Abstract
Background:
During the nutrition transition in Chile, dietary changes were marked by increased consumption of high-energy, nutrient-poor products, including sugar-sweetened beverages (SSBs). Obesity is now the primary nutritional problem in posttransitional Chile.
Objective:
We conducted a randomized controlled trial to examine the effects on body composition of delivering milk beverages to the homes of overweight and obese children to displace SSBs.
Design:
We randomly assigned 98 children aged 8–10 y who regularly consumed SSBs to intervention and control groups. During a 16-wk intervention, children were instructed to drink 3 servings/d (≈200 g per serving) of the milk delivered to their homes and to not consume SSBs. Body composition was measured by dual-energy X-ray absorptiometry. Data were analyzed by multiple regression analysis according to the intention-to-treat principle.
Results:
For the intervention group, milk consumption increased by a mean (± SEM) of 452.5 ± 37.7 g/d (P<0.0001), and consumption of SSBs decreased by −711.0 ± 33.7 g/d (P < 0.0001). For the control group, milk consumption did not change, and consumption of SSBs increased by 71.9 ± 33.6 g/d (P = 0.04). Changes in percentage body fat, the primary endpoint, did not differ between groups. Nevertheless, the mean (± SE) accretion of lean body mass was greater (P = 0.04) in the intervention (0.92 ± 0.10 kg) than in the control (0.62 ± 0.11 kg) group. The increase in height was also greater (P = 0.01) in the intervention group (2.50 ± 0.21 cm) than in the control group (1.77 ± 0.20 cm) for boys but not for girls.
Conclusion:
Replacing habitual consumption of SSBs with milk may have beneficial effects on lean body mass and growth in children, despite no changes in percentage body fat. This trial was registered at clinicaltrials.gov as NCT00149695.
INTRODUCTION
Historically, malnourished children have been underweight and stunted, largely because of dietary deficits in energy and protein (1, 2). The situation has changed over time in developed countries, and progressively in developing countries, such that malnutrition often coexists with excess energy intake and leads to obesity (3). Speculation regarding the underlying cause of this phenomenon has focused largely on the proliferation and marketing of foods with a high energy content and limited nutritional value (4). In particular, much debate has been directed toward the potentially unhealthful effects of consuming sugar-sweetened beverages (SSBs) (5-7).
With regard to developing countries, Chile is unique because the “nutrition transition” (8) occurred at a faster rate than in other countries (9). Modernization of society and improved living conditions across all socioeconomic levels elicited relatively rapid changes in dietary behaviors that now reflect those in developed countries. In parallel with changes in dietary behaviors, the prevalence of stunting among Chilean children dropped from 10% in 1985 to 2% in 2002 (10), but the prevalence of obesity increased from ≈5% to ≈18% over approximately the same time span (11). Thus, whereas some changes in dietary behaviors have been beneficial, others have been detrimental. The marked increase in consumption of SSBs is one of the dietary changes that has been identified as an adverse outcome of the nutrition transition (12). A survey evaluating food expenditures indicated that SSBs are among the top 3 items for which families spend the most money (12).
In a previous randomized controlled trial conducted in the United States, we found that delivery of noncaloric beverages (eg, bottled water and “diet” beverages) to the homes of adolescents for 25 wk almost completely eliminated the consumption of SSBs and promoted weight loss among those who had the highest body mass index (BMI) at baseline (13). Of particular note, this intervention focused only on eliminating consumption of SSBs, in contrast with most pediatric obesity treatment programs that have taken a comprehensive approach by targeting multiple behaviors believed to promote positive energy balance (4, 14, 15). If consumption of SSBs indeed has a high impact on body weight relative to other dietary behaviors, then a focused approach targeting this behavior may provide an especially pragmatic public health strategy for curbing the obesity epidemic in both developed and developing countries.
Moreover, replacing SSBs with milk rather than non-caloric beverages may be more effective for enhancing nutrition in children consuming diets with poor overall quality. In addition to its potential effects on bone health (16, 17), daily consumption has been shown to be inversely associated with body fat or BMI in several pediatric studies (18-21), and milk protein may contribute to the accretion of lean tissue (22). Nevertheless, the role of milk in optimizing body composition has come under scrutiny because of limited pediatric data on which to base guidelines (23-25).
In Chile and other countries that have recently experienced the nutrition transition, there is a great need to identify public health strategies to reduce obesity and optimize nutrition among children (9, 15). This challenge is usually complicated by the wide availability of high-energy, nutrient-poor foods and the higher cost of more healthful options. The purpose of the present study was to evaluate the effects of an intervention aimed at replacing habitual consumption of SSBs with milk in overweight and obese Chilean children. We hypothesized that the intervention would have beneficial effects on body composition and growth.
SUBJECTS AND METHODS
Overview
We conducted a randomized controlled trial comprising an initial baseline period followed by a 16-wk intervention period. Body composition and diet were assessed during the baseline period and at the end of the intervention period. The study protocol was approved by the institutional review boards at Children's Hospital Boston and the Institute of Nutrition and Food Technology (Instituto de Nutrición y Tecnología de los Alimentos, INTA), University of Chile. Written informed consent and assent were obtained from parents and children, respectively, before enrollment. Each child who completed the follow-up assessment received a gift certificate to a local department store in an amount equivalent to US$50. Data were collected between July 2004 and December 2005 in Santiago, Chile.
