Abstract
OBJECTIVE—Type 2 diabetes is the leading cause of end-stage renal disease worldwide. Aside from hyperglycemia and hypertension, other metabolic factors may determine renal outcome. We examined risk associations of metabolic syndrome with new onset of chronic kidney disease (CKD) in 5,829 Chinese patients with type 2 diabetes enrolled between 1995 and 2005.
RESEARCH DESIGN AND METHODS—Metabolic syndrome was defined by National Cholesterol Education Program Adult Treatment Panel III criteria with the Asian definition of obesity. Estimated glomerular filtration rate (eGFR) was calculated using the abbreviated Modification of Diet in Renal Disease formula modified for the Chinese population. New onset of CKD was defined as eGFR <60 ml/min per 1.73 m2 at the time of censor. Subjects with CKD at baseline were excluded from the analysis.
RESULTS—After a median follow-up duration of 4.6 years (interquartile range: 1.9–7.3 years), 741 patients developed CKD. The multivariable-adjusted hazard ratio (HR) of CKD was 1.31 (95% CI 1.12–1.54, P = 0.001) for subjects with metabolic syndrome compared with those without metabolic syndrome. Relative to subjects with no other components of metabolic syndrome except for diabetes, those with two, three, four, and five metabolic syndrome components had HRs of an increased risk of CKD of 1.15 (0.83–1.60, P = 0.407) 1.32 (0.94–1.86, P = 0.112), 1.64 (1.17–2.32, P = 0.004), and 2.34 (1.54–3.54, P < 0.001), respectively. The metabolic syndrome traits of central obesity, hypertriglyceridemia, hypertension, and low BMI were independent predictors for CKD.
CONCLUSIONS—The presence of metabolic syndrome independently predicts the development of CKD in subjects with type 2 diabetes.
There is now a pandemic of diabetes and obesity in both developing and developed countries (1,2). Apart from predicting type 2 diabetes and cardiovascular and all-cause mortality (3), metabolic syndrome has also been linked to chronic kidney disease (CKD) (4,5). However, most of these latter studies were either cross-sectional in nature or conducted in general populations. Diabetes and hypertension are the main driving forces for the rising epidemic of CKD and end-stage renal disease (ESRD) (6). Given that components of metabolic syndrome such as hypertension, hyperlipidemia, and obesity are common among subjects with type 2 diabetes, the impacts of clustering of these risk factors in the form of metabolic syndrome on CKD in individuals with type 2 diabetes remain to be established.
Furthermore, there is now consistent data showing that Asian diabetic populations, including Chinese, have a higher risk of renal complications than their Caucasian counterparts (7). In a large-scale multinational survey, up to 60% of Asian patients with type 2 diabetes had albuminuria, compared with 30–40% reported in the Western population (8).
Against this background, we examined the independent risk associations of metabolic syndrome and its components with CKD in Chinese patients with type 2 diabetes, using the Hong Kong Diabetes Registry.
RESEARCH DESIGN AND METHODS
The Hong Kong Diabetes Registry was established in 1995 as part of a continuous quality-improvement program at the Prince of Wales Hospital, Hong Kong. Between 1995 and 2005, a total of 7,838 patients from community and hospital clinics, as well as patients newly discharged from the hospital, was enrolled in this Registry. Patients with type 1 diabetes (n = 334) defined as acute presentation with diabetic ketoacidosis, heavy ketonuria (>3+), or a continuous requirement of insulin within 1 year of diagnosis were excluded from this analysis. Patients with estimated glomerular filtration rate (eGFR) <60 ml/min per 1.73 m2 (n = 540) and a preexisting macrovascular complication (n = 1,135) at enrollment were not included. A macrovascular complication was defined as ischemic heart disease, cerebrovascular disease, or peripheral vascular disease. A total of 5,829 Chinese patients with type 2 diabetes were included in the final analysis. All subjects had a comprehensive assessment of risk factors and complications based on the European DiabCare protocol (9). Ethics approval was obtained from the Chinese University of Hong Kong Clinical Research Ethics Committee, and written consent was obtained from all patients for data analysis and research purposes.
