Abstract
A single-group crossover design was used to examine the effects of a warm footbath on body temperatures, distal-proximal skin temperature gradient (DPG), and sleep outcomes in 15 Taiwanese elders with self-reported sleep disturbance. Body temperatures and polysomnography were recorded for 3 consecutive nights. Participants were assigned randomly to receive a 41°C footbath for 40 minutes before sleep onset on night 2 or night 3. Mean DPG before lights off was significantly elevated on the bathing night. There were no significant differences in sleep outcomes between the two nights. However, when the first two non-rapid eye movement (NREM) sleep periods were examined, the amount of wakefulness was decreased in the second NREM period on the bathing night.
Keywords: sleep/rest, aging
Insomnia is a common complaint in older adults. They have trouble falling asleep, as well as frequent or prolonged nocturnal or early morning awakenings with an inability to return to sleep (Ancoli-Israel, 1997; Ancoli-Israel & Roth, 1999; Floyd, Medler, Ager, & Janisse, 2000). The overall prevalence of insomnia in the elderly ranges from 14.0% - 42.2% based on epidemiological surveys (Chiu et al., 1999; Kim, Uchiyama, Okawa, Liu, & Ogihara, 2000; Ohayon, 1996). As many as 25% of healthy elderly men and women may have chronic insomnia (Ohayon & Smirne, 2002). Global sleep dissatisfaction, defined as overall dissatisfaction with sleep, ranges from 7.7 - 20.8% in the elderly, which is higher than in the general population (7.0-10.1%; Pallesen et al., 2001).
Sleep is periodic resting behavior characterized by “few body movements, a recumbent posture, and complex brain electroencephalographic activity” (Carskadon & Dement, 2000, p.15). Human sleep architecture is defined by polysomnography (PSG) and divided into stages: four of non-rapid eye movement (NREM) sleep (stage 1 to 4), and one stage of rapid-eye movement (REM) sleep (Rechtschaffen & Kales 1968). Stage 1 is a transitional stage between waking and other sleep stages. Stage 2 is characterized by periodic sleep spindles and comprises most NREM sleep. Stages 3 and 4 referred to as slow wave sleep (SWS) are defined by higher amplitude and slower electroencephalogram (EEG) frequency activity (delta wave). In a young adult, a typical night of sleep has 4 to 5 periods of NREM and REM sleep (approximately 90-120 minutes per period) and minimal wakefulness (Carskadon & Dement; Culebras, 2002).
PSG is the most valid and accurate way to assess sleep. Measures derived from PSG include: (a) total sleep time, (b) sleep efficiency (ratio of time spent asleep/time in bed), (c) sleep latency (time to fall asleep after lights out), (d) amount of wake time during sleep periods (waking after sleep onset, WASO), (e) number of awakenings, and (f) amount of each sleep stage (Landis, 2002). Diagnostic criteria for insomnia include a sleep latency or amount of WASO > 30 minutes and a sleep efficiency less than 85% of time in bed, occurring ≥ 3 nights per week for at least 1 month (Edinger et al., 2004).
Age has large effects on sleep architecture. Compared to young adults, PSG-derived sleep architecture in older adults shows increased amounts of nocturnal wakefulness, increased NREM stage 1 sleep with reduced amounts or complete absence of NREM stages 3 and 4 sleep (Floyd et al., 2000; Van Someren, 2000a). In some studies, the percentage of REM sleep was reduced slightly in older adults (Van Cauter, Leproult, & Plat, 2000), whereas findings from other studies showed that older adults experienced typical amounts of REM sleep (Campbell & Murphy, 1998). Long sleep latencies (e.g. > 30 minutes), reduced sleep efficiency (e.g. < 85%), and a sleep duration of ≤ 6 hours were common in older adults. These characteristics are consistent with the criteria of insomnia, although not all older adults complained of poor sleep (Vitiello, Larsen, & Moe, 2004).
In addition to changes in sleep architecture, there are changes in the circadian rhythm of body temperature in older adults compared to young adults. A circadian rhythm is the variation in body functions and behaviors that occur in a regular pattern around a 24 hour time interval (Aronson et al., 1993; Aschoff, 1984). Body temperature variation and the sleep wake cycle each follow a circadian rhythm. These rhythms are coupled or synchronized with each other in people who are active in the day and sleep at night. In the morning after waking up, core (rectal) body temperature rises throughout the day to reach its highest point (peak) in the afternoon or early evening. In the evening, body temperature declines prior to sleep onset and continues to fall during sleep to reach its lowest point (trough) in the early morning hours (Dijk & Czeisler, 1995; Lavie, 2001; Van Someren, Raymann, Scherder, Daanen, & Swaab, 2002). The difference between the peak and trough of a circadian rhythm is called the amplitude. Compared to young adults, the circadian rhythm of core body temperature in older adults shows decreased amplitude (Dijk, Duffy, & Czeisler, 2000; Van Someren, 2000a). This difference could result from a lower daytime peak, a higher nighttime trough, or a combination of both. Figure 1 shows the circadian core body temperature rhythm in young and older adults. The trough temperature during sleep is higher and the peak is lower in older adults compared to young adults (Carrier, Monk, Buysse, & Kupfer, 1996).
Figure 1.
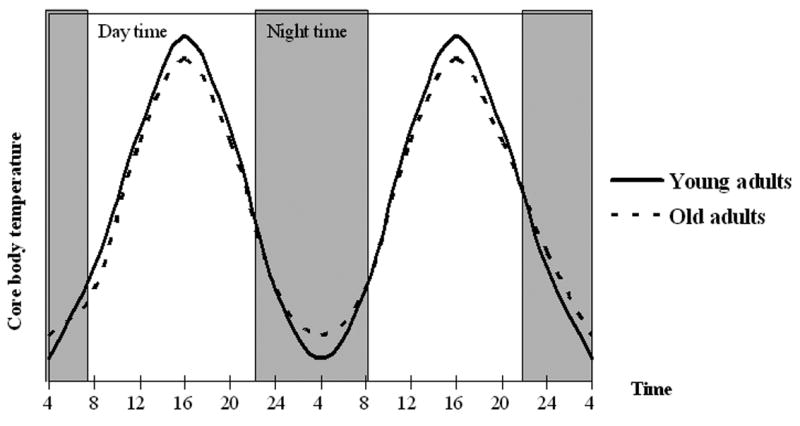
The fall in core body temperature before sleep onset and during sleep is associated with dilation of peripheral blood vessels, permitting heat dissipation from the body core to the periphery (Krauchi, 2002; Krauchi, Cajochen, Werth, & Wirz-Justice, 2000; Krauchi & Wirz-Justice, 2001). A lower core (rectal) temperature coupled with a higher distal (hands and feet) temperature before sleep is associated with a shorter sleep latency (Krauchi, Cajochen, & Wirz-Justice, 2004). The difference between core and peripheral temperatures is reflected by the distal-proximal skin temperature gradient (DPG) and has been shown to be a good predictor of sleepiness and the body's readiness for sleep (Krauchi, Cajochen, Werth, & Wirz-Justice, 1999). Treatments that enhance the peripheral gradient for heat loss have the potential to facilitate sleep onset and quality.
Based on evidence of the relation between body temperature and sleep, a warm bath (passive body heating) in the evening may promote sleep onset and improve overall sleep quality in older adults. Previous researchers have found that compared to a non-bathing night, a warm bath (40∼ 41°C) with whole body immersion in a bathtub taken 2 to 4 hours prior to sleep, shortened sleep latency, decreased the amount of wake time after sleep onset, and increased the amount of slow wave sleep, but also raised core (rectal) body temperature in older women with insomnia (Dorsey et al., 1996; Dorsey et al., 1999). Compared to whole body immersion in a tub, a warm bath (42°C) of only the lower legs taken within an hour of sleep onset improved sleep, but did not raise core body temperature in young women (Sung & Tochihara, 2000). A warm foot bath may increase peripheral blood flow and temperature gradient (DPG) to facilitate heat loss without increasing core body temperature and hereby improve sleep onset and quality. This hypothesis had not been tested in older adults with sleep disturbance.