Subjects
We enrolled 98 children (52 boys, 46 girls) aged 8–10 y with the assistance of teachers and staff at 2 schools. One of the inclusion criteria was a BMI greater than the 85th percentile for sex and age based on Centers for Disease Control and Prevention growth charts (26). Although developed in the United States, these growth charts have been used to evaluate the weight status of Chilean children (15, 27). All enrolled children were prepubertal (ie, Tanner Stage 1; 28) and reported consuming ≥2 servings/d of SSBs. We did not enroll any child known to have a serious underlying medical condition, lactose intolerance, or allergy to milk protein. We also excluded those who were taking prescription medications that might affect body weight.
The study statistician randomly assigned each eligible child to the intervention or control group using a computer-generated set of random numbers. Random assignment was stratified by height-for-age z score (HAZ ≤ 0 for sex and age, HAZ > 0) (26). The sequence of random numbers was concealed from personnel conducting recruitment until after the group assignment.
Intervention
A nutritionist visited the homes of children in the intervention group weekly to deliver the milk beverages (Soprole, San Bernardo, Chile), provide instructions to the family about consuming the delivered beverages, and encourage parents to remove SSBs from their homes. We selected flavored milk beverages to increase the likelihood that children would consume what was delivered to them (29). Each individually packaged portion (200 mL, ≈200 g) provided 80 kcal, 8 g protein, 3 g fat, 11 g carbohydrate, and 320 mg Ca. Children were counseled to drink 3 portions per day of the milk beverages and not to consume SSBs; no additional nutrition education regarding other foods or beverages was provided. Moreover, they were encouraged to take the milk beverages to school for consumption during lunch. To avoid competition between the subject and siblings for the milk beverages, the number of portions delivered to each home was based on household size. We provided 1 serving/d for each sibling in the household and offered additional servings if this allotment was not adequate on the basis of discussions with the mother. All members of each household were encouraged to support the subject by not drinking SSBs. This strategy was modeled after the intervention used in our previous trial (13). We gave no instructions regarding food or beverage choices to subjects in the control group and had no contact with them, other than to conduct assessments.
Weight, height, and body composition
All data were collected in a clinic at INTA. Weight was measured to the nearest 0.1 kg using an electronic scale (SECA Colorata 760; SECA, Hamburg, Germany), and height was measured to the nearest 0.5 cm using a stadiometer (SECA 214; SECA). For each child, BMI was calculated as total mass (kg) divided by height (m) squared, and HAZ was determined on the basis of sex- and age-specific growth curves (26).
Body composition was assessed by dual-energy X-ray absorptiometry (DXA) using a Lunar Prodigy whole-body scanner (Encore 2007, version 11.30.062; GE Medical Systems, Madison, WI) by certified clinic personnel who were masked to group assignment. Total body fat, trunk fat, and lean mass were the variables of primary interest in the present study. Body fat percentage, the primary endpoint, was calculated as the proportion of fat mass to total mass.
Food-frequency questionnaire
Diet was assessed by using a validated food-frequency questionnaire (FFQ), as previously described (27). The FFQ was administered to each subject at baseline and again at the end of the 16-wk intervention period. Mothers were present during administration of the FFQ and provided assistance as necessary. The beverage items of interest for this report included milk, carbonated beverages, juice drinks (often made by adding packaged sugary powders with fruit flavoring to water), and diet beverages. For each beverage category, a nutritionist asked the child the size of a usual portion and how many times the specified portion was consumed per week. Data were then converted to grams per day. Reported intakes for carbonated beverages and juice drinks were summed to obtain an overall value for consumption of SSBs. We also estimated daily intakes of protein, calcium, and energy according to the Chilean food-composition tables (30).
Statistical methods
The study was designed to provide 80% power to detect an effect size of 0.60 with the use of a 5% type I error rate. We compared baseline characteristics and changes in dietary variables between the intervention and control groups by Student's t test. Multiple linear regression analysis was used to analyze change scores for study outcomes. We included group as an indicator independent variable, age and sex as obligatory covariates, and a group × sex interaction term in the analytic model. According to the intention-to-treat principle, we included data from the 93 subjects who completed follow-up assessments irrespective of compliance with study protocols during the intervention period. We did not impute missing data for subjects who were lost to follow-up in light of the high retention rate and lack of an appropriate strategy for imputing data for growing children. Computations were performed with SAS software version 9.01 (SAS Institute Inc, Cary, NC). Statistical significance was defined as P < 0.05.
