Abstract
In normal rats callosal projections in striate cortex connect retinotopically corresponding, non mirror-symmetric cortical loci, whereas in rats bilaterally enucleated at birth, callosal fibers connect topographically mismatched, mirror-symmetric loci. Moreover, retina input specifies the topography of callosal projections by postnatal day 6 (P6). To investigate whether retinal input guides development of callosal maps by promoting either the corrective pruning of exuberant axon branches, or the specific ingrowth and elaboration of axon branches at topographically correct places, we studied the topography of emerging callosal connections at, and immediately after P6. After restricted intracortical injections of anterogradely and retrogradely transported tracers we observed that the normal, non-mirror symmetric callosal map, as well as the anomalous, mirror-symmetric map observed in neonatally enucleated animals, are present by P6–7, just as collateral branches of simple architecture emerge from their parental axons and grow into superficial cortical layers. Our results therefore do not support the idea that retinal input guides callosal map formation by primarily promoting the large-scale elimination of long, non-topographic branches and arbors. Instead, they suggest that retinal input specifies the sites on the parental axons from which interstitial branches will grow to invade middle and upper cortical layers, thereby ensuring that the location of invading interstitial branches is accurately related to the topographical location of the soma that gives rise to the parental axon. Moreover, our results from enucleated rats suggest that the cues that determine the mirror-symmetric callosal map exert only a weak control on the topography of fiber ingrowth.
Indexing terms: Striate Cortex, Corpus Callosum, Interhemispheric Commissure, Critical Period, Map
INTRODUCTION
Cortico-cortical neural projections are often organized into maps that preserve the topography of the areas giving rise to the projections. Understanding how ordered projections develop in the cerebral cortex remains a central issue in neurobiological research. A prevailing hypothesis about the development of topographic projections in the central nervous system poses that the initial targeting of axons and elaboration of terminal arbors is imprecise, and that topographic precision is achieved through axon branch elaboration at appropriate places, combined with the corrective removal of axonal branches located in inappropriate places (Katz and Shatz, 1996). A good illustration of this sequence of events is provided by the development of the projection from the retina to the superior colliculus in rodents (Simon and O’Leary, 1992; McLaughlin et al., 2003). In this pathway, the parental axon grows past the target zone (axon overshoot) and interstitial branches emerge from the axon shaft some distance (up to hundreds of microns) behind the primary growth cone (Simon and O’Leary, 1992; Yates et al., 2001). Experiments tracing the growth of axons from restricted retinal loci revealed that although the distribution of interstitial branches emerging along the parental axons is biased towards the location of the future termination zone in the superior colliculus, branches and axon arbors are also observed in an area several times larger than that of the mature termination zone, as well as virtually throughout the extent of the overshooting parental axons. Subsequent map refinement results from the large-scale elimination of topographically aberrant axons, axon segments, branches and arbors, including the overshooting portion of the parental axon, together with the preferential elaboration of arbors at the correct termination zone (Simon and O’Leary, 1992; McLaughlin et al., 2003).
In the cerebral cortex, the discovery by Wise and Jones (1976) and Innocenti et al. (1977) that the mature, discontinuous pattern of callosal neurons develops from a widespread neonatal distribution has been generalized to a variety of species and cortical areas (reviewed by Innocenti, 1991), with the exception of striate cortex in rhesus monkey (Dehay et al., 1988; Chalupa et al., 1989). However, in contrast to the widespread distribution of neurons, anterogradely labeled callosal axons are largely restricted to white matter at early stages of development, and at later ages they appear to invade gray matter only in those cortical regions where they are found in the adult (Olavarria and Van Sluyters, 1985; reviewed by Innocenti, 1991; Innocenti and Price 1995). In the cat, analysis of reconstructed callosal axons has further revealed that parental axons give off many short branches in the white matter underlying striate cortex. Moreover, the invasion into gray matter is restricted to branches located in the 17/18 callosal zone, while the remaining branches are eliminated before they penetrate gray matter to any significant extent (Aggoun-Zouaoui and Innocenti, 1994). In rodents, parental callosal axons navigate through lower cortical layers or white matter, often overshooting their target, and give rise to interstitial branches that grow towards the pia in regions that are callosally innervated in adults (Norris and Kalil, 1992; Hedin-Pereira et al., 1999; Ding and Elberger, 2001). These observations raise the possibility that development of callosal topography may not require extensive pruning of branches that have invaded the gray matter. Similar conclusions are suggested by studies of ipsilateral cortico-cortical connections in the cat (Kennedy et al., 1994; Caric and Price, 1996) and monkey (Barone et al., 1996). However, previous studies have not systematically investigated the emergence of fine topography within cortical target regions. At present it is not known whether the growth of interstitial branches and elaboration of axon arbors in gray matter is precise from the start, or whether a remodeling process similar to that observed in the rodent retinocollicular pathway is required for the establishment of fine topography in cortico-cortical projections.
In the present study we have addressed this issue by studying the development of topography in the rat visual callosal pathway. This system has unique topographic as well as plasticity features that make it ideal for investigating the development of cortical maps. We have previously proposed that the temporal retina, through a system of bilateral projections, promotes the formation of callosal linkages between opposite cortical loci in lateral striate cortex that are both retinotopically matched (i.e., they represent the same retinal locus), and non-mirror symmetric with respect to the brain midline (Lewis and Olavarria, 1995; Olavarria, 1996; 2001a, 2001b). Because of the non-symmetry of callosal linkages in lateral striate cortex, intracortical tracer injections separated by a few hundred microns typically produce distinctly different projection patterns, a property that can facilitate the detection of topographic maps in very young animals. Moreover, removal of retinal input at postnatal day 4 (P4) or earlier, but not at P6, reverses the topography of the callosal map: callosal fibers in rats enucleated at P4 or earlier connect opposite cortical loci that are largely mirror-symmetric with respect to the midline (Olavarria and Li, 1995; Olavarria and Hiroi, 2003). Comparing the emergence of topography in normal and enucleated animals therefore offers the unique opportunity of analyzing the role of retinal input in the specification of topography. Studying this role is more difficult in other organized cortical projection systems, such as the projection from area 17 to neighboring area 18, because experimental manipulations of retinal input often blur, but do not change, the gross point-to-point topography of the projections (Ruthazer and Stryker, 1996; Olavarria, unpubl. obs.). Finally, the fact that, in the rodent callosal pathway, most interstitial branches that invade the gray matter by P6 display simple architecture (Fish et al., 1991; Norris and Kalil, 1992; Hedin-Pereira et al., 1999; Ding and Elberger, 2001) makes it possible to analyze the topography of invading fibers in normal and neonatally enucleated animals before any significant elaboration of axon arbors has taken place.
If retinal input sculpts the normal callosal map from a diffuse, mirror-symmetric pattern, we expected the callosal topography to be mirror-symmetric in both normal and enucleated pups by P6. In subsequent days, a non-mirror symmetric topography would gradually emerge in normal pups, while in enucleated animals the topography of callosal projections would remain largely unchanged (i.e., mirror-symmetric). In contrast, if retinal input regulates the topographic accuracy of target invasion by interstitial branches, we expected that both the normal and anomalous maps would be recognizable by the end of the P4-P6 critical period. Since cellular and molecular mechanisms controlling the accuracy of interstitial branch invasion are likely to differ from those controlling the pruning of topographically incorrect axon branches, distinguishing between these two possibilities will help to identify of retinally-driven mechanisms that are involved in the specification of cortico-cortical maps. Using a combination of anterogradely and retrogradely transported tracers we found that the normal callosal map, as well as the anomalous map observed in neonatally enucleated animals, can be demonstrated when axonal branch invasion reaches superficial cortical layers and elaboration of terminal arbors in gray matter is just beginning. These results provide evidence that the establishment of cortical projection maps depends primarily on the topography displayed by simple fibers as they invade the gray matter, rather than on large-scale corrective processes that eliminate inappropriate projections resulting from inaccuracies in the invasion of interstitial branches and/or exuberant growth of axonal arbors.
MATERIALS AND METHODS
Our study is based on data obtained from a total of thirty six Long-Evans pigmented rats. Pregnant animals were monitored several times daily, and the births of the litters were determined to within 12 h. The first 24 hours were considered as postnatal day 0 (P0). Seven rat pups were anesthetized with Halothane (0.5 – 1.5% in air) and binocularly enucleated within 24 h of birth. After recovering from the anesthesia, pups were returned to their dams. All surgical procedures were performed according to protocols approved by the Institutional Animal Care and Use Committees at UW.
Tracer injections
Tracer injections were made in animals anesthetized with Halothane (0.5 – 1.5% in air). Single small (0.01 μl) injections of retrogradely transported tracers were used to analyze the distribution of the somas of callosal cells in fifteen pups. The tracers used included the fluorescent tracers Rhodamine and Green-beads (RB and GB, respectively, LumaFluor, Naples, Florida, concentrated stock solution). Small injections of RB and GB were separated by distances ranging from 310 to 650 μm. Biotinylated dextran amine (BDA, 10% in DW, Molecular Probes) was used to analyze the distribution of anterogradely labeled axons in twenty one pups. One or two small injections of BDA (about 0.01 μl each) were made in fifteen pups. In these pups, as well as in those that received small injections of retrograde tracers, the injections were placed in presumptive area 17, at a distance of 2.8–3.5 mm from the midline, and about 0.5 mm anterior to the lambda suture. All small injections analyzed were restricted to the gray matter. In the remaining six pups, the overall patterns of anterogradely labeled callosal axons were revealed by placing multiple (10–20; total volume of 1.0 – 1.5 μl) injections of BDA across the surface of one occipital lobe, in a pattern similar to that illustrated in Fig. 1B. Both anterograde and retrograde tracers were pressure-injected through glass micropipettes (50–100 μm tip diameter). For small, restricted injections, the pipette containing the tracers was lowered to about 600–800 μm and, about 1 min after the injection, it was slowly retracted. In determining the ages for injecting and perfusing the animals, we have assumed that the pattern of connections revealed with BDA is that present at the time of perfusion (Simon and O’Leary, 1992), whereas the pattern revealed with retrograde tracers is preferentially that present within a few hours after the injections.
