Abstract
Aging affects the function and structure of arteries and increases the risk of cardiovascular diseases (CVD). In healthy sedentary adults, aging is associated with increased stiffness (reduced compliance) of large elastic arteries; impaired vascular endothelial function, including reductions in endothelium-dependent dilation (EDD), release of tissue-type plasminogen activator (fibrinolytic capacity) and endothelial progenitor cell number and function; increased intima-media wall thickness (IMT); and peripheral vasoconstriction (decreased basal leg blood flow). Habitual physical activity/increased aerobic exercise capacity is associated with reduced risk of CVD. Compared with their sedentary peers, adults who regularly perform aerobic exercise demonstrate smaller or no age-associated increases in large elastic artery stiffness, reductions in vascular endothelial function, and increases in femoral artery IMT. A short-term, moderate-intensity aerobic exercise intervention (brisk daily walking for 12 wk) improves carotid artery compliance and can restore vascular endothelial function in previously sedentary middle-aged and older adults. Reduced oxidative stress may be an important mechanism contributing to these effects. Habitual resistance exercise increases (high-intensity) or does not affect (moderate-intensity) large elastic artery stiffness, and prevents/restores the age-associated reduction in basal leg blood flow independent of changes in leg fat-free mass. Habitual exercise favorably modulates several expressions of arterial aging, thus preserving vascular function and possibly reducing the risk of CVD.
Keywords: arterial stiffness, endothelial function, intima-media thickness, blood flow
we have known that aging is associated with changes in the function and structure of arteries since the late 1800s (84). Although of interest to physiologists, early observations were not of major biomedical importance because of the low prevalence of cardiovascular diseases (CVD) at the time. Today, CVD is a/the leading cause of morbidity and mortality in modern societies, and this is largely attributable to disorders of the arteries (4). Advancing age is a major risk factor for CVD and appears to exert its pathological influence primarily via adverse effects on arteries (54). Thus human aging is associated with arterial dysfunction and an increased risk of clinical vascular disease.
In contrast to age, regularly performed physical exercise in general, and aerobic exercise/fitness in particular, are associated with enhanced vascular function and reduced risk of CVD (7, 35, 99). The influence of regular resistance exercise (i.e., strength training, weight lifting, etc.) on arteries and CVD is less clear. However, the importance of resistance training in preserving strength suggests that it can play a significant role in the maintenance of functional capacity with aging. Taken together, these observations suggest that habitual exercise may exert its beneficial effects on physiological function and risk of CVD with aging at least in part by minimizing or preventing adverse changes in the structure and function of arteries.
This brief review will focus on the modulatory influence of regular exercise on selective changes in arterial function and structure with aging that we have studied over the last decade, especially large elastic artery stiffness and vascular endothelial function. Because of the larger amount of data available, the effects of aerobic exercise will be discussed in some detail. The influence of resistance exercise and combined aerobic and resistance training will be described where information exists. We will emphasize the results of investigations performed on healthy adult humans, using findings obtained in experimental animals and patients with CVD to extend insight into the underlying cellular and molecular mechanisms. Detailed descriptions of methodology and the integrative physiological mechanisms mediating arterial aging are available in the original research articles cited and the other brief reviews in this series. A discussion of aerobic training and age-associated changes in active muscle blood flow during acute exercise is available elsewhere (80, 119).
LARGE ELASTIC ARTERY STIFFNESS
A number of methodologies have been used to assess the elastic properties of arteries in humans. Pulse wave velocity (PWV) has been most frequently used as a measure of arterial stiffness (105, 113). Augmentation index, an index of wave reflection that can be influenced by arterial stiffness, also has been employed (105, 113). Arterial compliance has been measured by simultaneous application of ultrasound imaging and applanation tonometry (67, 106).
Changes with Age
Stiffening of large elastic arteries (e.g., aorta and carotid artery) with aging was originally documented by Charles Roy in 1881 (84) and became the basis for William Osler's famous saying that “man is as old as his arteries” (76). Based on cross-sectional observations, large elastic artery stiffness is progressively greater (compliance is lower) with advancing age even in healthy groups of men and women (5, 105, 113). We find 40–50% differences in large elastic artery stiffness and compliance between ∼age 25 and 75 yr in healthy adults without clinical disease or major coronary risk factors (67, 106). Based on aortic PWV, increases in large elastic artery stiffness with age appear to be similar in men and women (61, 94). In contrast to large elastic arteries, peripheral arteries do not obviously stiffen with aging in healthy humans (67, 105, 106).
Changes in the composition of the arterial wall, including fragmentation of elastin and increases in collagen deposition, collagen cross-linking (associated with greater accumulation of advanced glycation end products), interstitial cell adhesion molecules, and growth factors, as well as vascular smooth muscle cell hypertrophy, are believed to be important mechanisms in mediating arterial stiffness with aging (53, 74). Functional changes that result in increased vascular smooth muscle tone such as increased sympathetic nervous system activity and bioactivity of locally synthesized vasoconstrictor molecules (e.g., endothelin-1) and reduced endothelial dilator production (111), perhaps linked to oxidative stress (70), also likely contribute. Arterial stiffening with aging does not appear to depend on the presence of atherosclerosis because it is observed in rural Chinese populations who have a low prevalence of atherosclerotic diseases (5), in rigorously screened healthy men and women (67, 106), and in animal species resistant to the development of atherosclerosis (36, 83).
Influence of Habitual Exercise
The initial clue that habitual aerobic exercise might attenuate age-associated increases in large elastic artery stiffness was an observation from the Baltimore Longitudinal Study of Aging that older male endurance athletes demonstrated lower aortic PWV, augmentation index and systolic blood pressure than their sedentary peers (113). We subsequently found that aortic PWV, augmentation index, and 24-h systolic and pulse pressure were greater in postmenopausal compared with premenopausal sedentary, but not endurance exercise-trained, healthy women (90, 105). Results of follow-up investigations showed that age-associated reductions in carotid artery compliance were only ∼50% as great in healthy men and women who performed habitual aerobic exercise compared with sedentary adults (67, 106) (Fig. 1). Daily brisk walking for ∼3 mo improved carotid artery compliance in previously sedentary middle-aged/older men (106) (Fig. 1) and postmenopausal women (69) to levels observed in age-matched endurance exercise-trained adults, suggesting that even moderate aerobic exercise may produce an optimal effect.
Fig. 1.
Age-associated reductions in carotid artery compliance in sedentary and endurance exercise-trained men (left) and improvements in carotid artery compliance with 3 mo of aerobic exercise training (right). MA, middle aged. *P < 0.05 vs. young within same activity group. ‡P < 0.05 vs. sedentary of same age group. †P < 0.01 vs. before training. [From Tanaka et al. (106).]
There are limited data as to the mechanisms mediating these favorable modulatory effects of habitual aerobic exercise. One possibility is that regular exercise minimizes or reverses age-related structural changes in the arterial wall. This may contribute in settings of prolonged exercise training; however, there are no data available to support this mechanism. Reduced stiffness with aging in exercise-trained rodents is not dependent on structural changes in elastin and collagen (74). Functional adaptations may be involved, especially in response to shorter term exercise training (67, 106). Expression of genes associated with local vasodilatory signaling are modified in aorta of exercise-trained rats (60). Moreover, ascorbic acid (vitamin C) improves carotid artery compliance in sedentary, but not endurance exercise-trained postmenopausal women (70), suggesting reduced oxidative stress in the habitually exercising state. Improvements in carotid artery compliance in response to regular moderate-intensity exercise are independent of baseline compliance and changes in conventional risk factors for CVD, body composition, and aerobic fitness (67, 106).