The purpose of this single group, experimental crossover design study was to examine the effects of a warm footbath prior to sleep onset on body temperature and sleep in community dwelling older adults complaining of sleep disturbance. The conceptual framework for this study is shown in Figure 2. A warm foot bath was thought to facilitate heat dissipation to lower core (rectal) body and abdominal temperatures and raise foot temperature, and simultaneously to improve objective and perceived sleep outcomes. We hypothesized that a warm foot bath would raise the peripheral gradient for heat loss (increased DPG), improve PSG and perceived sleep patterns (increase total sleep time, shorten sleep latency, raise sleep efficiency, reduce WASO and number of awakenings, increase SWS), and improve perceived sleep quality (raise overall quality, improve sleep restoration and satisfaction) in older adults with complaints of sleep disturbance.
Figure 2.
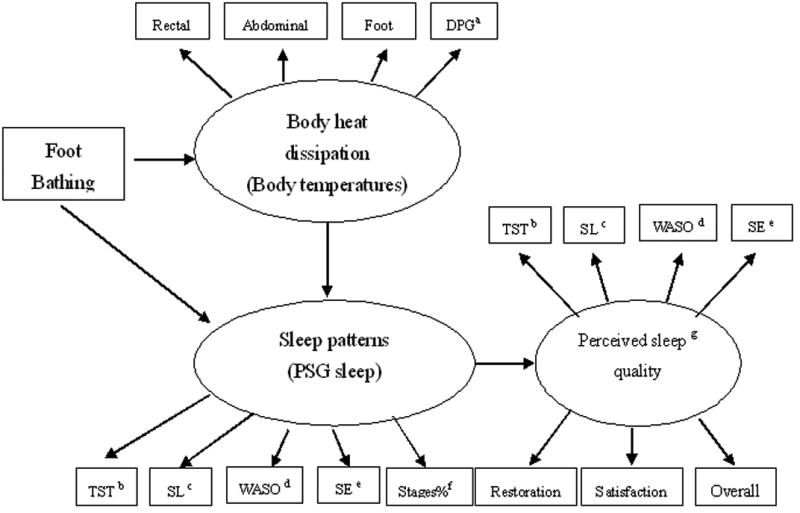
Methods
Participants
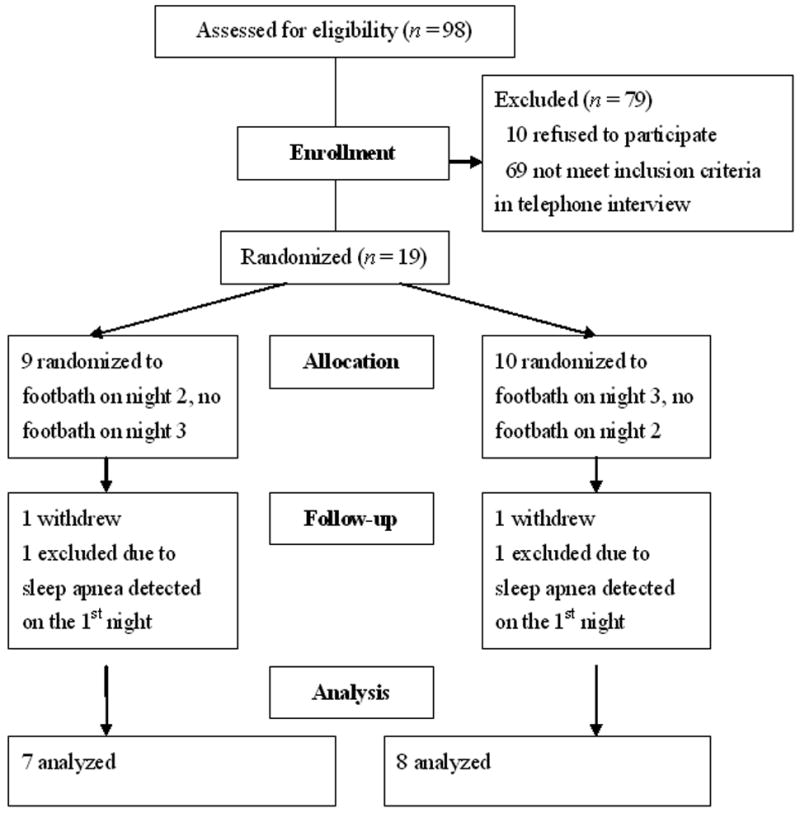
The University of Washington, Human Subjects Division and The National Taiwan University Hospital, Ethics Committee approved this study. Ninety-eight potential participants from North Taiwan, ages 60-75 years and complaining of sleep difficulties, responded to newspaper advertisements that were placed to recruit study volunteers. These potential participants were excluded if they had an acute illness, were undergoing chronic hemodialysis, or had diabetes, peripheral vascular or nerve diseases, major mental disorders, known sleep apnea, or were currently taking sleep medications. Participants with medical conditions that were considered under control (e.g. hypertension, heart arrhythmia, mild gastritis, and mild liver disease) were included in this study. Figure 3 shows the flow of participants through the trial. Sixty-nine participants were excluded during the initial telephone screening interview because they did not meet the inclusion criteria. An additional 10 participants refused to participate due to time conflicts or unwillingness to come to the hospital sleep laboratory. The remaining 19 potential participants were further screened by a research nurse for cognitive function, anxiety and depression, and subjective sleep disturbance as described below.
Figure 3.
Measurements
Cognitive function
The Chinese version of the Mini-Mental Status Examination (MMSE; Folstein, Folstein, & McHugh, 1975) was used to screen participants' cognitive function. The MMSE has 30 dichotomous items with a total possible score of 30; a score of < 24 is indicative of cognitive impairment (Folstein et al.; Tombaugh & McIntyre, 1992). The Chinese translation was modified slightly to adjust for Taiwanese social and cultural differences. Tests of the Chinese version demonstrated satisfactory internal consistency reliability (KR-20, α = .86) and inter-rater agreement (90%) in Taiwanese elder populations (Lou, Dai, Huang, & Yu, 2003). This questionnaire is suitable to differentiate cognitive impairment. All participants in this study had total scores of 30.
Anxiety and depression
The Chinese version of the Hospital Anxiety and Depression Scale (HADS; Zigmond & Snaith, 1983) was used to screen participants' anxiety and depression. The HADS contains 7 items to evaluate anxiety and 7 items to assess depression. Questions in both subscales are scored on a 3-point Likert scale with anchors of 0 (not at all) to 3 (most of the time). Each subscale is scored separately to derive an overall anxiety score and an overall depression score. The maximum score for each subscale is 21. A score of > 11 on either subscale indicates a case of anxiety or depression. The internal consistency reliability (Cronbach's alpha) of the HADS Chinese version were .84 for anxiety and .76 for depression in Taiwan's population (Chen, Chang, & Yeh, 2000). All participants in this study had scores of 10 or below in both anxiety and depression subscales, indicating they were not anxious or depressed. The overall Cronbach's alpha of the HADS in this study was .76 for anxiety and .81 for depression, respectively.
Sleep disturbance
The Chinese version of the Pittsburgh Sleep Quality Index (PSQI; Buysse, Reynolds, Monk, Berman, & Kupfer, 1989) was used to assess sleep disturbance. The PSQI contains seven components to evaluate participants' sleep over a 1-month time interval. A global PSQI score is derived by summing the score for each component. The maximum score is 21. A higher score indicates worse sleep quality. A score of 5 or greater is used to discriminate poor from good sleep. The internal consistency reliability (.82) and 14- to 21-day test-retest reliability (.85) of the PSQI Chinese version (CPSQI) was adequate in Taiwan's populations (Tsai et al., 2005). Participants had to score > 5 to be eligible for this study. The overall Cronbach's alpha of the global PSQI in this study was .72.
Sleep apnea
Screening for evidence of sleep apnea was done during the first night in the sleep lab. The evaluation criteria were based on the Report of the American Academy of Sleep Medicine Task Force (1999). Apnea was scored as the absence of airflow for at least 10 seconds, with a corresponding decrease in oxygen saturation of > 3%. The apnea index is the ratio of the total apnea events during sleep divided by the total sleep hours. An apnea index (AI) > 10 was used as the screening criterion in this study.