Fig. 1.
A: Diagram of the distribution of callosal connections in areas 17 and 18a in rat visual cortex produced by multiple tracer injections in the contralateral cortex. Gray areas correspond to cortical regions containing dense accumulations of callosal connections. The borders of area 17 are indicated by arrows. In the medial callosal region (MCR), labeled callosal cells are located preferentially in infragranular layers. The MCR extends from the medial border of area 17 to the medial edge (indicated by the bar) of the column-like accumulation of callosal connections at the 17/18a border region. The lateral callosal region (LCR) includes the portion of the 17/18a callosal zone that lies within striate cortex, excluding, therefore, the portion of this zone located in area 18a. In the LCR, callosal cells are distributed most densely in layers 2/3, 5a, 5c and 6a, less densely in layers 4, 5b and the remainder of layer 6, and are virtually absent in layer 1 (Olavarria and Van Sluyters, 1985; Olavarria et al., 1987). B: Diagram of the rat brain indicating the locations of areas 17 and 18a in occipital cortex, the injection sites on the right hemisphere (black dots), and the coronal level from which section in A was taken (horizontal line).
Histochemical processing
After a survival time that was generally 24 hours for RB and GB, and 48 hours for BDA, the animals were deeply anesthetized with pentobarbital sodium (100 mg/kg i.p.) and perfused through the heart with 0.9% saline followed by 4% paraformaldehyde in 0.1 M phosphate buffer (PB, pH 7.4). The brains were removed and left overnight in 30% sucrose and 0.1M PB. The data were analyzed in series of 80 μm thick coronal sections obtained using a freezing microtome. Sections to be examined for fluorescence retrograde labeling were mounted without further processing, and after the data had been collected, they were Nissl-stained and coverslipped to reveal the location of the borders of presumptive area 17 (Fig. 4A,B; Olavarria and Van Sluyters, 1985). BDA labeling was revealed using the standard Avidin-Biotin-Peroxidase protocol (Vectastain Elite ABC kit, Vector Laboratories, Burlingame, California) and 0.01% H2O2 in 0.05% 3-3′ diaminobenzidine, with cobalt or nickel intensification; sections were then mounted, dehydrated, defatted and coverslipped.
Data acquisition and analysis
The procedures for analysis of callosal maps were those employed in previous work from our laboratory (Lewis and Olavarria, 1995; Olavarria and Li, 1995; Olavarria and Hiroi, 2003). Labeling patterns in histological sections were analyzed using a microscope equipped with a motorized stage (LEPCO) controlled by a Dell XPS T500 computer, and a graphic system (Neurolucida, MicroBrightField). In both normal and enucleated animals, the location of the small injection sites in presumptive area 17 was determined by identifying the borders of this area in Nissl-stained sections. However, because the lesions produced by the injections often made it difficult to analyze local cytoarchitectonic changes, a more reliable way to assess the location of the tracer injections was provided by the analysis of the distribution of retrogradely or anterogradely labeled fields within the ipsilateral dorsal lateral geniculate nucleus (dLGN) of the thalamus (Montero et al., 1968; Godement et al., 1979; Kaiserman-Abramof et al., 1980; Guillery et al., 1985; Warton et al., 1988). This analysis was carried out in coronal sections passing through areas of dense geniculate labeling. A line extending from the mediodorsal tip of the dLGN to the center of the labeled field was drawn. This line segment was then projected onto an axial line drawn through the middle of the geniculate body, from the mediodorsal tip of the dLGN to its lateroventral border. The length of the projected distance to the labeled field was then expressed as a percentage of the length of the axial line.
Sections taken from the injected hemisphere were examined, and one or more sections passing through the center of the injections were scored to illustrate the location and size of the tracer deposits. Histological sections (2–8) passing through areas of highest labeling density were selected for detailed analysis of the distribution of retrograde or anterograde callosal labeling, and some of these sections were used to illustrate the results obtained. To analyze the distribution of retrogradely labeled cells, or of anterogradely labeled fibers in gray matter, the mediolateral axis was subdivided into bins approximately 166 μm wide. In the case of retrograde labeling, the labeled cells in each bin were counted. In the case of anterograde labeling, fibers were traced with lines of the same thickness, and the number of pixels for each bin was determined using Photoshop (Adobe Systems). Fibers extending mediolaterally in white matter or deep cortex were not included in this analysis. Data from neighboring sections were pooled by summing the counts from corresponding bins in a normalized density curve. The size of callosal fields in normal and enucleated animals were compared by measuring the half-widths of these plots. In some animals injected with RB and GB, double-labeled cells were scored in areas where there was a clear overlap in the distribution of cells labeled by each tracer. Within these overlap areas, the cells labeled by each tracer were counted and the incidence of double-labeled cells was expressed as a percentage of the smaller count. Figures were prepared using Photoshop, and all image processing used, including contrast enhancement and intensity level adjustments, was applied to the entire image and never locally.
RESULTS
Topography of neurons projecting through the corpus callosum
In area 17 of adult rats, two callosal regions, the medial and lateral callosal regions (MCR and LCR, respectively, Fig. 1), have been recognized in coronal sections through the occipital cortex (Lewis and Olavarria. 1995). These regions have distinctly different topographic organizations. In the MCR, callosal fibers connect loci that are mirror-symmetric with respect to the brain midline, whereas in the LCR, the mapping of callosal fibers is non mirror-symmetric. Thus, tracer injections into the 17/18a border, which lies within a narrow region known as the transition zone, produce labeled fields located immediately outside the transition zone, whereas LCR injections located outside the transition zone produce callosal labeling restricted to the transition zone (Lewis and Olavarria, 1995; Olavarria and Hiroi, 2003). Using small injections of two retrogradely transported fluorescent tracers, we found that the organization of callosally projecting cells in animals injected at P6 and perfused at P7 closely resembles the organization in adult rats (Fig. 2A). In these young animals, cells labeled after injections placed a few hundreds of microns medial to the 17/18a border accumulated preferentially at the 17/18a border, where they formed narrow clusters measuring about 260 μm in width (see labeled field produced by the injection of GB in Fig. 2A). In contrast, callosal cells labeled after injections at the 17/18a border occupied a broader region on both sides of the contralateral 17/18a border (see labeled fields produced by the injection of RB in Fig. 2A). The density of RB-labeled cells peaked both immediately medial and lateral to the 17/18a border, suggesting the existence of a RB-labeled field within area 17 and another one in area 18a (see graph at bottom of Fig. 2A). Similar results were found in another animal studied at the same age (not shown). If in Figure 2A one considers only the GB-, and RB-labeled fields lying within area 17, it is apparent that callosal linkages in the LCR of P6/P7 pups are not arranged as a mirror image of the injections sites. These results closely resemble those described in the LCR of normal adult rats (Lewis and Olavarria, 1995; Olavarria and Hiroi, 2003). In the case shown in Figure 2A, in addition to the field of GB-labeled cells centered at the 17/18a border (black dots), a cluster of GB-labeled cells were observed in more medial portions of area 17 (marked with an asterisk in Fig. 2A), probably within the prospective MCR. Unlike the LCR, the MCR is organized in a mirror-symmetric fashion with respect to the midline (Lewis and Olavarria, 1995). It is therefore possible that these few cells were labeled because the injection site (aimed at the LCR) encroached slightly onto the adjacent MCR.
Fig. 2.
Organization of callosal linkages revealed with retrogradely transported tracers in the LCR of normal pups. A: Pup injected on P6 and perfused on P7 (P6/P7, case L8J). This case received an injection of red beads (RB, indicated in gray in right panel) at the 17/18 border, and an injection of green beads (GB, indicated in black in right panel) placed about 650 μm more medially. Arrows indicate the 17/18a border. Top right inset illustrates the approximate location, relative to the border of area 17, of the injection sites (right), and of the corresponding labeled fields in the opposite hemisphere (left). At left, drawings show the distribution of GB-labeled cells (black dots, middle drawing) and RB-labeled cells (gray dots, bottom drawing) reconstructed from coronal sections taken from the region indicated by the parallel lines in top inset. These labeling patterns are superimposed in the top drawing. Labeled cells were observed throughout cortical layers 2–6. The asterisk in the middle drawing marks the location of a cluster of GB-labeled cells located in more medial portions of area 17 (presumptive MCR). The GB injection may have encroached onto the MCR, and these cells probably reflect the mirror-symmetric projection of the MCR. The relative locations of the RB- and GB-labeled cells in the LCR are represented in the bottom left graph. The curve for RB-labeling represents the distribution of RB-labeled cells located within area 17 and medial area 18a; the total numbers of RB- and GB-labeled cells counted are indicated. Bottom right inset illustrates the location of the labeled fields in the dorsal lateral geniculate nucleus (dLGN; medial is to the left). The location of labeled fields produced by the respective tracers in the ipsilateral LGN confirms the location of the injection sites in area 17. Scale bars = 1.00 mm. B: Pup injected on P8 and perfused on P9 (P8/P9, case L7B2). This case received an injection of red beads (RB, indicated in gray in right panel) at the 17/18 border, and an injection of green beads (GB, indicated in black in right panel) placed about 400 μm more medially. Other conventions as in A.
