Abstract
The workup of low back pain in children often results in overimaging so as not to miss organic back pain. The primary goal of this study was to identify which combination of imaging modalities provides the most sensitive and specific screening protocol for children with low back pain. Medical records from 100 consecutive patients between 2 and 18 years of age presenting with low back pain, without night pain or constitutional symptoms, were evaluated. A hyperextension test combined with a radiograph showed a negative predictive value of 0.81 and sensitivity of 0.90. The addition of a bone scan was highly effective in achieving good negative predictive value and sensitivity. Bone scans had perfect negative predictive value and sensitivity when symptom duration was less than 6 weeks. We identified a set of factors that is highly predictive for distinguishing organic back pain from mechanical back pain. Painless hyperextension combined with negative anteroposterior, lateral, and oblique lumbar radiographs and magnetic resonance images predicts mechanical back pain. For patients with nonneurologic back pain of less than 6 weeks duration, bone scan is the most useful screening test because it is accurate, accessible, inexpensive, and unlikely to require sedation.
Level of Evidence: Level III, diagnostic study. See the Guidelines for Authors for a complete description of levels of evidence.
Introduction
Numerous studies suggest children with low back pain may have serious spinal disease, including tumors and infections [14–16, 30]. These studies provided evidence for potentially worrisome sources of low back pain in children and, consequently, provide an argument for the extensive diagnostic workup of pediatric low back pain still commonly practiced today.
However, epidemiologic evidence has indicated pediatric low back pain may be much more prevalent than previously perceived [1, 9, 20, 26]. Among juvenile and adolescent athletes, back pain is common [4] with one study reporting a 79% rate of back pain compared with 38% in age-matched control subjects [28]. Despite the absence of worrisome hallmarks (eg, neurologic or constitutional symptoms), children often undergo extensive, costly, and time-consuming diagnostic workups that ultimately reveal no treatable organic disorder [7, 8, 29]. Such children are said to have mechanical back pain (MBP) [4, 9, 11].
When a thorough history and physical examination are not sufficient to reliably rule out an organic etiology of back pain (OBP), imaging is performed. Although multiple studies have examined the diagnostic accuracy of MRI, single photon emission computed tomography (SPECT), and plain film radiography in diagnosing specific conditions [2, 3, 5, 6, 10, 11, 19, 22–25, 31, 32], there is no evidence-based consensus regarding which of these tests should be used in evaluating low back pain [6, 22, 32]. Furthermore, no study to our knowledge has developed an approach that can distinguish children who have OBP (likely to require additional followup and treatment) from those with MBP (not likely to require additional followup and treatment). Such guidelines would provide physicians with a useful clinical tool that could streamline the workup of low back pain in children.
The purpose of this study was to analyze the value of information gained from clinical examination and imaging studies (plain radiography, MRI, and SPECT) in reaching a diagnosis in children who present with isolated low back pain. Our tangible goals from this analysis included (1) identification of the imaging modality or combinations of modalities best able to rule out a serious diagnosis (negative predictive value) and least likely to miss a serious diagnosis (sensitivity); (2) development of guidelines based on this identification to streamline evaluation (reduce imaging, cost, time, radiation, and sedation); and (3) contributing to a framework on which an evidence-based approach to evaluating back pain in children can be developed.
Materials and Methods
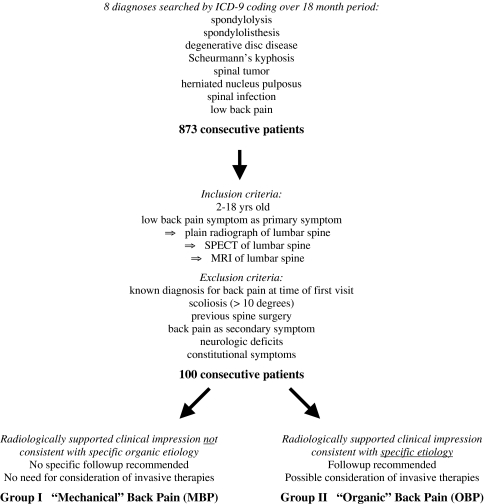
After obtaining Institutional Review Board approval, we performed a retrospective review of all children who presented (either primarily or as a consultation) to our pediatric orthopaedic surgery clinic with back pain between January 1, 2003, and June 1, 2004. Initial patient screening used computerized billing records and International Classification of Diseases, 9th Revision codes specific for any diagnosis that would identify patients with low back pain (Fig. 1). Patients were deemed to have low back pain (as opposed to midthoracic or upper back pain) if the patient vocalized and/or pointed to the low back, or, in cases in which the patient was not able to clearly vocalize the location of the pain, if the patient preferentially rubbed the lower lumbar region. We identified 873 children who presented with back pain during a period of 18 months. We evaluated the total number of bone scans and MRI scans performed at our institution during the same study period (2003–2004). There were 98 bone scans performed, 63 of which (64%) were isolated studies of the spine and 36 (37%) of which were of the lumbar spine. There were 1516 MRI examinations of the spine performed in 1394 patients during the same period. One hundred consecutive patients who met study criteria (Fig. 1) represented our working patient data set. Patients were excluded if they had any known previous diagnosis responsible for their current back pain, scoliosis (greater than 10° coronal plane curvature), neurologic symptoms (numbness, weakness, paresthesias, or leg pain radiating below the knee), night pain, or constitutional symptoms (weight loss, fevers, chills, rigors).