Core body and skin temperature recordings
Core body temperature was measured by a rectal thermal probe (Nikkiso-YSI Co., Ltd. Tokyo, Japan) that was lubricated with K-Y jelly and inserted approximately 13 cm into the rectum. This rectal thermal probe is a soft, thin silicon probe (0.5 cm caliber). Participants reported that they did not feel the probe when they were lying down in bed. Abdominal (proximal) temperature was measured with a skin thermal probe (Nikkiso-YSI Co., Ltd. Tokyo, Japan) attached to the abdomen close to the femoral artery. Foot (distal) skin temperature was measured from a skin thermal probe attached to the medial aspect of the ankle. DPG was obtained by subtracting the abdominal temperature from the foot temperature. The Mini-Logger monitor (Mini Mitter Co., Inc. Bend, Oregon USA) was used to record body temperatures in multiple locations simultaneously in 1-minute intervals. Body temperatures were measured prior to, during and after the foot bathing and until the participants awakened in the morning, except foot temperature was not measured during foot bathing. The range of the Mini-Logger monitor for thermometers was 30°C to 42°C, with resolution of 0.05°C and accuracy of 0.1°C.
Polysomnography (PSG) and sleep stage scoring
Electrodes for PSG including the EEG, electro-oculogram (EOG), and electromyogram (EMG) were placed according to standard procedures (Rechtschaffen & Kales, 1968) and Data were obtained with the PolySmith sleep data acquisition system (NeuroFax 1100 system, Nihon Konden Co., Tokyo, Japan). Chest parameters were added to the PSG recording system to screen sleep apnea in the first night. Chest and abdominal respiratory movements were measured with bands placed around the chest and the abdomen (Nihon Konden Co., Tokyo, Japan). Air flow was measured by a nasal cannula placed in the nose. Oxygen saturation was measured from the left index finger by a pulse oximeter (Ohmeda Biox 3740, Susquehanna Micro Inc., USA). Prior to each session, channels for all PSG recordings were calibrated.
Sleep stage scoring was based on standard criteria (Rechtschaffen & Kales, 1968) and performed by a single rater. Intra-rater reliability of sleep stages reflected an agreement of 85-90% in a 10% random sample of the recordings. Sleep variables were calculated for each participant. These included time in bed (TIB, lights out to final arising), sleep period time (SPT, time from the beginning of NREM stage 1 until final awakening), total sleep time (TST, the sum of sleep stages 1-4 and REM), sleep efficiency (SE, ratio of TST/TIB), sleep latency (SL, lights off to the first epoch of stage 1 sleep), NREM sleep (stages 1-4), REM sleep, and waking after sleep onset (WASO). Scored sleep data was partitioned into NREM periods (Armitage, Hoffmann, Trivedi, & Rush, 2000). A NREM period is the succession of NREM sleep stages greater than 15 minutes duration that terminated by stage REM or a wake period at least 5 minutes duration.
Perceived sleep quality
A Morning Questionnaire (MQ) was used to assess perceived sleep quality during the previous night. This 6-item questionnaire was modified from an instrument routinely used in previous laboratory sleep studies in adults (Landis, Lentz, Tsuji, Buchwald, & Shaver, 2004; Shaver et al., 1997). The MQ consists of three questions regarding sleep latency, sleep duration, and number and duration of awakenings, as well as three 10-centimeter visual analog scale (VAS) to gather the individual's perception of sleep restoration (rested to not rested at all), quality (very good to very poor), and satisfaction (very satisfied to very dissatisfied). The internal consistency reliability (Cronbach's alpha) of the three VASs for sleep perception in this study was .94.
Procedures
All 19 volunteers met the screening criteria and were scheduled to sleep 3 consecutive nights in the sleep laboratory at a medical center in Northern Taiwan. Written consent was obtained from the 19 enrolled participants. They were randomized to treatment order. The first night was used to allow all participants to adapt to the laboratory and to conduct PSG screening for evidence of sleep apnea. After the first night sleep in the laboratory, two participants were excluded because they showed evidence of sleep apnea, and two participants withdrew. The remaining 15 older adults completed the 3-night study. They were paid NT$ 1000 (US$ 30) for their effort and time.
Participants were randomly assigned to have a warm footbath on the second night (n=7) or on the third night (n=8). The footbath was scheduled 1 hour before their regular bedtime. Each participant had one foot bath in this study. Their feet and legs were immersed in warm water (41°C) up to 20 cm above the ankles for 40 minutes. A specially designed foot bathtub (Ten-Ta Co. Taipei, Taiwan) kept the water temperature constant at 41°C. This temperature was selected based on the results of a preliminary study indicating that the performance of elevating DPG to above 0°C in a water temperature of 41°C was better than a temperature of 40°C (Liao, Landis, Lentz, & Chiu, 2005).
Participants kept their routine activities during the daytime and came to the sleep laboratory 2 hours before their usual bedtime (according to participants' self-report). The schedule of lights off and on in this study was based on each individual's habitual routine, to control for individual variations in the circadian rhythm of sleep and wake. Participants wore their own pajamas and were covered with a standardized 100% cotton bed quilt during sleep. The sleeping environment was quiet and dark. An air conditioner was in use and set to keep the ambient temperature at 24°C during sleep. Data were collected from May to October 2003.
Data Analysis
Rectal, abdominal, and foot temperatures, and DPG at lights off were averaged for the last 3 minutes prior to lights off. All body temperatures and DPG during sleep were averaged for a 5 minute time intervals immediately after lights off and during sleep. Graphs were used to examine mean rectal, abdominal, and foot temperatures, and DPG across nights with and without footbath. Because participants had different total sleep times, only the first 7 hours were included in these graphs. Due to technical problems with the recordings, rectal temperatures for 3 participants during sleep were missing and excluded from the analysis of core body temperatures. Mean values of PSG sleep variables were calculated for the whole night, and for the first and the second NREM sleep periods. The latter half of one participant's PSG sleep records were excluded from the analysis of the whole night sleep because of technical problems with the recording, but retained in the analysis of the first and the second NREM sleep periods.
Data analyses were carried out in pre-determined conceptual blocks (e.g. body temperature, PSG sleep variables, and self-reported sleep outcomes). Each block was considered distinctly as a separate analysis. Each separate analysis was performed at the significance level of p < .05. A Bonferroni correction was applied for multiple comparisons within a block. Paired t-tests were used to test differences in body temperatures (rectal, abdominal, and foot temperatures and DPG), PSG and perceived sleep outcomes (TST SL, SE, WASO, number of awakenings), PSG sleep stage percents, and perceived sleep quality outcomes (restoration, sleep quality, and sleep satisfaction). Because previous researchers have shown that effects of passive body heating were most pronounced in the first half of a night of sleep (Horne & Reid, 1985), after examining the primary PSG sleep outcomes, we explored if there were any differences in wake and sleep stages during the first two NREM periods between the non-bathing and bathing nights.
Results
Clinical Characteristics
Participants' clinical characteristics are summarized in Table 1. Nine females and six males with a mean age of 65.7 years (SD = 3.7) and 70.0 years (SD = 6.1), respectively, participated in this study. The majority of participants were married, retired, and had 12 or more years of education. Additional information from the PSQI subscales showed that 40% of the participants reported a habitual sleep latency of > 30 minutes; 73.3% reported a sleep duration of < 6 hours, and 80% reported a sleep efficiency of <85 % (calculated from self-reported usual sleep duration/time in bed). Only two participants (13.3%) reported previous use of medications to aid sleep, and this use was prior to their participation in this study. No participants took any sleep medication during the study.
Table 1. Demographic and Clinical Characteristics.
| M | SD | Range | |
|---|---|---|---|
| Age, years | 67.4 | 5.1 | 60-75 |
| Depression a | 4.7 | 1.7 | 3-8 |
| Anxiety a | 4.8 | 3.6 | 0-10 |
| PSQI b Global Sleep quality | 10.7 | 3.9 | 5-19 |
|
| |||
| n | % | ||
|
| |||
| Gender | |||
| Female | 9 | 60.0 | |
| Male | 6 | 40.0 | |
| Marriage | |||
| Married | 11 | 73.3 | |
| Widow | 4 | 26.7 | |
| Education | |||
| 6-9 years | 4 | 26.7 | |
| >9 - <14 years | 5 | 33.3 | |
| 14-16 years | 6 | 40.0 | |
N = 15.