It should be noted that previous experiments using multiple injections of retrogradely transported tracers that infiltrate both gray and white matter have shown that callosal cells are widely distributed throughout the mediolateral extent of areas 17 and 18a in neonatal rats (Olavarria and Van Sluyters, 1985). In particular, these experiments have shown that by P6 there are many callosal cells in both supragranular and infragranular layers in medial portions of area 17 (Olavarria and Van Sluyters, 1985). While many infragranular cells remain in the MCR in mature animals, nearly all supragranular callosal cells in this region disappear by the third week of life (Olavarria and Van Sluyters, 1985). In this context, our present observation that the retrograde labeling was largely confined to the LCR in P6 rats injected into different regions within the contralateral LCR (Fig. 2A) indicate that supragranular callosal cells in the prospective MCR do not send projections into the contralateral LCR in young rats. As proposed before (Olavarria and Van Sluyters, 1985), the axons of these cells probably remain in the white matter and are later withdrawn.
By P6 callosal fibers of very simple architecture are observed extending up to supragranular cortical layers (Norris and Kalil, 1992; Hedin-Pereira et al., 1999; our own results). This raises the possibility that the subsequent elaboration of intracortical terminal arbors (Hedin-Pereira et al., 1999; Ding and Elberger; 2001; our Unpub. Obs.) may temporarily degrade the topography of callosal linkages. This could happen, for instance, due to unspecific emergence and growth of interstitial branches, or if intracortical terminal arbors go through a phase of exuberant growth. To determine if this was the case, we studied older animals with retrogradely transported tracers. Figure 2B shows that the organization of callosal linkages in animals injected at P8 and perfused at P9 closely resembles the organization we observed in P6/P7 animals (cf. Figs. 2A,B). Similar results were obtained in other animals studied either at P7/P8 (n = 1), or P9/P10 (n = 2) (data not shown). These findings indicate that elaboration of terminal arbors occurs at topographically appropriate places, and furthermore, that any unspecific growth of interstitial branches or exuberant growth of terminal arbors is not sufficient to degrade or obliterate the topography of the callosal map during the phases of development we studied.
Using retrogradely transported tracers we found that the normal callosal map is present as early as P6–7. To investigate if the same is true for the abnormal map induced by neonatal enucleation, we analyzed the labeling patterns in enucleated pups that were injected with two tracers at P6 and perfused at P7 (Fig. 3). In enucleated animals (n = 3) we observed that tracer injections of approximately the same size as those used in normal animals produced labeled fields that were much larger than those observed in normal animals. Injections sites were carefully examined to rule out an involvement of white matter. Moreover, although overlapping, the labeled fields were roughly arranged as a mirror image of the injection sites (see graph at bottom of Fig. 3). These characteristics are those we have described in enucleated rats studied as adults (Olavarria and Li, 1995; Olavarria and Hiroi, 2003). To summarize, our analysis of retrograde labeling in normal and enucleated pups provides evidence that both the normal callosal map, and the map induced by neonatal enucleation are present by the end of the critical period for callosal map specification.
Fig. 3.
Organization of callosal linkages revealed with retrogradely transported tracers in pups enucleated at birth and injected on P6 and perfused on P7 (case L22B). This case received an injection of red beads (RB, indicated in gray in right panel) at a location medial to the 17/18 border, and an injection of green beads (GB, indicated in gray in right panel) placed about 410 μm further medially. Injections sites are shown in Fig. 4C. Top right inset illustrates the approximate location, relative to the border of area 17, of the injection sites (right), and of the corresponding labeled fields in the opposite hemisphere (left). At left, drawings show the distribution of GB-labeled cells (black dots, top drawing) and RB-labeled cells (gray dots, bottom drawing) reconstructed from coronal sections taken from the region indicated by the parallel lines in top inset. Labeled cells were observed throughout cortical layers 2–6. The relative locations of the RB- and GB-labeled cells in the LCR are represented in the bottom left graph. The total numbers of RB- and GB-labeled cells counted are indicated. Bottom right inset illustrates the location of the labeled fields in the dorsal lateral geniculate nucleus (dLGN; medial is to the left). The location of labeled fields produced by the respective tracers in the ipsilateral LGN confirms the location of the injection sites in area 17. Scale bars = 1.00 mm.
One aspect of axonal morphology that is important in the establishment of topography is the spatial distribution of intracortical branches that derive from single parental axons. We estimated the spatial distribution of intracortical branches derived from single parental callosal axons by analyzing the proportion of cells that were double labeled after injections of two different retrogradely transported tracers. If parental axons give rise to widely separated interstitial branches, or if interstitial branches produce exuberant arbors in gray matter, a large proportion of double labeled cells would result after closely spaced injections of different tracers. Double-labeled cells were analyzed in two normal and two enucleated animals injected with GB and RB at P6 and perfused at P7. The injection sites did not compromise the white matter in these animals. Double-labeled cells were scored in areas where there was a clear overlap in the distribution of cells labeled by each tracer. Within these overlap areas, the cells labeled by each tracer were counted and the incidence of double-labeled cells was expressed as a percentage of the smaller count. In normal animals, the distances between the injections of GB and RB were 300 and 370 μm, and the proportions of double-labeled cells were 1/113 (0.9%) and1/73 (1.4%), respectively, yielding an average of 1.2%. In enucleated animals, the distances between the injections of GB and RB were 410 and 310 μm (Fig. 4C,D, respectively), and the proportions of double-labeled cells were 1/89 (1.1%) and1/60 (1.7%), respectively, yielding an average of 1.4%. The low incidence of double-labeled cells was not due to technical difficulties because we observed that the percentage of double-labeled cells averaged 73% in two pups in which the injections of GB and RB were aimed at the same place. The paucity of double-labeled cells we found after separate injections in normal and enucleated animals suggests that the intracortical area innervated by single parental callosal axons is similarly restricted in both groups of animals at about P7. Since formation of intracortical arbors begins by P6–P7, these results further suggest that parental axons in both normal and enucleated animals do not give off widely separated interstitial branches that grow to upper cortical layers.
Fig. 4.
Low (A) and high (B) power, bright-field views of Nissl-stained sections showing cytoarchitecture of posterior neocortex at P6 in normal rats. Arrow in B marks position at which the staining density in layer IV decreases upon passing from area 17 to area 18a. Cortical layers are indicated in B. Scale bars = 1.00 mm. C,D: Images of cortical injections of fluorescent tracers green beads (GB) and red beads (RB) in cases 22 B (C) and 22 C (D). Top edge of black background corresponds to the pia matter, while bottom edge corresponds to the border between gray and white matter. Data from case 22B (C) are shown in Fig. 3. Scale bar = 1.00 mm.
Topography of intracortical callosal fibers
To complement our results obtained with retrogradely transported tracers, we analyzed the distribution of intracortical fibers labeled by small contralateral injections of the anterogradely transported tracer BDA. Although labeled callosal axons form relatively dense bundles in the white matter and deep cortical layers, we observed that many radially oriented fibers originated at right angles from some of these axons. These observations are in agreement with previous studies indicating that callosal fibers in gray matter often form as interstitial branches from parental axons that navigate mediolaterally in deep cortical layers or white matter (Norris and Kalil, 1992; Hedin-Pereira et al., 1999). In normal pups, we found that the topography of intracortical labeled callosal fibers in young pups corresponds well with that we demonstrated with retrogradely transported tracers in animals of similar ages. Observations from initial experiments and our data from adult animals (Lewis and Olavarria, 1995; Olavarria and Hiroi, 2003) indicate that regions immediately medial and lateral to the 17/18a border region converge onto a narrow transition zone straddling the contralateral 17/18a border. Because callosal input to the 17/18a transition zone comes from a relatively broad cortical area, single small injections medial to this zone label relatively few callosal fibers. For this reason, in some cases in which the injections were targeted to regions medial to the 17/18a border, two small, adjacent BDA injections were placed in order to increase the number of labeled callosal fibers. Figures 5A, 6A show data from two animals injected a P4 and perfused at P6. The injections were placed about 400–500 μm medial to the 17/18a border. The location of the injections was confirmed by the labeling pattern in the ipsilateral dLGN (Figs. 5A, 6A). Contralaterally, these injections produced a narrow field of labeled fibers that was centered on the 17/18a border. The asterisks in Figs. 5A, and 6A mark the location of labeled fibers in more medial regions of area 17 (presumptive MCR, see Fig. legend). Similar results were obtained in two additional cases injected at P4 and perfused at P6 (data not shown). In contrast, fibers labeled after injections of BDA at the 17/18a border occupied a broader region on both sides of the 17/18a border (Figs. 5B, 6B). The graphs at the bottom of Figs 5B, 6B show a reduction of the labeling density at the 17/18a border, suggesting that fibers accumulate preferentially both immediately medial and lateral to the 17/18a border. Virtually no labeled fibers were observed in other regions of area 17. Consistent results were obtained in two additional cases injected at P4 and perfused at P6 (data not shown). Comparison of the projections patterns in panels A and B in Figures 5, 6 provides evidence that, as in normal adult rats (Olavarria and Hiroi, 2003), the origin and termination of callosal projections in lateral area 17 are arranged in a non mirror-symmetric fashion with respect to the midline.
Fig. 5.
Organization of callosal projections revealed with the anterogradely transported tracer BDA in the lateral callosal region of normal pups injected on P4 and perfused on P6. A: Pup (case L46 NC) that received two adjacent injections of BDA at locations medial to the prospective 17/18a border. A: drawing and a low-power micrograph of the injections are shown in the top and bottom panels at right, respectively. At left, drawing shows the distribution of BDA-labeled fibers reconstructed from coronal sections. The asterisk marks the location of labeled fibers located in more medial portions of area 17 (presumptive MCR). The BDA injections may have encroached onto the MCR, and these fibers probably reflect the mirror-symmetric projection of the MCR. The distribution of labeling density is represented in the bottom left graph. Bottom right inset shows an outline of the anterogradely labeled field in the right dorsal lateral geniculate nucleus (dLGN; medial is to the left). The location of labeled field produced by the BDA injection in the ipsilateral LGN confirms the location of the injection sites in area 17. Scale bars = 1.00 mm. B: In this case (L46 NA), the BDA injection was placed at the 17/18a border. Conventions as in A.
Fig. 6.
