Abstract
Ethanol drinking was assessed in the P/NP, HAD1/LAD1, and HAD2/LAD2 lines of rats under environmental conditions that produce schedule-induced polydipsia. Female rats (n = 8/line), maintained at 85% of free-feeding body weights, underwent daily 1-h sessions during which 45-mg food pellets were delivered every 60 s. Water, 2, 4, 8, 16, or 32% w/v ethanol solution was available from a single bottle for 8 consecutive sessions at each concentration, with blood-ethanol levels (BELs) determined after selected sessions. P and HAD2 rats drank more water and ethanol than their non-preferring counterparts, while HAD1 and LAD1 rats did not differ. Ethanol intake and BELs were positively correlated (r = 0.75) across lines. Finally, rats were allowed 14 daily choice sessions with 8% ethanol and water concurrently available. Water intake generally exceeded ethanol intake in all lines, while P rats drank similar amounts of both fluids. These line differences indicate pleiotropic effects of genes that mediate ethanol intake and schedule-induced behaviors.
Keywords: Adjunctive behavior, Schedule-induced polydipsia, Alcohol-preferring rats, Alcohol-nonpreferring rats, Genetic correlation, P rats, NP rats, HAD rats, LAD rats
It is well accepted that alcoholism is a complex behavioral genetic trait accounted for by the interaction effects of multiple genes and environmental factors (Crabbe 2002; Enoch and Goldman 2001). A useful approach to the development of an animal model of the genetic contribution to alcohol drinking has been the development of programs in which rats are selectively bred for extremes of high and low voluntary oral alcohol consumption (Lumeng et al. 1995). Perhaps the most powerful, albeit controversial, animal model of the contribution of environmental factors to excessive alcohol drinking is drinking engendered by schedule-induced polydipsia (Falk 1998). The purpose of the present investigation is to study gene-environment interactions by examining alcohol drinking by selectively bred rats under conditions that produce schedule-induced polydipsia.
The alcohol-preferring (P) and-nonpreferring (NP) lines were developed at Indiana University through bidirectional selective breeding from a randomly bred closed colony of Wistar rats from the Walter Reed Army Institute of Research, Washington, D.C. (Lumeng et al. 1977; Li et al. 1991). The selection criterion for P rats is intake greater than 5 g ethanol/kg body weight/day and the selection criterion for NP rats is intake less than 1.5 g ethanol/kg body weight/day with 10% (v/v) ethanol, water, and food continuously available (Lumeng et al. 1977). The P rats have characteristics important for an animal model of alcoholism (Cicero 1979). Voluntary oral consumption of ethanol by P rats results in high blood ethanol concentrations (50–250 mg%) even in the presence of food and water (Murphy et al. 1986). P rats consume ethanol for its pharmacological effects rather than its taste, smell or caloric content, as evidenced by intragastric (Waller et al. 1984) and intracerebral (Gatto et al. 1994) self-administration of ethanol. Finally, chronic voluntary ethanol consumption results in tolerance (Gatto et al. 1987) and physical dependence (Waller et al. 1982) in P rats.
The high alcohol-drinking (HAD) and low alcohol-drinking (LAD) rat lines also were developed at Indiana University and have been selectively bred for high and low alcohol preference and consumption using similar phenotypic criteria to those used for the P and NP lines (Li et al. 1993). However, the HAD and LAD lines were derived from the N/Nih stock rat, which was developed from crossing eight inbred rat strains with a wide range of alcohol intakes (Li and Lumeng 1984). The ethanol consumption of the N/Nih stock animals is intermediate in the range of the eight inbred strains from which the N/Nih rats were derived (Li and Lumeng 1984). Thus, the HAD and LAD rat lines were derived from a more heterogeneous foundation stock than the Wistar stock rats used to derive the P and NP rat lines, and should have greater genetic diversity than the P and NP lines. Within-family selection and a rotational breeding design were used to discourage inbreeding. Replicate lines (HAD1/LAD1, HAD2/LAD2) were independently developed.
In a classic study of schedule-induced polydipsia, Falk (1961) reduced rats to 80% of their free-feeding body weight and allowed them to lever-press on a variable interval 1-min (VI 1) schedule for single 45-mg food pellets, with water available. Previously normal water intake by these rats increased to nearly one half of body weights, a level of intake far beyond that which is necessary to maintain water balance (Falk 1961). Schedule-induced polydipsia has since been demonstrated in a variety of species ranging from macaques to humans (see Falk 1998). In this procedure, substitution of ethanol solution for water results in substantial ethanol intake (Lester 1961; Freed and Lester 1970).
In a study designed to maximize daily ethanol intake and induce physical dependence on ethanol (Falk et al. 1972), weight-reduced rats were exposed daily to six 1-h polydipsia sessions across each 24-h period, with 5% (v/v) ethanol available. During three months of exposure, rats consumed more than 13 grams ethanol per kilogram body weight (g/kg) per day, and exhibited audiogenic seizures following discontinuation of polydipsia sessions. Perhaps more impressive, rats exposed daily to 3-h polydipsia sessions with 5% (v/v) ethanol available consumed about 6 g/kg ethanol per session, and exhibited signs of hyperexcitable behavior following the removal of ethanol (Tang and Falk 1983).
Schedule-induced polydipsia is the most well-researched example of adjunctive behavior, defined explicitly as a behavior maintained at high probability not by variables which directly affect its unconditioned, conditioned, or operant probability, but rather by variables which determine the probability of some other class of behavior (Falk 1971). That is, adjunctive behaviors are normal behaviors that occur in excess due to seemingly unrelated conditions of intermittence (Falk 1981). There has been considerable debate in the literature as to whether schedule-induced polydipsia is capable of providing a good animal model of alcoholism. The primary issues of concern have been whether food-deprived animals are drinking ethanol strictly for its caloric value (Freed and Lester 1970), that the effects of intermittent schedules are not specific for ethanol drinking (Falk 1971), and whether or not schedule induction can reliably cause ethanol dependence in animals (Heintzelman et al. 1976).