Hospital Anxiety and Depression Scale, overall rating for anxiety and depression.
Pittsburgh Sleep Quality Index, global score
Effects of Foot Bathing on Core and Skin Body Temperatures, and DPG
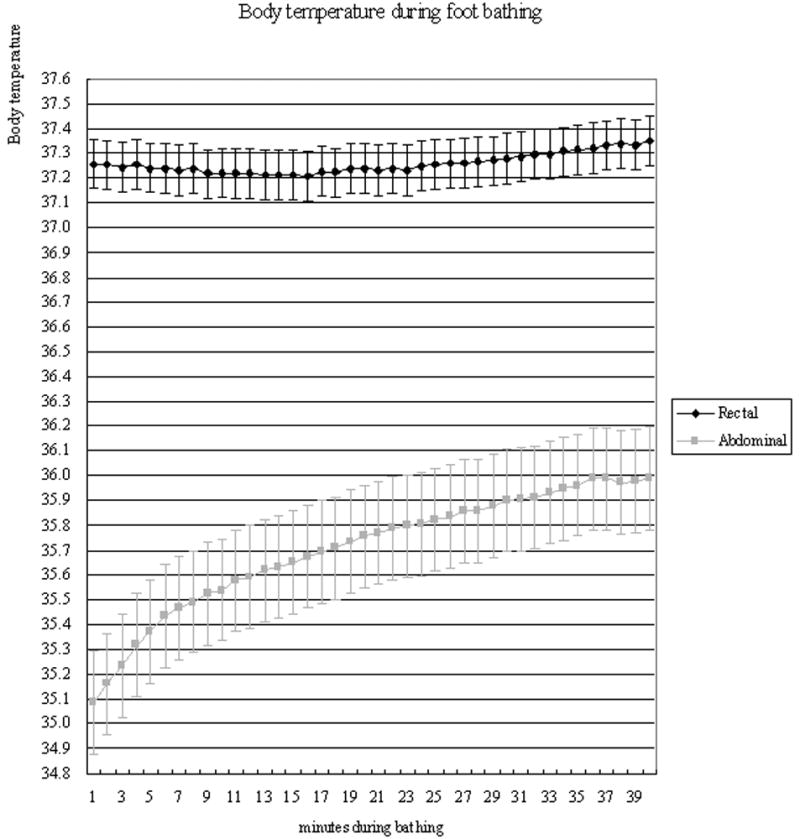
Figure 4 shows the mean rectal and abdominal temperatures during foot bathing. While abdominal temperature steadily increased, rectal temperature actually decreased slightly during the first 15 minutes of foot bathing and then increased. Compared to the beginning of foot bathing, abdominal (M = 0.63°C, SD = 0.75), t(14) = 3.0, p = .01 and foot skin temperatures (M = 2.83°C, SD = 0.98), t(14) = 11.2, p < .001 increased significantly after foot bathing, but rectal temperatures did not change significantly (M = 0.10°C, SD = 0.20), t(14) = 1.62, p = .13. DPG increased significantly from −3.11°C (SD = 1.06) before bathing to −0.81°C (SD = 1.06) after bathing t(14)= 5.68, p < .001. Footbath increased all skin temperatures significantly.
Figure 4.
Effects of Foot Bathing on Body Temperatures During Sleep
Mean rectal, abdominal, and foot temperatures, and DPG at lights off on bathing and non-bathing nights are summarized in Table 2. Compared to the non-bathing night, mean rectal and abdominal temperatures were 0.23°C (SD = 0.30) and 0.38°C (SD = 0.57) higher on the bathing night, respectively, but these increases did not reach statistical significance. However, mean DPG on the bathing night was significantly higher than on the non-bathing night. Mean foot temperature was significantly higher (M = 2.09°C, SD = 0.73) on the bathing night than on the non-bathing night. It decreased during the first 120 minutes of sleep on the bathing night, which was opposite to what was observed on the non-bathing night (Figure 5).
Table 2. Body Temperatures and Distal-Proximal Skin Temperature Gradient at Lights Off on Non-Bathing and Bathing Nights.
| Non-bathing | Bathing | |||||
|---|---|---|---|---|---|---|
| M | SD | M | SD | t | p | |
| Rectal a (°C) | 37.05 | 0.19 | 37.28 | 0.38 | 1.80 | .095 |
| Abdominal (°C) | 34.95 | 0.60 | 35.33 | 0.54 | 2.26 | .040 |
| Foot (°C) | 32.82 | 0.77 | 34.91 | 0.69 | 8.73 | .000 |
| DPG (°C) | -2.14 | 0.57 | -0.42 | 0.89 | 6.81 | .000 |
N = 15. Data = average of the last 3 minutes before lights off. Paired-t test, 2 tailed, with Bonferroni correction alpha = .05/4 = .0125.
n = 12 for rectal temperature data, 3 subjects were excluded due to missing data.
Figure 5.
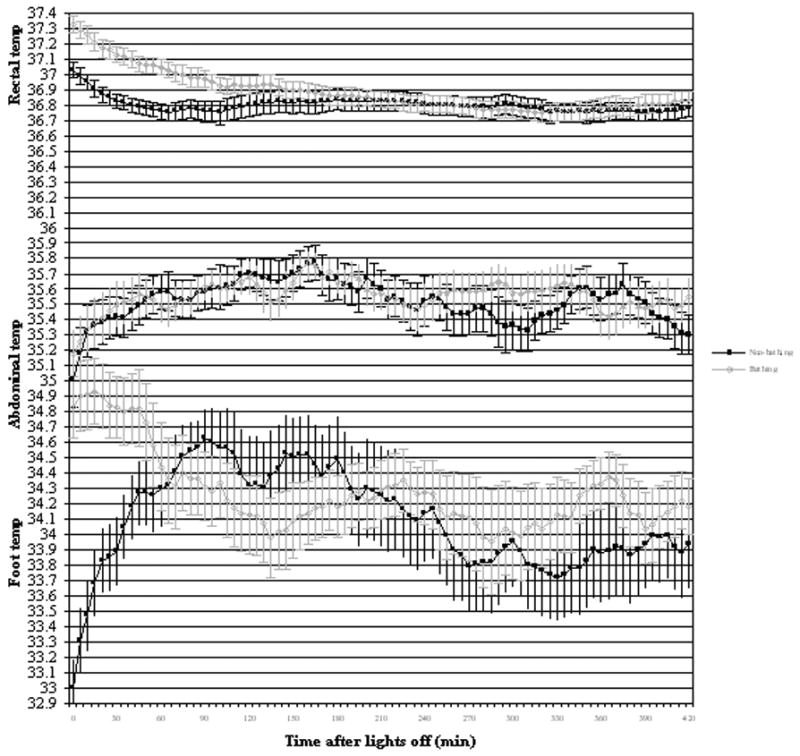
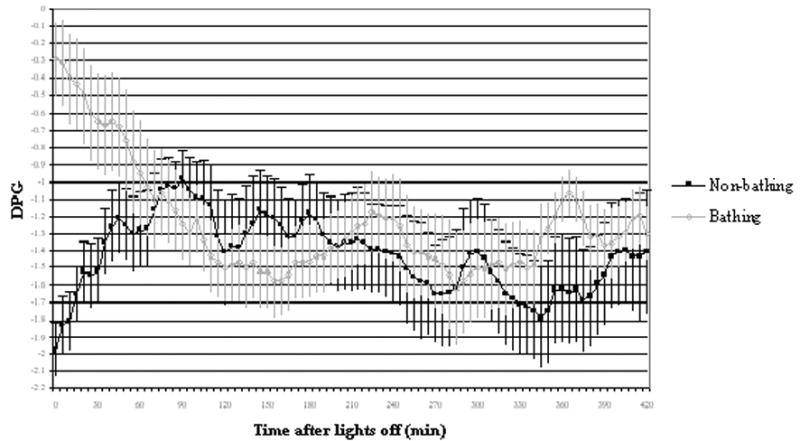
DPG has been used as an indirect index of peripheral heat loss (Krauchi et al., 2000). A higher DPG is associated with better sleep (Krauchi et al., 1999; Nakao, McGinty, Szymusiak, & Yamamoto, 1995; Van Someren, 2000b). Figure 6 shows the mean DPG during sleep for the non-bathing and bathing nights. DPG on the non-bathing night gradually increased after lights off, indicative of gradually increasing distal heat loss. DPG on the bathing night approached zero, representing little difference between foot and abdominal temperatures, and was indicative of high distal heat loss. After lights off, DPG gradually decreased on the bathing night, which was indicative of a reduced rate of distal heat loss. Warm foot bathing significantly elevated DPG, but this effect was not retained during sleep.