Organization of callosal projections revealed with the anterogradely transported tracer BDA in the lateral callosal region of normal pups injected on P4 and perfused on P6. Cases additional to those shown in Fig. 5 further illustrate the patterns of callosal axons labeled after injections of BDA placed medial to the 17/18a border (A) or on the 17/18a border (B). The asterisk in A marks the location of labeled fibers located in more medial portions of area 17 (presumptive MCR). The BDA injections may have encroached onto the MCR, and these fibers probably reflect the mirror-symmetric projection of the MCR. Low-power micrographs at bottom right show BDA-labeled fields (arrowheads) in the dLGN ipsilateral to the injections. Other conventions as in Fig. 5.
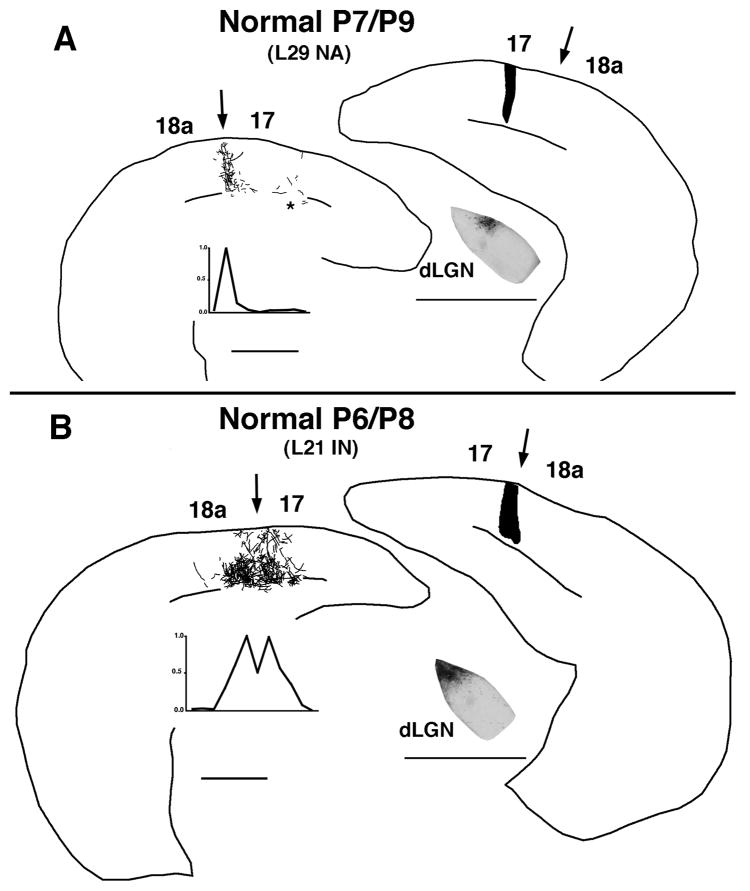
We also studied older animals to determine whether subsequent growth of callosal innervation and elaboration of terminal arbors temporarily degrade the topography of callosal fibers that we observed at P6. As in younger pups, we found that injections placed a few hundred microns medial to the 17/18a border produced fields of labeled fibers that were narrow and centered on the 17/18a border. Figure 7A illustrates this result in a case injected at P7 and perfused at P9. The asterisk in Figs. 7A marks the location of labeled fibers in more medial regions of area 17 (presumptive MCR, see Fig. legend). Similar results were obtained in two additional animals injected at P6 and perfused at P8 (data not shown). Likewise, we found that an injection of BDA at the 17/18a border in older animals produced a labeling pattern that was similar to that that observed in younger animals, that is, the labeling field was broader, and labeling density peaked on both sides of the 17/18a border (cf. Figs. 7B and 5B,6B). Figure 7B illustrates this labeling pattern in a case injected at P6 and perfused at P8. In summary, the results we obtained in animals injected at P6–7 and perfused at P8–9 are similar to those we obtained in younger animals (P4/P6), as well as in adult normal rats (Olavarria and Hiroi, 2003). These data are in agreement with our experiments using retrograde tracers (Fig. 2), and provide further evidence that the topography of the callosal map is not temporarily degraded by the progressive invasion of callosal fibers and elaboration of terminal arbors.
Fig. 7.
Organization of callosal projections revealed with the anterogradely transported tracer BDA in the lateral callosal region of normal pups injected on P6–7 and perfused on P8–9. A: Case (L29 NA) injected on P7 and perfused on P9. The BDA injection was placed at a location medial to the 17/18a border (right panel). At left, drawing shows the distribution of BDA-labeled fibers reconstructed from coronal sections. The asterisk marks the location of labeled fibers located in more medial portions of area 17 (presumptive MCR). The BDA injections may have encroached onto the MCR, and these fibers probably reflect the mirror-symmetric projection of the MCR. The distribution of labeling density is represented in the bottom left graph. Low-power micrograph shows the location of the anterogradely labeled field (area of dark labeling) in the right dorsal lateral geniculate nucleus (dLGN; medial is to the left). Scale bars = 1.00 mm. B: Case (L21 IN) injected on P6 and perfused on P8. In this case the BDA injection was placed at the 17/18a border, as confirmed by the pattern of dLGN labeling shown in the low-power micrograph at bottom right. Other conventions as in A.
In addition to injections into the presumptive LCR, we also injected BDA into medial regions of area 17, within the presumptive MCR. When the BDA injection reached layers 5 and 6, we observed short labeled fibers that were largely restricted to infragranular layers and distributed in regions roughly mirror-symmetric with the injection sites (data not shown). These results indicate that the MCR in young animals, as in adults, project in a mirror-symmetric fashion, and that the projections are largely restricted to infragranular layers. Moreover, when the BDA injections did not reach beyond layer 4, very few or no labeled fibers were observed in contralateral area 17, confirming our observations made with retrograde tracers that most callosally projecting supragranular cells in medial area 17 of young animals do not give off callosal fibers that invade gray matter to a significant extent.
In enucleated animals we found that the organization of anterogradely labeled fibers presented anomalies that are similar to those observed with retrograde tracers in enucleated pups of the same ages. Figure 8 shows the projection patterns in animals injected with BDA at P4 and perfused at P6, while Figure 9 shows the results from animals injected at P6 and perfused at P8. As in normal pups, we observed that, by P6, fibers of simple architecture extend to supragranular layers, often reaching layers 1 and 2. Our data show that restricted injections of BDA produce relatively broad and sparse fields of labeled fibers that are in rough correspondence with the location of the injection sites. For instance, in Figures 8A and 9B, the injections fell within 400 μm from the 17/18a border. Whereas in normal animals such injections would have produced narrow accumulations of labeled fibers at the 17/18a border (Figs. 5A, 6A, 7A), injections in the enucleated animals produced relatively broad labeled fields located in the lateral half of area 17, in rough correspondence with the location of the injection site. In Figure 9A, the injection was displaced further medially, and the resulting labeled field also extended into more medial portions of area 17 (cf. panels A,B in Fig. 9). Finally, Figure 8B shows a case in which the injection of BDA fell approximately on the 17/18a border. This injection produced an extensive field of labeled fibers that occupied significant portions of areas 17 and 18a. This field is much larger than that produced by a similar injection in normal pups (cf. Fig. 8B and Figs. 5B,6B,7B). Moreover, the decrease in the density of labeled fibers observed at the 17/18a border in this case likely reflects the fact that, unlike normal animals, callosal connections are often reduced or absent within a narrow region centered at the 17/18a border in enucleated animals (Olavarria et al., 1987).
Fig. 8.
Organization of callosal projections revealed with the anterogradely transported tracer BDA in pups enucleated at birth. Tracer injections on P4 and perfusion on P6. A: Pup (case L42–3A) that received an injection of BDA at a location medial to the prospective 17/18a border. Right panels show drawing (top) and low-power micrograph (bottom) of injection site. At left, drawing shows the distribution of BDA-labeled fibers reconstructed from coronal sections. Labeled fibers were observed throughout cortical layers 1–6. The distribution of labeling density is represented in the bottom left graph. Bottom right inset illustrates the location (outline) of the anterogradely labeled field in the right dorsal lateral geniculate nucleus (dLGN; medial is to the left). Scale bars = 1.00 mm. B: In this case (L44), the BDA injection was placed at the 17/18a border. Other conventions as in A.
Fig. 9.
Organization of callosal projections revealed with the anterogradely transported tracer BDA in pups enucleated at birth. Tracer injection on P6 and perfusion on P8. A: Pup (case L22F) that received an injection of BDA at a location medial to 17/18a border. Right panels show drawing (top) and low-power micrograph (bottom) of injection site. At left, drawing shows the distribution of BDA-labeled fibers reconstructed from coronal sections. The distribution of labeling density is represented in the bottom left graph. Bottom right inset illustrates the location (outline) of the anterogradely labeled field in the right dorsal lateral geniculate nucleus (dLGN; medial is to the left). Scale bars = 1.00 mm. B: In this case (L27A), the BDA injection was placed closer to the 17/18a border than in the case shown in A. Drawing at left shows the distribution of BDA-labeled fibers reconstructed from coronal sections. Graph (black line) at bottom left shows the distribution of labeling density. Graph from A (gray line) has been superimposed for comparison. Other conventions as in A.
Correlating width of callosal fields and location of injection sites
In both normal and enucleated animals we found that the data obtained with retrograde tracers was in good agreement with that obtained with BDA, not only in regard to topography, but also in the size of the labeled fields. In normal animals, injections placed a few hundred microns medial to the 17/18a border produced labeled fields that were narrow and centered on the 17/18a border, whereas injections at the 17/18a border produced broader fields whose labeling density peaked at both sides of the 17/18a border. In contrast, injections of similar size in enucleated animals produced labeled fields that appeared to be much broader than those observed in normal animals. To quantify these observations, we measured the half-widths of the distributions of labeling density in normal and enucleated pups, and pooled the results from retrograde and anterograde experiments in each group (Fig. 10A). In normal animals (black bars in Fig. 10A), we found that the width of the labeled fields centered on the 17/18a border (left black bar, mean: 266 μm) was significantly smaller (p < 0.01) than the width of the fields produced by injections on the 17/18a border (right black bar, mean: 766 μm). The latter fields typically peaked on either side of the 17/18a border (Figs. 5B, 6B, 7B). The width of the callosal fields in enucleated animals (mean:1,421 μm, gray bar in Fig. 10A) was significantly larger (p < 0.01) than the width of the fields in normal animals.