In contrast to findings on healthy middle-aged and older men and women, regular aerobic exercise may not improve arterial stiffness in certain groups of adults with chronically elevated arterial blood pressure (32, 91). Moreover, higher intensity resistance exercise training is associated with greater rather than lower large elastic artery stiffness in young and middle-aged adults (6, 62), although the addition of aerobic exercise may counteract this negative influence (50). Moderate-intensity resistance exercise training does not influence arterial stiffness in healthy young men (82) or postmenopausal women (10).
VASCULAR ENDOTHELIAL FUNCTION
Endothelium-Dependent Dilation
Endothelium-Dependent Dilation (EDD) has been assessed primarily using two models: increases in forearm blood flow to intra-brachial artery infusions of endothelial dilators like acetylcholine (20) and brachial artery flow-mediated dilation (12). The former examines resistance vessel EDD in response to a pharmacological stimulus, whereas the latter model assesses large conduit artery EDD in response to the physiological stimulus of increased shear stress.
Changes with age.
Both expressions of EDD demonstrate reductions with age, even in healthy sedentary adults, although the temporal pattern of decline may differ (11, 103). The decrease in EDD may be delayed to an older absolute age in women than men, possibly because of menopause-associated estrogen deficiency (11, 103). In contrast to EDD, endothelium-independent dilation is not reduced with aging in healthy adults (11, 103).
In humans, the age-associated decline in EDD is mediated in part by an oxidative stress-dependent reduction in nitric oxide (NO) production and bioavailability (27, 30, 101). Based on data from experimental animals, the reduction in EDD is not consistently associated with reductions in the expression or activity of endothelial NO synthase (eNOS) (95, 115), the enzyme responsible for endothelial production of NO. Rather, superoxide reaction with NO and oxidation of tetrahydrobiopterin (BH4), the key cofactor for NO synthase production of NO, appear to be important mechanisms (16, 31, 39, 40, 57). Altered cyclooxygenase signaling (e.g., increased bioactivity of vasoconstrictor prostaglandins) also may contribute to impaired EDD with aging (96, 102). Other mechanisms discussed in this highlighted series include inflammation, arginase, and p66Shc signaling (9, 14, 86).
Influence of habitual exercise.
EDD is greater in middle-aged and older men who regularly perform aerobic exercise compared with their sedentary peers (17, 30, 100), and it is similar to (17, 30) or modestly lower than (100) that of young healthy sedentary and exercising men (Fig. 2). Three months of moderate-intensity aerobic exercise training improves EDD in previously sedentary middle-aged and older healthy men (17) (Fig. 2) and in patients with the metabolic syndrome but no clinical disease (58). It is unknown whether aerobic exercise training is associated with enhanced EDD with aging in women, although EDD is increased after a single bout of aerobic exercise in postmenopausal, but not premenopausal, women (41). Aerobic exercise training improves EDD in middle-aged and older patients with CVD (38), but consistent effects have not been observed in healthy young men and women with normal baseline function (13, 17, 51, 65). Moderate-intensity resistance exercise training has no obvious effect on EDD in healthy postmenopausal women (10).
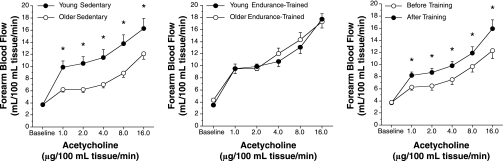
Fig. 2.
Forearm blood flow (FBF) responses to intrabrachial artery infusion of acetylcholine (i.e., endothelium-dependent dilation) in sedentary (left) and endurance exercise-trained (middle) healthy young and middle-aged/older men, and before and after an aerobic exercise intervention in middle-aged/older men (right). *P < 0.05. [From DeSouza et al. (17).]
The greater EDD in middle-aged and older men who regularly perform aerobic exercise is mediated in part by enhanced NO bioavailability (100). Tonic NO production also appears to be enhanced in these men (100) (Fig. 3), as well as in previously sedentary middle-aged and older men who walk for 12 wk (Fig. 3). Aerobic training-induced improvements in EDD and NO bioavailability are associated with an increase in eNOS gene and protein expression in older rats (95) and an increase in eNOS protein and serine 1177 phosphorylation of eNOS in patients with coronary artery disease (37), but no information is available on healthy older humans. The production and bioactivity of other endothelial-derived vasodilators such as prostacyclin also are increased with aerobic exercise training in older animals (96).
Fig. 3.
Reductions in FBF in response to intra-brachial artery infusion of NG-monomethyl l-arginine (l-NMMA) (i.e., tonic nitric oxide release) in sedentary (left) and endurance exercise-trained (middle) healthy young and middle-aged/older men, and before and after an aerobic exercise intervention in middle-aged/older men (right). *P < 0.05.
The greater NO-mediated improvements in EDD in middle-aged and older adults who perform aerobic exercise may be secondary to reduced oxidative stress. Acute administration of supraphysiological concentrations of antioxidants (vitamin C) improves EDD in sedentary, but not in endurance exercise-trained, men (30, 100). Habitually exercising middle-aged and older adults have lower malondialdehyde and higher total oxyradical scavenging capacity compared with age-matched sedentary adults, and this is related to enhanced EDD (34). In patients with coronary disease, aerobic exercise training is associated with decreased production of reactive oxygen species and expression of the oxidant-producing enzyme, NADPH oxidase, in the mammary artery (1). That acute BH4 administration restores EDD in older sedentary adults without affecting EDD in their exercising peers suggests that the lower oxidative stress-related increase in NO bioavailability in older adults who exercise may be linked to reduced oxidation and enhanced bioactivity of BH4 (31).
Exercise-induced increases in shear stress as a result of increased blood flow and pulse pressure to both active and nonactive limbs are thought to be important stimuli for the molecular endothelial adaptations to aerobic training because improvements in EDD are observed in arteries outside the exercising limbs (35, 52, 109). Increases in EDD in response to an aerobic exercise intervention are not dependent on improvements in conventional risk factors for CVD and are not related to increases in maximal aerobic exercise capacity (17).
Endothelial Fibrinolytic Capacity
The hemostatic mechanism responsible for the proteolytic degradation of intravascular fibrin is the fibrinolytic system. This enzymatic pathway maintains vascular patency by converting the inactive proenzyme plasminogen to the active enzyme, plasmin, which lyses fibrin into soluble degradation products (77). The vascular endothelium plays a prominent role in modulating fibrinolytic system function through its synthesis and release of tissue-type plasminogen activator (t-PA), the primary plasminogen activator, and its biological inhibitor plasminogen activator inhibitor-1 (PAI-1) (59). t-PA is the key enzyme involved in initiating an endogenous fibrinolytic response as a result of its ability to preferentially activate plasminogen on the surface of developing thrombi (29). The capacity of the endothelium to rapidly release t-PA from intracellular storage pools, rather than circulating plasma concentrations, determines the efficacy of endogenous fibrinolysis (78, 114).
t-PA is released by endothelial cells in response to a variety of stimuli including thrombin, hypoxia, and inflammatory cytokines (77, 104). Endothelial agonists such as substance P, bradykinin and methacholine also stimulate the release of t-PA in a dose-dependent fashion. The use of these vasoactive agents at subsystemic, locally active concentrations in the forearm coupled with measures of blood flow and arterial-venous differences in t-PA provide a reproducible method for directly assessing the rate of t-PA release from the endothelium in vivo (8, 49, 93).
Changes with age.
The capacity of the endothelium to release t-PA declines with age in healthy sedentary men as indicated by a marked reduction in endothelial t-PA release across the forearm vasculature in response to bradykinin (93). The underlying mechanisms are unknown. Data from animal models suggest that t-PA expression may decline with age, resulting in reduced intracellular bioavailability (2, 79). Increased oxidative stress also may be a contributing factor (116). Indeed, oxygen free radicals inhibit t-PA release from endothelial cells (92). In contrast to men, endothelial t-PA release does not differ in healthy sedentary premenopausal and postmenopausal women (45, 46), and it is greater in middle-aged and older women compared with men of similar age (97).