Details of the clinical assessments and laboratory assays were described previously (10). In brief, sitting blood pressure was measured in both arms using a Dinamap machine after at least 5 min of resting, and the mean value was used for analysis. Waist circumference (to the nearest centimeter) was measured by a plastic tape at the narrowest level between the xiphisternum and umbilicus. Funduscopy was performed by physicians with training in diabetes or ophthalmologists through dilated pupils. Retinopathy was defined by the presence of dot and blot hemorrhages, hard exudates, cotton wool spots, neovascularization, laser scars, or a history of vitrectomy. Sensory neuropathy was diagnosed by two of these three features: reduced sensation to monofilament examination in any part of the sole with normal skin, a score of ≤6 of 8 (aged <65 years) or ≤4 of 8 (aged ≥65 years old) using a graduated tuning fork, or abnormal sensation in lower limbs.
Fasting blood samples were taken for measurement of plasma glucose, A1C, lipid profile (total cholesterol, HDL cholesterol, triglycerides, and calculated LDL cholesterol), and renal function. A sterile random spot urine sample was used to measure the albumin-to-creatinine ratio (ACR). Microalbuminuria was defined as either ACR of 2.5–30 mg/mmol in women or 3.5–30 mg/mmol in men. Macroalbuminuria was defined as ACR >30 mg/mmol. All laboratory assays were performed at the Department of Chemical Pathology, the Prince of Wales Hospital, which is accredited by the Royal College of Pathologists of Australasia. Glomerular filtration rate was calculated using the abbreviated equation developed by the Modification of Diet in Renal Disease (MDRD) study with modification for the Chinese population: eGFR = 186 × [SCR × 0.011]−1.154 × [age]−0.203 × [0.742 if female] × 1.233, where SCR is serum creatinine expressed as micromoles per liter (original milligrams per deciliter converted to micromoles per liter) and 1.233 is the adjusting coefficient for Chinese (11).
Metabolic syndrome was diagnosed according to the National Cholesterol Education Program Adult Treatment Panel III criteria with Asian modifications for waist circumferences (12). Because patients already had diabetes, metabolic syndrome was considered to be present if two or more of the following four criteria were met: waist circumference >80 cm in women and >90 cm in men, fasting plasma triglyceride ≥1.7 mmol/l, fasting HDL cholesterol <1.0 mmol/l in men and <1.3 mmol/l in women, and blood pressure ≥130/85 mmHg. Patients who were taking antihypertensive drugs including ACE inhibitors and angiotensin receptor blockers were recorded as having elevated blood pressure. Patients who were using fibrates and/or statins were recorded as fulfilling one lipid criterion.
Clinical outcomes
All clinical end points including hospital admissions and mortality were censored on 30 July 2005 using databases from the HA Central Computer System, which records admissions to all public hospitals. These databases, including the Hong Kong Death Registry, were matched by a unique identification number, the Hong Kong Identity Card number, which is compulsory for all residents in Hong Kong and used by all government departments and major organizations. Serum creatinine measurements collected within 6 months of the censored date and hospitalization discharge diagnoses were used to derive eGFR for end point definition. Hospital discharge diagnoses were coded by the ICD-9. Hospitalization with CKD event was defined as 1) hospitalization with a diagnosis of diabetes with renal manifestations (code 250.4), CKD (code 585), or unspecified renal failure (code 586) or 2) dialysis (ICD-9 procedure code 39.95) or peritoneal dialysis (ICD-9 procedure code 54.98). In this study, the end point was defined as the first eGFR <60 ml/min per 1.73 m2 or the first hospitalization with CKD event.
