Abstract
Olfaction is generally assumed to be critical for survival because this sense allows animals to detect food and pheromonal cues. While the ability to sense sex pheromones [1–3] is likely to be important for insects, the contribution of general odor detection to survival is unknown. We investigated the extent to which the olfactory system confers a survival advantage on Drosophila larvae foraging for food under conditions of limited resources and competition from other larvae.
This study utilized anosmic Or83b mutant larvae, which lack the essential olfactory co-receptor OR83b [4, 5]. Although Or83b mutant larvae lack behavioral responses to odors [4, 5], they show normal viability under standard laboratory rearing conditions that feature abundant food resources. We reasoned that a survival advantage of the olfactory system might be revealed in semi-natural conditions in which Or83b mutant larvae are challenged to forage for limited food resources.
We developed a survival assay in which either 10 or 50 embryos were introduced into 100 mg of fly food in the center of a 150 mm circular arena at 1 day after egg laying (AEL). At 3 days AEL, a second 100 mg food source supplemented with 70 mg live yeast paste was introduced 70 mm away from the first food source (see Figure S1A and Experimental Procedures in the Supplement). Larvae hatch on the first food source and if this is exhausted by 3 days AEL, must locate the second food source to continue eating. Because the assay is carried out in the dark, we presume that larvae use primarily chemosensory cues to find the second food source. The number of newly emerged adult flies was counted daily from 10 to 20 days AEL and these data were used to generate cumulative eclosion rate plots, a direct measure of embryo-to-adult survival.
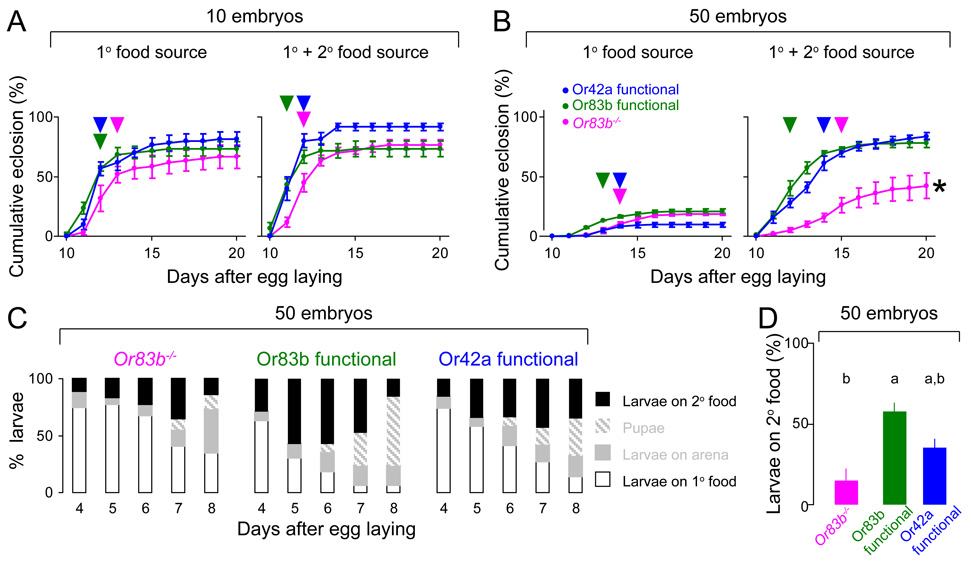
We compared the survival of anosmic Or83b mutants to control animals in which Or83b was genetically rescued in all olfactory neurons (“Or83b functional”). Normal olfactory behavior is known to be restored in Or83b functional animals [4, 5] and their survival did not differ from wild type strains in our assays (data not shown). Both anosmic Or83b mutant and control Or83b functional animals showed indistinguishable high adult survival rates at low population density whether or not a second food source was supplied (Figure 1A). This result demonstrates that Or83b mutants do not have a non-specific survival deficit independent of the known olfactory phenotype. When food was limited and larval density was high, both Or83b mutant and Or83b functional animals showed severely reduced survival (Figure 1B, left). When a second food source was provided at a distance, Or83b functional animals showed significantly higher survival than Or83b mutants (Figure 1B, right). To test whether restoring partial olfactory function sufficed for survival when a second food source was supplied, we rescued Or83b function only in the pair of olfactory neurons expressing Or42a (“Or42a functional”) [5]. Or42a functional larvae showed survival indistinguishable from Or83b functional animals in all conditions tested (Figure 1A–B), which we attribute to the broad tuning of the generalist Or42a receptor, which is sensitive to fly food [6] and a large number of fruit odors [7].
Figure 1. Anosmic Or83b mutant larvae show reduced survival and fail to exploit a secondary food source.