Influence of habitual exercise.
In contrast to their healthy sedentary peers, aerobic exercise-trained men do not demonstrate an age-related decline in the capacity of the endothelium to release t-PA (93) (Fig. 4). Consistent with this, 12 wk of moderate-intensity aerobic exercise training (daily walking) improves endothelial t-PA release in previously sedentary middle-aged and older men to levels of young men, independent of changes in body composition, CVD risk factors and maximal exercise capacity (93) (Fig. 4). There is no information regarding the effects of habitual resistance exercise on endothelial t-PA release.
Fig. 4.
Net release rate of tissue-type plasminogen activator (t-PA) antigen across the forearm in response to intrabrachial artery infusion of bradykinin in sedentary (left) and endurance exercise-trained (middle) healthy young and middle-aged/older men, and before and after an aerobic exercise intervention in middle-aged/older men (right). *P < 0.05. [From Smith et al. (93).]
The mechanisms mediating the beneficial effects of regular aerobic exercise on endothelial t-PA release with aging in healthy men have not been established. In cultured endothelial cells, shear stress induces t-PA transcription and protein synthesis (18, 19). Exercise-training associated increases in cytoplasmic calcium levels may potentiate t-PA release (56). Reduced oxidative stress also could contribute, although no data are available to support this possibility.
Endothelial Progenitor Cells
Currently there are no clear, consensus criteria for identifying bone marrow-derived circulating endothelial progenitor cells (EPC). Nevertheless, EPC are believed to play a critical role in maintaining, repairing, and regenerating the vascular endothelial monolayer and restoring functional activity (22, 28, 112). Reduced number and impaired function of EPC are associated with endothelial dysfunction (43, 73) and increased risk and severity of CVD (43, 88).
Changes with age.
Aging is associated with reduced number and function of circulating EPCs (21, 42, 47, 87, 110). EPC colony-forming capacity and migratory activity decline with age even in healthy men (42, 47), and they are related to both reductions in EDD and increased Framingham risk score (42, 43). EPC colony formation and migration are greater in middle-aged and older women compared with men (44), suggesting that EPC function may be better preserved with aging in women.
Influence of habitual exercise.
In patients with cardiovascular risk factors and CVD, aerobic exercise intervention increases EPC number (55, 85, 98), and this is strongly related to improvements in EDD (98). Increases in both EPC colony forming and migratory capacity are observed after 12 wk of brisk walking in the absence of changes in body composition, CVD risk factors, and maximal exercise capacity in previously sedentary healthy middle-aged and/or older men (47), although unchanged EPC number also has been reported after endurance training (110). In general, these findings support the idea that increases in EPC number and function may contribute to improvements in vascular endothelial function with regular aerobic exercise in middle-aged and older adults.
ARTERIAL WALL THICKNESS
Changes with Age
The intima-media wall thickness (IMT) of the carotid and femoral arteries are independent predictors of future CVD (75), and both increase with age even in healthy men and women (68, 72, 89, 107). The increases in IMT are mediated by thickening of both the intimal and medial layers (81, 117). In healthy adults, these age-associated changes likely reflect arterial remodeling in response to intravascular hemodynamic changes related in part to increases in local arterial blood pressure (107). Lumen diameter also widens as part of this remodeling (54, 107). In patients with major risk factors or clinical CVD, increased IMT also may reflect atherosclerotic plaques (118).
Influence of Habitual Exercise
Early cross-sectional studies using questionnaires to assess habitual physical activity reported lower, not different and even greater carotid IMT in physically active compared with sedentary adults (15, 33). In rigorously screened healthy men and women in whom maximal aerobic exercise capacity is objectively assessed, the age-associated increase in carotid IMT does not differ among sedentary, moderately active, and endurance exercise-trained adults (68, 108). In agreement with these cross-sectional observations, carotid IMT is unaffected by 3 mo of daily walking (68, 108). Similar findings have been reported for resistance exercise training (62, 63). The absence of habitual exercise effects may be explained by the fact that carotid artery pressure, a strong independent predictor of carotid IMT in healthy adults (107), is not associated with exercise status (62, 63, 68, 108). Carotid lumen diameter does not differ with age among groups differing in exercise behavior, nor does it change in response to aerobic or resistance exercise interventions (62, 63, 68, 108).
In contrast to carotid IMT, femoral IMT is smaller in both moderately active and endurance exercise-trained compared with sedentary healthy middle-aged and older adults, and the age-associated increases in femoral IMT are smaller in exercising than in sedentary adults (25, 68). Femoral artery IMT is reduced after 12 wk of daily walking in middle-aged and older men in the absence of changes in CVD risk factors (25). The greatest modulatory effects of habitual aerobic exercise on femoral IMT are observed in older adults (72). Habitual exercise also is associated with greater femoral lumen diameter (25, 68). Together, these changes may reflect expansive arterial remodeling to normalize wall stress in response to increases in femoral blood flow evoked by daily aerobic leg exercise (25). They are not related to changes in the aerobic exercise stimulus, exercise capacity or CVD risk factors (25). Femoral IMT is not affected by resistance exercise training (3, 64).
PERIPHERAL VASOCONSTRICTION (REDUCTIONS IN BASAL LEG BLOOD FLOW)
Changes with Age
Basal leg blood flow decreases linearly between the ages of 25 and 65 yr in healthy men (24, 48, 64) and is 25–30% lower in postmenopausal compared with premenopausal healthy women (66, 69). These age-associated reductions in blood flow are mediated by corresponding decreases in vascular conductance (23, 24, 48, 64, 66, 69). These changes are most closely related to decreases in estimated leg oxygen consumption, but they are not completely explained by reductions in leg fat-free mass, i.e., blood flow per kg fat-free mass decreases with age (23, 26, 69). In men, the reductions in leg blood flow and vascular conductance with age are mediated by increased sympathetic α-adrenergic tone (26), perhaps linked to increased oxidative stress (48), whereas the latter appears to play a significant but lesser role in women (71).
Influence of Habitual Exercise
Despite the apparent mechanistic role for oxidative stress and evidence that regular aerobic exercise is associated with reduced vascular oxidative stress, reductions in basal leg blood flow and vascular conductance with age are similar in healthy sedentary, moderately active, and endurance exercise-trained men (24, 25). In contrast, reductions in basal leg blood flow and vascular conductance are not observed in middle-aged compared with young resistance exercise-trained men, independent of leg fat-free mass (64). Consistent with this, 13 wk of resistance exercise that has no effect on leg fat-free mass or cardiac output restores basal leg blood flow in healthy middle-aged and older adults (3). Thus resistance exercise training may be associated with preserved basal whole-leg blood flow with aging.
SUMMARY AND CONCLUSIONS
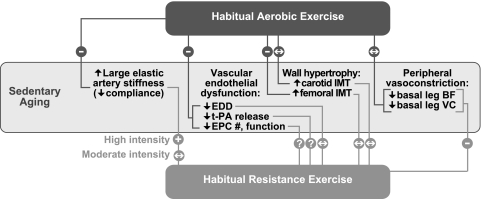
The influence of habitual aerobic and resistance exercise on arterial aging is summarized in Fig. 5. Regular aerobic exercise is associated with smaller increases in arterial stiffness with aging and improves stiffness in previously sedentary middle-aged and older healthy men and women. Reduced oxidative stress may contribute to these effects, but little insight is available as to mechanisms of action. Higher, but not moderate, intensity resistance exercise may exert a negative influence on arterial stiffness when performed without aerobic training.
Fig. 5.