Fig. 1.
This diagram depicts the selection process of the final study population. ICD-9 = International Classification of Diseases, 9th Revision; SPECT = single photon emission computed tomography.
The study population of 100 included 38 boys and 62 girls with a mean age of 13.5 years (SD, 3.3; range, 3–17 years). The average duration of back pain before presentation was 42 weeks (SD, 48; range, 1–260 weeks). For each of the 100 patients, the following historical data were obtained: age at presentation, gender, duration of pain, location of pain (midline, paraspinal), description of pain (constant, intermittent, radiating, buttock), history of preceding traumatic event, smoking history, and sports participation. On physical examination, data collected included tenderness to palpation, pain with hyperextension, pain with forward bending, hamstring tightness, gait abnormalities, and results from straight leg raise testing. Radiologic data included the timing of the imaging study in relation to the duration and onset of symptoms and the study findings. Technical data regarding MRI studies (eg, Tesla strength, sectioning distance) typically were not available and, therefore, not collected or analyzed. Because children often need sedation (ages 0–4 years for SPECT and 0–10 years for MRI at our institution), we also assessed the potential for reduction in sedation procedures. The number of sedation procedures was obtained from each patient’s medical record. All clinical and radiographic results were acquired by four observers (JDA, JA, MHZ, SCR). The clinical and radiographic chart review was performed at the same time and thus the observers were not blinded to the results.
Group I (MBP) was defined as back pain without clinical or radiographic evidence (negative radiograph, SPECT, and MRI) of a specific organic etiology. Group II (OBP) was defined by back pain with clinical and radiographic evidence suggesting a specific organic etiology (Fig. 1).
The sensitivity (Sens) and negative predictive value (NPV) of chosen factors (history, physical examination, positive imaging as defined previously) were calculated. These factors were analyzed for their use in statistically distinguishing between Group I and Group II by univariate analysis (chi square). No factor from the patient history or physical examination met criteria (p < 0.10) for inclusion in constructing a multivariate logistic regression model. A statistician performed chi square, Fisher’s exact, and Mann Whitney U tests using SAS v9.1 software (Cary, NC) for all analyses shown. Average values with standard deviations (SDs) and ranges are given when appropriate. Statistical significance was set at p < 0.05. A power analysis was not performed because one of our study goals was to stringently define the differences that would provide clinical use in achieving a diagnosis.
Results
None of the elements of patient demographics, history, or physical examination were different between Groups I and II (Table 1) and were not useful as distinguishing factors between the two groups. Fifty-seven children had negative radiographs, SPECT, and MRI and no identified organic diagnosis; they were assigned to Group I, MBP (Fig. 1). The diagnoses that led to assignment of patients in Group II, OBP (Fig. 1), were spondylolysis (21% [21 of 100]), spondylolisthesis (6%), herniated nucleus pulposus (4%), and other rare causes (12%, spina bifida occulta, Bertolotti’s syndrome, lumbar Scheuermann’s, pathologic compression fracture, osteomyelitis, L4 pedicle stress reaction, large Schmorl’s node). The examination findings common to both groups included tenderness to palpation of the lower back, hyperextension pain of the lumbar spine, and hamstring tightness (Table 1).
Table 1.