(A–B) Cumulative eclosion rates of Or83b functional (green), Or83b mutant (magenta), and Or42a functional (blue) flies from either 10 (A) or 50 (B) embryos in the absence (left) or presence (right) of a second food source are plotted against days AEL (mean ± S.E.M., n=6). Half-maximal eclosion rates are indicated with an arrowhead. The cumulative eclosion rate did not differ across genotypes with 10 embryos (A), but the cumulative eclosion rate of Or83b mutants was significantly lower than that of the three other genotypes when the second food source was supplied to 50 embryos (B) (ANOVA and post-hoc Tukey’s HSD test; * 99% confidence level).
(C) Distributions of Or83b mutant (n=7), Or83b functional (n=4), and Or42a functional (n=5) animals from 4 to 8 days AEL are shown in stacked bar plots.
(D) Percentage of larvae accumulating on the second food source at 5 days AEL (mean ± S.E.M.). Bars that are statistically different are labeled with different letters (ANOVA and post-hoc Tukey’s HSD test; 99% confidence level).
While Or83b functional control larvae left the first food, located the second food, and pupated, most Or83b mutant larvae failed to exploit this secondary resource (Figure 1C–D). This confirms that olfactory impairment directly impacts the survival ability of Or83b mutants. Or42a functional larvae showed intermediate migration to the second food source, suggesting that a partially functioning olfactory system is slightly less efficient than Or83b functional controls in finding the second food source. The survival of Or83b mutants was not affected by switching the location of the first and second food sources (Figure S2A) or by omitting propionic acid, a taste chemical that Or83b mutants can perceive [4], from the fly food (Figure S2B).
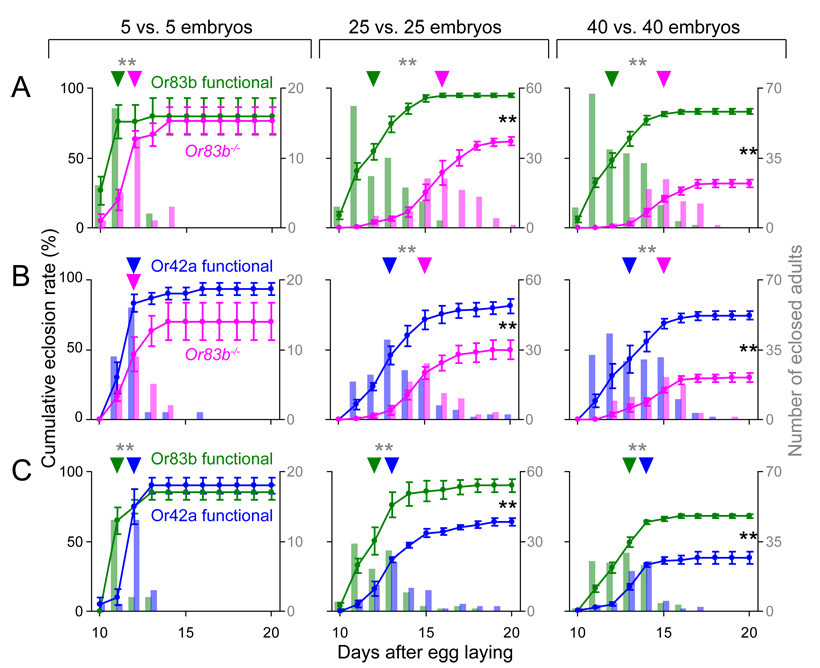
The poor eclosion rate of Or83b mutants in conditions of high density and limited food suggests that the lack of a functioning olfactory system could prove to be a disadvantage when larvae are challenged to compete. We designed a competition assay that was carried out exactly as the survival assay except that equal numbers of embryos from both wild type and Or83b mutant strains were introduced together onto 100 mg of food and supplied with a second food source at 3 days AEL (Figure S1B). Newly eclosed adults of both strains were genotyped and counted in the same manner as in the survival assay.
When 5 Or83b functional control embryos and 5 Or83b mutant embryos were introduced, both strains showed indistinguishable cumulative survival, but Or83b mutants eclosed more slowly (Figure 2A, left). When 25 or 40 embryos of each strain were introduced, both the eclosion rate and total number of eclosed adults of Or83b mutants were significantly different from that of Or83b functional animals (Figure 2A, center and right), Thus, anosmic Or83b mutants were selectively eliminated in competition with control animals at high density.
Figure 2. Survival of anosmic Or83b mutants is further reduced by competition with larvae that detect odors.