Statistical analysis
All data are means ± SD or median (interquartile range). For between-group comparisons, the χ2 test was used for categorical variables, and the Student's t test or ANOVA was used for continuous variables. Cox regression analysis was applied by the backward stepwise method with CKD as the dependent variable. The metabolic syndrome (or its components) and other known risk factors for CKD were included in the model. Hazard ratios (HRs) with 95% CIs were calculated. Before analysis, the skewed distribution of ACR and triglyceride was normalized by logarithmic transformation. A Kaplan-Meier curve was used to show the risk relationship between patients with and without the metabolic syndrome and patients with different components of the metabolic syndrome. P (two sided) <0.05 was considered to be significant. Statistical analysis was performed using the Statistical Package for Social Science (version 13.0 for Windows; SPSS, Chicago, IL).
RESULTS
The mean ± SD age of the cohort was 54.1 ± 13.0 years, and 45.5% were men. The mean duration of diabetes was 6.23 ± 6.17 years. The frequency of metabolic syndrome was 51.2% (n = 2,985). After a median observation period of 4.6 years (interquartile range 1.9–7.3), 741 patients (12.7%) developed CKD. Table 1 compares the baseline demographic, clinical, and biochemical characteristics of patients with and without metabolic syndrome. Patients in the former group were older and had a longer duration of diabetes and worse glycemic control. In addition, compared with the group without metabolic syndrome, those with metabolic syndrome had twice the frequency of albuminuria and had a lower eGFR at baseline.
Table 1.
Baseline clinical and biochemical characteristics of 5,829 Chinese patients with type 2 diabetes with and without metabolic syndrome as defined by National Cholesterol Education Program Adult Treatment Panel III criteria
| Metabolic syndrome |
P value | ||
|---|---|---|---|
| No | Yes | ||
| n | 2,844 | 2,985 | |
| Age (years) | 52.6 ± 13.1 | 55.6 ± 12.8 | <0.001 |
| Men (%) | 56.3 | 43.7 | <0.001 |
| Smoking status (%) | |||
| Nonsmoker | 70.2 | 73.9 | |
| Ex-smoker | 15.6 | 13.0 | |
| Current smoker | 14.2 | 13.1 | 0.003* |
| Duration of diabetes (years) | 6.0 ± 6.0 | 6.5 ± 6.3 | 0.003 |
| BMI (kg/m2 ) | 23.71 ± 3.3 | 26.5 ± 4.2 | <0.001 |
| Waist circumference (cm) | |||
| Men | 84.8 ± 8.3 | 92.4 ± 10.0 | <0.001 |
| Women | 77.8 ± 7.6 | 87.6 ± 9.7 | <0.001 |
| Systolic blood pressure (mmHg) | 127 ± 19 | 139 ± 18 | <0.001 |
| Diastolic blood pressure (mmHg) | 74 ± 10 | 78 ± 11 | <0.001 |
| A1C (%) | 7.58 ± 1.93 | 7.78 ± 1.70 | <0.001 |
| Fasting plasma glucose (mmol/l) | 8.50 ± 3.27 | 8.91 ± 3.20 | <0.001 |
| Triglycerides (mmol/l) | 1.02 (0.77–1.32) | 1.90 (1.32–2.62) | <0.001 |
| HDL cholesterol (mmol/l) | |||
| Men | 1.36 ± 0.33 | 1.06 ± 0.28 | <0.001 |
| Women | 1.56 ± 0.37 | 1.25 ± 0.31 | <0.001 |
| LDL cholesterol (mmol/l) | 3.18 ± 0.91 | 3.25 ± 0.99 | <0.001 |
| Plasma creatinine (μmol/l) | 74.0 (62.0–88.0) | 76.0 (63.0–90.0) | <0.001 |
| eGFR (ml/min per 1.73m2) | 117.8 ± 29.4 | 109.0 ± 29.2 | <0.001 |
| Urinary ACR | 1.15 (0.61–3.17) | 2.42 (085–11.52) | <0.001 |
| Albuminuria status | |||
| Normal (%) | 73.3 | 52.7 | |
| Microalbuminuria (%) | 21.0 | 32.4 | |
| Macroalbuminuria (%) | 5.7 | 14.9 | <0.001* |
| Neuropathy (%) | 21.0 | 23.1 | 0.047* |
| Retinopathy (%) | 19.7 | 23.8 | <0.001* |
Data are means ± SD or median (interquartile range) unless otherwise indicated.