RESULTS
Subject flow
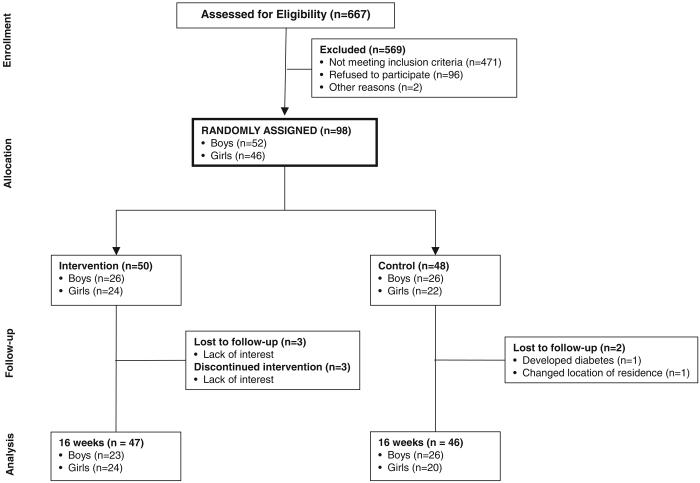
The flow of subjects through the trial is presented in Figure 1. Of the 50 children who were randomly assigned to the intervention group, 3 boys were lost to follow-up, and 3 girls discontinued the intervention but were available for follow-up measures. Of the 48 children who were randomly assigned to the control group, 1 girl developed type 2 diabetes mellitus and was dismissed from the study, and another girl was lost to follow-up. On study completion, we noted that 3 of the girls who were randomly assigned to the control group did not meet the BMI inclusion criterion because of miscalculation of BMI percentiles. Nevertheless, we opted to include the collected data from all randomized subjects in the statistical analyses, particularly given that inclusion versus exclusion of data from the 3 subjects in question did not alter the results or conclusions. The retention rate, calculated based on the number of children who completed the follow-up body-composition assessment, was 95%. There were no serious adverse events deemed to be directly related to study participation.
FIGURE 1.
Flow of subjects throughout the trial.
Baseline measures
Baseline subject characteristics are presented in Table 1. There were no significant differences between the intervention and control groups for age, weight, height, or body composition. As expected, given that Chile is in the posttransitional phase of the nutrition transition (9), we found no evidence of severe stunting in that the HAZ was > −2 for all subjects, ranging from −1.83 to 2.12. Regarding baseline beverage consumption (Table 2), there were no significant differences between groups for milk and SSB consumption, but the intervention group consumed more diet beverages than did the control group (P = 0.02). There were no group differences in protein, calcium, or energy intakes at baseline (Table 2).
TABLE 1.
Baseline characteristics of subjects in the intervention and control groups
| Variable | Intervention (n = 50) |
Control (n = 48) |
P1 |
|---|---|---|---|
| Age (y) | 9.26 ± 0.102 | 9.43 ± 0.09 | 0.23 |
| Weight (kg) | 42.03 ± 0.91 | 41.83 ± 1.03 | 0.89 |
| Height (cm) | 136.42 ± 0.92 | 136.66 ± 0.92 | 0.863 |
| Height-for-age z score | 0.29 ± 0.13 | 0.18 ± 0.10 | 0.523 |
| BMI (kg/m2) | 22.47 ± 0.29 | 22.29 ± 0.39 | 0.71 |
| BMI z score | 1.74 ± 0.04 | 1.64 ± 0.06 | 0.18 |
| Percentage body fat | 36.46 ± 0.75 | 35.99 ± 0.71 | 0.65 |
| Total fat mass (kg) | 15.28 ± 0.61 | 15.00 ± 0.61 | 0.74 |
| Trunk fat mass (kg) | 7.55 ± 0.36 | 7.28 ± 0.35 | 0.59 |
| Lean mass (kg) | 24.71 ± 0.41 | 24.85 ± 0.46 | 0.82 |
| Bone mass (kg) | 1.30 ± 0.03 | 1.28 ± 0.03 | 0.56 |
From Student's t test comparing the intervention and control groups.
x̄ ± SEM (all such values).
Random group assignment stratified by height-for-age z score.
TABLE 2.
Dietary intake in the intervention and control groups
| Variable | Intervention (n = 44) |
Control (n = 46) |
P1 |
|---|---|---|---|
| Milk beverages (g/d) | |||
| Baseline | 662.5 ± 33.02 | 678.3 ± 24.2 | 0.70 |
| Change | 452.5 ± 37.73 | 11.3 ± 11.3 | <0.0001 |
| Sugar-sweetened beverages (g/d) | |||
| Baseline | 742.8 ± 29.4 | 802.1 ± 20.5 | 0.10 |
| Change | −711.0 ± 33.73 | 71.9 ± 33.64 | <0.0001 |
| Diet beverages (g/d) | |||
| Baseline | 37.5 ± 13.3 | 3.7 ± 2.6 | 0.02 |
| Change | 298.9 ± 46.13 | 15.2 ± 15.2 | <0.0001 |
| Protein (g/d) | |||
| Baseline | 87.1 ± 2.5 | 85.5 ±2.1 | 0.64 |
| Change | 11.5 ± 1.83 | 1.8 ± 0.75 | <0.0001 |
| Calcium (mg/d) | |||
| Baseline | 983.7 ± 39.6 | 982.7 ± 32.0 | 0.98 |
| Change | 670.8 ± 37.73 | 13.4 ± 14.8 | <0.0001 |
| Energy (kcal/d) | |||
| Baseline | 2644.7 ± 50.5 | 2577.8 ± 63.9 | 0.42 |
| Change | −91.0 ± 33.05 | 9.7 ± 29.8 | 0.03 |
From Student's t test comparing the intervention and control groups.
x̄ ± SEM (all such values).
Significant change from baseline: P < 0.0001
Significant change from baseline: P = 0.04
Significant change from baseline: P ≤ 0.01.