Fig. 10.
A: Comparison of the sizes of the callosal fields in normal (black bars) and in enucleated (gray bar) rats. For each animal, the size of the callosal field corresponds to the half-width of the distribution of callosal labeling density. The means and SEM are graphed. In normal animals, the size of the callosal fields resulting from BDA injections located medial to the 17/18a border (left black bar) are significantly smaller (P < 0.01) than the size of the fields resulting from BDA injections on the 17/18a border (right black bar). The size of callosal fields in enucleated animals (gray bar) are significantly larger (P < 0.01) than the fields in normal animals. B: Scatter plot correlating the width of the callosal fields with the location of the injection sites in area 17 of normal (black dots), and enucleated (gray dots) pups. The location of the injection sites in area 17 was estimated from the pattern of BDA labeling in the dLGN ipsilateral to the cortical injections (see Materials and Methods).
We then correlated the width of the callosal fields with the location of the injection sites in area 17. The location of the injection sites in area 17 was estimated by measuring the distance from the center of the labeled field in the ipsilateral dLGN to the mediodorsal tip of this nucleus. This distance was expressed as a percentage of the length of a line extending through the middle of the nucleus from its mediodorsal to lateroventral borders (see Materials and Methods). Figure 10B shows that the data points from normal animals (black dots) form two separate clusters. The right cluster derives from injections judged to have fallen within the presumptive LCR, but largely outside the putative transition zone. For these injections, the labeled fields in the dLGN are located at distances that range from 28.7 to 44.4% (average: 35.5%) of the length of the nucleus in its mediodorsal to lateroventral axis. The left cluster of black dots in Figure 10B derives from injections judged to have fallen on the 17/18a border, and the locations of the dLGN labeled fields for these injections range from 8.3 to 14.5% (average: 11.4%). In order to compare these results with data from adult animals, we performed similar calculations of the location of the labeled geniculate fields in animals from previous studies (Lewis and Olavarria, 1995; Olavarria and Hiroi, 2003). We found that the locations of the labeled fields in the dLGN for injections falling on the transition zone at the 17/18a border in adult animals (n = 6) were very similar to the locations for the corresponding injections in pups, ranging from 7.14 to 16% (average: 12.15%). Likewise, the locations of the labeled fields in the dLGN for injections falling away from the transition zone in adult animals (n = 7) were similar to the locations for the corresponding injections in pups, ranging from 28.6 to 44% (average: 37.5%). This comparison strengthens our results in normal pups and provides further evidence that the topography of callosal connections is present at early stages of pathway development. In contrast to our data from normal pups, the data from enucleated pups did not show a correlation between the width of the callosal fields and the location of the injection sites (gray dots in Fig. 10B). Moreover, injections falling medial to the 17/18 border never resulted in fields that were as narrow as those observed after similarly located injections in normal pups.
Callosal projections before P6
Our analysis with retrograde and anterograde tracers provides evidence that the normal callosal map, as well as the anomalous map observed in neonatally enucleated animals, can be demonstrated by P6–7, just as growing fibers reach superficial layers of area 17. However, it is possible that a significant number of fibers have already reached superficial layers of gray matter before P6, and that elimination of many of these fibers contributes to the formation of the maps we observed at P6. To examine this possibility we compared the overall pattern of labeled callosal fibers that is present at P6 with that present at earlier days. We performed multiple injections of BDA across area 17 in P1, P2, and P3 pups, and analyzed the distribution of anterogradely labeled fibers at P3, P4, and P5, respectively. While a rich contingent of labeled fibers was present in white matter below area 17 at P3, we observed only a few, short fibers invading deep aspects of the gray matter (data not shown). By P4 there were a few more fibers invading the gray matter and most of them were short and largely restricted to cortical layer 6 (Fig. 11, top panel). Moreover, these fibers were located throughout the mediolateral extent of presumptive area 17, without an obvious bias towards the location of the future callosal region in lateral area 17. By P5 the number of fibers in layer 6 had increased to some extent, and a few longer fibers extended to layer 5, and occasionally up to the cortical plate from which layers 2 and 3 will differentiate. At this age, most short fibers were found throughout area17, while the longer ones were located preferentially in lateral area 17, in the presumptive 17/18a region (Fig. 11, bottom panel). This progressive ingrowth of callosal fibers closely parallels the cytoarchitectural differentiation of the cortical layers from the dense cortical plate. Our data from animals perfused at P6 indicate that, by the time all cortical layers have differentiated from the cortical plate (Ignacio et a., 1995; Fig 4B), a greater number of callosal fibers reach superficial cortical layers, including layer 1. These observations are in general agreement with previous reports in rodents on the growth of callosal fibers in visual cortex (Fish et al., 1991; Ding and Elberger, 2001) and other cortical areas (Norris and Kalil, 1992; Hedin-Pereira et al., 1999). Based on our results, it is unlikely that the topographic maps we observed by P6–7 result from the elimination of callosal fibers that have reached supragranular layers, since there are few callosal fibers in gray matter prior to P6, and most of these are short and restricted to deep cortical layers. These observations are also consistent with previous results showing that tracer injections restricted to gray matter in occipital cortex of P4 pups fail to produce significant retrograde or anterograde labeling in the contralateral hemisphere (Olavarria and Van Sluyters, 1985).
Fig. 11.

Overall distribution of callosal projections revealed after multiple injections of the anterogradely transported tracer BDA in normal pups. Drawings illustrate the distribution of BDA-labeled callosal fibers in representative coronal sections in a case injected at P2 and perfused at P4 (top drawing), and in a case injected at P3 and perfused at P5 (bottom drawing). Medial is to the right. The location of presumptive area 17 is indicated in the bottom drawing. Cortical layers are indicated at left. CP = cortical plate; scale bar = 1.00 mm.
DISCUSSION
Our results show that the normal callosal map, as well as the anomalous map observed in neonatally enucleated animals, can be demonstrated by P6–7, when growing fibers reach superficial layers of area 17. In normal P6-P7 pups we were able to recognize the topographical features that are characteristic of the LCR and MCR in adult rats (Lewis and Olavarria, 1995): injections into medial area 17 (presumptive MCR) produced labeling in mirror-symmetric locations in contralateral area 17, while injections into the presumptive LCR produced non mirror-symmetric labeled fields. As in adult rats, injections in the presumptive LCR placed a few hundred microns away from the 17/18a border produced restricted fields of labeling centered on the contralateral 17/18a border, while injections falling on the 17/18a border of normal pups produced relatively dense, focused labeled fields located immediately medial and lateral to the 17/18a border on either side of the transition zone. The latter fields were significantly wider than those centered on the 17/18a border. These observations, summarized in Figure 10, show that, as it occurs in adult rats, the location and width of the callosal fields resulting from restricted injections into lateral area 17 of normal pups are highly dependent on the precise location of the injection sites with respect to the 17/18a border.
Compelling evidence that the topography of callosal connections is present at early stages of development comes from our observation that the fields of labeled callosal fibers resulting from injections located medial to the 17/18a border were tightly focused on the contralateral 17/18a border even when two BDA injections were placed adjacent to each other (Figs, 5A, 6A). These results are consistent with observations in adults indicating that a narrow transition zone located at the 17/18a border receives convergent projections from regions in the LCR located outside the transition zone (Lewis and Olavarria, 1995). However, while tracers injections into the 17/18a border of adult rats typically produced well-defined labeled fields located immediately medial and lateral to the 17/18a border, on either side of the transition zone (Lewis and Olavarria, 1995; Olavarria and Hiori, 2003), the separation of these fields was often less complete in pups due to the presence of either labeled cells or labeled fibers between them. Rather than a lack of topography, the reduced definition of these fields may be due to the small width of the transition zone in young pups relative to the size of the injections. Even if tracer injections were centered in the transition zone, some regions outside the transition zone may have become involved, producing some labeling inside the transition zone in the contralateral hemisphere. Something similar is observed in adult mice, whose transition zones are narrower than in adult rats (see Fig. 4B in Olavarria and Hiroi, 2003). Nevertheless, the possibility that removal of some, incorrectly placed, cells and fibers in the transition zone contributes to the sharpening of the projection after injections into the 17/18a border cannot be ruled out.
In addition to the main labeled field centered on the 17/18a border, tracer injections placed a few hundred microns medial to the 17/18a border produced a number of labeled cells, or anterogradely labeled fibers, in medial area 17 of some normal pups, in the prospective MCR (fields marked with asterisks in Figs. 2A, 5A, 6A,7A). It is unlikely that these fields correspond to diffuse labeling in pups because labeling in this region after similarly placed injections can also occur in the mature callosal pathway (see Fig. 7 in Lewis and Olavarria, 1995; and Fig. 8A in Olavarria and Hiroi, 2003). It is therefore more likely that in all these young and adult cases the injections encroached from the medial border of the LCR into the MCR, and the labeling in medial area 17 simply reflects the mirror-symmetric projection of the MCR. If this interpretation is correct, then these observations in young rats provide further support to the idea that the topography of callosal connections in lateral (LCR) and medial (MCR) area 17 is present as early as P6, several days before the overall distribution of callosal cells in area 17 matures (Olavarria and Van Sluyters, 1985).