Figure 6.
Objective and Perceived Sleep Outcomes on Non-Bathing and Bathing Nights
Objective and perceived sleep outcomes between non-bathing and bathing nights for the whole night are summarized in Table 3. There were no significant differences in PSG-derived (objective) or perceived sleep outcomes between the non-bathing and bathing nights. We partitioned sleep into NREM periods and explored the extent to which sleep stages might differ between the first two NREM periods for the non-bathing and bathing nights (Table 4). In the first NREM sleep period, there were no significant differences in any sleep variables. However, the percentage awake was significantly decreased during the second NREM sleep period on the bathing night compared to the non-bathing night (Table 4).
Table 3. Polysomnography and Perceived Sleep Quality for the Whole Night Sleep.
| Non-bathing | Bathing | |||||
|---|---|---|---|---|---|---|
| M | SD | M | SD | t | p | |
| Polysomnography a | ||||||
| Total sleep time (min) | 408.8 | 58.5 | 406.3 | 49.2 | -0.06 | .95 |
| Sleep latency (min) (to stage 1) | 11.7 | 12 | 8.6 | 5.8 | -1.22 | .28 |
| Wake after sleep onset (min) | 50.5 | 23.7 | 46.4 | 40.4 | -0.47 | .65 |
| Sleep efficiency b (%) | 84.8 | 7.1 | 86.4 | 7.8 | 0.63 | .54 |
| Sleep stages (% sleep period time c) | ||||||
| Stage 1 | 20.3 | 6.0 | 19.7 | 9.2 | -0.77 | .45 |
| Stage 2 | 49.6 | 7.4 | 47.2 | 10.1 | -0.72 | .48 |
| Stage 3 & 4 | 3.4 | 3.2 | 4.2 | 3.4 | 1.27 | .23 |
| REM | 15.7 | 3.1 | 19.0 | 4.9 | 2.43 | .03 |
| Perceived sleep | ||||||
| Total sleep time (min) | 330.0 | 58.9 | 342.0 | 89.7 | 0.28 | .78 |
| Sleep latency (min) | 34.3 | 21.0 | 29.6 | 19.0 | -1.54 | .15 |
| Number of Awakenings | 3.1 | 1.4 | 2.5 | 1.6 | -1.31 | .21 |
| Wake after sleep (min) | 63.2 | 53.7 | 25.8 | 17.9 | -1.33 | .21 |
| Sleep efficiency (%) | 71.5 | 13.6 | 74.6 | 17.8 | 0.37 | .72 |
| Perceived sleep quality | ||||||
| Morning restoration (rested-no) | 4.2 | 1.9 | 3.5 | 1.9 | -1.32 | .21 |
| Overall sleep quality (good-poor) | 4.2 | 2.0 | 3.9 | 2.6 | -0.48 | .64 |
| Sleep satisfaction (satisfied-dissatisfied) | 3.6 | 1.6 | 3.6 | 2.4 | -0.11 | .92 |
N = 15. Paired-t test, 2 tailed, with Beferroni correction alpha = .05/8=.006 for polysomnography, and for perceived sleep and sleep quality, respectively.
n = 14, 1 subject was excluded from whole night PSG sleep due to technical problem.
Sleep efficiency = total sleep time/time in bed * 100.
Sleep period time = total sleep time + wake after sleep onset.
Table 4. Polysomnography Sleep in the First and the Second NREM Sleep Periods.
| Non-bathing | Bathing | |||||
|---|---|---|---|---|---|---|
| M | SD | M | SD | t | p | |
| First NREM period | ||||||
| NREM period time (minutes) | 115.3 | 38.9 | 119.4 | 37.0 | -0.38 | .71 |
| Wake (%, NREM period time) | 7.2 | 7.3 | 9.0 | 10.9 | 0.58 | .57 |
| Sleep stages (%, NREM period time) | ||||||
| Stage 1 | 15.8 | 7.3 | 20.4 | 14.7 | 1.50 | .15 |
| Stage 2 | 51.5 | 12.7 | 42.4 | 13.9 | 1.89 | .08 |
| Stage 3 & 4 | 9.2 | 13.8 | 7.9 | 8.4 | -0.35 | .73 |
| REM | 16.2 | 7.1 | 20.3 | 8.7 | 2.00 | .07 |
| Second NREM period | ||||||
| NREM period time (minutes) | 109.2 | 33.7 | 105.5 | 29.9 | 0.42 | .68 |
| Wake (%, NREM period time) | 10.3 | 8.8 | 3.7 | 5.0 | -2.95 | .01 |
| Sleep stages (%, NREM period time) | ||||||
| Stage 1 | 15.1 | 6.7 | 12.9 | 8.1 | -1.05 | .31 |
| Stage 2 | 52.2 | 10.6 | 54.7 | 14.2 | 0.65 | .53 |
| Stage 3 & 4 | 4.3 | 5.5 | 6.5 | 8.6 | 0.99 | .34 |
| REM | 18.1 | 10.1 | 22.2 | 8.0 | 1.37 | .19 |
Note. N = 15. Paired-t test, 2 tailed, with Beferroni correction alpha = .05/5 = .01 for each NREM period.
Discussion
In this study we examined the effects of a warm footbath prior to sleep on body temperatures and sleep outcomes in a small group of older adults with complaints of sleep disturbance. It was found that a warm footbath shortly before sleep onset raised foot temperature and the gradient for heat loss (e.g. DPG) considerably, but had little effect on core body temperature or objective and perceived sleep outcomes. REM sleep was increased on the bathing night, but the change did not reach statistical significance after correcting for multiple comparisons. The amount of wakefulness was reduced in the second NREM sleep period, but no increases in any sleep stages were observed. Participants also perceived that they had less wakefulness after sleep onset, but these differences did not reach statistical significance. In this small sample of older adults, a warm footbath taken shortly before bedtime did not have large effects upon sleep.
The results of this passive heating study are different from prior studies of older adults with insomnia. Previous researchers have found that a whole body bath taken 1.5-2 hours before bedtime was associated with decreased wakefulness (WASO, 21-30 minutes) and increased slow wave sleep (SWS, 5.3%; Dorsey et al., 1996; Dorsey et al., 1999). Participants in prior studies and the present footbath study had similar sleep complaints based on diagnostic criteria for insomnia. However, participants in this footbath study had better mean PSG sleep efficiency (SE = 86%) and less wakefulness (WASO = 46 minutes) on the bathing night compared to a prior study (SE = 74%, WASO = 92 minutes; Dorsey, 1996 #60}. Participants in this study perceived their sleep efficiency as 75% on the bathing night, but their PSG-recorded sleep efficiency was much higher. The discrepancies between objective and perceived sleep suggests that participants may have had sleep state misperception (Bonnet & Arand, 1997; Dorsey & Bootzin, 1997), a type of paradoxical insomnia in which individuals perceive their sleep as poor, despite having good sleep as measured by PSG. Behavioral therapy that focuses on correcting distorted perceptions of poor sleep may be an effective way to manage this type of insomnia (Tang & Harvey, 2004).