Summary of modulatory effects of habitual aerobic and resistance exercise on selective features of arterial aging in healthy adults. −, inhibition of; +, exacerbation of; ↔, no influence on the effects of sedentary aging; ?, no published data available; EDD, endothelium-dependent dilation; t-PA, tissue-type plasminogen activator (fibrinolytic capacity); EPC, endothelial progenitor cells; IMT, intima-media thickness; BF, blood flow; VC, vascular conductance.
Habitual aerobic exercise acts to preserve several vascular endothelial functions that decline with age, and it can restore lost function in older adults, although definitive data only are available on men. Reduced oxidative stress and, in the case of EDD, greater NO bioavailability associated with increased eNOS expression/activation and BH4 bioactivity, may be involved, but more mechanistic insight is needed.
Carotid IMT is not affected by chronic exercise status, but femoral IMT is smaller in aerobic exercise-trained adults across age, probably as part of expansive arterial remodeling in response to acute exercise-related changes in intravascular wall stress. Resistance, but not aerobic, exercise training may preserve and restore basal whole leg blood flow with aging.
Collectively, these findings support the view that regular physical activity, particularly large-muscle aerobic exercise, is an effective strategy for combating several adverse physiological changes associated with arterial aging, especially increases in large-elastic artery stiffness and vascular endothelial dysfunction. This modulatory influence may explain in part why physically active and aerobically fit middle-aged and older adults have increased cardiovascular functional capacity and a lower prevalence of CVD than sedentary adults.
FUTURE DIRECTIONS
There is extensive need for both applied/clinical and basic research on this topic. Because regular resistance exercise is now recognized as a fundamental strategy for maintenance of muscle strength with advancing age, a much better understanding is needed of how this type of training, alone and combined with complementary aerobic and perhaps other forms of physical exercise, affects the function and structure of arteries as we age. A practical, but important issue is the volume and intensity of exercise required to minimize arterial aging, and whether this is function-specific. For example, our work indicates that 12 wk of brisk walking can restore EDD in some middle-aged and older adults, but it did not restore carotid artery compliance (17, 106). Little is known about the efficacy of habitual exercise for treating vascular dysfunction in adults over 70–80 yr of age and those with clinical CVD or risk factors for CVD. In some cases (e.g., aging, aerobic exercise and EDD), no data are available on women. Moreover, in clinical practice exercise is not prescribed in isolation but rather with other lifestyle (e.g., weight loss; low-fat diet; smoking cessation) and pharmacological strategies. Does exercise exert additive or synergistic effects with other treatments or is there redundancy? Cross-sectional (comparison of sedentary and exercise-trained adults), exercise intervention, and longitudinal (following sedentary and exercising adults over time) studies all can be used to provide insight. In particular, more information is needed as to the effects of aging and habitual exercise on EPC-mediated (or assisted) vascular repair.
Presently, we have some understanding, albeit incomplete, of the integrative (whole artery to molecular) physiological mechanisms underlying the effects of regular aerobic exercise on EDD with aging. In comparison, very little is known about the mechanisms by which habitual exercise influences arterial stiffness and other functions as we age. The main limitation with human investigations is a lack of access to internal (nonperipheral) arteries for tissue sampling and manipulation of signaling pathways, particularly in healthy adults. In vivo and in vitro animal and vascular cell models, as well as innovative methods in humans, will be needed to pursue these issues. Mechanisms previously implicated in age- and CVD-related physiological dysfunction represent potential targets as to how exercise may impact arterial aging. A major candidate in this context is inflammation, including its interaction with oxidative stress. The roles played by exercise-induced changes in the bioactivity of novel signaling molecules and the transcriptional and posttranscriptional regulation of the genes involved in mediating functional improvements represent important goals of future research in this area.
CLINICAL SIGNIFICANCE
As the populations in the United States and other modern societies age, greater resources will be required to meet health care needs. The associated costs and demand pose a real and increasingly imminent threat to our health care systems. A key strategy will be delaying the onset and development of age-associated physiological dysfunction and disease. Habitual exercise, combined with other lifestyle and pharmacological interventions, can be a powerful tool to achieve this goal. Along with cancer, CVDs are the leading cause of morbidity and premature mortality in industrialized nations. Much of the etiology of CVD is attributable to disorders of the arteries. As such, the use of regular exercise to delay, slow and, in some cases, prevent the development of arterial dysfunction and disease can play a key role in promoting healthy aging.
GRANTS
This work was supported by National Institutes of Health Grants AG-006537, AG-00847, AG-013038, AG-022241, AG-029337, AG-000279, AG-20966, HL-03840, HL-068030, HL-076434, and RR-00051, and by American Heart Association Grants 0060430Z, 0110221Z, 0255921Z, and 0555678Z.
REFERENCES
- 1.Adams V, Linke A, Krankel N, Erbs S, Gielen S, Mobius-Winkler S, Gummert JF, Mohr FW, Schuler G, Hambrecht R. Impact of regular physical activity on the NAD(P)H oxidase and angiotensin receptor system in patients with coronary artery disease. Circulation 111: 555–562, 2005. [DOI] [PubMed] [Google Scholar]
- 2.Ahn MY, Zhang ZG, Zhang L, Chopp M. The effect of age on expression of endogenous plasminogen activators after focal cerebral ischemia in mice. Brain Res 833: 112–116, 1999. [DOI] [PubMed] [Google Scholar]
- 3.Anton MM, Cortez-Cooper MY, DeVan AE, Neidre DB, Cook JN, Tanaka H. Resistance training increases basal limb blood flow and vascular conductance in aging humans. J Appl Physiol 101: 1351–1355, 2006. [DOI] [PubMed] [Google Scholar]
- 4.Amercian Heart Association. Heart and Stroke Facts: 2006 Statistical Supplement. Dallas, TX: American Heart Association, 2007.