In our experiments using retrograde tracers, the pipette containing the tracer was lowered to about 600–800 μm deep in order to avoid involvement of the white matter. Since RB and GB do not diffuse significantly from the injection site (Katz et al., 1984), in at least some cases little tracer was deposited in deep cortical layers. As a result, we may have not been able to systematically label callosal cells giving rise to short fibers that only penetrated into deep cortical layers at the time of the injection. Likewise, some of our BDA injections may have failed to reach deep neurons that give rise to short fibers restricted to deep cortical layers in the contralateral cortex. Thus, it remains possible that several short branches may emerge in a diffuse pattern from a given parental axon, and that only those fibers that emerge from the “correct” location continue to grow into upper cortical layers. This possibility is consistent with studies of the development of both callosal connections (Aggoun-Zouaoui and Innocenti, 1994, see Introduction) and the projection from area 17 to ipsilateral area 18 in the kitten (Caric and Price, 1996). Using retrograde and anterograde tracers, Caric and Price (1996) found that area 17 projections to deep layers in area 18 were highly convergent/divergent, whereas projections to more superficial layers were more accurate from the outset. Preliminary, high resolution time-lapse observations of the development of the 17 to 18a projection in rat pups revealed that many small branches do emerge and retract over short time intervals from a given segment of the parental axon (Olavarria et al., 2003), and it is likely that highly dynamic short branches also exist in the developing callosal projection (Halloran and Kalil, 1994). However, because of their short length, it is not technically feasible to restrict tracer uptake to very small branches without also compromising nearby parental axons that navigate within lower cortical layers or white matter.
Finally, it should be noted that while the analysis of fixed tissue can provide information about the fine topography of emerging connections, this approach does not have the temporal resolution that is needed for a detailed description of local, dynamic processes that may occur during map formation. Studies in tadpoles using time-lapse techniques and high-resolution imaging methods show that retinotectal axon arbors develop by a continuous process of iterative addition and retraction of very small branches (e.g., O’Rourke et al., ’94; Alsina et al., ’01; Ruthazer et al., ’03). Branch addition and retraction are concurrent and, in time, biased toward addition in topographically correct places and toward elimination in incorrect places, with the result that topographic projections emerge through small incremental changes in arbor architecture. Preliminary time-lapse data from rat pups show that a continuous process of iterative addition and retraction of small axonal branch tips also occurs during the development of cortico-cortical connections (Olavarria et al., 2003). If callosal connections develop by a similarly dynamic process, analysis of labeled fibers in fixed tissue at different ages may give the impression of directed growth. It will therefore be important to extend the present analysis with high-resolution time-lapse studies in order to determine the extent to which the role that retinal input has on the development of organized cortico-cortical maps depends on the regulation of rapid, small-scale dynamic processes that are not detectable in fixed tissue.
Retinotopic precision of interstitial branch invasion depends on retinal input
Restricted injections of anterograde or retrograde tracers in normal pups produced focused labeled fields that conformed to the topography of the mature callosal map, whereas single restricted injections in enucleated animals produced highly divergent/convergent projections. The diffuse projection patterns in enucleated animals may result from parental axons giving off exuberant projections. For instance, multiple, widely separated interstitial branches may extend from single parental axons and/or highly exuberant terminal arbors may develop from each interstitial branch. If the hypothesis that the normal, non mirror-symmetric map emerges from an initial mirror-symmetric map through corrective pruning processes driven by retinal input is correct, then elimination of subsets of these exuberant projections would produce the normal topography. We tested whether projections from single axons in enucleated animals are exuberant by analyzing the incidence of double-labeled cells after closely placed injections of two fluorescent tracers. Contrary to this idea, we found very few double-labeled cells even when there was a large area of overlap between the populations of cells labeled by each tracer. In fact, the incidence of double-labeled cells in enucleated animals was as low as that in normal pups of the same age. Since at the age these experiments were performed interstitial fibers in both normal or enucleated animals still had relatively simple architecture, the paucity of double-labeled cells suggests that multiple, widely distributed interstitial branches that grow from single parental axons into middle and supragranular area 17 are rare in both normal and enucleated animals. Moreover, if multiple branches do grow from single axons into middle or upper cortical layers, these data suggest that the points of emergence of these branches are close to each other, or that the fibers converge to the same terminal region as they grow toward the pia (see Hedin-Pereira et al., 1999). This conclusion does not preclude the possibility that the parental axons give off additional interstitial branches in other, more distant, cortical areas, although this appears to be rare (Hedin-Pereira et al., 1999).
Our results therefore do not support the idea that retinal input guides callosal map formation primarily through the promotion of large-scale elimination of long, non-topographic branches. Instead, our analysis suggests that retinal input specifies the sites on the parental axons from which interstitial branches will grow to invade middle and upper cortical layers. In this manner, retinal input would ensure that the location where long, invading interstitial branches form on the parental axon is accurately related to the topographical location of the soma that gives rise to the parental axon. In contrast, the absence of retinal input appears to result in a merely loose relation between the emergence sites of long side branches and the actual position of the cell soma in the contralateral hemisphere. This suggests that the cues that determine the mirror-symmetric callosal map in enucleated pups exert only a weak control on the topography of fiber ingrowth. These ideas are illustrated in the diagram shown in Figure 12. What prevents the growth and extension of multiple, widely separated interstitial branches from single parental axons in both normal and enucleated pups is not known, but it is possible that the preferential growth of branches from one locus on the axon both inhibits the extension and promotes the destabilization of other branches through mechanisms that are intrinsic to the axon. In addition to specifying the sites on the parental axons from which interstitial branches will grow, retinal input appears to influence the subsequent elaboration of terminal arbors, as suggested by the report that a significant proportion of callosal arbors in striate cortex of neonatally enucleated hamsters are more widespread than callosal arbors in normal hamsters (Fish et al., 1991).
Fig. 12.
Diagram illustrates hypothesis regarding the emergence of callosal maps. The top panels in A and B summarize the development of callosal projections observed by P6 in both normal (A) and bilaterally enucleated (B) animals. The arrows mark the location of area 17. At right, injections placed at the 17/18a border and medial to it are represented in black and gray, respectively. At left, the corresponding colors are used to represent the patterns of callosal interstitial branches labeled by each tracer. These diagrams illustrate that both the normal (non-mirror symmetric, A), as well as the abnormal (mirror symmetric, B) callosal maps are present by P6. Middle panels in A and B illustrate the emergence of long interstitial branches from 3 representative callosal cells labeled by each tracer. In normal animals (middle panel in A), our results suggest that retinal input specifies the sites on the parental axons from which interstitial branches will grow to invade middle and upper cortical layers. Retinal input would ensure that the location where invading interstitial branches form on the parental axon is accurately related to the topographical location of the soma that gives rise to the parental axon. Note that interstitial branches in 18a in the normal animal are not connected to parental axons to indicate that, at present, it is not known whether single parental axons can give rise to branches on either side of the 17/18a border. In enucleated animals (middle panel in B), our results suggest that the cues that determine the mirror-symmetric callosal map in enucleated pups exert only a weak control on the topography of fiber ingrowth. These cues would only establish a loose relationship between the emergence sites of long side branches and the actual position of the cell soma in the contralateral hemisphere. Bottom panels in A and B represent the relative distributions of axons labeled by each tracer.
Previous studies in the somatosensory system provide evidence that early thalamocortical projections show little or no branching and a high degree of topographical precision (Dawson and Killackey, 1985; Naegele et al., 1988; Agmon et al., 1993; 1995; Catalano et al., 1996; but see Krug et al., 1998). In addition, it has been suggested that the abnormal thalamic projections that develop in animals with neonatal infraorbital nerve cut result from an effect of the nerve cut on the initial growth of thalamic axons into the cortex, rather than on a subsequent remodeling process (Jensen and Killackey, 1987; Catalano et al., 1995). Similarly, in the visual cortex, recent studies suggest that formation of eye-specific thalamocortical projections is best characterized as a progressive developmental event rather than a regressive remodeling of an initially diffuse projection (Crowley and Katz, 2000). These comparisons suggest that the processes that characterize the development of normal and abnormal callosal maps are not fundamentally different from those characterizing the development of thalamocortical maps. However, as noted in the Introduction and discussed below, there seems to be marked differences between the development of callosal and retinofugal maps.
Comparison with the development of retinofugal maps in rodents
During development of the normal retino-collicular projection in rodents, interstitial branches and arbors develop not only in the future termination zone, but also in areas several times larger than the mature termination zone, as well as virtually throughout the extent of the overshooting parental axons (Simon and O’Leary, 1992; McLaughlin et al., 2003). Simon and O’Leary (1992) emphasize that “The development of topographic order is achieved by a major remodeling of the initially disordered projection involving the rapid elimination of aberrantly positioned axons, axon segments, branches, and arbors, concurrent with the preferential elaboration of arbors at topographically appropriate positions”. In contrast, our data provide evidence that emergence of the callosal map does not depend on the removal of topographically aberrant branches and arbors. What may account for these differences? An important difference between the development of retinofugal (Godement et al., 1984; Simon and O’Leary, 1992; McLaughlin et al., 2003) and corticocortical projections in rodents relates to the timing of invasion of axonal branches into the target. In the superior colliculus of rats, for instance, retinal axons have begun to give off branches at topographically correct and incorrect places as early as embryonic age 20–21 (Simon and O’Leary, 1992). By P2, axon branches and nascent arbors are present throughout the SC, although a bias towards the correct termination zone is observed. At P4, branches emerging from the overshooting retinal parental axons form relatively dense fields that, while biased towards the location of the correct termination zone, are several times larger than the size of future termination zone. At this age, collateral branches are also seen in clearly wrong positions along the overshooting axon (Simon and O’Leary, 1992). In sharp contrast, we found that collateral branches emerging from callosal parental axons are virtually absent prior to P4. However, after P4, the changes in the retinocollicular pathway resemble those we observed in the callosal pathway in the sense that there is a rapid emergence of precise topography (See Fig. 4 in Simon and O’Leary, 1992).