Warm foot bathing prior to sleep in this study did not increase slow wave sleep as has been reported in previous studies of whole body bathing in older adults with insomnia (Dorsey et al., 1996; Dorsey et al., 1999). Participants in our footbath study averaged 19 minutes of SWS after the footbath compared to 73 minutes of SWS reported in a previous study (Dorsey et al., 1996). Even when sleep stages were examined by NREM periods, SWS was not increased after the footbath, although the amount of wakefulness was reduced in the second NREM period. A previous passive body heating study in young adults showed similar findings of SWS increases (Horne & Reid, 1985). In these studies, investigators had participants take a warm bath several hours before sleep onset, which was closer to the typical peak of the circadian rhythm of body temperature. Parameters related to the type of bath (feet and legs versus whole body), timing of the bath relative to sleep onset, and ambient temperature are important in considering results from passive heating studies.
Based on previous studies, local warming of the feet was not expected to provide a sufficient heat load to increase core body temperature (Aoki, Kondo, Shibasaki, Takano, & Katsuura, 1997; Sung & Tochihara, 2000; Taylor, Johnson, O'Leary, & Park, 1984) as would be expected with whole body emersion in a bath. However, when the ambient temperature surrounding the skin exceeds body temperature (e.g. 37°C), vasodilation will transfer heat from the environment to the body (Van Someren et al., 2002). Core body temperature was slightly elevated after 15 minutes of a warm footbath (Figure 4) suggesting that some heat was transferred to the body during the footbath. Furthermore, waking was not decreased until the second NREM period. This delay implies that this footbath could have been administered too close to sleep onset. There is a paucity of passive heating studies in older adults, but results from previous studies in young, healthy adults and those from older women with insomnia show that body temperature is raised and SWS is increased when the warm bath is taken 3 to 4 hours prior to sleep onset (Dorsey et al., 1996; Dorsey et al., 1999; Mishima, Hozumi, Shimizu, Hishikawa, & Mishima, 2005). After a warm bath and before going to bed, the body may need time to cool down. Taking time to cool down after a warm bath may be very important for older adults if mechanisms involved in heat dissipation from the core to the periphery are less efficient with aging. A higher trough of the circadian temperature rhythm during sleep in older adults (Carrier et al., 1996) provides some evidence that heat dissipation mechanisms are less effective compared to young adults. Future research focused on comparing different time intervals between the footbath relative to sleep onset and effects on both body temperature and sleep would increase understanding about passive heating effects on sleep and in particular, effects on slow wave sleep.
In addition to altered heat dissipation capacity in older adults, ambient air temperature may have effects on skin temperature and heat dissipation (Hirosawa et al., 1984; Krauchi et al., 2000). High ambient temperature (> 28°C) is associated with disturbed sleep (Ohnaka, Tochihara, & Kanda, 1995). Our study was conducted in the summer months when the ambient temperature in Taiwan is seldom < 25°C. Although the ambient temperature of the laboratory in this study was maintained at 24°C, which is considered a comfortable and neutral ambient temperature (15°C - 25°C) for body temperature regulation, it was warmer than the ambient temperature in a previous footbath study (10°C; Sung & Tochihara, 2000). DPG results in this study indicated that heat dissipation to the periphery was slowed during sleep after the footbath. Although ambient temperature can not be ignored, it should be noted that passive heating with a warm footbath in this study had minimal effects on either objective or perceived sleep outcomes in older adults. Reduced heat dissipation capacity in older adults (Dorsey et al., 1999; Minson, Holowatz, Wong, Kenney, & Wilkins, 2002; Okamoto-Mizuno, Tsuzuki, & Mizuno, 2004), rather than effects of ambient temperature, may be a more plausible explanation for our observed effects of a foot bath on body temperature in older adults.
The rationale for this study was derived from previous observational studies linking distal vasodilation and heat loss with sleepiness in studies of young adults (Krauchi et al., 1999, 2000). Our observations in older adults that foot temperature and DPG increased and rectal temperature decreased after lights off on non-bathing nights are consistent with reports in the literature of increased heat loss from the core to the periphery shortly after sleep onset. After a warm footbath, we found that mean core (rectal) temperature was slightly higher and foot temperature was considerably elevated, and there was no change in abdominal temperature. The net result was a large increased DPG, which could be accounted for primarily by changes in peripheral blood flow. A current review of circadian thermoregulation mechanisms suggests that sympathetic vasoconstriction under cold stimuli is modulated in the distal portions of limbs, but the trunk and proximal portions play more important roles in the activation of vasodilation to warming (Van Someren et al., 2002). Warming proximal sites of the body is suggested as a more effective way to induce heat dissipation and improve sleep compared to warming distal sites (Ebben & Spielman, 2006; Raymann, Swaab, & Van Someren, 2005). Heating proximal parts of the body, as occurs with a whole body bath, may actually be necessary to trigger heat dissipation from the core to the periphery. Further study examining the effect of proximal warming, such as using a warm pad/bolster on the trunk or on proximal portions of limbs, on sleep in older adults may provide additional insights to enhance sleep.
There are several limitations worth noting in this study. First, although a large number of adults responded to the advertisement and were screened for this study, the small sample study limits the generalizability of the results. The small sample also precluded an examination of gender differences in the findings. For this initial study of foot bathing effects on body temperature and sleep in older adults, we included both men and women who were relatively healthy, but complained of poor sleep. Many older adults, however, have chronic conditions where the use of passive heating interventions may be contraindicated. For example, it will be important in future studies to screen older participants for signs of peripheral vascular disease because it could retard heat dissipation. Second, some of adults who were eligible for the study refused to come to the hospital where the sleep laboratory is located. Many healthy older adults in Taiwan often hesitate to sleep outside of their home. Home monitoring of sleep and body temperature may increase the willingness of older adults in Taiwan to participate in future studies. Third, we used the PSQI as the only screening measure to quantify the extent of poor sleep in participants. Recently published recommendations for the assessment of participants' perceptions of sleep include completion of several screening questionnaires in addition to the PSQI, and completion of a sleep diary for two weeks to verify the extent of insomnia (Buysse, Ancoli-Israel, Edinger, Lichstein, & Morin, 2006; Monk et al., 2006; Morin et al., 2006). In addition to assessing the effects of interventions on sleep outcomes, it is also important to include assessments of how sleepiness or wakefulness affect daytime function.
In summary, passive body heating prior to sleep onset significantly elevated foot temperature and the peripheral gradient for heat loss, but, unlike previous studies, did not change objective and perceived sleep outcomes. The results of this study raise important questions about heat load, type and timing of the bath, and age differences in response to passive body warming. More studies examining the application of footbath or other passive body heating interventions on good and poor sleepers will provide crucial information about managing sleep and benefit those who are suffering from sleep difficulties.
Acknowledgments
This work was supported by the Hester McLaws Nursing Scholarship, University of Washington, School of Nursing; the Center for Women's Health and Gender Research, Grants NR04001, NR081346, AT002108; and the National Taiwan University Hospital, Department of Neurology. We thank Mr. Jim Rothermel for his assistance with sleep scoring and the NREM program implementation, Dr. Martha Lentz for her vast patience and knowledge in managing and analyzing data, and Dr. Patricia Prinz for her guidance in the early drafts of this manuscript. We also express our sincere appreciation to the elders who participated in this study.