Pearson χ2 test.
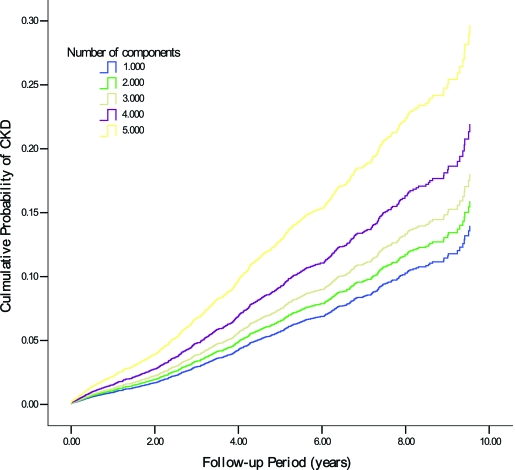
The relationship between CKD and metabolic syndrome was analyzed using Cox regression analysis, with adjustment for confounding variables including age, sex, smoking status, duration of diabetes, A1C, BMI, and albuminuria. Patients with two, three, four, and five metabolic syndrome components had sequential increases in multivariable-adjusted HRs of CKD of 1.15 (95% CI 0.83–1.60, P = 0.407), 1.32 (0.94–1.86, P = 0.112), 1.64 (1.17–2.32, P = 0.004), and 2.34 (1.54–3.54, P < 0.001), respectively (Fig. 1). The multivariable-adjusted HR of CKD for subjects with metabolic syndrome compared with that without metabolic syndrome was 1.31 (1.12–1.54, P = 0.001).
Figure 1.
Cumulative probability of new-onset CKD according to number of metabolic syndrome components.
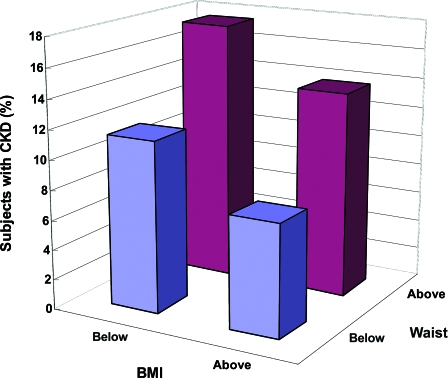
Among the components of metabolic syndrome, central obesity, hypertension, and hypertriglyceridemia, but not HDL cholesterol (HR 1.12 [95% CI 0.95–1.32]), were associated with increased risk of CKD. Other risk factors included age, male sex, disease duration, A1C, retinopathy, microalbuminuria, and macroalbuminuria, with the latter two conferring two- and fivefold increased risk of CKD, respectively (Table 2). Paradoxically, low BMI was also associated with increased risk of CKD. Figure 2 shows the risk association of CKD in these patients stratified by median values of waist and BMI. Compared with patients with the highest BMI and lowest waist circumference, those with the highest waist circumference and lowest BMI had the highest HR of CKD.
Table 2.
Cox regression analysis showing the association of CKD with individual metabolic syndrome traits and covariables in Chinese patients with type 2 diabetes
| Independent variables | HR (95% CI) | P value |
|---|---|---|
| Male sex | 1.30 (1.10–1.53) | 0.002 |
| Age per year | 1.04 (1.04–1.05) | <0.001 |
| Duration of diabetes (year) | 1.02 (1.01–1.03) | <0.001 |
| A1C | 1.11 (1.07–1.16) | <0.001 |
| Microalbuminuria | 2.16 (1.77–2.63) | <0.001 |
| Macroalbuminuria | 5.82 (4.75–7.14) | <0.001 |
| BMI | 0.97 (0.94–0.99) | 0.008 |
| Retinopathy | 1.79 (1.52–2.10) | <0.001 |
| Metabolic syndrome traits | ||
| Hypertension | 1.37 (1.09–1.72) | 0.007 |
| Central obesity | 1.58 (1.26–1.98) | <0.001 |
| Hypertriglyceridemia | 1.24 (1.06–1.44) | 0.006 |
Figure 2.