This comparison suggests the intriguing possibility that, as in the cortex, the eyes influence the topography of the retinocollicular map during a limited period starting at about P4. If this is the case, then it is possible that other, less precise cues guide the emergence of collicular interstitial branches before P4, resulting in the emergence of topographically incorrect branches and arbors. In this respect, the imprecise development of retinocollicular branches up to P4 would resemble how callosal projections develop in older pups in the absence of the eyes. After P4, the retinocollicular pathway would be under the guidance of retinal cues, which would promote the preferential invasion of fibers and elaboration of terminal arbors at the correct termination zone, as well as the destabilization of topographically incorrect fibers and arbors. Evidence for two distinct modes of growth in the retinocollicular projection has been described in the hamster (Jhaveri et al., 1991). Unlike the scenario in which retinal cues would operate on an existing diffuse distribution of retinocollicular projections, our data suggest that retinal mechanisms that specify normal callosal topography between P4 and P6 do not operate on an existing exuberant distribution of intracortical callosal fibers. Rather, the eyes specify callosal topography just as interstitial branches form and elongate, thereby obviating the need for the removal of exuberant branches that, had they existed before P4, would have developed under the influence of less precise cues. Further support to the idea that the eyes influence the topography of the retinocollicular map during a limited time window comes from the recent demonstration that development of precise retonocollicular maps requires spontaneous retinal waves during a well-defined postnatal period (McLaughlin et al., 2003). These authors showed that diffuse retinocollicular projections caused by the lack of retinal activity waves remain diffuse even after retinal waves are reinstated at about P8. The above discussion raises the possibility that what might appear as different strategies in the development of topographical projections among species, or among different stages of the visual pathway in a given species, may be at least partly explained by differences in the developmental time course of the projections in question with respect to the timing of mechanisms specifying the topography of the projections.
Timing of interstitial branches emergence and growth does not depend on retinal input
Our data provide evidence that retinal input specifies the location on the parental axon where interstitial branches will grow towards the pia. Does retinal input also regulate the timing of interstitial branch emergence and growth? We observed that callosal fibers begin a progressive invasion of callosal fibers into the gray matter at about P4, and a population of these fibers extends to upper cortical layers in lateral area 17 of normal pups by P6. The coincidence of the timing of this progressive growth with the P4-P6 critical period might suggest that retinal input not only specifies the callosal map but also triggers the outgrowth of interstitial branches. However, our observation that callosal fibers in enucleated animals also reach superficial cortical layers by P6 provides evidence that the timing of interstitial branch emergence and growth does not depend on retinal input. Moreover, the progressive invasion of fibers into gray matter closely parallels the maturation of cortical layers (Ignacio et al., 1995). Indeed, as interstitial branches penetrate the cortical layers, they tend to remain largely restricted to those lower layers that have differentiated from the cortical plate (Norris and Kalil, 1992). Interstitial branches reach upper cortical layers by P6, at a time when differentiation of cortical layers from the cortical plate is virtually complete (Hicks and D’Amato, 1968; Norris and Kalil, 1992). Previous findings that the timing of layer differentiation and callosal maturation is not affected by enucleation (Olavarria et al., 1987) further support the idea that retinal input regulates the retinotopic precision of interstitial branch invasion, but not the timing of interstitial branch emergence and growth. The eyes therefore seem to act at precisely the appropriate window of time to guide the retinotopy of interstitial branch invasion. The mechanisms that determine the precise timing for this retinal action remain unknown.
In conclusion, our results do not support the hypothesis that the mirror-symmetric pattern of callosal connections observed in neonatally enucleated rats represents an immature stage of callosal development, from which the normal, non mirror-symmetric pattern emerges through large-scale corrective pruning processes driven by retinal input. Rather, our results from normal pups suggest that the invasion and growth of most callosal fibers into middle and upper layers are in good topographic correspondence with the location of the parental cell soma in the contralateral hemisphere. How does retinal input regulate the topographic accuracy with which interstitial branches invade the cortical target? We have previously proposed that the invasion of callosal fibers may be guided by a blueprint laid down during the P4-P6 window by influences delivered to both hemispheres through a system of bilateral projections emanating from temporal retina (Olavarria and Hiroi, 2003). This blueprint could be demarcated by chemical cues that remain effective after P6. For instance, topographic chemical cues of retinal origin delivered through thalamocortical projections could be “read” by the parental axon’s growth cone as it navigates past the target region, leading to the emergence/growth of interstitial branches at the appropriate location (Szebenyi et al., 1998), or the chemical cues could act directly at specific locations along the parental axon shaft to induce the emergence/growth of side branches independently from growth cone recognition mechanisms. Alternatively, interhemispherically correlated activity driven by spontaneously active ganglion cells (Maffei and Galli-Resta, 1990) in temporal retina could promote the stabilization of retinotopically matched callosal linkages. Under this influence, the callosal interstitial branches that invade the cortex during the P4-P6 critical period may establish a sufficient number of retinotopically matched synaptic contacts to form a blueprint of the callosal map. After P6, this blueprint would guide the growth of additional, correctly placed interstitial branches, and the strengthening of topographically correct synaptic contacts (Olavarria and Hiroi, 2003). If the eyes are removed at P4 or earlier, the blueprint established by either chemical or activity cues would not be laid down and callosal branches would instead invade cortical layers in a mirror-symmetric fashion guided by less accurate cues that may be already present prior to P4. Determining the extent to which chemical or activity cues are involved in the development of callosal maps will be an important step toward understanding the retinally-driven cellular and molecular processes that specify the topography of cortico-cortical maps.
Acknowledgments
We thank E. Ruthazer and M. Yanos for comments on the article, and J. Lasiene for helping with some experiments.
LITERATURE CITED
- Aggoun-Zouaoui D, Innocenti GM. Juvenile visual callosal axons in kittens display origin-and fate-related morphology and distribution of arbors. Eur J Neurosci. 1994;6:1846–1863. doi: 10.1111/j.1460-9568.1994.tb00577.x. [DOI] [PubMed] [Google Scholar]
- Agmon A, Yang LT, O’Dowd DK, Jones EG. Organized Growth of thalamocortical axons from the deep tier of terminations into layer IV of developing mouse barrel cortex. J Neurosci. 1993;13:5365–5382. doi: 10.1523/JNEUROSCI.13-12-05365.1993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Agmon A, Yang LT, Jones EG, O’Dowd DK. Topological precision in the thalamic projection of neonatal mouse barrel cortex. J Neurosci. 1995;15:549–565. doi: 10.1523/JNEUROSCI.15-01-00549.1995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Alsina B, Vu T, Cohen-Cory S. Visualizing synapse formation in arborizing optic axons in vivo: dynamics and modulation by BDNF. Nat Neurosci. 2001;4:1093–1101. doi: 10.1038/nn735. [DOI] [PubMed] [Google Scholar]
- Barone P, Dehay C, Berland M, Kennedy H. Role of directed growth and target selection in the formation of cortical pathways: prenatal development of the projection of area V2 to area V4 in the monkey. J Comp Neurol. 1996;374:1–20. doi: 10.1002/(SICI)1096-9861(19961007)374:1<1::AID-CNE1>3.0.CO;2-7. [DOI] [PubMed] [Google Scholar]
- Catalano SM, Robertson RT, Killackey HP. Rapid alteration of thalamocortical axon morphology follows peripheral damage in the neonatal rat. Proc Natl Acad Sci. 1995;92:2549–2552. doi: 10.1073/pnas.92.7.2549. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Catalano SM, Robertson RT, Killackey HP. Individual axon morphology and thalamocortical topography in developing rat somatosensory cortex. J Comp Neurol. 1996;366:36–53. doi: 10.1002/(SICI)1096-9861(19960325)367:1<36::AID-CNE4>3.0.CO;2-K. [DOI] [PubMed] [Google Scholar]
- Caric D, Price DJ. The organization of visual corticocortical connections in early postnatal kittens. Neurosci. 1996;73:817–829. doi: 10.1016/0306-4522(96)00059-0. [DOI] [PubMed] [Google Scholar]
- Chalupa LM, Killackey HP, Snider CJ, Lia B. Callosal projection neurons in area 17 of the fetal rhesus monkey. Dev Brain Res. 1989;46:303–308. doi: 10.1016/0165-3806(89)90294-0. [DOI] [PubMed] [Google Scholar]
- Crowley JC, Katz LC. Early development of ocular dominance columns. Science. 2000;290:1321–1324. doi: 10.1126/science.290.5495.1321. [DOI] [PubMed] [Google Scholar]