Comparison of patient characteristics
| Characteristic | Group I | Group II | Test | p Value |
|---|---|---|---|---|
| Age (years ± SD) | 13 ± 3.5 | 14 ± 2.9 | t test | NS |
| Female (%) | 61 | 63 | Chi square | NS |
| Comorbidity (%) | 9 | 14 | Fisher’s exact | NS |
| Pain duration (weeks ± SD) | 42 ± 46 | 43 ± 51 | t test | NS |
| Tender to palpation (%) | 60 | 57 | Chi square | NS |
| Hyperextension pain (%) | 67 | 83 | Chi square | NS |
| Hamstring tightness (%) | 33 | 42 | Chi square | NS |
| + SLR (%) | 4 | 6 | Fisher’s exact | NS |
| Number of patients | 53 | 47 |
MBP = mechanical back pain; OBP = organic back pain; SD = standard deviation; t test = two-tailed Student’s t-test; SLR = straight leg raise; NS = not statistically significant at p > 0.05.
Evaluation of the use of imaging alone in distinguishing between Groups I and II revealed that all three modalities (radiography, SPECT, MRI) were able to discriminate between the two groups (p < 0.001). Magnetic resonance imaging was the most useful with an overall NPV (predicting MBP) of 0.79 compared with 0.75 for SPECT and 0.68 for radiographs (Table 2). The overall sensitivity (identification of OBP) of MRI alone in detecting patients with Group II diagnoses was 0.65, which was superior to SPECT (0.56) and radiographs (0.38).
Table 2.
Radiologic studies
| Imaging | Group I NPV | Group II Sens | p Value |
|---|---|---|---|
| Radiography | 0.68 | 0.38 | < 0.001 |
| SPECT | 0.75 | 0.56 | < 0.001 |
| MRI | 0.79 | 0.65 | < 0.001 |
| Imaging + hamstring | |||
| Radiography and SPECT | 0.87 | 0.90 | < 0.001 |
| Radiography and MRI | 0.90 | 0.90 | < 0.001 |
| Imaging + hyperextension | |||
| Radiography and SPECT | 0.98 | 0.94 | < 0.001 |
| Radiography and MRI | 1.0 | 1.0 | < 0.001 |
SPECT = single photon emission computed tomography; MRI = magnetic resonance imaging; MBP = mechanical back pain; OBP = organic back pain; NPV = negative predictive value; Sens = sensitivity.
All combinations of anteroposterior, lateral, and oblique plain film radiography (routinely the first imaging obtained in evaluation) with either SPECT or MRI showed the ability to distinguish between Groups I and II (p < 0.001) (Table 2) with high NPV and Sens values (Table 2). The most useful combination of imaging and physical examination findings was MRI in conjunction with radiography and hyperextension testing, NPV and Sens of 1.0. Combining any imaging modality alone with hyperextension pain or hamstring tightness resulted in lower NPV and Sens values (data not shown).
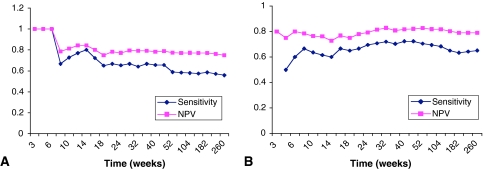
A stepwise analysis showed a significant association between SPECT use (NPV and Sens) and time less than or equal to 6 weeks (p < 0.001). A SPECT scan performed within the first 6 weeks of symptom duration (n = 6), regardless of physical examination findings, had a NPV and Sens of 1.0. After 6 weeks, there was a significant dropoff in both values (Fig. 2A). To validate this finding in a small population, we rescreened our initial population of 873 for all patients with a SPECT scan. Among 220 such patients, we again found a NPV and Sens of 1.0 (n = 29). In contrast, the use of MRI scans did not show time dependence (p > 0.05) (Fig. 2B).
Fig. 2A–B.
These figures show the time-dependent value of (A) SPECT and (B) MRI and in the diagnostic workup of back pain in children. Negative predictive values (NPVs) and sensitivities (Y-axis) are shown as a function of symptom duration (X-axis) for (A) SPECT and (B) MRI studies.
No child who received a bone scan required sedation (zero of 63). In contrast, 685 (49.1%) of the 1394 patients (1516 MRIs) who had MRI at our institution during the same period required sedation.