The P/NP, HAD1/LAD1, and HAD2/LAD2 pairs of lines are selected on the basis of homecage ethanol drinking (preference under 2-bottle free-choice continuous access conditions). However, the genes which have been selected for homecage ethanol preference are likely not identical to the genes which account for ethanol drinking under different environmental conditions. For example, the selectively bred rat lines were tested for operant self-administration, during which delivery of small amounts of ethanol solution was contingent on lever press responding during daily 30-min sessions (Samson et al. 1998). About two-thirds of the genetic variance across these pairs of lines in operant self-administration is accounted for by the genes selected for homecage drinking, indicating that some genes have pleiotropic effects on both operant and homecage ethanol drinking, and that multiple genes contribute to each individual behavior (i.e., epistasis; Samson et al. 1998). When one behavior differs between lines selectively bred to diverge on a second behavior, a genetic correlation is said to exist between these two behavioral traits. Therefore, it is of interest to examine whether selected differences in homecage ethanol drinking are genetically correlated with differences in susceptibility to adjunctive behavior (i.e. schedule-induced polydipsia). Inbred strains of mice known to drink large (C57BL/6J) and small (DBA/2J) amounts of ethanol (Crabbe 2002) have been tested for polydipsic water and ethanol intake using 1-h sessions with intermittent food delivery (Mittleman et al. 2003). When presented with 5 10, or 20% (v/v) ethanol in a single-bottle test, C57BL/6J mice drank greater amounts than DBA/2J mice, but these two strains did not differ in fluid intake when water was presented in a single-bottle test (Mittleman et al. 2003).
The purpose of the present study was to examine the effects of response-independent intermittent scheduled delivery of food pellets on water- and ethanol-drinking behavior in lines of rats selectively bred for high and low alcohol preference. This approach was used to reveal any genetic correlations that exist between selection for homecage ethanol drinking and susceptibility to schedule-induced polydipsia. Behavioral differences were compared between the lines of each selected pair and also across pairs of selectively bred lines.
Method
Subjects
Subjects were 48 experimentally naïve female rats bred at the Indiana University School of Medicine. Following reduction to 85% of free-feeding body weight, P (n = 8) and NP (n = 8) rats from the 52nd generation of selective breeding weighed 243.3 (±3.7) g and 254.8 (±3.0) g, respectively; HAD1 (n = 8) and LAD1 (n = 8) rats from the 40th generation of selective breeding weighed 154.2 (±3.4) g and 176.0 (±16.2) g, respectively; and HAD2 (n = 8) and LAD2 (n = 8) rats from the 38th generation of selective breeding weighed 157.5 (±4.2) g and 174.1 (±4.7) g, respectively. All rats were double-housed in plastic tub-style cages in a vivarium maintained on a 12:12 h light/dark cycle (lights off at 1900 h). Water was always available ad libitum. Food (Lab Diet 5001, PMI Nutrition International Inc., Brentwood, MO) was available immediately following operant sessions (see below). The protocol for this study was approved by the IUPUI School of Science IACUC and was conducted in accordance with NIH guidelines (National Research Council 1996).
Apparatus
Sessions took place in Habitest operant cages (Coulbourn Instruments; Allentown, PA) with dimensions of approximately 26 cm L × 29 cm W × 29 cm H. The floor of the operant cages consisted of parallel metal bars, and 2 sides of the boxes were metal while 2 sides were transparent plexiglass. No levers were available in the operant box during sessions. A food receptacle was located on one wall of the box. On the wall opposite the food receptacle were two holes through which sipper tubes attached to graduated cylinders were inserted. During the concentration response phase, only one sipper tube was available, and during the two-bottle choice phase, two sipper tubes were available.
Sessions
For 1 h each day, rats were placed in an operant chamber where single 45-mg food pellets were delivered on a fixed-time (FT)-1 schedule. Rats were not required to perform an operant response to receive food pellets. This FT-1 schedule was used for both the concentration response phase and the two-bottle choice phase (see below). There was a house light within the operant chamber. Following each daily session in the operant chamber, rats were weighed, placed in tub-style cages alone, and given an amount of food appropriate to maintain them at 85% of their free-feeding body weight. All sessions for each individual rat occurred in the same operant chamber. The walls of the operant chambers, floor grids, and trays were cleaned between all rats.
Concentration response phase
Rats were food-deprived to 85% of their free-feeding body weight and then maintained at this body weight for several days before operant sessions began. Each day, during the vivarium light cycle, rats were placed in operant chambers as described above. The side of the chamber on which the available fluid was placed was alternated daily.
Rats underwent between 16 and 20 daily operant sessions during which water was available from a single sipper tube. Thereafter, rats were administered consecutive blocks of 8 daily sessions in which ascending concentrations (2, 4, 8, 16, 32% [w/v]) of ethanol solution were available from a single sipper tube. The 8% (w/v) ethanol solution was then reintroduced as the sole fluid available during daily sessions in order to determine blood-ethanol levels (BELs). This ethanol concentration remained the only fluid available during daily sessions until rats met criterion for BEL assessment. Criterion required that each rat consume an amount (ml) of 8% (w/v) ethanol solution no less than the mean volume consumed (ml) from its previous 8-day block of sessions at the same ethanol concentration.
Two-bottle choice test
Finally, 14 consecutive daily choice sessions were given in which 8% (w/v) ethanol and water were concurrently available from two sipper tubes. The side of the chamber on which the ethanol was placed was alternated daily.
Blood-ethanol level determination
In order to determine BELs, blood was collected from animals by cutting the tip of the tail. Blood samples were collected immediately following selected sessions in which only 8% (w/v) ethanol was available. BELs were determined with an alko-analyzer (AM1 series, Analox Instruments Ltd., MA) that measures oxygen levels (proportional to ethanol concentrations) following oxidation of ethanol to acetaldehyde and hydrogen peroxide by alcohol oxidase.
Data analysis
The amount of fluid consumed was determined according to the graduated marks on hanging bottles attached to the sides of operant cages. Volumes of water (ml) and ethanol (ml) consumed as well as ethanol intake (g/kg) during the ethanol concentration response phase were analyzed using 2-way and 3-way mixed-design (line and/or replicate as between-subjects factors and ethanol concentration as a within-subjects factor) ANOVAs. Volumes of water and ethanol (ml) consumed during the choice phase were compared using similar 2-way and 3-way mixed-design ANOVAs. Planned paired-samples t-tests were used to compare water and ethanol intake within lines. Comparison of ethanol intake (g/kg) and ethanol preference ratio (ethanol/total; E:T) during the choice phase were analyzed using 2-way and 3-way mixed-design (fluid as a within-subjects factor) ANOVAs and independent-samples t-tests. The relationship between ethanol intake (g/kg) and BELs were analyzed using simple regression analysis. Comparison of mean BELs between replicates and lines were analyzed using 1-way and 2-way ANOVAs and independent-samples t-tests. In all cases, significance was determined at P < 0.05. Post-hoc analyses were conducted using the Student-New-man-Keuls method where appropriate.