- 5.Avolio AP, Deng FQ, Li WQ, Luo YF, Huang ZD, Xing LF, O'Rourke MF. Effects of aging on arterial distensibility in populations with high and low prevalence of hypertension: comparison between urban and rural communities in China. Circulation 71: 202–210, 1985. [DOI] [PubMed] [Google Scholar]
- 6.Bertovic DA, Waddell TK, Gatzka CD, Cameron JD, Dart AM, Kingwell BA. Muscular strength training is associated with low arterial compliance and high pulse pressure. Hypertension 33: 1385–1391, 1999. [DOI] [PubMed] [Google Scholar]
- 7.Blair SN, Kohl HW 3rd, Paffenbarger RS Jr, Clark DG, Cooper KH, Gibbons LW. Physical fitness and all-cause mortality A prospective study of healthy men and women. JAMA 262: 2395–2401, 1989. [DOI] [PubMed] [Google Scholar]
- 8.Brown NJ, Nadeau JH, Vaughan DE. Selective stimulation of tissue-type plasminogen activator (t-PA) in vivo by infusion of bradykinin. Thromb Haemost 77: 522–525, 1997. [PubMed] [Google Scholar]
- 9.Camici GG, Cosentino F, Tanner FC, Lüscher TF. P66Shc deletion and age-associated arterial dysfunction. J Appl Physiol; doi: 10.1152/japplphysiol.90579.2008. [DOI] [PubMed]
- 10.Casey DP, Pierce GL, Howe KS, Mering MC, Braith RW. Effect of resistance training on arterial wave reflection and brachial artery reactivity in normotensive postmenopausal women. Eur J Appl Physiol 100: 403–408, 2007. [DOI] [PubMed] [Google Scholar]
- 11.Celermajer D, Sorensen K, Bull C, Robinson J, Deanfield J. Endothelium-dependent dilation in the systemic arteries of asymptomatic subjects relates to coronary risk factors and their interaction. J Am Coll Cardiol 24: 1468–1474, 1994. [DOI] [PubMed] [Google Scholar]
- 12.Celermajer DS, Sorensen KE, Gooch VM, Spiegelhalter DJ, Miller OI, Sullivan ID, Lloyd JK, Deanfield JE. Non-invasive detection of endothelial dysfunction in children and adults at risk of atherosclerosis. Lancet 340: 1111–1115, 1992. [DOI] [PubMed] [Google Scholar]
- 13.Clarkson P, Montgomery HE, Mullen MJ, Donald AE, Powe AJ, Bull T, Jubb M, World M, Deanfield JE. Exercise training enhances endothelial function in young men. J Am Coll Cardiol 33: 1379–1385, 1999. [DOI] [PubMed] [Google Scholar]
- 14.Csiszar A, Lakatta EG, Ungvari Z. Inflammation and arterial dysfunction in aging: role of NF-kB. J Appl Physiol (July 3, 2008). doi: 10.1152/japplphysiol.90470.2008. [DOI] [PMC free article] [PubMed]
- 15.Cuspidi C, Lonati L, Sampieri L, Leonetti G, Zanchetti A. Similarities and differences in structural and functional changes of left ventricle and carotid arteries in young borderline hypertensives and in athletes. J Hypertens 14: 759–764, 1996. [DOI] [PubMed] [Google Scholar]
- 16.Delp MD, Behnke BJ, Spier SA, Wu G, Muller-Delp JM. Ageing diminishes endothelium-dependent vasodilatation and tetrahydrobiopterin content in rat skeletal muscle arterioles. J Physiol 586: 1161–1168, 2008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.DeSouza CA, Shapiro LF, Clevenger CM, Dinenno FA, Monahan KD, Tanaka H, Seals DR. Regular aerobic exercise prevents and restores age-related declines in endothelium-dependent vasodilation in healthy men. Circulation 102: 1351–1357, 2000. [DOI] [PubMed] [Google Scholar]
- 18.Diamond SL, Eskin SG, McIntire LV. Fluid flow stimulates tissue plasminogen activator secretion by cultured human endothelial cells. Science 243: 1483–1485, 1989. [DOI] [PubMed] [Google Scholar]
- 19.Diamond SL, Sharefkin JB, Dieffenbach C, Frasier-Scott K, McIntire LV, Eskin SG. Tissue plasminogen activator messenger RNA levels increase in cultured human endothelial cells exposed to laminar shear stress. J Cell Physiol 143: 364–371, 1990. [DOI] [PubMed] [Google Scholar]
- 20.Dietz NM, Rivera JM, Eggener SE, Fix RT, Warner DO, Joyner MJ. Nitric oxide contributes to the rise in forearm blood flow during mental stress in humans. J Physiol 480: 361–368, 1994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Dimmeler S, Vasa-Nicotera M. Aging of progenitor cells: limitation for regenerative capacity? J Am Coll Cardiol 42: 2081–2082, 2003. [DOI] [PubMed] [Google Scholar]
- 22.Dimmeler S, Zeiher AM. Vascular repair by circulating endothelial progenitor cells: the missing link in atherosclerosis? J Mol Med 82: 671–677, 2004. [DOI] [PubMed] [Google Scholar]
- 23.Dinenno FA, Jones PP, Seals DR, Tanaka H. Limb blood flow and vascular conductance are reduced with age in healthy humans: relation to elevations in sympathetic nerve activity and declines in oxygen demand. Circulation 100: 164–170, 1999. [DOI] [PubMed] [Google Scholar]
- 24.Dinenno FA, Seals DR, DeSouza CA, Tanaka H. Age-related decreases in basal limb blood flow in humans: time course, determinants and habitual exercise effects. J Physiol 531: 573–579, 2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Dinenno FA, Tanaka H, Monahan KD, Clevenger CM, Eskurza I, DeSouza CA, Seals DR. Regular endurance exercise induces expansive arterial remodelling in the trained limbs of healthy men. J Physiol 534: 287–295, 2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Dinenno FA, Tanaka H, Stauffer BL, Seals DR. Reductions in basal limb blood flow and vascular conductance with human ageing: role for augmented alpha-adrenergic vasoconstriction. J Physiol 536: 977–983, 2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Donato AJ, Eskurza I, Silver AE, Levy AS, Pierce GL, Gates PE, Seals DR. Direct evidence of endothelial oxidative stress with aging in humans: relation to impaired endothelium-dependent dilation and upregulation of nuclear factor-kappaB. Circ Res 100: 1659–1666, 2007. [DOI] [PubMed] [Google Scholar]
- 28.Dzau VJ, Gnecchi M, Pachori AS, Morello F, Melo LG. Therapeutic potential of endothelial progenitor cells in cardiovascular diseases. Hypertension 46: 7–18, 2005. [DOI] [PubMed] [Google Scholar]
- 29.Emeis J The control of t-PA and PAI-1 secretion from the vessel wall. Vascular Med Rev 6: 153–166, 1995. [Google Scholar]
- 30.Eskurza I, Monahan KD, Robinson JA, Seals DR. Effect of acute and chronic ascorbic acid on flow-mediated dilatation with sedentary and physically active human ageing. J Physiol 556: 315–324, 2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Eskurza I, Myerburgh LA, Kahn ZD, Seals DR. Tetrahydrobiopterin augments endothelium-dependent dilatation in sedentary but not in habitually exercising older adults. J Physiol 568: 1057–1065, 2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Ferrier KE, Waddell TK, Gatzka CD, Cameron JD, Dart AM, Kingwell BA. Aerobic exercise training does not modify large-artery compliance in isolated systolic hypertension. Hypertension 38: 222–226, 2001. [DOI] [PubMed] [Google Scholar]
- 33.Folsom AR, Eckfeldt JH, Weitzman S, Ma J, Chambless LE, Barnes RW, Cram KB, Hutchinson RG. Relation of carotid artery wall thickness to diabetes mellitus, fasting glucose and insulin, body size, and physical activity. Atherosclerosis Risk in Communities (ARIC) Study Investigators. Stroke 25: 66–73, 1994. [DOI] [PubMed] [Google Scholar]