Dietary data
As shown in Table 2, daily milk consumption increased by ≈450 g/d for the intervention group (P < 0.0001) and did not change for the control group. In the intervention group, increased milk consumption was accompanied by a 96% reduction in consumption of SSBs (P < 0.0001) and increased consumption of diet beverages (P < 0.0001). Protein and calcium intakes increased (P < 0.0001) and energy intake decreased (P = 0.009) with the intervention. In the control group, consumption of SSBs increased (P = 0.04), but the consumption of milk and diet beverages did not change. Protein intake increased slightly but significantly (P = 0.01), whereas calcium and energy intakes did not change from baseline to follow-up.
Body-composition outcomes
Body-composition outcomes are presented in Table 3. Change in percentage body fat, the primary endpoint, did not differ significantly groups. Nevertheless, accretion of lean mass was greater in the intervention group than in the control group (P = 0.04), despite no difference in fat mass. For boys, but not for girls, height increased more in the intervention group than in the control group (P = 0.01).
TABLE 3.
Changes in body composition in the intervention and control groups
|
P |
||||
|---|---|---|---|---|
| Characteristic | Intervention (n = 47) |
Control (n = 46) |
Group1 | Group × sex2 |
| Weight (kg) | 1.57 ± 0.243 | 1.13 ± 0.24 | 0.20 | 0.20 |
| Height (cm) | 2.27 ± 0.15 | 2.16 ± 0.15 | 0.61 | 0.004 |
| Boys | 2.50 ± 0.21 | 1.77 ± 0.20 | 0.01 | — |
| Girls | 2.04 ± 0.21 | 2.55 ± 0.23 | 0.10 | — |
| BMI (kg/m2) | 0.08 ± 0.12 | −0.09 ± 0.12 | 0.33 | 0.82 |
| BMI z score | 0.01 ± 0.02 | −0.01 ± 0.02 | 0.58 | 0.78 |
| Percentage body fat (%) | 0.36 ± 0.24 | 0.78 ± 0.25 | 0.22 | 0.89 |
| Total fat mass (kg) | 0.84 ± 0.15 | 0.89 ± 0.15 | 0.81 | 0.65 |
| Trunk fat mass (kg) | 0.31 ± 0.09 | 0.39 ± 0.09 | 0.52 | 0.97 |
| Lean mass (kg) | 0.92 ± 0.10 | 0.62 ± 0.11 | 0.04 | 0.43 |
| Bone mass (kg) | 0.06 ± 0.01 | 0.05 ± 0.01 | 0.47 | 0.48 |
Tests for group difference that is consistent between the sexes by multiple linear regression analysis.
Tests for interaction (ie, inconsistent group difference between the 2 sexes) by multiple linear regression analysis.
Least-squares x̄ ± SE from regression analysis, adjusted for age and sex (all such values).
DISCUSSION
An intervention designed to change the availability of beverages in the home environment increased consumption of milk while almost completely eliminating consumption of SSBs in overweight and obese Chilean children. These changes in beverage consumption increased the accretion of lean mass but did not significantly affect fat mass. The increased lean mass was accompanied by greater linear growth for the boys, but not for the girls.
The American Academy of Pediatrics recommends that children consume 2 (aged ≤8 y) to 3 (aged 9 y and older) servings of milk or dairy products per day (31), consistent with the Dietary Guidelines for Americans, 2005 (32). According to these guidelines, a single serving of milk is specified as 8 fluid ounces (1 cup), which is equal to ≈240 g and slightly greater than the 200-g portion that was considered a serving in the present study. Recommendations are calculated from idealized dietary patterns in which the nutrients provided in milk, with a particular focus on calcium, also are obtained from other foods such as dark green leafy vegetables and legumes. However, most children do not consume idealized diets, and some experts argue that recommendations for milk and dairy products should be increased by one serving (≈240 g) per day, because this food group provides a readily available and relatively low-cost source of essential nutrients (33, 34). At follow-up in the present study, children in the intervention group reported drinking >1100 g milk/d (ie, between 4 and 5 servings/d). Thus, study outcomes reflect the effects of consuming milk in excess of current recommendations. Nevertheless, calcium intake for all study participants at follow-up was less than the recommended upper limit of 2500 mg for children (34).
Studies examining the time course of postprandial aminoacidemia and protein turnover after milk consumption provide a plausible physiologic basis for interpreting the greater accretion of lean mass in the intervention group than in the control group (22, 35, 36). The 2 major protein fractions in milk are whey and casein (37). Whey is digested rapidly and causes an abrupt and transient increase in aminoacidemia that stimulates whole-body protein synthesis (35). In contrast, casein is digested more slowly and does not stimulate protein synthesis, but suppresses protein breakdown (35). Postprandial metabolism after milk consumption may lead to increased accretion of lean mass over time because of the distinct and synergistic effects of whey and casein in promoting net anabolism.
Another potential mechanism may involve the direct adverse effects of SSBs on body composition. SSBs are among the greatest contributors to dietary glycemic load (mathematical product of dietary glycemic index and total carbohydrate) (38, 39) in children's diets. A high-glycemic-load meal has been shown to elicit reactive hypoglycemia and counterregulatory hormone secretion in the late postprandial period (40). Counterregulatory hormones have proteolytic actions that may adversely affect lean body mass if persistently elevated (41), as previously proposed (42, 43). Indeed, rodents fed a high-glycemic-index diet had significantly less lean tissue at the same mean body weight than did rodents fed a low-glycemic-index diet (44).