References
- American Academy of Sleep Medicine Task Force. Sleep-related breathing disorders in adults: Recommendations for syndrome definition and measurement techniques in clinical research. The Report of an American Academy of Sleep Medicine Task Force. Sleep. 1999;22:667–689. [PubMed] [Google Scholar]
- Ancoli-Israel S. Sleep problems in older adults: Putting myths to bed. Geriatrics. 1997;52(1):20–30. [PubMed] [Google Scholar]
- Ancoli-Israel S, Roth T. Characteristics of insomnia in the United States: Results of the 1991 National Sleep Foundation Survey. I. Sleep. 1999;22 2:S347–S353. [PubMed] [Google Scholar]
- Aoki K, Kondo N, Shibasaki M, Takano S, Katsuura T. Circadian variation in skin blood flow responses to passive heat stress. Physiology & Behavior. 1997;63:1–5. doi: 10.1016/s0031-9384(97)00348-x. [DOI] [PubMed] [Google Scholar]
- Armitage R, Hoffmann R, Trivedi M, Rush AJ. Slow-wave activity in NREM sleep: Sex and age effects in depressed outpatients and healthy controls. Psychiatry Research. 2000;95:201–213. doi: 10.1016/s0165-1781(00)00178-5. [DOI] [PubMed] [Google Scholar]
- Aronson BD, Bell-Pedersen D, Block GD, Bos NP, Dunlap JC, Eskin A, et al. Circadian rhythms. Brain Research Brain Research Review. 1993;18:315–333. doi: 10.1016/0165-0173(93)90015-r. [DOI] [PubMed] [Google Scholar]
- Aschoff J. Circadian timing. Annals of the New York Academy of Sciences. 1984;423:442–468. doi: 10.1111/j.1749-6632.1984.tb23452.x. [DOI] [PubMed] [Google Scholar]
- Bonnet MH, Arand DL. Physiological activation in patients with sleep state misperception. Psychosomatic Medicine. 1997;59:533–540. doi: 10.1097/00006842-199709000-00011. [DOI] [PubMed] [Google Scholar]
- Buysse DJ, Ancoli-Israel S, Edinger JD, Lichstein KL, Morin CM. Recommendations for a standard research assessment of insomnia. Sleep. 2006;29:1155–1173. doi: 10.1093/sleep/29.9.1155. [DOI] [PubMed] [Google Scholar]
- Buysse DJ, Reynolds CF, 3rd, Monk TH, Berman SR, Kupfer DJ. The Pittsburgh Sleep Quality Index: A new instrument for psychiatric practice and research. Psychiatry Research. 1989;28:193–213. doi: 10.1016/0165-1781(89)90047-4. [DOI] [PubMed] [Google Scholar]
- Campbell SS, Murphy PJ. Relationships between sleep and body temperature in middle-aged and older subjects. Journal of the American Geriatrics Society. 1998;46:458–462. doi: 10.1111/j.1532-5415.1998.tb02466.x. [DOI] [PubMed] [Google Scholar]
- Carrier J, Monk TH, Buysse DJ, Kupfer DJ. Amplitude reduction of the circadian temperature and sleep rhythms in the elderly. Chronobiology International. 1996;13:373–386. doi: 10.3109/07420529609012661. [DOI] [PubMed] [Google Scholar]
- Carskadon M, Dement W. Normal human sleep: An overview. In: Kryger M, Roth T, Dement W, editors. Principles and practice of sleep medicine. 3rd. Philadelphia: W.B. Saunders; 2000. pp. 15–25. [Google Scholar]
- Chen ML, Chang HK, Yeh CH. Anxiety and depression in Taiwanese cancer patients with and without pain. Journal of Advanced Nursing. 2000;32:944–951. [PubMed] [Google Scholar]
- Chiu HF, Leung T, Lam LC, Wing YK, Chung DW, Li SW, et al. Sleep problems in Chinese elderly in Hong Kong. Sleep. 1999;22:717–726. doi: 10.1093/sleep/22.6.717. [DOI] [PubMed] [Google Scholar]
- Culebras A. Normal sleep. In: Lee-Chiong T Jr, Sateia M, Carskadon MA, editors. Sleep Medicine. Philadelphia: Hanley & Belfus, Inc.; 2002. pp. 1–6. [Google Scholar]
- Dijk DJ, Czeisler CA. Contribution of the circadian pacemaker and the sleep homeostat to sleep propensity, sleep structure, electroencephalographic slow waves, and sleep spindle activity in humans. Journal of Neuroscience. 1995;15:3526–3538. doi: 10.1523/JNEUROSCI.15-05-03526.1995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dijk DJ, Duffy JF, Czeisler CA. Contribution of circadian physiology and sleep homeostasis to age-related changes in human sleep. Chronobiology International. 2000;17:285–311. doi: 10.1081/cbi-100101049. [DOI] [PubMed] [Google Scholar]
- Dorsey CM, Bootzin RR. Subjective and psychophysiologic insomnia: An examination of sleep tendency and personality. Biological Psychiatry. 1997;41:209–216. doi: 10.1016/0006-3223(95)00659-1. [DOI] [PubMed] [Google Scholar]
- Dorsey CM, Lukas SE, Teicher MH, Harper D, Winkelman JW, Cunningham SL, et al. Effects of passive body heating on the sleep of older female insomniacs. Journal of Geriatric Psychiatry & Neurology. 1996;9:83–90. doi: 10.1177/089198879600900203. [DOI] [PubMed] [Google Scholar]
- Dorsey CM, Teicher MH, Cohen-Zion M, Stefanovic L, Satlin A, Tartarini W, et al. Core body temperature and sleep of older female insomniacs before and after passive body heating. Sleep. 1999;22:891–898. doi: 10.1093/sleep/22.7.891. [DOI] [PubMed] [Google Scholar]
- Ebben MR, Spielman AJ. The effects of distal limb warming on sleep latency. International Journal of Behavioral Medicine. 2006;13:221–228. doi: 10.1207/s15327558ijbm1303_5. [DOI] [PubMed] [Google Scholar]
- Edinger JD, Bonnet MH, Bootzin RR, Doghramji K, Dorsey CM, Espie CA, et al. Derivation of research diagnostic criteria for insomnia: Report of an American Academy of Sleep Medicine Work Group. Sleep. 2004;27:1567–1596. doi: 10.1093/sleep/27.8.1567. [DOI] [PubMed] [Google Scholar]
- Floyd JA, Medler SM, Ager JW, Janisse JJ. Age-related changes in initiation and maintenance of sleep: A meta-analysis. Research in Nursing & Health. 2000;23:106–117. doi: 10.1002/(sici)1098-240x(200004)23:2<106::aid-nur3>3.0.co;2-a. [DOI] [PubMed] [Google Scholar]
- Folstein MF, Folstein SE, McHugh PR. “Mini-mental state”. A practical method for grading the cognitive state of patients for the clinician. Journal of Psychiatric Research. 1975;12:189–198. doi: 10.1016/0022-3956(75)90026-6. [DOI] [PubMed] [Google Scholar]
- Hirosawa I, Dodo H, Hosokawa M, Watanabe S, Nishiyama K, Fukuchi Y. Physiological variations of warm and cool sense with shift of environmental temperature. International Journal of Neuroscience. 1984;24:281–288. doi: 10.3109/00207458409089817. [DOI] [PubMed] [Google Scholar]
- Horne JA, Reid AJ. Night-time sleep EEG changes following body heating in a warm bath. Electroencephalography and Clinical Neurophysiology. 1985;60:154–157. doi: 10.1016/0013-4694(85)90022-7. [DOI] [PubMed] [Google Scholar]
- Kim K, Uchiyama M, Okawa M, Liu X, Ogihara R. An epidemiological study of insomnia among the Japanese general population. Sleep. 2000;23:41–47. [PubMed] [Google Scholar]
- Krauchi K. How is the circadian rhythm of core body temperature regulated? Clinical Autonomic Research. 2002;12:147–149. doi: 10.1007/s10286-002-0043-9. [DOI] [PubMed] [Google Scholar]
- Krauchi K, Cajochen C, Werth E, Wirz-Justice A. Warm feet promote the rapid onset of sleep. Nature. 1999;401:36–37. doi: 10.1038/43366. [DOI] [PubMed] [Google Scholar]
- Krauchi K, Cajochen C, Werth E, Wirz-Justice A. Functional link between distal vasodilation and sleep-onset latency? American Journal of Physiology. Regulatory, Integrative and Comparative Physiology. 2000;278:R741–R748. doi: 10.1152/ajpregu.2000.278.3.R741. [DOI] [PubMed] [Google Scholar]
- Krauchi K, Cajochen C, Wirz-Justice A. Waking up properly: Is there a role of thermoregulation in sleep inertia? Journal of Sleep Research. 2004;13:121–127. doi: 10.1111/j.1365-2869.2004.00398.x. [DOI] [PubMed] [Google Scholar]
