, Finnell RH, Wlodarczyk B, Jordan VC, Safe S. 3-Methylcholanthrene and other aryl hydrocarbon receptor agonists directly activate estrogen receptor α. Cancer Res. 2006;66:2459–2467. doi: 10.1158/0008-5472.CAN-05-3132. [DOI] [PubMed] [Google Scholar]
- 104.Liu S, Abdelrahim M, Khan S, Ariazi E, Jordan VC, Safe S. Aryl hydrocarbon receptor agonists directly activate estrogen receptor α in MCF-7 breast cancer cells. Biol Chem. 2006;387:1209–1213. doi: 10.1515/BC.2006.149. [DOI] [PubMed] [Google Scholar]
- 105.Liu Y, Zhong X, Li W, Brattain MG, Banerji SS. The role of Sp1 in the differential expression of transforming growth factor-β receptor type II in human breast adenocarcinoma MCF-7 cells. J Biol Chem. 2000;275:12231–12236. doi: 10.1074/jbc.275.16.12231. [DOI] [PubMed] [Google Scholar]
- 106.Le HT, Schaldach CM, Firestone GL, Bjeldanes LF. Plant-derived 3,3′-diindolylmethane is a strong androgen antagonist in human prostate cancer cells. J Biol Chem. 2003;278:21136–21145. doi: 10.1074/jbc.M300588200. [DOI] [PubMed] [Google Scholar]
- 107.McDougal A, Sethi-Gupta M, Ramamoorthy K, Sun G, Safe S. Inhibition of carcinogen-induced rat mammary tumor growth and other estrogen-dependent responses by symmetrical dihalo-substituted analogs of diindolylmethane. Cancer Lett. 2000;151:169–179. doi: 10.1016/s0304-3835(99)00406-1. [DOI] [PubMed] [Google Scholar]
- 108.McDougal A, Gupta MS, Morrow D, Ramamoorthy K, Lee YE, Safe S. Methyl-substituted diindolylmethanes as inhibitors of estrogen-induced growth of T47D cells and mammary tumors in rats. Breast Cancer Res Treat. 2001;66:147–157. doi: 10.1023/a:1010608000074. [DOI] [PubMed] [Google Scholar]
- 109.Qin C, Morrow D, Stewart J, Spencer K, Porter W, Smith R, III, Phillips T, Abdelrahim M, Samudio I, Safe S. A new class of peroxisome proliferator-activated receptor γ (PPARγ) agonists that inhibit growth of breast cancer cells: 1,1-bis(3′-indolyl)-1-(p-substituted phenyl)methanes. Mol Cancer Therap. 2004;3:247–259. [PubMed] [Google Scholar]
- 110.Hong J, Samudio I, Liu S, Abdelrahim M, Safe S. Peroxisome proliferator-activated receptor γ-dependent activation of p21 in Panc-28 pancreatic cancer cells involves Sp1 and Sp4 proteins. Endocrinology. 2004;145:5774– 5785. doi: 10.1210/en.2004-0686. [DOI] [PubMed] [Google Scholar]
- 111.Chintharlapalli S, Smith R, III, Samudio I, Zhang W, Safe S. 1,1-Bis(3′-indolyl)-1-(p-substituted phenyl)methanes induce peroxisome proliferator-activated receptor γ-mediated growth inhibition, transactivation and differentiation markers in colon cancer cells. Cancer Res. 2004;64:5994– 6001. doi: 10.1158/0008-5472.CAN-04-0399. [DOI] [PubMed] [Google Scholar]
- 112.Contractor R, Samudio I, Estrov Z, Harris D, McCubrey JA, Safe S, Andree M, Konopleva M. A novel ring-substituted diindolylmethane 1,1-bis[3′-(5-methoxyindolyl)]-1-(p-t-butylphenyl)methane inhibits ERK activation and induces apoptosis in acute myeloid leukemia. Cancer Res. 2005;65:2890–2898. doi: 10.1158/0008-5472.CAN-04-3781. [DOI] [PubMed] [Google Scholar]