With increased consumption of milk and decreased consumption of SSBs occurring concurrently, energy intake decreased in the intervention group. The significance of this finding, based on self-report of diet, is not readily apparent given that we observed no changes in body weight or BMI. Nevertheless, milk may be more satiating than SSBs because of its mixed macronutrient composition (45), low glycemic load (46), and other intrinsic properties, such as micronutrient profiles, that influence insulin signaling and glucose homeostasis (47). An increase in satiety may lead to lower energy intakes over time.
Our data are consistent with previous randomized controlled trials conducted in developed countries, indicating no changes in fat mass with increased consumption of milk and dairy products (48-50). In a study of 9- to 13-y-old girls, Chan et al (48) reported no effect with supplementation of dairy products providing 1200 mg Ca/d (equivalent to ≈4 servings of milk/d) over 1 y. Likewise, Cadogan et al (49) found no effect with provision of 568 mL/d (≈2.5 servings/d) of extra milk to 12-y-old girls for 18 mo, and Merrilees et al (50) observed no effect with an increase in dairy foods to provide an extra 1000 mg Ca/d (equivalent to ≈3.3 servings of milk/d) to teenage girls over 2 y. In a 1-y study of young women, Gunther et al (51) found no differences in fat mass between 3 study groups with daily consumption of milk or dairy products providing calcium in average amounts of 742, 1026, or 1131 mg (approximating 2.5, 3.4, and 3.8 servings of milk, respectively). Moreover, a secondary regression analysis of 6-mo of follow-up data from the latter study indicated that the estimated net beneficial effect on fat mass of relatively high (≈4 servings) compared with low (≈1.7 servings) daily intake of milk or dairy products is <2 kg over 18 mo (52). Thus, associations between intake of milk or dairy products and body weight and composition reported in several (18-21), although not all (53, 54), observational studies may be due to small but cumulative effects over time.
None of the randomized controlled trials cited above (48-51) showed an increase in accretion of lean mass with increased consumption of milk or dairy products, as observed in the present study. Previous studies were conducted in developed, rather than in developing, countries. Moreover, discrepant findings may relate to differences in subject characteristics or details of the intervention. Previous studies focused on girls who were at various stages of puberty and not overweight (48-50). In contrast, we enrolled boys and girls who were prepubertal and overweight or obese at baseline. Regarding interventions, some of the previous studies provided a variety of dairy products to subjects (48, 50), whereas we provided only milk given the focus of the present study on beverage consumption. The present study also was unique in that we aimed to not only increase the consumption of milk but also to decrease the consumption of SSBs. The dual focus of the intervention may have conferred added benefit.
Study strengths and limitations warrant comment. The strengths included a state-of-the-art method for assessing body composition, an environmental intervention that relied largely on beverage deliveries to the home, and a high retention rate. Families were receptive to opening their homes to the study nutritionist for delivery of the milk beverages, and only 6 of 50 children in the intervention group requested discontinuation of beverage delivery. Follow-up DXA assessments were completed in 3 of these 6 children, such that analyses could be conducted according to the intention-to-treat principle using data from 95% of the subjects who were randomly assigned to a study group. Moreover, on the basis of data derived from the FFQ, the intervention had the anticipated effect, ie, it significantly decreased the consumption of SSBs and increased the consumption of milk. Limitations of the study included a relatively small sample size and short intervention period. Reliance on self-report for dietary assessment was an additional limitation, as in all trials of free-living subjects. In addition, we recognize that concerns have been raised for the long-term adverse health effects of high levels of dairy consumption (55, 56)—an issue we were unable to address because of the study's short duration.
In conclusion, replacing the habitual consumption of SSBs with milk may have beneficial effects on lean body mass and growth in children. We did not observe a change in fat mass or percentage body fat, either because of the study's short duration or because replacement of one energy-containing beverage for another does not affect these endpoints. Larger studies are needed to evaluate the long-term effects of altering beverage consumption on body composition in children and to determine which beverages promote optimum growth and chronic disease risk reduction among pediatric populations in different regions of the world.
Acknowledgments
We thank the clinic personnel at INTA for assessing body composition, Henry A Feldman for advice regarding the statistical analyses and data presentation, and Erica Garcia-Lago for assistance with translation and data management.
The authors' responsibilities were as follows: CA: designed the study, supervised the conduct of the study, interpreted the data, and wrote the manuscript; CBE: designed the study, provided consultation regarding the conduct of the study, performed the statistical analyses, interpreted the data, and wrote the manuscript; MC: designed the study and supervised the conduct of the study; LL: randomly assigned subjects to groups, managed the data, and performed the statistical analyses; NB: recruited the study subjects and visited the homes of children in the intervention group; and DSL: designed the study, secured funding, provided consultation regarding the conduct of the study, and wrote the manuscript. DSL is the author of a book on childhood obesity (Ending the Food Fight: Guide Your Child to a Healthy Weight in a Fast Food/Fake Food World). None of the other authors reported any conflicts of interest.
Footnotes
The content is solely the responsibility of the authors and does not necessarily represent the official views of the Fogarty International Center or the National Institutes of Health.
Supported by grant TW006818 from the Fogarty International Center, National Institutes of Health.