Results
Fluid consumption during ethanol concentration response phase
P and NP rats
A 2-way (line × ethanol concentration) ANOVA revealed that P rats consumed significantly more fluid (ml) during daily sessions than NP rats, F(1, 14) = 48.77, P < 0.001, as well as a significant effect of ethanol concentration, F(5, 70) = 59.81, P < 0.001, and a significant line × concentration effect, F(5, 70) = 3.05, P = 0.015, on fluid intake (see Fig. 1). Post-hoc analyses revealed that, across 8-day blocks, P rats consumed significantly more water, 2% ethanol, 4% ethanol, and 8% ethanol than NP rats (P < 0.01 in all cases). An independent-samples t-test indicated that P rats weighed significantly less than NP rats throughout the concentration response phase, t(14) = 2.59, P = 0.022.
Fig. 1.

(Upper panel) Mean (±SEM) consumption of water (ml; open symbols) and solutions containing various ethanol concentrations (w/v; solid symbols) in P rats (circles) and NP rats (triangles) across 8-day blocks of the ethanol concentration response phase during which a single fluid was available during test sessions. (Lower panel) Mean (±SEM) ethanol intake (g/kg) from solutions of various ethanol concentrations (w/v) by P rats (circles) and NP rats (triangles) across 8-day blocks of the ethanol concentration response phase during which a single fluid was available during test sessions. * P < 0.01 significant difference from NP rats
Also shown in Fig. 1, a separate 2- way (line × ethanol concentration) ANOVA revealed that P rats exhibited significantly higher ethanol intake (g/kg) during daily sessions than NP rats, F(1, 14) = 131.47, P < 0.001, as well as a significant effect of ethanol concentration, F(4, 56) = 9.18, P < 0.001, and a significant line × ethanol concentration effect, F(4, 56) = 10.29, P < 0.001. Post-hoc analyses revealed that, across 8-day blocks, P rats exhibited significantly higher ethanol intake (g/kg) than NP rats at every concentration tested (P < 0.001 in all cases).
HAD and LAD replicate lines of rats
A 3-way (line × replicate × ethanol concentration) ANOVA on volume of fluid (ml) consumed revealed a significant effect of ethanol concentration, F(5, 140) = 125.50, P < 0.001, a line × replicate interaction effect, F(1, 28) = 19.85, P < 0.001, and a line × replicate × ethanol concentration interaction effect, F(5, 140) = 8.49, P < 0.001. A separate 3-way (line × replicate × ethanol concentration) ANOVA on ethanol intake (g/kg) revealed that HAD rats consumed significantly more ethanol than LAD rats, F(1, 28) = 8.02, P = 0.008, as well as a significant effect of ethanol concentration, F(4, 112) = 5.52, P < 0.001, and significant interaction effects of replicate × ethanol concentration, F(4, 112) = 7.30, P < 0.001, and line × replicate, F(1, 28) = 6.19, P = 0.019. These interaction effects were a result of less divergence in fluid and ethanol intake in the HAD1/LAD1 pair of lines as compared to the HAD2/LAD2 pair of lines (see below). Finally, a 2-way (line × replicate) ANOVA on water intake revealed a significant line × replicate interaction effect, F(1, 28) = 16.80, P < 0.001, but no significant main effects. A 2-way (line × replicate) ANOVA indicated that HAD rats weighed less than LAD rats throughout the concentration response phase, F(1, 28) = 45.28, P < 0.001.
Figure 2 illustrates that a 2-way ANOVA (line × ethanol concentration) on volume of fluid (ml) consumed in only HAD1 and LAD1 rats revealed a significant effect of ethanol concentration, F(5, 70) = 67.03, P < 0.001, but not rat line. Also shown in Fig. 2, a separate 2-way ANOVA (line × ethanol concentration) on ethanol intake (g/kg) in only HAD1 and LAD1 rats revealed a significant effect of ethanol concentration, F(4, 56) = 5.37, P < 0.001, but not rat line. A planned independent-samples t-test indicated that HAD1 and LAD1 rats drank similar amounts of water when it was the only fluid available during test sessions, t(14) = 1.82, P > 0.05.
Fig. 2.
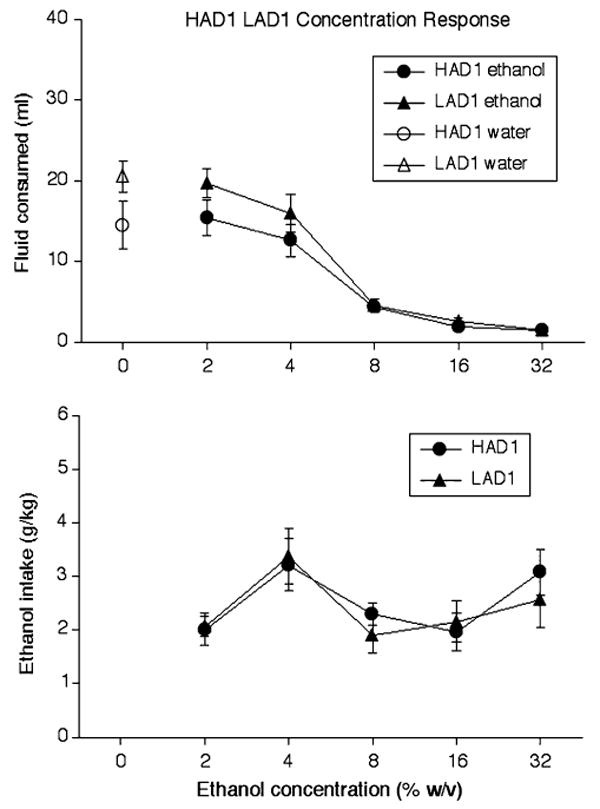
(Upper panel) Mean (±SEM) consumption of water (ml; open symbols) and solutions containing various ethanol concentrations (w/v; solid symbols) in HAD1 rats (circles) and LAD1 rats (triangles) across 8-day blocks of the ethanol concentration response phase during which a single fluid was available during test sessions. (Lower panel) Mean (±SEM) ethanol intake (g/kg) from solutions of various ethanol concentrations (w/v) by HAD1 rats (circles) and LAD1 rats (triangles) across 8-day blocks of the ethanol concentration response phase during which a single fluid was available during test sessions
Figure 3 illustrates that a 2-way ANOVA (line × ethanol concentration) on volume of fluid (ml) consumed in only HAD2 and LAD2 rats revealed significant effects of rat line, F(1, 14) = 21.74, P < 0.001, and ethanol concentration, F(5, 70) = 60.68, P < 0.001, as well as a significant line × ethanol concentration interaction effect, F(5, 70) = 8.24, P < 0.001. Post-hoc analyses revealed that, across 8-day blocks, HAD2 rats consumed significantly more water, 2% ethanol, and 4% ethanol than LAD2 rats (P < 0.05 in all cases). Also shown in Fig. 3, a separate 2-way ANOVA (line × ethanol concentration) in only HAD2 and LAD2 rats revealed that HAD2 rats exhibited significantly higher ethanol intake (g/kg) than LAD2 rats, F(1, 14) = 8.99, P < 0.01, as well as a significant effect of ethanol concentration, F(4, 56) = 5.37, P < 0.001, on ethanol intake (g/kg). Finally, a planned independent-samples t-test indicated that HAD2 rats drank significantly more water than LAD2 rats when it was the only fluid available during test sessions, t(14) = 3.81, P < 0.01.