- 113.Kassouf W, Chintharlapalli S, Abdelrahim M, Nelkin G, Safe S, Kamat AM. Inhibition of bladder tumor growth by 1,1-bis(3′-indolyl)-1-(p-substituted phenyl)methanes: a new class of peroxisome proliferator-activated receptor γ agonists. Cancer Res. 2006;66:412–418. doi: 10.1158/0008-5472.CAN-05-2755. [DOI] [PubMed] [Google Scholar]
- 114.Calabro P, Samudio I, Safe SH, Willerson JT, Yeh ET. Inhibition of tumor necrosis factor-α-induced endothelial cell activation by a new class of PPARγ agonists: an in vitro study. J Vascular Res. 2005;42:509–516. doi: 10.1159/000088260. [DOI] [PubMed] [Google Scholar]
- 115.Chintharlapalli S, Papineni S, Baek SJ, Liu S, Safe S. 1,1-Bis(3′-indolyl)-1-(p-substituted phenyl)methanes are peroxisome proliferator-activated receptor gamma agonists but decrease HCT-116 colon cancer cell survival through receptor-independent activation of early growth response-1 and NAG-1. Mol Pharmacol. 2005;68:1782– 1792. doi: 10.1124/mol.105.017046. [DOI] [PubMed] [Google Scholar]
- 116.Chintharlapalli S, Papineni S, Safe S. 1,1-Bis(3′-indolyl)- 1-(p-substituted phenyl)methanes inhibit colon cancer cell and tumor growth through PPARγ-dependent and PPARγ independent pathways. Mol Cancer Ther. 2006;5:1362– 1370. doi: 10.1158/1535-7163.MCT-06-0002. [DOI] [PubMed] [Google Scholar]
- 117.Lei P, Abdelrahim M, Safe S. 1,1-Bis(3′-indolyl)-1- (p-substituted phenyl)methanes inhibit ovarian cancer cell growth through peroxisome proliferator-activated receptor-dependent and -independent pathways. Mol Cancer Ther. 2006;5:2324–2336. doi: 10.1158/1535-7163.MCT-06-0184. [DOI] [PubMed] [Google Scholar]
- 118.Chintharlapalli S, Papineni S, Safe SH. 1,1-Bis(3′-indolyl)- 1-(p-substituted phenyl)methanes inhibit growth, induce apoptosis, and decrease the androgen receptor in LNCaP prostate cancer cells through PPARγ-independent pathways. Mol Pharmacol. 2007;71:558–569. doi: 10.1124/mol.106.028696. [DOI] [PubMed] [Google Scholar]
- 119.Chintharlapalli S, Burghardt R, Papineni S, Ramaiah S, Yoon K, Safe S. Activation of Nur77 by selected 1,1- bis(3′-indolyl)-1-(p-substituted phenyl)methanes induces apoptosis through nuclear pathways. J Biol Chem. 2005;280:24903–24914. doi: 10.1074/jbc.M500107200. [DOI] [PubMed] [Google Scholar]
- 120.Cho SD, Yoon K, Chintharlapalli S, Abdelrahim M, Pei P, Hamilton S, Khan S, Ramaiah SK, Safe S. Nur77 agonists induce proapoptotic genes and responses in colon cancer cells through nuclear receptor-dependent and - independent pathways. Cancer Res. 2007;67:674–683. doi: 10.1158/0008-5472.CAN-06-2907. [DOI] [PubMed] [Google Scholar]
- 121.York M, Abdelrahim M, Chintharlapalli S, Lucero SD, Safe S. 1,1-Bis(3′-indolyl)-1-(p-substituted phenyl)methanes induce apoptosis and inhibit renal cell carcinoma growth. Clin Cancer Res. 2007;13:6743–6752. doi: 10.1158/1078-0432.CCR-07-0967. [DOI] [PubMed] [Google Scholar]
- 122.Vanderlaag K, Su Y, Frankel AE, Grage H, Smith R, III, Khan S, Safe S. 1,1-Bis(3′-indolyl)-1-(p-substituted phenyl)methanes inhibit proliferation of estrogen receptor-negative breast cancer cells by activation of multiple pathways. Breast Cancer Res Treat. 2007 doi: 10.1007/s10549-007-9648-y. [DOI] [PubMed] [Google Scholar]