REFERENCES
- 1.Chase HP, Martin HP. Undernutrition and child development. N Engl J Med. 1970;282:933–9. doi: 10.1056/NEJM197004232821701. [DOI] [PubMed] [Google Scholar]
- 2.Hansen JD, Freesemann C, Moodie AD, Evans DE. What does nutritional growth retardation imply? Pediatrics. 1971;47(suppl 2):299+. [PubMed] [Google Scholar]
- 3.Langevin DD, Kwiatkowski C, McKay MG, et al. Evaluation of diet quality and weight status of children from a low socioeconomic urban environment supports “at risk” classification. J Am Diet Assoc. 2007;107:1973–7. doi: 10.1016/j.jada.2007.08.008. [DOI] [PubMed] [Google Scholar]
- 4.Ebbeling CB, Pawlak DB, Ludwig DS. Childhood obesity: public-health crisis, common sense cure. Lancet. 2002;360:473–82. doi: 10.1016/S0140-6736(02)09678-2. [DOI] [PubMed] [Google Scholar]
- 5.Bachman CM, Baranowski T, Nicklas TA. Is there an association between sweetened beverages and adiposity? Nutr Rev. 2006;64:153–74. doi: 10.1111/j.1753-4887.2006.tb00199.x. [DOI] [PubMed] [Google Scholar]
- 6.Drewnowski A, Bellisle F. Liquid calories, sugar, and body weight. Am J Clin Nutr. 2007;85:651–61. doi: 10.1093/ajcn/85.3.651. [DOI] [PubMed] [Google Scholar]
- 7.Malik VS, Schulze MB, Hu FB. Intake of sugar-sweetened beverages and weight gain: a systematic review. Am J Clin Nutr. 2006;84:274–88. doi: 10.1093/ajcn/84.1.274. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Popkin BM. The nutrition transition: an overview of world patterns of change. Nutr Rev. 2004;62(suppl):S140–3. doi: 10.1111/j.1753-4887.2004.tb00084.x. [DOI] [PubMed] [Google Scholar]
- 9.Uauy R, Albala C, Kain J. Obesity trends in Latin America: transiting from under- to overweight. J Nutr. 2001;131(suppl):893S–9S. doi: 10.1093/jn/131.3.893S. [DOI] [PubMed] [Google Scholar]
- 10.Kain J, Uauy R, Lera L, Taibo M, Albala C. Trends in height and BMI of 6-year-old children during the nutrition transition in Chile. Obes Res. 2005;13:2178–86. doi: 10.1038/oby.2005.270. [DOI] [PubMed] [Google Scholar]
- 11.Vio F, Albala C, Kain J. Nutrition transition in Chile revisited: mid-term evaluation of obesity goals for the period 2000-2010. Public Health Nutr. 2008;11:405–12. doi: 10.1017/S136898000700050X. [DOI] [PubMed] [Google Scholar]
- 12.Albala C, Vio F, Kain J, Uauy R. Nutrition transition in Chile: determinants and consequences. Public Health Nutr. 2002;5:123–8. doi: 10.1079/PHN2001283. [DOI] [PubMed] [Google Scholar]
- 13.Ebbeling CB, Feldman HA, Osganian SK, Chomitz VR, Ellenbogen SJ, Ludwig DS. Effects of decreasing sugar-sweetened beverage consumption on body weight in adolescents: a randomized, controlled pilot study. Pediatrics. 2006;117:673–80. doi: 10.1542/peds.2005-0983. [DOI] [PubMed] [Google Scholar]
- 14.Baranowski T, Cullen KW, Nicklas T, Thompson D, Baranowski J. School-based obesity prevention: a blueprint for taming the epidemic. Am J Health Behav. 2002;26:486–93. doi: 10.5993/ajhb.26.6.9. [DOI] [PubMed] [Google Scholar]
- 15.Kain J, Uauy R, Albala, Vio F, Cerda R, Leyton B. School-based obesity prevention in Chilean primary school children: methodology and evaluation of a controlled study. Int J Obes Relat Metab Disord. 2004;28:483–93. doi: 10.1038/sj.ijo.0802611. [DOI] [PubMed] [Google Scholar]
- 16.Black RE, Williams SM, Jones IE, Goulding A. Children who avoid drinking cow milk have low dietary calcium intakes and poor bone health. Am J Clin Nutr. 2002;76:675–80. doi: 10.1093/ajcn/76.3.675. [DOI] [PubMed] [Google Scholar]
- 17.Kalkwarf HJ, Khoury JC, Lanphear BP. Milk intake during childhood and adolescence, adult bone density, and osteoporotic fractures in US women. Am J Clin Nutr. 2003;77:257–65. doi: 10.1093/ajcn/77.1.257. [DOI] [PubMed] [Google Scholar]
- 18.Carruth BR, Skinner JD. The role of dietary calcium and other nutrients in moderating body fat in preschool children. Int J Obes Relat Metab Disord. 2001;25:559–66. doi: 10.1038/sj.ijo.0801562. [DOI] [PubMed] [Google Scholar]
- 19.Dixon LB, Pellizzon MA, Jawad AF, Tershakovec AM. Calcium and dairy intake and measures of obesity in hyper- and normocholesterolemic children. Obes Res. 2005;13:1727–38. doi: 10.1038/oby.2005.211. [DOI] [PubMed] [Google Scholar]