- Dawson DR, Killackey HP. Distinguishing topography and somatotopy in the thalamocortical projection of the developing rat. Dev Brain Res. 1985;17:309–313. doi: 10.1016/0165-3806(85)90162-2. [DOI] [PubMed] [Google Scholar]
- Dehay C, Kennedy H, Bullier J, Berland M. Absence of interhemispheric connections of area 17 during development in the monkey. Nature. 1988;331:348–350. doi: 10.1038/331348a0. [DOI] [PubMed] [Google Scholar]
- Ding S-L, Elberger AJ. Postnatal development of biotinylated dextran amine-labeled corpus callosum axons projecting from the visual and auditory cortices to the visual cortex of the rat. Exp Brain Res. 2001;136:179–193. doi: 10.1007/s002210000576. [DOI] [PubMed] [Google Scholar]
- Fish SE, Rhoades RW, Bennett-Clarke CA, Figley B, Mooney RD. Organization, development and enucleation-induced alterations in the visual callosal projection of the hamster: single axon tracing with phaseolus vulgaris leucoagglutinin and Di-I. Eur J Neurosci. 1991;3:1255–1270. doi: 10.1111/j.1460-9568.1991.tb00059.x. [DOI] [PubMed] [Google Scholar]
- Godement P, Saillour P, Imbert M. Thalamic afferents to the visual cortex in congenitally anophthalmic mice. Neurosci Lett. 1979;13:271–278. doi: 10.1016/0304-3940(79)91506-4. [DOI] [PubMed] [Google Scholar]
- Godement P, Salaun J, Imbert M. Prenatal and postnatal development of retinogeniculate and retinocollicular projections in the mouse. J Comp Neurol. 1984;230:552–575. doi: 10.1002/cne.902300406. [DOI] [PubMed] [Google Scholar]
- Guillery RW, Ombrellaro M, LaMantia AL. The organization of the lateral geniculate nucleus and of the geniculocortical pathway that develops without retinal afferents. Dev Brain Res. 1985;20:221–233. doi: 10.1016/0165-3806(85)90109-9. [DOI] [PubMed] [Google Scholar]
- Hedin-Pereira C, Lent R, Jhaveri S. Morphogenesis of callosal arbors in the parietal cortex of hamsters. Cer Cortex. 1999;9:50–64. doi: 10.1093/cercor/9.1.50. [DOI] [PubMed] [Google Scholar]
- Hicks SP, D’Amato CJ. Cell migration to the isocortex in the rat. Anat Rec. 1968;160:619–634. doi: 10.1002/ar.1091600311. [DOI] [PubMed] [Google Scholar]
- Halloran MC, Kalil K. Dynamic behaviors of growth cones extending in the corpus callosum of living cortical brain slices observed with video microscopy. J Neurosci. 1994;14:2161–2177. doi: 10.1523/JNEUROSCI.14-04-02161.1994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ignacio MPD, Kimm EJ, Kageyama GH, Yu J, Robertson RT. Postnatal migration of neurons and formation of laminae in rat cerebral cortex. Anat Embryol. 1995;191:89–100. doi: 10.1007/BF00186782. [DOI] [PubMed] [Google Scholar]
- Innocenti GM. The development of projections from cerebral cortex. Prog Sensory Physiol. 1991;12:65–114. [Google Scholar]
- Innocenti GM, Fiore L, Caminiti R. Exuberant projection into the corpus callosum from the visual cortex of newborn cats. Neurisci Lett. 1977;4:237–242. doi: 10.1016/0304-3940(77)90185-9. [DOI] [PubMed] [Google Scholar]
- Innocenti GM, Price DJ. Exuberance in the development of cortical networks. Nat Rev Neurosci. 2005;6:955–965. doi: 10.1038/nrn1790. [DOI] [PubMed] [Google Scholar]
- Jensen KF, Killackey HP. Terminal arbor of axons projecting to the somatosensory cortex of the adult rat: II. The altered morphology of thalamocortical afferents following neonatal infraorbital nerve cut. J Neurosci. 1987;7:3544–3553. doi: 10.1523/JNEUROSCI.07-11-03544.1987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jhaveri S, Edwards MA, Schneider GE. Initial stages of retinofugal axon development in the hamster: evidence for two distinct modes of growth. Exp Brain Res. 1991;87:371–382. doi: 10.1007/BF00231854. [DOI] [PubMed] [Google Scholar]
- Kaiserman-Abramof IR, Graybiel AM, Nauta WJH. The thalamic projection to cortical area 17 in a congenitally anophthalmic mouse strain. Neurosci. 1980;5:41–52. doi: 10.1016/0306-4522(80)90069-x. [DOI] [PubMed] [Google Scholar]
- Katz LC, Burkhalter A, Dreyer WJ. Fluorescent latex microspheres as a retrograde neuronal marker for in vivo and in vitro studies of visual cortex. Nature. 1984;310:498–500. doi: 10.1038/310498a0. [DOI] [PubMed] [Google Scholar]
- Katz LC, Shatz CJ. Synaptic activity and the construction of cortical circuits. Science. 1996;274:1133–1138. doi: 10.1126/science.274.5290.1133. [DOI] [PubMed] [Google Scholar]
- Kennedy H, Salin P, Bullier J, Horsburgh Topography of developing thalamic and cortical pathways in the visual system of the cat. J Comp neurol. 1994;348:298–319. doi: 10.1002/cne.903480211. [DOI] [PubMed] [Google Scholar]
- Krug K, Smith AL, Thompson ID. The development of topography in the hamster geniculo-cortical projection. J Neurosci. 1998;18:5766–5776. doi: 10.1523/JNEUROSCI.18-15-05766.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lewis JW, Olavarria JF. Two rules for callosal connectivity in striate cortex of the rat. J Comp Neurol. 1995;361:119–137. doi: 10.1002/cne.903610110. [DOI] [PubMed] [Google Scholar]
- Maffei L, Galli-Resta L. Correlation in the discharges of neighboring rat retinal ganglion cells during prenatal life. Proc Natl Acad Sci USA. 1990;87:2861–2864. doi: 10.1073/pnas.87.7.2861. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McLaughlin T, Torborg CL, Feller MB. Retinotopic map refinement requires spontaneous retinal waves during a brief period of development. Neuron. 2003;40:1147–1160. doi: 10.1016/s0896-6273(03)00790-6. [DOI] [PubMed] [Google Scholar]
- Montero VM, Brugge JF, Beitel RE. Relation of the visual field to the lateral geniculate body of the albino rat. J Neurophysiol. 1968;31:221–136. doi: 10.1152/jn.1968.31.2.221. [DOI] [PubMed] [Google Scholar]
- Naegele JR, Jhaveri S, Schneider GE. Sharpening of topographical projections and maturation of geniculocortical axon arbors in the hamster. J Comp Neurol. 1988;277:593–607. doi: 10.1002/cne.902770411. [DOI] [PubMed] [Google Scholar]
- Norris CR, Kalil K. Development of callosal connections in the sensorimotor cortex of the hamster. J Comp Neurol. 1992;326:121–132. doi: 10.1002/cne.903260111. [DOI] [PubMed] [Google Scholar]
- Olavarria JF. Non-mirror-symmetric patterns of callosal linkages in areas 17 and 18 in cat visual cortex. J Comp Neurol. 1996;366:643–655. doi: 10.1002/(SICI)1096-9861(19960318)366:4<643::AID-CNE6>3.0.CO;2-4. [DOI] [PubMed] [Google Scholar]
- Olavarria JF. Callosal connections correlate preferentially with ipsilateral cortical domains in cat areas 17 and 18, and with contralateral domains in the 17/18 transition zone. J Comp Neurol. 2001a;433:441–457. doi: 10.1002/cne.1152. [DOI] [PubMed] [Google Scholar]
- Olavarria JF. Influence of topography and ocular dominance on the functional organization of callosal connections in cat striate cortex. In: Payne B, Peters A, editors. The cat primary visual cortex. New York: Academic Press; 2001b. pp. 259–294. [Google Scholar]
- Olavarria JF, Hiroi R. Retinal influences specify cortico-cortical maps by postnatal day six in rats and mice. J Comp Neurol. 2003;459:156–172. doi: 10.1002/cne.10615. [DOI] [PubMed] [Google Scholar]
- Olavarria JF, Li Ch-P. Effects of neonatal enucleation on the organization of callosal linkages in striate cortex of the rat. J Comp Neurol. 1995;361:138–151. doi: 10.1002/cne.903610111. [DOI] [PubMed] [Google Scholar]
- Olavarria J, Malach R, Van Sluyters RC. Development of visual callosal connections in neonatally enucleated rats. J Comp Neurol. 1987;260:321–348. doi: 10.1002/cne.902600302. [DOI] [PubMed] [Google Scholar]
- Olavarria JF, Ruthazer ES, Lasiene J, Cline HT. Program No.35.11.2003 Abstract Viewer/Itinerary Planner. Washington, DC: Society for Neuroscience; 2003. Time-lapse confocal analysis reveals highly dynamic changes in the development of axonal projections from striate to extrastriate visual cortex in the rat. [Google Scholar]
- Olavarria J, Van Sluyters RC. Organization and postnatal development of callosal connections in the visual cortex of the rat. J Comp Neurol. 1985;239:1–26. doi: 10.1002/cne.902390102. [DOI] [PubMed] [Google Scholar]
- O’Rourke NA, Cline HT, Fraser SE. Rapid remodeling of retinal arbors in the tectum with and without blockade of synaptic transmission. Neuron. 1994;12:921–934. doi: 10.1016/0896-6273(94)90343-3. [DOI] [PubMed] [Google Scholar]
- Ruthazer ES, Akerman CJ, Cline HT. Control of axon dynamics by correlated activity in vivo. Science. 2003;301:66–70. doi: 10.1126/science.1082545. [DOI] [PubMed] [Google Scholar]
- Ruthazer ES, Stryker MP. The role of activity in the development of long-range horizontal connections in area 17 of the ferret. J Neurosci. 1996;16:7253–7269. doi: 10.1523/JNEUROSCI.16-22-07253.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Simon DK, O’Leary DDM. Development of topographic order in the mammalian retinocollicular projection. J Neurosci. 1992;12:1212–1232. doi: 10.1523/JNEUROSCI.12-04-01212.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Szebenyi G, Callaway JL, Dent EW, Kalil K. Interstitial branches develop from active regions of the the axon demercated by the primary growth cone during pausing behavior. J Neurosci. 1998;18:7930–7940. doi: 10.1523/JNEUROSCI.18-19-07930.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Warton SS, Dyson SE, Harvey AR. Visual thalamocortical projections in normal and enucleated rats: HRP and fluorescent dye studies. Exp Neurol. 1988;100:23–39. doi: 10.1016/0014-4886(88)90198-7. [DOI] [PubMed] [Google Scholar]
- Wise SP, Jones EG. The organization and postnatal development of the commissural projection of the rat somatic sensory cortex. J Comp Neurol. 1976;168:313–344. doi: 10.1002/cne.901680302. [DOI] [PubMed] [Google Scholar]
- Yates PA, Roskies AL, McLaughlin T, O’Leary DM. Topographic-specific axon branching controlled by ephrin-As is the critical event in retinotectal map development. J Neurosci. 2001;21:8548–8563. doi: 10.1523/JNEUROSCI.21-21-08548.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]