- 123.Ohkura N, Hijikuro M, Yamamoto A, Miki K. Molecular cloning of a novel thyroid/steroid receptor superfamily gene from cultured rat neuronal cells. Biochem Biophys Res Commun. 1994;205:1959–1965. doi: 10.1006/bbrc.1994.2900. [DOI] [PubMed] [Google Scholar]
- 124.Winoto A. Genes involved in T-cell receptor-mediated apoptosis of thymocytes and T-cell hybridomas. Semin Immunol. 1997;9:51–58. doi: 10.1006/smim.1996.0053. [DOI] [PubMed] [Google Scholar]
- 125.He YW. Orphan nuclear receptors in T lymphocyte development. J Leukoc Biol. 2002;72:440–446. [PubMed] [Google Scholar]
- 126.Cheng LE, Chan FK, Cado D, Winoto A. Functional redundancy of the Nur77 and Nor-1 orphan steroid receptors in T-cell apoptosis. EMBO J. 1997;16:1865– 1875. doi: 10.1093/emboj/16.8.1865. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 127.Calnan BJ, Szychowski S, Chan FK, Cado D, Winoto A. A role for the orphan steroid receptor Nur77 in apoptosis accompanying antigen-induced negative selection. Immunity. 1995;3:273–282. doi: 10.1016/1074-7613(95)90113-2. [DOI] [PubMed] [Google Scholar]
- 128.Li H, Kolluri SK, Gu J, Dawson MI, Cao X, Hobbs PD, Lin B, Chen G, Lu J, Lin F, Xie Z, Fontana JA, Reed JC, Zhang X. Cytochrome c release and apoptosis induced by mitochondrial targeting of nuclear orphan receptor TR3. Science. 2000;289:1159–1164. doi: 10.1126/science.289.5482.1159. [DOI] [PubMed] [Google Scholar]
- 129.Lin B, Kolluri SK, Lin F, Liu W, Han YH, Cao X, Dawson MI, Reed JC, Zhang XK. Conversion of Bcl-2 from protector to killer by interaction with nuclear orphan receptor Nur77/TR3. Cell. 2004;116:527–540. doi: 10.1016/s0092-8674(04)00162-x. [DOI] [PubMed] [Google Scholar]
- 130.Wu Q, Liu S, Ye XF, Huang ZW, Su WJ. Dual roles of Nur77 in selective regulation of apoptosis and cell cycle by TPA and ATRA in gastric cancer cells. Carcinogenesis. 2002;23:1583–1592. doi: 10.1093/carcin/23.10.1583. [DOI] [PubMed] [Google Scholar]
- 131.Holmes WF, Soprano DR, Soprano KJ. Early events in the induction of apoptosis in ovarian carcinoma cells by CD437: activation of the p38 MAP kinase signal pathway. Oncogene. 2003;22:6377–6386. doi: 10.1038/sj.onc.1206694. [DOI] [PubMed] [Google Scholar]
- 132.Holmes WF, Soprano DR, Soprano KJ. Comparison of the mechanism of induction of apoptosis in ovarian carcinoma cells by the conformationally restricted synthetic retinoids CD437 and 4-HPR. J Cell Biochem. 2003;89:262–278. doi: 10.1002/jcb.10505. [DOI] [PubMed] [Google Scholar]
- 133.Wilson AJ, Arango D, Mariadason JM, Heerdt BG, Augenlicht LH. TR3/Nur77 in colon cancer cell apoptosis. Cancer Res. 2003;63:5401–5407. [PubMed] [Google Scholar]
- 134.Mu X, Chang C. TR3 orphan nuclear receptor mediates apoptosis through up-regulating E2F1 in human prostate cancer LNCaP cells. J Biol Chem. 2003;278:42840– 42845. doi: 10.1074/jbc.M305594200. [DOI] [PubMed] [Google Scholar]
- 135.Weng JR, Tsai CH, Kulp SK, Wang D, Lin CH, Yang HC, Ma Y, Sargeant A, Chiu CF, Tsai MH, Chen CS. A potent indole-3-carbinol derived antitumor agent with pleiotropic effects on multiple signaling pathways in prostate cancer cells. Cancer Res. 2007;67:7815–7824. doi: 10.1158/0008-5472.CAN-07-0794. [DOI] [PubMed] [Google Scholar]