- 20.Moore LL, Bradlee ML, Gao D, Singer MR. Low dairy intake in early childhood predicts excess body fat gain. Obesity (Silver Spring) 2006;14:1010–8. doi: 10.1038/oby.2006.116. [DOI] [PubMed] [Google Scholar]
- 21.Novotny R, Daida YG, Acharya S, Grove JS, Vogt TM. Dairy intake is associated with lower body fat and soda intake with greater weight in adolescent girls. J Nutr. 2004;134:1905–9. doi: 10.1093/jn/134.8.1905. [DOI] [PubMed] [Google Scholar]
- 22.Lacroix M, Bos C, Leonil J, et al. Compared with casein or total milk protein, digestion of milk soluble proteins is too rapid to sustain the anabolic postprandial amino acid requirement. Am J Clin Nutr. 2006;84:1070–9. doi: 10.1093/ajcn/84.5.1070. [DOI] [PubMed] [Google Scholar]
- 23.Huang TT, McCrory MA. Dairy intake, obesity, and metabolic health in children and adolescents: knowledge and gaps. Nutr Rev. 2005;63:71–80. doi: 10.1111/j.1753-4887.2005.tb00124.x. [DOI] [PubMed] [Google Scholar]
- 24.Lanou AJ, Berkow SE, Barnard ND. Calcium, dairy products, and bone health in children and young adults: a reevaluation of the evidence. Pediatrics. 2005;115:736–43. doi: 10.1542/peds.2004-0548. [DOI] [PubMed] [Google Scholar]
- 25.Winzenberg T, Shaw K, Fryer J, Jones G. Calcium supplements in healthy children do not affect weight gain, height, or body composition. Obesity (Silver Spring) 2007;15:1789–98. doi: 10.1038/oby.2007.213. [DOI] [PubMed] [Google Scholar]
- 26.Kuczmarski RJ, Ogden CL, Grummer-Strawn LM, et al. CDC growth charts: United States. Adv Data. 2000;(314):1–27. [PubMed] [Google Scholar]
- 27.Olivares S, Kain J, Lera L, Pizarro F, Vio F, Moron C. Nutritional status, food consumption and physical activity among Chilean school children: a descriptive study. Eur J Clin Nutr. 2004;58:1278–85. doi: 10.1038/sj.ejcn.1601962. [DOI] [PubMed] [Google Scholar]
- 28.Tanner JM. Growth at adolescence. Blackwell Scientific Publications; Oxford, United Kingdom: 1962. [Google Scholar]
- 29.Johnson RK, Frary C, Wang MQ. The nutritional consequences of flavored-milk consumption by school-aged children and adolescents in the United States. J Am Diet Assoc. 2002;102:853–6. doi: 10.1016/s0002-8223(02)90192-6. [DOI] [PubMed] [Google Scholar]
- 30.Schmidt-Hebbel H, Pennacchiotti I, Masson L, Mella MA. Tabla de Composición Química de Alimentos Chilenos. (Table of the chemical composition of Chilean foods.) 8th ed. Facultad de Ciencias Químicas y Farmacéuticas, Universidad de Chile; Santiago, Chile: 1992. [Google Scholar]
- 31.Gidding SS, Dennison BA, Birch LL, et al. Dietary recommendations for children and adolescents: a guide for practitioners. Pediatrics. 2006;117:544–59. doi: 10.1542/peds.2005-2374. [DOI] [PubMed] [Google Scholar]
- 32.US Department of Health and Human Services. US Department of Agriculture . Dietary guidelines for Americans, 2005. 6th ed. US Government Printing Office; Washington, DC: 2005. [Google Scholar]
- 33.Fulgoni VL, III, Huth PJ, DiRienzo DB, Miller GD. Determination of the optimal number of dairy servings to ensure a low prevalence of inadequate calcium intake in Americans. J Am Coll Nutr. 2004;23:651–9. doi: 10.1080/07315724.2004.10719407. [DOI] [PubMed] [Google Scholar]
- 34.Greer FR, Krebs NF. Optimizing bone health and calcium intakes of infants, children, and adolescents. Pediatrics. 2006;117:578–85. doi: 10.1542/peds.2005-2822. [DOI] [PubMed] [Google Scholar]
- 35.Boirie Y, Dangin M, Gachon P, Vasson MP, Maubois JL, Beaufrere B. Slow and fast dietary proteins differently modulate postprandial protein accretion. Proc Natl Acad Sci U S A. 1997;94:14930–5. doi: 10.1073/pnas.94.26.14930. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Dangin M, Boirie Y, Garcia-Rodenas C, et al. The digestion rate of protein is an independent regulating factor of postprandial protein retention. Am J Physiol Endocrinol Metab. 2001;280:E340–8. doi: 10.1152/ajpendo.2001.280.2.E340. [DOI] [PubMed] [Google Scholar]
- 37.Miller GD, Jarvis JK, McBean LD. Handbook of dairy foods and nutrition. 3rd ed. CRC Press; Boca Raton, FL: 2006. [Google Scholar]
- 38.Salmeron J, Manson JE, Stampfer MJ, Colditz GA, Wing AL, Willett WC. Dietary fiber, glycemic load, and risk of non-insulin-dependent diabetes mellitus in women. JAMA. 1997;277:472–7. doi: 10.1001/jama.1997.03540300040031. [DOI] [PubMed] [Google Scholar]