- 34.Franzoni F, Ghiadoni L, Galetta F, Plantinga Y, Lubrano V, Huang Y, Salvetti G, Regoli F, Taddei S, Santoro G, Salvetti A. Physical activity, plasma antioxidant capacity, and endothelium-dependent vasodilation in young and older men. Am J Hypertens 18: 510–516, 2005. [DOI] [PubMed] [Google Scholar]
- 35.Green DJ, Maiorana A, O'Driscoll G, Taylor R. Effect of exercise training on endothelium-derived nitric oxide function in humans. J Physiol 561: 1–25, 2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Haidet GC, Wennberg PW, Finkelstein SM, Morgan DJ. Effects of aging per se on arterial stiffness: systemic and regional compliance in beagles. Am Heart J 132: 319–327, 1996. [DOI] [PubMed] [Google Scholar]
- 37.Hambrecht R, Adams V, Erbs S, Linke A, Krankel N, Shu Y, Baither Y, Gielen S, Thiele H, Gummert JF, Mohr FW, Schuler G. Regular physical activity improves endothelial function in patients with coronary artery disease by increasing phosphorylation of endothelial nitric oxide synthase. Circulation 107: 3152–3158, 2003. [DOI] [PubMed] [Google Scholar]
- 38.Hambrecht R, Fiehn E, Weigl C, Gielen S, Hamann C, Kaiser R, Yu J, Adams V, Niebauer J, Schuler G. Regular physical exercise corrects endothelial dysfunction and improves exercise capacity in patients with chronic heart failure. Circulation 98: 2709–2715, 1998. [DOI] [PubMed] [Google Scholar]
- 39.Hamilton CA, Brosnan MJ, McIntyre M, Graham D, Dominiczak AF. Superoxide excess in hypertension and aging: a common cause of endothelial dysfunction. Hypertension 37: 529–534, 2001. [DOI] [PubMed] [Google Scholar]
- 40.Harrison DG Endothelial function and oxidant stress. Clin Cardiol 20: II-11–17, 1997. [PubMed] [Google Scholar]
- 41.Harvey PJ, Morris BL, Kubo T, Picton PE, Su WS, Notarius CF, Floras JS. Hemodynamic after-effects of acute dynamic exercise in sedentary normotensive postmenopausal women. J Hypertens 23: 285–292, 2005. [DOI] [PubMed] [Google Scholar]
- 42.Heiss C, Keymel S, Niesler U, Ziemann J, Kelm M, Kalka C. Impaired progenitor cell activity in age-related endothelial dysfunction. J Am Coll Cardiol 45: 1441–1448, 2005. [DOI] [PubMed] [Google Scholar]
- 43.Hill JM, Zalos G, Halcox JP, Schenke WH, Waclawiw MA, Quyyumi AA, Finkel T. Circulating endothelial progenitor cells, vascular function, and cardiovascular risk. N Engl J Med 348: 593–600, 2003. [DOI] [PubMed] [Google Scholar]
- 44.Hoetzer GL, MacEneaney OJ, Irmiger HM, Keith R, Van Guilder GP, Stauffer BL, DeSouza CA. Gender differences in circulating endothelial progenitor cell colony-forming capacity and migratory activity in middle-aged adults. Am J Cardiol 99: 46–48, 2007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Hoetzer GL, Stauffer BL, Greiner JJ, Casas Y, Smith DT, DeSouza CA. Influence of oral contraceptive use on endothelial t-PA release in healthy premenopausal women. Am J Physiol Endocrinol Metab 284: E90–E95, 2003. [DOI] [PubMed] [Google Scholar]
- 46.Hoetzer GL, Stauffer BL, Irmiger HM, Ng M, Smith DT, DeSouza CA. Acute and chronic effects of oestrogen on endothelial tissue-type plasminogen activator release in postmenopausal women. J Physiol 551: 721–728, 2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Hoetzer GL, Van Guilder GP, Irmiger HM, Keith RS, Stauffer BL, DeSouza CA. Aging, exercise, and endothelial progenitor cell clonogenic and migratory capacity in men. J Appl Physiol 102: 847–852, 2007. [DOI] [PubMed] [Google Scholar]
- 48.Jablonski KL, Seals DR, Eskurza I, Monahan KD, Donato AJ. High-dose ascorbic acid infusion abolishes chronic vasoconstriction and restores resting leg blood flow in healthy older men. J Appl Physiol 103: 1715–1721, 2007. [DOI] [PubMed] [Google Scholar]
- 49.Jern S, Selin L, Bergbrant A, Jern C. Release of tissue-type plasminogen activator in response to muscarinic receptor stimulation in human forearm. Thromb Haemost 72: 588–594, 1994. [PubMed] [Google Scholar]
- 50.Kawano H, Tanaka H, Miyachi M. Resistance training and arterial compliance: keeping the benefits while minimizing the stiffening. J Hypertens 24: 1753–1759, 2006. [DOI] [PubMed] [Google Scholar]
- 51.Kingwell BA, Tran B, Cameron JD, Jennings GL, Dart AM. Enhanced vasodilation to acetylcholine in athletes is associated with lower plasma cholesterol. Am J Physiol Heart Circ Physiol 270: H2008–H2013, 1996. [DOI] [PubMed] [Google Scholar]
- 52.Kojda G, Hambrecht R. Molecular mechanisms of vascular adaptations to exercise. Physical activity as an effective antioxidant therapy? Cardiovasc Res 67: 187–197, 2005. [DOI] [PubMed] [Google Scholar]
- 53.Lakatta EG Arterial and cardiac aging: major shareholders in cardiovascular disease enterprises: part III: cellular and molecular clues to heart and arterial aging. Circulation 107: 490–497, 2003. [DOI] [PubMed] [Google Scholar]
- 54.Lakatta EG, Levy D. Arterial and cardiac aging: major shareholders in cardiovascular disease enterprises: part I: aging arteries: a “set up” for vascular disease. Circulation 107: 139–146, 2003. [DOI] [PubMed] [Google Scholar]
- 55.Laufs U, Werner N, Link A, Endres M, Wassmann S, Jurgens K, Miche E, Bohm M, Nickenig G. Physical training increases endothelial progenitor cells, inhibits neointima formation, and enhances angiogenesis. Circulation 109: 220–226, 2004. [DOI] [PubMed] [Google Scholar]
- 56.Laughlin MH, Oltman CL, Bowles DK. Exercise training-induced adaptations in the coronary circulation. Med Sci Sports Exerc 30: 352–360, 1998. [DOI] [PubMed] [Google Scholar]
- 57.Laursen JB, Somers M, Kurz S, McCann L, Warnholtz A, Freeman BA, Tarpey M, Fukai T, Harrison DG. Endothelial regulation of vasomotion in apoE-deficient mice: implications for interactions between peroxynitrite and tetrahydrobiopterin. Circulation 103: 1282–1288, 2001. [DOI] [PubMed] [Google Scholar]
- 58.Lavrencic A, Salobir BG, Keber I. Physical training improves flow-mediated dilation in patients with the polymetabolic syndrome. Arterioscler Thromb Vasc Biol 20: 551–555, 2000. [DOI] [PubMed] [Google Scholar]
- 59.Lijnen HR, Collen D. Endothelium in hemostasis and thrombosis. Prog Cardiovasc Dis 39: 343–350, 1997. [DOI] [PubMed] [Google Scholar]
- 60.Maeda S, Iemitsu M, Miyauchi T, Kuno S, Matsuda M, Tanaka H. Aortic stiffness and aerobic exercise: mechanistic insight from microarray analyses. Med Sci Sports Exerc 37: 1710–1716, 2005. [DOI] [PubMed] [Google Scholar]
- 61.Mitchell GF, Parise H, Benjamin EJ, Larson MG, Keyes MJ, Vita JA, Vasan RS, Levy D. Changes in arterial stiffness and wave reflection with advancing age in healthy men and women: the Framingham Heart Study. Hypertension 43: 1239–1245, 2004. [DOI] [PubMed] [Google Scholar]
- 62.Miyachi M, Donato AJ, Yamamoto K, Takahashi K, Gates PE, Moreau KL, Tanaka H. Greater age-related reductions in central arterial compliance in resistance-trained men. Hypertension 41: 130–135, 2003. [DOI] [PubMed] [Google Scholar]