Fig. 3.
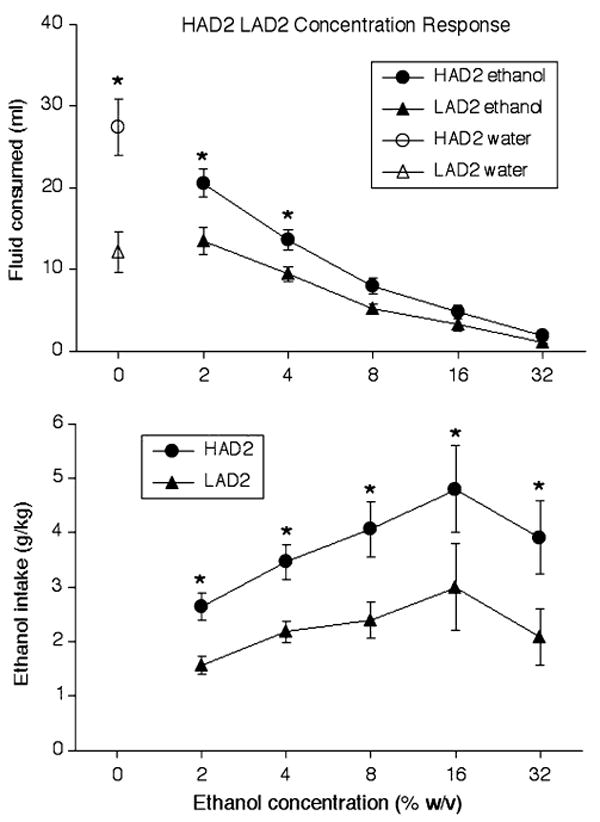
(Upper panel) Mean (±SEM) consumption of water (ml; open symbols) and solutions containing various ethanol concentrations (w/v; solid symbols) in HAD2 rats (circles) and LAD2 rats (triangles) across 8-day blocks of the ethanol concentration response phase during which a single fluid was available during test sessions. (Lower panel) Mean (±SEM) ethanol intake (g/kg) from solutions of various ethanol concentrations (w/v) by HAD2 rats (circles) and LAD2 rats (triangles) across 8-day blocks of the ethanol concentration response phase during which a single fluid was available during test sessions. * P < 0.05 significant difference from LAD2 rats
Fluid consumption during two-bottle choice test
Figure 4 shows average intake of 8% ethanol and water, as well as ethanol (E:T; ethanol/total fluid) preference, across the 14-day choice phase. A 2-way (line × fluid) ANOVA of the P and NP data yielded no significant effects (P > 0.05 in all cases). A 3-way (line × replicate × fluid) ANOVA of the HAD and LAD data revealed that, across both replicate and lines, rats drank more water than ethanol, F(1, 28) = 35.22, P < 0.001. Planned paired-samples t-tests indicated that HAD1 rats, t(7) = 2.52, P < 0.05, LAD1 rats, t(7) = 3.51, P < 0.01, and LAD2 rats, t(7) = 4.27, P < 0.001, drank significantly more water than 8% ethanol across the 14-day choice phase. HAD2 rats and NP rats exhibited a tendency toward drinking more water than ethanol (P < 0.08), while P rats drank similar amounts of water and ethanol over the same time period.
Fig. 4.
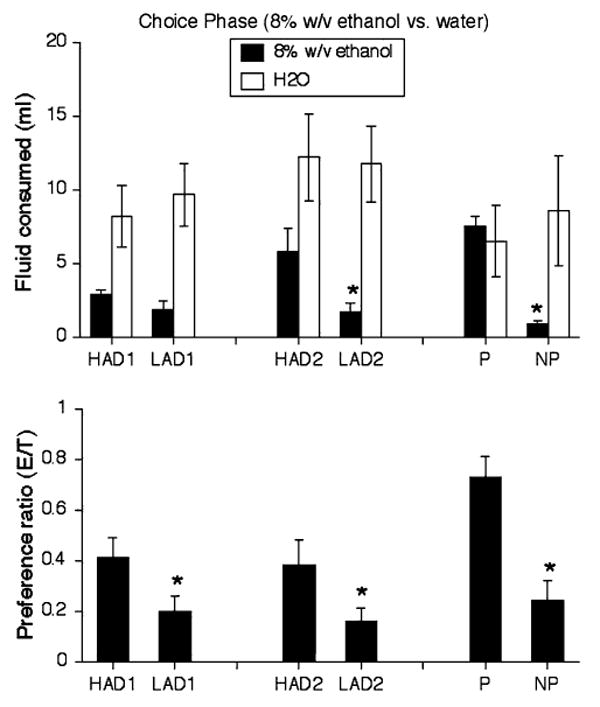
(Upper panel) Mean (±SEM) consumption of water (ml; open bars) and 8% (w/v) ethanol (solid bars) in P/NP rats, HAD/LAD1 rats, and HAD2/LAD2 rats across test sessions of the 14-day 2-bottle choice phase. (Lower panel) Mean (±SEM) ethanol preference (ethanol/total fluid) in P/NP rats, HAD/LAD1 rats, and HAD2/LAD2 rats across test sessions of the 14-day 2-bottle choice phase. * P < 0.05 significant difference from water intake
Table 1 shows mean ethanol intake (g/kg) during the choice phase. A 2-way (line × replicate) ANOVA indicated that, across replicates, HAD rats exhibited significantly higher ethanol intake (g/kg) than LAD rats, F(1, 28) = 9.30, P < 0.01. A separate set of independent-samples t-tests indicated that HAD2 rats exhibited significantly higher ethanol intake (g/kg) than LAD2 rats, t(14) = 2.58, P < 0.05, and P rats exhibited significantly higher ethanol intake (g/kg) than NP rats, t(14) = 12.12, P < 0.001. There was no significant difference in ethanol intake (g/kg) between HAD1 and LAD1 rats (P > 0.05).
Table 1.