- 39.Ebbeling CB, Ludwig DS. Treating obesity in youth: should dietary glycemic load be a consideration? Adv Pediatr. 2001;48:179–212. [PubMed] [Google Scholar]
- 40.Ludwig DS, Majzoub JA, Al-Zahrani A, Dallal GE, Blanco I, Roberts SB. High glycemic index foods, overeating, and obesity. Pediatrics. 1999;103:E26. doi: 10.1542/peds.103.3.e26. [DOI] [PubMed] [Google Scholar]
- 41.Gelfand RA, Matthews DE, Bier DM, Sherwin RS. Role of counterregulatory hormones in the catabolic response to stress. J Clin Invest. 1984;74:2238–48. doi: 10.1172/JCI111650. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Agus MS, Swain JF, Larson CL, Eckert EA, Ludwig DS. Dietary composition and physiologic adaptations to energy restriction. Am J Clin Nutr. 2000;71:901–7. doi: 10.1093/ajcn/71.4.901. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Ludwig DS. The glycemic index: physiological mechanisms relating to obesity, diabetes, and cardiovascular disease. JAMA. 2002;287:2414–23. doi: 10.1001/jama.287.18.2414. [DOI] [PubMed] [Google Scholar]
- 44.Pawlak DB, Kushner JA, Ludwig DS. Effects of dietary glycaemic index on adiposity, glucose homoeostasis, and plasma lipids in animals. Lancet. 2004;364:778–85. doi: 10.1016/S0140-6736(04)16937-7. [DOI] [PubMed] [Google Scholar]
- 45.St-Onge MP, Rubiano F, DeNino WF, et al. Added thermogenic and satiety effects of a mixed nutrient vs a sugar-only beverage. Int J Obes Relat Metab Disord. 2004;28:248–53. doi: 10.1038/sj.ijo.0802560. [DOI] [PubMed] [Google Scholar]
- 46.Ball SD, Keller KR, Moyer-Mileur LJ, Ding YW, Donaldson D, Jackson WD. Prolongation of satiety after low versus moderately high glycemic index meals in obese adolescents. Pediatrics. 2003;111:488–94. doi: 10.1542/peds.111.3.488. [DOI] [PubMed] [Google Scholar]
- 47.Barbagallo M, Dominguez LJ. Magnesium metabolism in type 2 diabetes mellitus, metabolic syndrome and insulin resistance. Arch Biochem Biophys. 2007;458:40–7. doi: 10.1016/j.abb.2006.05.007. [DOI] [PubMed] [Google Scholar]
- 48.Chan GM, Hoffman K, McMurry M. Effects of dairy products on bone and body composition in pubertal girls. J Pediatr. 1995;126:551–6. doi: 10.1016/s0022-3476(95)70348-9. [DOI] [PubMed] [Google Scholar]
- 49.Cadogan J, Eastell R, Jones N, Barker ME. Milk intake and bone mineral acquisition in adolescent girls: randomised, controlled intervention trial. BMJ. 1997;315:1255–60. doi: 10.1136/bmj.315.7118.1255. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Merrilees MJ, Smart EJ, Gilchrist NL, et al. Effects of diary food supplements on bone mineral density in teenage girls. Eur J Nutr. 2000;39:256–62. doi: 10.1007/s003940070004. [DOI] [PubMed] [Google Scholar]
- 51.Gunther CW, Legowski PA, Lyle RM, et al. Dairy products do not lead to alterations in body weight or fat mass in young women in a 1-y intervention. Am J Clin Nutr. 2005;81:751–6. doi: 10.1093/ajcn/81.4.754. [DOI] [PubMed] [Google Scholar]
- 52.Eagan MS, Lyle RM, Gunther CW, Peacock M, Teegarden D. Effect of 1-year dairy product intervention on fat mass in young women: 6-month follow-up. Obesity (Silver Spring) 2006;14:2242–8. doi: 10.1038/oby.2006.263. [DOI] [PubMed] [Google Scholar]
- 53.Berkey CS, Rockett HR, Willett WC, Colditz GA. Milk, dairy fat, dietary calcium, and weight gain: a longitudinal study of adolescents. Arch Pediatr Adolesc Med. 2005;159:543–50. doi: 10.1001/archpedi.159.6.543. [DOI] [PubMed] [Google Scholar]
- 54.Phillips SM, Bandini LG, Cyr H, Colclough-Douglas S, Naumova E, Must A. Dairy food consumption and body weight and fatness studied longitudinally over the adolescent period. Int J Obes Relat Metab Disord. 2003;27:1106–13. doi: 10.1038/sj.ijo.0802370. [DOI] [PubMed] [Google Scholar]
- 55.Willett W. Lessons from dietary studies in Adventists and questions for the future. Am J Clin Nutr. 2003;78(suppl):539S–43S. doi: 10.1093/ajcn/78.3.539S. [DOI] [PubMed] [Google Scholar]
- 56.van der Pols JC, Bain C, Gunnell D, Smith GD, Frobisher C, Martin RM. Childhood dairy intake and adult cancer risk: 65-y follow-up of the Boyd Orr cohort. Am J Clin Nutr. 2007;86:1722–9. doi: 10.1093/ajcn/86.5.1722. [DOI] [PubMed] [Google Scholar]