Percentage of patients with new-onset CKD stratified by medians of waist circumference and BMI.
CONCLUSIONS
In this prospective analysis of a relatively large cohort of patients with type 2 diabetes, we confirmed the risk association between metabolic syndrome and CKD independent of conventional risk factors including age, male sex, glycemic control, disease duration, and albuminuria. This finding is strengthened by the stepwise increase in HR for CKD with an increasing number of metabolic syndrome components. Among the latter, central obesity, hypertriglyceridemia, and hypertension were the three main metabolic syndrome traits related to CKD in this cohort. These three factors were also found to predict all-cause and cardiovascular mortality in a community-based Hong Kong Chinese cohort (13).
Of note, we observed the additive effects of high waist circumference and low BMI on risk of CKD independent of metabolic control. There is now general consensus that waist circumference as a surrogate of visceral adiposity (14) is more strongly correlated with adverse metabolic risks including high blood pressure, dyslipidemia, and increased insulin resistance (15). Lamacchia et al. (16) observed recently in subjects with type 2 diabetes that waist circumference but not BMI was associated with an increased renal resistive index, a parameter that reflects intrarenal arteriosclerosis and glomerulosclerosis. Increased adipose tissue, especially that of visceral origin, is a rich source of inflammatory cytokines and growth factors including leptin, tumor necrosis factor-α, and interleukin-6 (17). In a separate cohort of Chinese patients with type 2 diabetes with nephropathy, we reported aberrant activation and expression of inflammatory cytokines or adhesion molecules, which can have direct effects on renal hemodynamics and glomerular cellularity (18). Although low BMI may reflect lipolysis and muscle wasting due to hyperglycemia and insufficient insulin action, the risk conferred by low BMI on CKD in our cohort was independent of A1C. In the Japanese population, obesity is an independent predictor for end-stage renal disease in the general population (19), although low BMI and increased waist circumference are independent predictors for diabetes (20). Thus, with the onset of diabetes, the effect of body composition and/or distribution may take on different prognostic significance.
In this cohort, hypertriglyceridemia but not a low HDL cholesterol level was predictive of CKD development. In keeping with the clinical observations on lipotoxicity and renal damage, there are experimental studies showing the stimulatory effects of lipids on mesangial cell proliferation and matrix deposition, in addition to recruitment of inflammatory cells and synthesis of inflammatory cytokines (21). In a recent meta-analysis of 40,000 individuals, the use of hydroxymethylglutaryl-CoA inhibitors attenuated the rate of progression of renal impairment and reduced proteinuria (22). In a post hoc analysis of a clinical study comparing the effects of fenofibrate and placebo treatment on progression of coronary atherosclerosis, fenofibrate was associated with a greater reduction in albuminuria in patients with type 2 diabetes (23).
Metabolic syndrome is now known to be associated with development of cardiovascular diseases (3). In our current study of the Chinese diabetic population, we observed that metabolic syndrome increased the risk of CKD. Moreover, CKD per se is an independent determinant of atherosclerotic diseases and the two conditions often coexist. In a separate analyses of this cohort, we have demonstrated that albuminuria (24) and estimated GFR (25) were strongly predictive of cardiovascular outcome. We proposed that the same metabolic risk factors are operative in the progression of both conditions. In this connection, increased oxidative stress, chronic inflammation, increased fibrogenic activity, and endothelial dysfunction are common underlying pathological events.
Our study has several limitations. First, baseline measurements of serum creatinine were based on a single collection and may not fully reflect the background renal function, especially in the presence of intercurrent illness. At baseline, all clinical and biochemical assessments were performed in an outpatient setting when the patient was clinically stable. Second, although diabetes may be the main culprit for the new onset of CKD in this prospective cohort, other causes such as ischemic nephrosclerosis, obstructive uropathy, and glomerulonephritis may also contribute to deterioration of renal function. Third, some investigators have suggested overestimation of the prevalence of stage 4 to 5 CKD using the original MDRD equations. However, the current modified MDRD formula has been validated in Chinese populations (11).