Mean (±SEM) intake of ethanol (g/kg) in P/NP, HAD1/LAD1, and HAD2/LAD2 rats across test sessions of the 14-day 2-bottle choice phase
| Rat line | ||||||
|---|---|---|---|---|---|---|
| P | NP | HAD1 | LAD1 | HAD2 | LAD2 | |
| Intake | 2.5 (0.2) | 0.3 (0.1)* | 1.5 (0.2) | 0.9 (0.3) | 3.0 (0.1) | 0.8 (0.3)* |
P < 0.05 significant difference between P and NP rats or between HAD2 and LAD2 rats
Figure 4 also illustrates ethanol preference ratios in the P/NP, HAD1/LAD1, and HAD2/LAD2 pairs of rat lines. An independent-samples t-tests indicated that P rats had significantly higher ethanol preference than NP rats, t(14) = 4.63, P < 0.001. A 2-way (line × replicate) ANOVA indicated that, across replicates, HAD rats exhibited significantly higher ethanol preference than LAD rats, F(1, 28) = 9.47, P < 0.01. Planned independent-samples t-tests further revealed that HAD1 rats had significantly higher E:T preference than LAD1 rats, t(14) = 2.19, P < 0.05, and HAD2 rats had significantly higher E:T preference than LAD2 rats, t(14) = 2.16, P < 0.05.
Blood-ethanol levels
Figure 5 consists of scatter plots of ethanol intake (g/kg) and BELs (mg%) in individual rats of the P/NP pair of rat lines, the HAD1/LAD1 pair of rat lines, and the HAD2/LAD2 pair of rat lines. Regression analysis indicated that, within the P/NP pair of rat lines, ethanol intake (g/kg) and BELs were positively correlated, r(14) = 0.96, P < 0.001. Within the HAD1/LAD1 pair of rat lines, ethanol intake (g/kg) and BELs were positively correlated, r(14) = 0.69, P < 0.01. Also, within the HAD2/LAD2 pair of rat lines, ethanol intake (g/kg) and BELs were positively correlated, r(14) = 0.53, P < 0.05. Finally, across all rats, ethanol intake (g/kg) and BELs were positively correlated, r(14) = 0.68, P < 0.001.
Fig. 5.
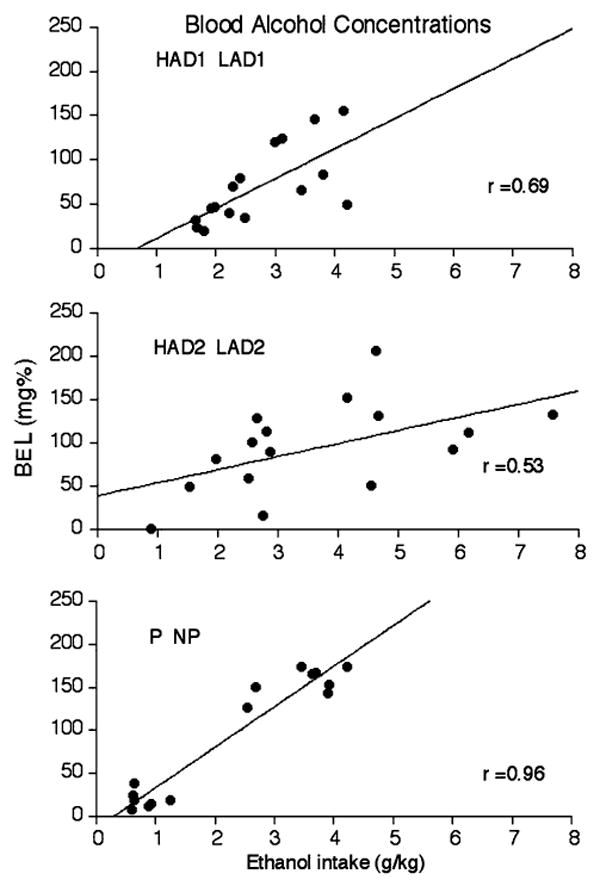
Scatter plots of ethanol intake (g/kg) and blood-ethanol levels (mg%) by individual rats of the P/NP, HAD/LAD1, and HAD2/LAD2 pairs of lines on criterion day of the 8% (w/v) ethanol reintroduction phase
For sessions during which blood samples were collected, mean BELs as well as mean ethanol intake (g/kg) for all lines of rats are shown in Table 2. A 2-way (line × replicate) ANOVA indicated that, across replicates, HAD rats had higher BELs than LAD rats, F(1, 28) = 14.94, P < 0.001). An independent samples t-test showed that P rats had higher BELs than NP rats, t(14) = 20.57, P < 0.001).
Table 2.
Mean (±SEM) ethanol intake (g/kg) and blood ethanol levels (BEL; mg%) in P/NP, HAD1/LAD1, and HAD2/LAD2 rats for sessions when blood samples were taken
| Rat line | ||||||
|---|---|---|---|---|---|---|
| P | NP | HAD1 | LAD1 | HAD2 | LAD2 | |
| Intake | 3.5 (0.2) | 0.8 (0.1) | 3.2 (0.3) | 2.3 (0.3) | 4.4 (0.7) | 2.9 (0.6) |
| BEL | 155.9 (6.3) | 18.1 (3.5) | 101.9 (14.7) | 38.8 (5.9) | 117.7 (17.3) | 71.5 (19.1) |
Discussion
In the present investigation, three pairs of rat lines selectively bred for high and low ethanol preference were tested for differences in polydipsic behavior induced by an intermittent schedule of food deliveries. In a single-bottle situation, P rats and HAD2 rats consumed more fluid relative to their nonpreferring counterparts when that fluid was water or a low to moderate ethanol concentration. P rats and HAD2 rats also exhibited significantly higher ethanol intake (g/kg) under these conditions than NP and LAD2 rats, respectively. In contrast, HAD1 rats did not drink more fluid (water or ethanol), nor did they differ from LAD1 rats on ethanol intake (g/kg). When a choice between water and ethanol was available, P rats drank similar amounts of both fluids, whereas all other lines preferred water to ethanol. P rats and HAD2 rats exhibited significantly higher ethanol intake (g/kg) during the choice phase than their nonpreferring counterparts, whereas HAD1 rats did not. These results indicate that the genes that govern ethanol preference might also play a role in susceptibility to schedule-induced polydipsia.