(A–C) Cumulative eclosion rates (mean ± S.E.M.) from competition of Or83b functional vs. Or83b mutants (n=6) (A), Or42a functional vs. Or83b null mutant larvae (n=6) (B) and Or42a functional vs. Or83b functional larvae (n=4) (C) for 10 (left), 50 (middle), and 80 (right) total embryos. Values of cumulative eclosion that differ statistically between genotypes are indicated with black asterisks (Student’s t-test; **P<0.01). Overlaid bar plots indicate the total number of adults eclosing on each day from 10 to 20 days AEL. Half-maximal eclosion rates for each genotype are indicated with an arrowhead and eclosion distributions that differ statistically between competing strains are indicated with gray asterisks (Kolmogorov-Smirnov; **P<0.01). Unmarked comparisons are not significant by the same tests (P>0.05).
To investigate whether a single pair of OSNs could restore larval competitiveness, we competed Or42a functional animals against Or83b mutants. When 25 or 40 embryos of each strain were introduced, the survival of Or83b mutant animals was severely reduced in the presence of Or42a functional animals (Figure 2B). Finally, we observed that Or42a functional animals showed delayed eclosion and reduced survival when competing against control Or83b functional animals at high population densities. Therefore, while larvae with one pair of functional Or42a-expressing OSNs are competitive against anosmic Or83b mutant animals, they perform less well when compared to larvae with 21 functional OSNs. Fly food is likely to emit a complex mixture of volatile chemicals, many of which would not be detected by the Or42a OSN. This suggests that a fully functional olfactory system confers additional advantages in exploiting a distant food source for survival.
Our results provide the first direct evidence that the sense of smell is necessary for effective foraging and survival to adulthood in Drosophila. Although these animals are deposited on food sources by their mothers, our results suggest that the larval sense of smell is indeed useful for dispersal to find food. In conditions of high population density and limited food, the loss of a functional olfactory system selectively impairs survival. We believe this is because resources at the site of oviposition are depleted under such conditions, making it necessary for animals to disperse to obtain food. Such competitive conditions may be faced by these animals in their natural environment [8, 9]. Although our experimental conditions are artificial, we speculate that many insects face resource scarcity in their natural environment and therefore our findings are likely to be relevant beyond laboratory-reared fruit flies. We suggest that impairment of olfactory function will reduce insect competitiveness and thus may be useful in insect control strategies that target the sense of smell.
Supplementary Material
Supplemental data are available at http://www.current-biology.com/cgi/content/full/XXX
Acknowledgments
We thank Emilie Russler for expert technical assistance. This research was supported by National Institutes of Health grant RO1 DC006711. KA designed and carried out experiments, analyzed data, and supervised VP, who carried out experiments under the auspices of the Bard-Rockefeller Semester in Science program. LBV supervised the project and wrote the paper.
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
References Cited
- 1.Moore AJ, Gowaty PA, Wallin WG, Moore PJ. Sexual conflict and the evolution of female mate choice and male social dominance. Proc Biol Sci. 2001;268:517–523. doi: 10.1098/rspb.2000.1399. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Widemo F, Johansson BG. Male-male pheromone signalling in a lekking Drosophila. Proc Biol Sci. 2006;273:713–717. doi: 10.1098/rspb.2005.3379. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Grillet M, Dartevelle L, Ferveur JF. A Drosophila male pheromone affects female sexual receptivity. Proc Biol Sci. 2006;273:315–323. doi: 10.1098/rspb.2005.3332. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Larsson MC, Domingos AI, Jones WD, Chiappe ME, Amrein H, Vosshall LB. Or83b encodes a broadly expressed odorant receptor essential for Drosophila olfaction. Neuron. 2004;43:703–714. doi: 10.1016/j.neuron.2004.08.019. [DOI] [PubMed] [Google Scholar]
- 5.Fishilevich E, Domingos AI, Asahina K, Naef F, Vosshall LB, Louis M. Chemotaxis behavior mediated by single larval olfactory neurons in Drosophila. Curr Biol. 2005;15:2086–2096. doi: 10.1016/j.cub.2005.11.016. [DOI] [PubMed] [Google Scholar]
- 6.Ditzen M, Pellegrino M, Vosshall LB. Insect odorant receptors are molecular targets of the insect repellent DEET. Science. 2008;319:1838–1842. doi: 10.1126/science.1153121. [DOI] [PubMed] [Google Scholar]
- 7.Kreher SA, Kwon JY, Carlson JR. The molecular basis of odor coding in the Drosophila larva. Neuron. 2005;46:445–456. doi: 10.1016/j.neuron.2005.04.007. [DOI] [PubMed] [Google Scholar]
- 8.Lindsay SL. Food preferences of Drosophila larvae. American Naturalist. 1958;92:279–285. [Google Scholar]
- 9.Fogleman JC, Starmer WT, Heed WB. Larval selectivity for yeast species by Drosophila mojavensis in natural substrates. Proc Natl Acad Sci U S A. 1981;78:4435–4439. doi: 10.1073/pnas.78.7.4435. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Supplemental data are available at http://www.current-biology.com/cgi/content/full/XXX