Despite these limitations, the relatively large sample size of the present cohort with detailed phenotyping and a relatively long observational period, averaging 5 years, is an important strength of our study to support the causal relationship between metabolic factors and renal dysfunction. In summary, the presence of metabolic syndrome is associated with an increased risk of development of CKD. Our study has expanded the current body of knowledge on the possible causes of diabetic nephropathy and highlights metabolic syndrome as a major problem for which individuals with diabetes who are at high risk of cardiorenal complications need treatment.
Acknowledgments
This project was partially supported by the Li Ka Shing Institute of Health Sciences and the Hong Kong Foundation for Research and Development in Diabetes of the Chinese University of Hong Kong. R.C.W.M. is the recipient of a Croucher Foundation Fellowship.
We thank all medical and nursing staff at the Diabetes Mellitus and Endocrine Centre at the Prince of Wales Hospital for their dedication and commitment in implementing structured patient care.
Published ahead of print at http://care.diabetesjournals.org on 3 October 2008.
The costs of publication of this article were defrayed in part by the payment of page charges. This article must therefore be hereby marked “advertisement” in accordance with 18 U.S.C Section 1734 solely to indicate this fact.
References
- 1.Yoon KH, Lee JH, Kim JW, et al.: Epidemic obesity and type 2 diabetes in Asia. Lancet 368:1681–1688, 2006 [DOI] [PubMed] [Google Scholar]
- 2.Zimmet P, Alberti KG, Shaw J: Global and societal implications of the diabetes epidemic. Nature 414:782–787, 2001 [DOI] [PubMed] [Google Scholar]
- 3.Ford ES: Risks for all-cause mortality, cardiovascular disease, and diabetes associated with the metabolic syndrome: a summary of the evidence. Diabetes Care 28:1769–1778, 2005 [DOI] [PubMed] [Google Scholar]
- 4.Chen J, Muntner P, Hamm LL, et al.: The metabolic syndrome and chronic kidney disease in US adults. Ann Intern Med 140:167–174, 2004 [DOI] [PubMed] [Google Scholar]
- 5.Costa LA, Canani LH, Lisboa HR, et al.: Aggregation of features of the metabolic syndrome is associated with increased prevalence of chronic complications in type 2 diabetes. Diabet Med 21:252–255, 2004 [DOI] [PubMed] [Google Scholar]
- 6.Cooper M: Pathogenesis, prevention and treatment of diabetic nephropathy. Lancet 352:213–219, 1998 [DOI] [PubMed] [Google Scholar]
- 7.Karter A, Ferrara A, Liu J, et al.: Ethnic disparities in diabetic complications in an insured population. JAMA 287:2519–2527, 2002 [DOI] [PubMed] [Google Scholar]
- 8.Wu AY, Kong NC, de Leon FA, et al.: An alarmingly high prevalence of diabetic nephropathy in Asian type 2 diabetic patients: the MicroAlbuminuria Prevalence (MAP) Study. Diabetologia 48:1674–1675, 2005 [DOI] [PubMed]
- 9.Piwernetz K, Home PD, Snorgaard O, et al.: monitoring the targets of the St. Vincent Declaration and the implementation of quality management in diabetes care: the DIABCARE initiative. Diabet Med 10:371–377, 1993 [DOI] [PubMed] [Google Scholar]
- 10.Tong PC, Kong AP, So WY, et al.: The usefulness of the International Diabetes Federation and the National Cholesterol Education Program's Adult Treatment Panel III Definitions of the metabolic syndrome in predicting coronary heart disease in subjects with type 2 diabetes. Diabetes Care 30:1206–1211, 2007 [DOI] [PubMed] [Google Scholar]