In the single-bottle situation, P and HAD2 rats drank significantly more water than NP and LAD2 rats under intermittent schedule conditions, and HAD1 and LAD1 rats did not differ in water intake, despite the fact that P/HAD1/HAD2 rats weighed less than their nonpreferring counterparts. Alcohol-preferring rats do not drink more water than alcohol-nonpreferring rats under normal environmental conditions (unpublished observations from this lab). Since all lines of rats were similarly maintained at 85% of their free-feeding body weight and had ad libitum access to water in the home cage, it is unlikely that alcohol-preferring rats were more motivated to consume water during test sessions because of food deprivation schedules inherent to this procedure. It is also unlikely that increased water intake by P/HAD2 rats relative to NP/LAD2 rats was due to prandial (post-feeding) effects because all rats received equivalent amounts of food on identical delivery schedules during test sessions, and also alcohol-preferring rats weighed less than non-preferring rats. Taken together, these results suggest that a genetic correlation exists between ethanol preference and schedule-induced water intake.
P rats and HAD2 rats also exhibited significantly higher ethanol intake (g/kg) than NP rats and LAD2 rats during test sessions in which it was the only fluid available. This result was anticipated since these pairs of rat lines are selectively bred to diverge on this behavioral trait. Unexpectedly, HAD1 rats did not consume more ethanol than LAD1 rats during single-bottle sessions because (a) HAD1 rats exhibited lower ethanol intake (g/kg) than rats of the HAD2 replicate line (although consumption by HAD1 rats more closely resembled consumption by P rats), and (b) LAD1 rats consumed the same amount of ethanol as rats in the LAD2 replicate line (and both of these lines consumed more ethanol than NP rats). This result is especially peculiar because another study that examined limited-access intake by all three pairs of lines indicated that the HAD1/LAD1 pair of lines is at least as divergent, if not more divergent than the P/NP and HAD2/LAD2 pairs of lines (Samson et al. 1998). In that study, operant intakes (g/kg) by HAD1 and HAD2 rats were similar to each other and greater than intake by P rats across multiple ethanol concentrations; intakes by LAD1 and LAD2 rats were similar to each other and less than intake by NP rats across multiple ethanol concentrations. Therefore, in that study, the P/NP pair of lines was less divergent than the HAD/LAD replicate pairs of lines. This discrepancy may be attributable to the different reinforcement schedules in the two studies, or the sucrose-substitution procedure employed by Samson et al. (1998).
It is beyond the scope of these data to explicitly calculate heritability (h2) estimates of schedule-induced polydipsia, mainly because rats from a single generation of each selected line were tested. However, studies in mice have also suggested a genetic component of schedule-induce ethanol polydipsia (Mittleman et al. 2003; Goldowitz et al. 2006). It would be of great interest to repeat the present experiment in rats from subsequent breeding generations to determine how the genetic correlation between ethanol preference and schedule-induced polydipsia may have changed over time.
Perhaps the most impressive result of this experiment was the amount of ethanol (g/kg) consumed by rats during 1-h sessions under intermittent schedule conditions. All of the alcohol-preferring rat lines surpassed 3 g/kg ethanol intake at some point during the ethanol concentration response phase, and HAD2 rats even consumed amounts of ethanol in 1 h that approached the selection criterion 24-h intake (∼5 g/kg ethanol). Likewise, LAD1 and LAD2 rats, which are selected to drink less than 1.5 g/kg ethanol per day, consumed substantial amounts of ethanol (2–3 g/kg) during 1-h test sessions (NP rats never reached 1 g/kg ethanol intake). These results indicate that polydipsic behavior induced by intermittent reinforcer schedules works additively with genetic susceptibilities to drink ethanol, cooperatively in the case of alcohol-preferring rats and antagonistically in the case of alcohol-nonpreferring rats. This study reflects the powerful control that intermittent food schedules can exert over ethanol drinking, regardless of genetic selection. This result is partly consistent with another study that showed the ability of intermittent ethanol sipper tube presentations to produce increases in ethanol intake (g/kg), although that effect was not augmented by response-non-contingent intermittent food deliveries (Tomie et al. 2006).
During the choice phase of this experiment, rats either consumed similar amounts of water and ethanol (P, HAD2, and NP rats) or significantly preferred water over ethanol (HAD1, LAD1, and LAD2 rats). This pattern of results seems to contradict the high- and low-ethanol-preference traits presumably instilled in these animals by many generations of selective breeding. However, these lines of rats are selected for high and low ethanol preference on the basis of continuous-access home cage drinking, and the genes that mediate ethanol drinking under different environmental conditions are not necessarily identical (Samson et al. 1998).
An alternative explanation for the lack of ethanol preference in the P and HAD lines might be that fluids were presented to these animals in single-bottle situations prior to the choice phase. Quantities of ethanol consumption by an organism are dependent on prior ethanol drinking initiation procedures (Samson and Czachowski 2003). Furthermore, prior experience with a single adjunctive behavioral option may attenuate the expression of a second adjunctive behavior when both behavioral options are available together (Williams et al. 1992). More specifically, all rats in this study were first trained in sessions during which water was the sole fluid available, followed by the ethanol concentration response phase, and then the two-bottle choice phase. It is possible that schedule-induced ethanol intake and preference ratio during the choice phase were affected by the availability of water as the sole fluid during the training phase.
The lack of preference for ethanol over water by any line of rats argues against the caloric hypothesis regarding polydipsic ethanol intake by rats, since rats reliably chose the noncaloric fluid over the caloric fluid. It is likely that at the doses and BELs obtained, many of the rats tested in this experiment are experiencing the pharmacological effects of ethanol, although this does not necessarily indicate that those effects are the primary cause of the high ethanol intake. Tang et al. (1982) showed a complete reversal of robust ethanol (and water) polydipsia (only one fluid available at a time) when food is given to rats in a single ration following extensive training with those fluids in the presence of intermittent food delivery schedules. These results provide further evidence against the hypotheses that the caloric and/or pharmacological effects of ethanol are the lone motivating factors for high ethanol intake under conditions of intermittence.
Few studies have simultaneously examined aspects of ethanol self-administration in all three of the Indiana University selectively bred pairs of rat lines (Murphy et al. 2002). In a study published in two parts, the P/NP, HAD1/LAD1 and HAD2/LAD2 pairs of rat lines were tested for ethanol initiation and ethanol self-administration in a limited and continuous access operant paradigm (Files et al. 1998; Samson et al. 1998). Those studies concluded that the genes that are selected for home cage ethanol preference during the selective breeding process overlap with, but are not identical to those which contribute to operant ethanol self-administration. This notion is supported by the results of the present investigation since the P/NP and HAD2/LAD2 rats diverged on schedule-induced water and ethanol polydipsia, but the HAD1/LAD1 rats did not diverge on schedule-induced polydispic intake of water or ethanol.