- 63.Miyachi M, Kawano H, Sugawara J, Takahashi K, Hayashi K, Yamazaki K, Tabata I, Tanaka H. Unfavorable effects of resistance training on central arterial compliance: a randomized intervention study. Circulation 110: 2858–2863, 2004. [DOI] [PubMed] [Google Scholar]
- 64.Miyachi M, Tanaka H, Kawano H, Okajima M, Tabata I. Lack of age-related decreases in basal whole leg blood flow in resistance-trained men. J Appl Physiol 99: 1384–1390, 2005. [DOI] [PubMed] [Google Scholar]
- 65.Moe IT, Hoven H, Hetland EV, Rognmo O, Slordahl SA. Endothelial function in highly endurance-trained and sedentary, healthy young women. Vasc Med 10: 97–102, 2005. [DOI] [PubMed] [Google Scholar]
- 66.Moreau KL, DePaulis AR, Gavin KM, Seals DR. Oxidative stress contributes to chronic leg vasoconstriction in estrogen-deficient postmenopausal women. J Appl Physiol 102: 890–895, 2007. [DOI] [PubMed] [Google Scholar]
- 67.Moreau KL, Donato AJ, Seals DR, DeSouza CA, Tanaka H. Regular exercise, hormone replacement therapy and the age-related decline in carotid arterial compliance in healthy women. Cardiovasc Res 57: 861–868, 2003. [DOI] [PubMed] [Google Scholar]
- 68.Moreau KL, Donato AJ, Seals DR, Dinenno FA, Blackett SD, Hoetzer GL, Desouza CA, Tanaka H. Arterial intima-media thickness: site-specific associations with HRT and habitual exercise. Am J Physiol Heart Circ Physiol 283: H1409–H1417, 2002. [DOI] [PubMed] [Google Scholar]
- 69.Moreau KL, Donato AJ, Tanaka H, Jones PP, Gates PE, Seals DR. Basal leg blood flow in healthy women is related to age and hormone replacement therapy status. J Physiol 547: 309–316, 2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Moreau KL, Gavin KM, Plum AE, Seals DR. Ascorbic acid selectively improves large elastic artery compliance in postmenopausal women. Hypertension 45: 1107–1112, 2005. [DOI] [PubMed] [Google Scholar]
- 71.Moreau KL, Gavin KM, Plum AE, Seals DR. Oxidative stress explains differences in large elastic artery compliance between sedentary and habitually exercising postmenopausal women. Menopause 13: 951–958, 2006. [DOI] [PubMed] [Google Scholar]
- 72.Moreau KL, Silver AE, Dinenno FA, Seals DR. Habitual aerobic exercise is associated with smaller femoral artery intima-media thickness with age in healthy men and women. Eur J Cardiovasc Prev Rehabil 13: 805–811, 2006. [DOI] [PubMed] [Google Scholar]
- 73.Murphy C, Kanaganayagam GS, Jiang B, Chowienczyk PJ, Zbinden R, Saha M, Rahman S, Shah AM, Marber MS, Kearney MT. Vascular dysfunction and reduced circulating endothelial progenitor cells in young healthy UK South Asian men. Arterioscler Thromb Vasc Biol 27: 936–942, 2007. [DOI] [PubMed] [Google Scholar]
- 74.Nosaka T, Tanaka H, Watanabe I, Sato M, Matsuda M. Influence of regular exercise on age-related changes in arterial elasticity: mechanistic insights from wall compositions in rat aorta. Can J Appl Physiol 28: 204–212, 2003. [DOI] [PubMed] [Google Scholar]
- 75.O'Leary DH, Polak JF, Kronmal RA, Manolio TA, Burke GL, Wolfson SK Jr. Carotid-artery intima and media thickness as a risk factor for myocardial infarction and stroke in older adults. Cardiovascular Health Study Collaborative Research Group. N Engl J Med 340: 14–22, 1999. [DOI] [PubMed] [Google Scholar]
- 76.Olser W The Principles and Practice of Medicine. New York: Appleton, 1898.
- 77.Ouimet H Fibrinolysis. Boston, MA: Blackwell Scientific Publications, 1994, p. 127–143.
- 78.Parmer RJ, Mahata M, Mahata S, Sebald MT, O'Connor DT, Miles LA. Tissue plasminogen activator (t-PA) is targeted to the regulated secretory pathway. Catecholamine storage vesicles as a reservoir for the rapid release of t-PA. J Biol Chem 272: 1976–1982, 1997. [DOI] [PubMed] [Google Scholar]
- 79.Popa-Wagner A, Fischer B, Platt D, Schmoll H, Kessler C. Delayed and blunted induction of mRNA for tissue plasminogen activator in the brain of old rats following pentylenetetrazole-induced seizure activity. J Gerontol A Biol Sci Med Sci 55: B242–B248, 2000. [DOI] [PubMed] [Google Scholar]
- 80.Proctor DN, Parker BA. Vasodilation and vascular control in contracting muscle of the aging human. Microcirculation 13: 315–327, 2006. [DOI] [PubMed] [Google Scholar]
- 81.Qiu H, Depre C, Ghosh K, Resuello RG, Natividad FF, Rossi F, Peppas A, Shen YT, Vatner DE, Vatner SF. Mechanism of gender-specific differences in aortic stiffness with aging in nonhuman primates. Circulation 116: 669–676, 2007. [DOI] [PubMed] [Google Scholar]
- 82.Rakobowchuk M, McGowan CL, de Groot PC, Bruinsma D, Hartman JW, Phillips SM, MacDonald MJ. Effect of whole body resistance training on arterial compliance in young men. Exp Physiol 90: 645–651, 2005. [DOI] [PubMed] [Google Scholar]
- 83.Reddy AK, Li YH, Pham TT, Ochoa LN, Trevino MT, Hartley CJ, Michael LH, Entman ML, Taffet GE. Measurement of aortic input impedance in mice: effects of age on aortic stiffness. Am J Physiol Heart Circ Physiol 285: H1464–H1470, 2003. [DOI] [PubMed] [Google Scholar]
- 84.Roy C The elastic properties of the arterial wall. J Physiol 3: 125–162, 1881. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Sandri M, Adams V, Gielen S, Linke A, Lenk K, Krankel N, Lenz D, Erbs S, Scheinert D, Mohr FW, Schuler G, Hambrecht R. Effects of exercise and ischemia on mobilization and functional activation of blood-derived progenitor cells in patients with ischemic syndromes: results of 3 randomized studies. Circulation 111: 3391–3399, 2005. [DOI] [PubMed] [Google Scholar]
- 86.Santhanam L, Christianson DW, Nyhan D, Berkowitz DE. Arginase and vascular aging. J Appl Physiol (August 21, 2008); doi: 10.1152/japplphysiol.90627.2008. [DOI] [PMC free article] [PubMed]
- 87.Scheubel RJ, Zorn H, Silber RE, Kuss O, Morawietz H, Holtz J, Simm A. Age-dependent depression in circulating endothelial progenitor cells in patients undergoing coronary artery bypass grafting. J Am Coll Cardiol 42: 2073–2080, 2003. [DOI] [PubMed] [Google Scholar]
- 88.Schmidt-Lucke C, Rossig L, Fichtlscherer S, Vasa M, Britten M, Kamper U, Dimmeler S, Zeiher AM. Reduced number of circulating endothelial progenitor cells predicts future cardiovascular events: proof of concept for the clinical importance of endogenous vascular repair. Circ 111: 2981–2987, 2005. [DOI] [PubMed] [Google Scholar]
- 89.Schmidt-Trucksass A, Grathwohl D, Schmid A, Boragk R, Upmeier C, Keul J, Huonker M. Structural, functional, and hemodynamic changes of the common carotid artery with age in male subjects. Arterioscler Thromb Vasc Biol 19: 1091–1097, 1999. [DOI] [PubMed] [Google Scholar]
- 90.Seals DR, Stevenson ET, Jones PP, DeSouza CA, Tanaka H. Lack of age-associated elevations in 24-h systolic and pulse pressures in women who exercise regularly. Am J Physiol Heart Circ Physiol 277: H947–H955, 1999. [DOI] [PubMed] [Google Scholar]
- 91.Seals DR, Tanaka H, Clevenger CM, Monahan KD, Reiling MJ, Hiatt WR, Davy KP, DeSouza CA. Blood pressure reductions with exercise and sodium restriction in postmenopausal women with elevated systolic pressure: role of arterial stiffness. J Am Coll Cardiol 38: 506–513, 2001. [DOI] [PubMed] [Google Scholar]