- 11.Ma YC, Zuo J, Chen JH, et al.: Modified glomerular filtration rate estimating equation for Chinese patients with chronic kidney disease. J Am Soc Nephrol 17:2937–2944, 2006 [DOI] [PubMed] [Google Scholar]
- 12.Grundy SM, Cleeman JI, Daniels SR, et al.: American Heart Association, National Heart, Lung and Blood Institute: diagnosis and management of the metabolic syndrome: an American Heart Association/National Heart, Lung, and Blood Institute Scientific Statement. Circulation 112:2735–2752, 2005 [DOI] [PubMed] [Google Scholar]
- 13.Thomas GN, Schooling CM, McGhee SM, et al.: Metabolic syndrome increases all-cause and vascular mortality: the Hong Kong Cardiovascular Risk Factor Study. Clin Endocrinol 66:666–671, 2007 [DOI] [PubMed] [Google Scholar]
- 14.Janssen I, Heymsfield SB, Allison DB, et al.: Body mass index and waist circumference independently contribute to the prediction of nonabdominal, abdominal subcutaneous and visceral fat. Am J Clin Nutr 75:683–688, 2002 [DOI] [PubMed] [Google Scholar]
- 15.Janssen I, Katzmarzyk PT, Ross R: Waist circumference and not body mass index explains obesity-related health risk. Am J Clin Nutr 79:379–384, 2004 [DOI] [PubMed] [Google Scholar]
- 16.Lamacchia O, Nicastro V, Camarchio D, et al.: Waist circumference is strongly associated with renal resistive index in normoalbuminuria patients with type 2 diabetes. Am J Nephrol 28:54–58, 2008 [DOI] [PubMed] [Google Scholar]
- 17.Wisse BE: The inflammatory syndrome: the role of adipose tissue cytokines in metabolic disorders linked to obesity. J Am Soc Nephrol 15:2792–2800, 2004 [DOI] [PubMed] [Google Scholar]
- 18.Wong CK, Ho AW, Tong PC, et al.: Aberrant expression of soluble co-stimulatory molecules and adhesion molecules in type 2 diabetic patients with nephropathy. J Clin Immunol 28:36–43, 2007 [DOI] [PubMed] [Google Scholar]
- 19.Iseki K, Ikemiya Y, Kinjo K, et al.: Body mass index and the risk of development of end-stage renal disease in a screened cohort. Kidney Int 54:1870–1876, 2004 [DOI] [PubMed] [Google Scholar]
- 20.Boyko EJ, Fujumoto WY, Leonetti DL, et al.: Visceral adiposity and risk of type 2 diabetes: a prospective study among Japanese Americans. Diabetes Care 23:465–471, 2000 [DOI] [PubMed] [Google Scholar]
- 21.Nishida Y, Oda H, Yorioka N: Effect of lipoproteins on mesangial cell proliferation. Kidney Int 71 (Suppl.):S51–S53, 1999 [DOI] [PubMed] [Google Scholar]
- 22.Sandhu S, Wiebe N, Fried LF, et al.: Statins for improving renal outcomes: a meta-analysis. J Am Soc Nephrol 17:2006–2016, 2006 [DOI] [PubMed] [Google Scholar]
- 23.Ansquer J, Foucher C, Rattier S, et al.: Fenofibrate reduces progression to microalbuminuria over 3 years in a placebo-controlled study in type 2 diabetes: results from the Diabetes Atherosclerosis Intervention Study (DAIS). Am J Kidney Dis 45:485–493, 2005 [DOI] [PubMed] [Google Scholar]
- 24.Tong PC, Kong AP, So WY, et al.: Interactive effect of retinopathy and macroalbuminuria on all-cause mortality, cardiovascular and renal end points in Chinese patients with type 2 diabetes mellitus. Diabet Med 24:741–746, 2007 [DOI] [PubMed] [Google Scholar]
- 25.So WY, Kong AP, Ma RC, et al.: Glomerular filtration rate, cardiorenal end points, and all-cause mortality in type 2 diabetic patients. Diabetes Care 29:2046–2052, 2006 [DOI] [PubMed] [Google Scholar]