The behavioral profiles of the HAD1/LAD1 and HAD2/LAD2 replicate pairs of rat lines were not identical under conditions of an intermittent schedule in this study. When lines of a pair selectively bred for responses on a single trait respond differently on a second trait, it is said that a genetic correlation exists between the two traits and that the genes contributing to each response are having pleiotropic effects (Crabbe 1999). Further, proposed genetic correlations between two traits in one pair of lines are corroborated or negated by the response profiles of a replicate pair of lines selected for the same trait. According to a scheme introduced by Crabbe et al. (1990), the strength of these genetic correlations is dependent on the statistical significance and direction of differences between lines and replicates, as well as line × replicate interactions. For example, strong evidence for a genetic correlation is indicated by a significant main effect of line and the presence of the effect in both replicates (regardless of line × replicate interaction effects). Moderate evidence for a genetic correlation is indicated by a significant main effect of line and a significant interaction effect, where the effect is present in only one of the two replicates. Finally, weak evidence for a genetic correlation is indicated when there is no main effect of line, but there is a significant interaction effect, where the effect is present in only one of the two replicates.
The strength of genetic correlations between criteria responses for selection traits and fluid intake under conditions of intermittency were analyzed in the HAD/LAD replicate pair of lines according to the scheme devised by Crabbe et al. (1990). There was a strong positive genetic correlation between criteria responses and ethanol preference as well as ethanol intake (g/kg) during the 2-bottle choice phase of this experiment. This finding indicates that similar genetic loci mediate ethanol preference under normal environmental conditions and under conditions of intermittent reinforcer deliveries. There was also a moderate genetic correlation between criteria responses and ethanol intake (g/kg) during the concentration response phase of the experiment, again suggesting that similar genetic loci contribute to ethanol intake under normal environmental conditions and under conditions of intermittent reinforcer deliveries. During the concentration response phase, there was also a weak genetic correlation between criteria responses and fluid (ml) as well as water (ml) consumed. These latter two findings reflect the increased water intake and fluid intake in HAD2 rats relative to LAD2 rats, in the absence of differences between HAD1 and LAD1 rats. Still, according to Crabbe et al. (1990), this result allows for the “cautious” conclusion that there exists a genetic correlation between schedule-induced polydipsic water intake and criteria responses on selection traits in HAD/LAD rats. Further evidence for such a notion is provided by the increased water intake observed in P rats relative to NP rats under conditions of intermittent reinforcer deliveries.
In summary, this study examined schedule-induced polydipsic intake of water and ethanol in the P/NP, HAD1/LAD1, and HAD2/LAD2 pairs of rat lines selectively bred for high and low ethanol preference. Food pellets were delivered to food-deprived rats on a FT-1 schedule in a response-non-contingent manner. The P/NP and HAD2/LAD2 pairs of rat lines behaved similarly since P rats and HAD2 rats consumed more water, more ethanol, and had higher ethanol preference ratios than their nonpreferring counterparts in the presence of an intermittent schedule. Although HAD1 rats did exhibit a higher ethanol preference ratio than LAD1 rats when both fluids were available, this ethanol preference ratio was lower than those of P rats and HAD2 rats. Further, HAD1 rats did not drink more water than LAD1 rats when it was only the fluid available, and also they did not differ on ethanol intake (g/kg) relative to LAD1 rats at any point during the experiment. All rat lines consumed impressive amounts of ethanol during 1-h test sessions (approaching 24-h selection criterion in alcohol-preferring rats and far exceeding 24-h selection criterion in alcohol-nonpreferring rats). Finally, according to the scheme presented by Crabbe et al. (1990), results from the HAD/LAD replicate pairs of lines indicate that genetic correlations may exist between the selected traits in these lines of rats and behavioral traits indicative of susceptibility to schedule-induced polydipsia. Therefore, polydipsic behavior induced by intermittent reinforcer schedules may work additively with genetic susceptibilities to drink ethanol.
Acknowledgments
Supported by National Institutes of Alcohol Abuse and Alcoholism grants AA07611, AA10722.
References
- Cicero TJ. A critique of animal analogues of alcoholism. In: Majchrowicz E, Noble EP, editors. Biochemistry and pharmacology of ethanol. Vol. 2. Plenum Press; New York: 1979. pp. 533–560. [Google Scholar]
- Crabbe JC. Animal models in neurobehavioral genetics: methods for estimating genetic correlation. In: Mormède P, Jones BC, editors. Neurobehavioral genetics: methods and applications. CRC press; Boca Raton, FL: 1999. pp. 121–138. [Google Scholar]
- Crabbe JC. Alcohol and genetics: new models. Am J Med Gen. 2002;114:969–974. doi: 10.1002/ajmg.b.10984. [DOI] [PubMed] [Google Scholar]
- Crabbe JC, Phillips TJ, Kosobud A, Belknap JK. Estimation of genetic correlation: interpretation of experiments using selectively bred and inbred animals. Alcohol Clin Exp Res. 1990;14:141–151. doi: 10.1111/j.1530-0277.1990.tb00461.x. [DOI] [PubMed] [Google Scholar]
- Enoch MA, Goldman D. The genetics of alcoholism and alcohol abuse. Curr Psychiat Rep. 2001;3:144–151. doi: 10.1007/s11920-001-0012-3. [DOI] [PubMed] [Google Scholar]
- Falk JL. Production of polydipsia in normal rats by an intermittent food schedule. Science. 1961;133:195–196. doi: 10.1126/science.133.3447.195. [DOI] [PubMed] [Google Scholar]