- 92.Shatos MA, Doherty JM, Stump DC, Thompson EA, Collen D. Oxygen radicals generated during anoxia followed by reoxygenation reduce the synthesis of tissue-type plasminogen activator and plasminogen activator inhibitor-1 in human endothelial cell culture. J Biol Chem 265: 20443–20448, 1990. [PubMed] [Google Scholar]
- 93.Smith DT, Hoetzer GL, Greiner JJ, Stauffer BL, DeSouza CA. Effects of ageing and regular aerobic exercise on endothelial fibrinolytic capacity in humans. J Physiol 546: 289–298, 2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Smulyan H, Asmar RG, Rudnicki A, London GM, Safar ME. Comparative effects of aging in men and women on the properties of the arterial tree. J Am Coll Cardiol 37: 1374–1380, 2001. [DOI] [PubMed] [Google Scholar]
- 95.Spier SA, Delp MD, Meininger CJ, Donato AJ, Ramsey MW, Muller-Delp JM. Effects of ageing and exercise training on endothelium-dependent vasodilatation and structure of rat skeletal muscle arterioles. J Physiol 556: 947–958, 2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Spier SA, Delp MD, Stallone JN, Dominguez JM 2nd, Muller-Delp JM. Exercise training enhances flow-induced vasodilation in skeletal muscle resistance arteries of aged rats: role of PGI2 and nitric oxide. Am J Physiol Heart Circ Physiol 292: H3119–H3127, 2007. [DOI] [PubMed] [Google Scholar]
- 97.Stauffer BL, Hoetzer GL, Van Guilder GP, Smith DT, DeSouza CA. Gender differences in endothelial tissue-type plasminogen activator release in middle-aged adults. J Am Coll Cardiol 45: 1547–1549, 2005. [DOI] [PubMed] [Google Scholar]
- 98.Steiner S, Niessner A, Ziegler S, Richter B, Seidinger D, Pleiner J, Penka M, Wolzt M, Huber K, Wojta J, Minar E, Kopp CW. Endurance training increases the number of endothelial progenitor cells in patients with cardiovascular risk and coronary artery disease. Atherosclerosis 181: 305–310, 2005. [DOI] [PubMed] [Google Scholar]
- 99.Sui X, LaMonte MJ, Laditka JN, Hardin JW, Chase N, Hooker SP, Blair SN. Cardiorespiratory fitness and adiposity as mortality predictors in older adults. JAMA 298: 2507–2516, 2007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Taddei S, Galetta F, Virdis A, Ghiadoni L, Salvetti G, Franzoni F, Giusti C, Salvetti A. Physical activity prevents age-related impairment in nitric oxide availability in elderly athletes. Circulation 101: 2896–2901, 2000. [DOI] [PubMed] [Google Scholar]
- 101.Taddei S, Virdis A, Ghiadoni L, Salvetti G, Bernini G, Magagna A, Salvetti A. Age-related reduction of NO availability and oxidative stress in humans. Hypertension 38: 274–279, 2001. [DOI] [PubMed] [Google Scholar]
- 102.Taddei S, Virdis A, Mattei P, Ghiadoni L, Fasolo CB, Sudano I, Salvetti A. Hypertension causes premature aging of endothelial function in humans. Hypertension 29: 736–743, 1997. [DOI] [PubMed] [Google Scholar]
- 103.Taddei S, Virdis A, Mattei P, Ghiadoni L, Gennari A, Fasolo CB, Sudano I, Salvetti A. Aging and endothelial function in normotensive subjects and patients with essential hypertension. Circulation 91: 1981–1987, 1995. [DOI] [PubMed] [Google Scholar]
- 104.Takada A, Takada Y, Urano T. The physiological aspects of fibrinolysis. Thromb Res 76: 1–31, 1994. [DOI] [PubMed] [Google Scholar]
- 105.Tanaka H, DeSouza CA, Seals DR. Absence of age-related increase in central arterial stiffness in physically active women. Arterioscler Thromb Vasc Biol 18: 127–132, 1998. [DOI] [PubMed] [Google Scholar]
- 106.Tanaka H, Dinenno FA, Monahan KD, Clevenger CM, DeSouza CA, Seals DR. Aging, habitual exercise, and dynamic arterial compliance. Circulation 102: 1270–1275, 2000. [DOI] [PubMed] [Google Scholar]
- 107.Tanaka H, Dinenno FA, Monahan KD, DeSouza CA, Seals DR. Carotid artery wall hypertrophy with age is related to local systolic blood pressure in healthy men. Arterioscler Thromb Vasc Biol 21: 82–87, 2001. [DOI] [PubMed] [Google Scholar]
- 108.Tanaka H, Seals DR, Monahan KD, Clevenger CM, DeSouza CA, Dinenno FA. Regular aerobic exercise and the age-related increase in carotid artery intima-media thickness in healthy men. J Appl Physiol 92: 1458–1464, 2002. [DOI] [PubMed] [Google Scholar]
- 109.Tanaka H, Shimizu S, Ohmori F, Muraoka Y, Kumagai M, Yoshizawa M, Kagaya A. Increases in blood flow and shear stress to nonworking limbs during incremental exercise. Med Sci Sports Exerc 38: 81–85, 2006. [DOI] [PubMed] [Google Scholar]
- 110.Thijssen DH, Vos JB, Verseyden C, van Zonneveld AJ, Smits P, Sweep FC, Hopman MT, de Boer HC. Haematopoietic stem cells and endothelial progenitor cells in healthy men: effect of aging and training. Aging Cell 5: 495–503, 2006. [DOI] [PubMed] [Google Scholar]
- 111.Tschudi MR, Barton M, Bersinger NA, Moreau P, Cosentino F, Noll G, Malinski T, Luscher TF. Effect of age on kinetics of nitric oxide release in rat aorta and pulmonary artery. J Clin Invest 98: 899–905, 1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.Urbich C, Dimmeler S. Endothelial progenitor cells: characterization and role in vascular biology. Circ Res 95: 343–353, 2004. [DOI] [PubMed] [Google Scholar]
- 113.Vaitkevicius PV, Fleg JL, Engel JH, O'Connor FC, Wright JG, Lakatta LE, Yin FC, Lakatta EG. Effects of age and aerobic capacity on arterial stiffness in healthy adults. Circ 88: 1456–1462, 1993. [DOI] [PubMed] [Google Scholar]
- 114.van den Eijnden-Schrauwen Y, Kooistra T, de Vries RE, Emeis JJ. Studies on the acute release of tissue-type plasminogen activator from human endothelial cells in vitro and in rats in vivo: evidence for a dynamic storage pool. Blood 85: 3510–3517, 1995. [PubMed] [Google Scholar]
- 115.van der Loo B, Labugger R, Skepper JN, Bachschmid M, Kilo J, Powell JM, Palacios-Callender M, Erusalimsky JD, Quaschning T, Malinski T, Gygi D, Ullrich V, Luscher TF. Enhanced peroxynitrite formation is associated with vascular aging. J Exp Med 192: 1731–1744, 2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116.Van Guilder GP, Hoetzer GL, Smith DT, Irmiger HM, Greiner JJ, Stauffer BL, DeSouza CA. Endothelial t-PA release is impaired in overweight and obese adults but can be improved with regular aerobic exercise. Am J Physiol Endocrinol Metab 289: E807–E813, 2005. [DOI] [PubMed] [Google Scholar]
- 117.Wang M, Zhang J, Jiang LQ, Spinetti G, Pintus G, Monticone R, Kolodgie FD, Virmani R, Lakatta EG. Proinflammatory profile within the grossly normal aged human aortic wall. Hypertension 50: 219–227, 2007. [DOI] [PubMed] [Google Scholar]
- 118.Wendelhag I, Wiklund O, Wikstrand J. Atherosclerotic changes in the femoral and carotid arteries in familial hypercholesterolemia. Ultrasonographic assessment of intima-media thickness and plaque occurrence. Arterioscler Thromb 13: 1404–1411, 1993. [DOI] [PubMed] [Google Scholar]
- 119.Wray DW, Richardson RS. Aging, exercise, and limb vascular heterogeneity in humans. Med Sci Sports Exerc 38: 1804–1810, 2006. [DOI] [PubMed] [Google Scholar]