- Krauchi K, Wirz-Justice A. Circadian clues to sleep onset mechanisms. Neuropsychopharmacology. 2001;25:S92–S96. doi: 10.1016/S0893-133X(01)00315-3. [DOI] [PubMed] [Google Scholar]
- Landis CA. Sleep and methods of assessment. Nursing Clinics of North America. 2002;37:583–597. doi: 10.1016/s0029-6465(02)00027-0. [DOI] [PubMed] [Google Scholar]
- Landis CA, Lentz MJ, Tsuji J, Buchwald D, Shaver JL. Pain, psychological variables, sleep quality, and natural killer cell activity in midlife women with and without fibromyalgia. Brain, Behavior, and Immunity. 2004;18:304–313. doi: 10.1016/j.bbi.2003.11.001. [DOI] [PubMed] [Google Scholar]
- Lavie P. Sleep-wake as a biological rhythm. Annual Review of Psychology. 2001;52:277–303. doi: 10.1146/annurev.psych.52.1.277. [DOI] [PubMed] [Google Scholar]
- Liao WC, Landis CA, Lentz MJ, Chiu MJ. Effect of foot bathing on distal-proximal skin temperature gradient in elders. International Journal of Nursing Studies. 2005;42:717–722. doi: 10.1016/j.ijnurstu.2004.11.011. [DOI] [PubMed] [Google Scholar]
- Lou MF, Dai YT, Huang GS, Yu PJ. Postoperative cognitive changes among older Taiwanese patients. Journal of Clinical Nursing. 2003;12:579–588. doi: 10.1046/j.1365-2702.2003.00753.x. [DOI] [PubMed] [Google Scholar]
- Minson CT, Holowatz LA, Wong BJ, Kenney WL, Wilkins BW. Decreased nitric oxide- and axon reflex-mediated cutaneous vasodilation with age during local heating. Journal of Applied Physiology. 2002;93:1644–1649. doi: 10.1152/japplphysiol.00229.2002. [DOI] [PubMed] [Google Scholar]
- Mishima Y, Hozumi S, Shimizu T, Hishikawa Y, Mishima K. Passive body heating ameliorates sleep disturbances in patients with vascular dementia without circadian phase-shifting. American Journal of Geriatric Psychiatry. 2005;13:369–376. doi: 10.1176/appi.ajgp.13.5.369. [DOI] [PubMed] [Google Scholar]
- Monk TH, Buysse DJ, Hall M, Nofzinger EA, Thompson WK, Mazumdar SA, et al. Age-related differences in the lifestyle regularity of seniors experiencing bereavement, care-giving, insomnia, and advancement into old-old age. Chronobiology International. 2006;23:831–841. doi: 10.1080/07420520600827152. [DOI] [PubMed] [Google Scholar]
- Morin CM, Bootzin RR, Buysse DJ, Edinger JD, Espie CA, Lichstein KL. Psychological and behavioral treatment of insomnia:Update of the recent evidence (1998-2004) Sleep. 2006;29:1398–1414. doi: 10.1093/sleep/29.11.1398. [DOI] [PubMed] [Google Scholar]
- Nakao M, McGinty D, Szymusiak R, Yamamoto M. Dynamical features of thermoregulatory model of sleep control. Japanese Journal of Physiology. 1995;45:311–326. doi: 10.2170/jjphysiol.45.311. [DOI] [PubMed] [Google Scholar]
- Ohayon M. Epidemiological study on insomnia in the general population. Sleep. 1996;19:S7–15. doi: 10.1093/sleep/19.suppl_3.s7. [DOI] [PubMed] [Google Scholar]
- Ohayon M, Smirne S. Prevalence and consequences of insomnia disorders in the general population of Italy. Sleep Medicine. 2002;3:115–120. doi: 10.1016/s1389-9457(01)00158-7. [DOI] [PubMed] [Google Scholar]
- Ohnaka T, Tochihara Y, Kanda K. Body movements of the elderly during sleep and thermal conditions in bedrooms in summer. Applied Human Science. 1995;14:89–93. doi: 10.2114/ahs.14.89. [DOI] [PubMed] [Google Scholar]
- Okamoto-Mizuno K, Tsuzuki K, Mizuno K. Effects of mild heat exposure on sleep stages and body temperature in older men. International Journal of Biometeorology. 2004;49:32–36. doi: 10.1007/s00484-004-0209-3. [DOI] [PubMed] [Google Scholar]
- Pallesen S, Nordhus IH, Nielsen GH, Havik OE, Kvale G, Johnsen BH, et al. Prevalence of insomnia in the adult Norwegian population. Sleep. 2001;24:771–779. [PubMed] [Google Scholar]
- Raymann RJ, Swaab DF, Van Someren EJ. Cutaneous warming promotes sleep onset. American Journal of Physiology - Regulatory Integrative & Comparative Physiology. 2005;288:R1589–R1597. doi: 10.1152/ajpregu.00492.2004. [DOI] [PubMed] [Google Scholar]
- Rechtschaffen A, Kales A. A manual of standardized terminology, techniques and scoring system for sleep stages of human subjects. Los Angels: Brain information service/Brain research institute, University of California; 1968. [Google Scholar]
- Shaver JL, Lentz M, Landis CA, Heitkemper MM, Buchwald DS, Woods NF. Sleep, psychological distress, and stress arousal in women with fibromyalgia. Research in Nursing & Health. 1997;20:247–257. doi: 10.1002/(sici)1098-240x(199706)20:3<247::aid-nur7>3.0.co;2-i. [DOI] [PubMed] [Google Scholar]
- Sung EJ, Tochihara Y. Effects of bathing and hot footbath on sleep in winter. Journal of Physiological Anthropology and Applied Human Science. 2000;19:21–27. doi: 10.2114/jpa.19.21. [DOI] [PubMed] [Google Scholar]
- Tang NK, Harvey AG. Effects of cognitive arousal and physiological arousal on sleep perception. Sleep. 2004;27:69–78. doi: 10.1093/sleep/27.1.69. [DOI] [PubMed] [Google Scholar]
- Taylor WF, Johnson JM, O'Leary D, Park MK. Effect of high local temperature on reflex cutaneous vasodilation. Journal of Applied Physiology. 1984;57:191–196. doi: 10.1152/jappl.1984.57.1.191. [DOI] [PubMed] [Google Scholar]
- Tombaugh TN, McIntyre NJ. The mini-mental state examination: A comprehensive review. Journal of the American Geriatrics Society. 1992;40:922–935. doi: 10.1111/j.1532-5415.1992.tb01992.x. [DOI] [PubMed] [Google Scholar]
- Tsai PS, Wang SY, Wang MY, Su CT, Yang TT, Huang CJ, et al. Psychometric evaluation of the Chinese version of the Pittsburgh Sleep Quality Index (CPSQI) in primary insomnia and control subjects. Quality of Life Research. 2005;14:1943–1952. doi: 10.1007/s11136-005-4346-x. [DOI] [PubMed] [Google Scholar]
- Van Cauter E, Leproult R, Plat L. Age-related changes in slow wave sleep and REM sleep and relationship with growth hormone and cortisol levels in healthy men. JAMA. 2000;284:861–868. doi: 10.1001/jama.284.7.861. [DOI] [PubMed] [Google Scholar]
- Van Someren EJ. Circadian rhythms and sleep in human aging. Chronobiology International. 2000a;17:233–243. doi: 10.1081/cbi-100101046. [DOI] [PubMed] [Google Scholar]
- Van Someren EJ. More than a marker: Interaction between the circadian regulation of temperature and sleep, age-related changes, and treatment possibilities. Chronobiology International. 2000b;17:313–354. doi: 10.1081/cbi-100101050. [DOI] [PubMed] [Google Scholar]
- Van Someren EJ, Raymann RJ, Scherder EJ, Daanen HA, Swaab DF. Circadian and age-related modulation of thermoreception and temperature regulation: mechanisms and functional implications. Ageing Research Review. 2002;1:721–778. doi: 10.1016/s1568-1637(02)00030-2. [DOI] [PubMed] [Google Scholar]
- Vitiello MV, Larsen LH, Moe KE. Age-related sleep change: Gender and estrogen effects on the subjective-objective sleep quality relationships of healthy, noncomplaining older men and women. Journal of Psychosomatic Research. 2004;56:503–510. doi: 10.1016/S0022-3999(04)00023-6. [DOI] [PubMed] [Google Scholar]
- Zigmond AS, Snaith RP. The hospital anxiety and depression scale. Acta Psychiatrica Scandinavica. 1983;67:361–370. doi: 10.1111/j.1600-0447.1983.tb09716.x. [DOI] [PubMed] [Google Scholar]