- Falk JL. The nature and determinants of adjunctive behavior. Physiol Behav. 1971;6:577–588. doi: 10.1016/0031-9384(71)90209-5. [DOI] [PubMed] [Google Scholar]
- Falk JL. The place of adjunctive behavior in drug abuse research. NIDA Res Monograph. 1981;37:271–278. [PubMed] [Google Scholar]
- Falk JL. Drug abuse as an adjunctive behavior. Drug Alc Dep. 1998;52:91–98. doi: 10.1016/s0376-8716(98)00084-2. [DOI] [PubMed] [Google Scholar]
- Falk JL, Samson HH, Winger G. Behavioral maintenance of high concentrations of blood ethanol and physical dependence in the rat. Science. 1972;177:811–813. doi: 10.1126/science.177.4051.811. [DOI] [PubMed] [Google Scholar]
- Files FJ, Samson HH, Denning CE, Marvin S. Comparison of alcohol-preferring and nonpreferring selectively bred rat lines, II Operant self-administration in a continuous-access situation. Alcohol Clin Exp Res. 1998;22:2147–2158. [PubMed] [Google Scholar]
- Freed EX, Lester D. Schedule-induced consumption of alcohol: calories or chemotherapy? Physiol Behav. 1970;5:555–560. doi: 10.1016/0031-9384(70)90080-6. [DOI] [PubMed] [Google Scholar]
- Gatto GJ, Murphy JM, Waller MB, McBride WJ, Lumeng L, Li TK. Persistence of tolerance to a single dose of ethanol in the selectively bred alcohol-preferring P rats. Pharamcol Biochem Behav. 1987;28:111–115. doi: 10.1016/0091-3057(87)90020-7. [DOI] [PubMed] [Google Scholar]
- Gatto GJ, McBride WJ, Murphy JM, Lumeng L, Li TK. Ethanol self-infusion into the ventral tegmental area by alcohol-preferring rats. Alcohol. 1994;11:557–564. doi: 10.1016/0741-8329(94)90083-3. [DOI] [PubMed] [Google Scholar]
- Goldowitz D, Matthews DB, Hamre KM, Mittleman G, Chesler EJ, Becker HC, Lopez MF, Jones SR, Mathews TA, Miles MF, Kerns R, Grant KA. Progress in using mouse inbred strains, consomics, and mutants to identify genes related to stress, anxiety, and alcohol phenotypes. Alcohol Clin Exp Res. 2006;30:1066–1078. doi: 10.1111/j.1530-0277.2006.00125.x. [DOI] [PubMed] [Google Scholar]
- Heintzelman ME, Best J, Senter RJ. Polydipsia-induced alcohol dependency in rats: a reexamination. Science. 1976;191:482–483. doi: 10.1126/science.942801. [DOI] [PubMed] [Google Scholar]
- Lester D. Self-maintenance of intoxication in the rat. Q J Stud Alcohol. 1961;22:223–231. [PubMed] [Google Scholar]
- Li TK, Lumeng L. Alcohol preference and voluntary alcohol intakes of inbred rat strains and the National Institutes of Health heterogenous stock of rats. Alcohol Clin Exp Res. 1984;8:485–486. doi: 10.1111/j.1530-0277.1984.tb05708.x. [DOI] [PubMed] [Google Scholar]
- Li TK, Lumeng L, McBride WJ, Murphy JM. Rodent lines selected for factors affecting alcohol consumption. Alcohol Alcohol Suppl. 1991;1:91–96. [PubMed] [Google Scholar]
- Li TK, Lumeng L, Doolittle DP. Selective breeding for alcohol preference and associated responses. Behav Gen. 1993;23:163–170. doi: 10.1007/BF01067421. [DOI] [PubMed] [Google Scholar]
- Lumeng L, Hawkins DT, Li TK. New strains of rats with alcohol preference and nonpreference. In: Thurman RG, editor. Alcohol and aldehyde metabolizing systems. Vol. 3. Alcohol and Aldehyde Metabolizing Systems Academic Press; New York: 1977. pp. 537–544. [Google Scholar]
- Lumeng L, Murphy JM, McBride WJ, Li TK. Genetic influences on alcohol preference in animals. In: Begleiter H, Kissin B, editors. The genetics of alcoholism. Oxford University Press; New York: 1995. pp. 165–201. [Google Scholar]
- Mittleman G, Van Brunt CL, Matthews DB. Schedule-induced ethanol self-administration in DBA/2 J and C57BL/6 J mice. Alcohol Clin Exp Res. 2003;27:918–925. doi: 10.1097/01.ALC.0000071930.48632.AE. [DOI] [PubMed] [Google Scholar]
- Murphy JM, Gatto GJ, Waller MB, McBride WJ, Lumeng L, Li TK. Effects of scheduled access on ethanol intake by the alcohol-preferring P lines of rats. Alcohol. 1986;3:331–336. doi: 10.1016/0741-8329(86)90010-8. [DOI] [PubMed] [Google Scholar]
- Murphy JM, Stewart RB, Bell RL, Badia-Elder NE, Carr LG, McBride WJ, Lumeng L, Li TK. Phenotypic and genotypic characterization of the Indiana University rat lines selectively bred for high and low alcohol preference. Behav Gen. 2002;32:363–388. doi: 10.1023/a:1020266306135. [DOI] [PubMed] [Google Scholar]
- National Research Council. Guide for the care and use of laboratory animals. National Research Council; Washington DC: 1996. [Google Scholar]
- Samson HH, Czachowski CL. Behavioral measures of alcohol self-administration and intake control: rodent models. Int Rev Neurobiol. 2003;54:107–143. doi: 10.1016/s0074-7742(03)54004-1. [DOI] [PubMed] [Google Scholar]
- Samson HH, Files FJ, Denning C, Marvin S. Comparison of alcohol-preferring and nonpreferring selectively bred rat lines. I. Ethanol initiation and limited access operant self-administration. Alcohol Clin Exp Res. 1998;22:2133–2146. [PubMed] [Google Scholar]
- Tang M, Falk JL. Production of physical dependence on ethanol by a short drinking episode each day. Pharmacol Biochem Behav. 1983;19:53–55. doi: 10.1016/0091-3057(83)90311-8. [DOI] [PubMed] [Google Scholar]
- Tang M, Brown C, Falk JL. Complete reversal of chronic ethanol polydipsia by schedule withdrawal. Pharmacol Biochem Behav. 1982;16:155–158. doi: 10.1016/0091-3057(82)90028-4. [DOI] [PubMed] [Google Scholar]
- Tomie A, Miller WC, Dranoff E, Pohorecky LA. Intermittent presentations of ethanol sipper tube induce ethanol drinking in rats. Alcohol Alcohol. 2006;41:225–230. doi: 10.1093/alcalc/agl002. [DOI] [PubMed] [Google Scholar]
- Waller MB, McBride WJ, Lumeng L, Li TK. Induction of dependence on ethanol by free-choice drinking in alcohol-preferring rats. Pharmacol Biochem Behav. 1982;16:501–507. doi: 10.1016/0091-3057(82)90459-2. [DOI] [PubMed] [Google Scholar]
- Waller MB, McBride WJ, Gatto GJ, Lumeng L, Li TK. Intragastric self-infusion of ethanol by ethanol-preferring and-nonpreferring lines of rats. Science. 1984;225:78–80. doi: 10.1126/science.6539502. [DOI] [PubMed] [Google Scholar]
- Williams SL, Tang M, Falk JL. Prior exposure to a running wheel and scheduledfood attenuates polydipsia acquisition. Physiol Behav. 1992;52:481–483. doi: 10.1016/0031-9384(92)90334-x. [DOI] [PubMed] [Google Scholar]