Discussion
Traditionally, when clinical findings alone cannot sufficiently rule out organic causes of back pain in children, plain film radiography of the lumbar spine (anteroposterior, lateral, and oblique views) is the most common first diagnostic test [12, 14, 16, 22, 23, 27]. With persistent pain and a negative radiograph, however, the clinician then must choose which test may be needed to rule out suspected disorders. Traditionally, a SPECT/bone scan would be the next test of choice [2, 3, 10–12, 19, 22, 24]. With improved techniques, however, MRI has been proffered by numerous authors as a potential first-line test to rule out suspected disorders such as spondylolysis [5, 6, 31, 32]. The objective of the current study was to determine the optimal imaging modality, or combination of imaging modalities, that can rule out a serious diagnosis (NPV) and be least likely to miss a serious diagnosis (Sens).
This study has several limitations. First, our study was retrospective and, therefore, is reflective of only our study population. Only a prospective cohort study in which every patient obtains an MRI and SPECT scan can truly abrogate the potentially considerable selection bias in a retrospective diagnostic study. Such a study may not be feasible or ethical. Second, we did not have a true gold standard for diagnosis nor did we have potential for one; MBP is an overall clinical diagnosis and often one of exclusion. Third, our population represents a potentially skewed referral population and may not reflect the average child visiting their local pediatrician. Although this may be true, many of these patients came to us with multiple studies already in hand in which, on retrospective review, one study provided a correct and clear diagnosis. Another potential limitation is that we chose to analyze all ages of children together as a single group, similar to the methods used by Feldman et al., and not substratified according to age [11]. Current research efforts in our group, however, focus on application of our findings and algorithm in the adolescent population, the most common group we encountered. Finally, relying on even the best study does not ensure a diagnosis. Therefore, all guidelines have to be used in context; if the best study fails, then the next appropriate step should be taken based on the clinical situation.
Because there is a high cumulative prevalence of pediatric back pain and close to one-tenth of all pediatric patients with this condition seek medical attention [4, 9, 20, 26], traditional comprehensive back pain workups, without clear published guidelines, have great potential for inefficiency and waste. The potential for unnecessary or redundant studies increases because the majority of these children have MBP without an identifiable organic disorder [11]. Our study selection process (Fig. 1) revealed at least 11.5% (100 of 873) of patients may have been overimaged.
With the goals of reducing redundancy in imaging, we reviewed our experience with uncomplicated pediatric back pain during an 18-month period in patients who had radiographs, SPECT, and MRI performed as part of a comprehensive imaging workup for back pain. We limited our study group to patients with isolated back pain without a prior known etiology and excluded patients with neurologic or constitutional symptoms because these patients represent a different patient population than that addressed by our study hypotheses [11, 14–16, 30]. By requiring all three imaging studies (radiography, SPECT, MRI) to be negative for a diagnosis of MBP, and without an accepted gold standard, we also made it less likely that an organic diagnosis would be missed. For analysis, we divided our population into two clinically relevant groups; Group I patients typically do not require close followup or consideration of interventional therapy, whereas Group II patients typically do require specific followup and consideration of interventional therapies as treatment options.
With these patient selection characteristics, it is notable that we found a high prevalence of MBP (53%) among our population. Our data indicated history and physical findings alone had limited use in obtaining a diagnosis (data not shown) (Table 1). Pain with hyperextension, the most useful of all the examination findings, had a Sens of 0.83 in identifying an OBP diagnosis, a value of 0.71 in predicting MBP, and did not have a significant association with either group (p > 0.05). Evaluation of imaging studies alone revealed overall, MRI is the most useful in making an organic diagnosis and predicting MBP (Table 2). Spondylolysis, our most common diagnosis in Group II, however, was detected by SPECT in 71 cases compared with only 57 with MRI and 38 with radiography.
Surprisingly, we found negative SPECT was 100% predictive of MBP for patients with less than 6 weeks of symptoms regardless of examination findings. After 6 weeks, the test was considerably less predictive of MBP. This finding may be partially explained by the ability of SPECT to detect high rates of bone turnover as would be seen in acute spondylolysis or spondylolisthesis, the two most common diagnoses in our series. The literature suggests in a chronic pars fracture or nonunion in which there is little to no bone turnover or healing processes occurring, SPECT is less capable of detecting any abnormality [10, 13, 21, 25]. However, we cannot know how this predictability would be affected (if at all) if the proportion of diagnoses other than pars disorders was much higher. Nonetheless, from these results, we can conclude that a SPECT scan is a good diagnostic option for early presentation of low back pain in children, especially if the physical examination is not reliable.
Our findings also show a negative combination of MRI, radiography, and absent hyperextension pain had a NPV of 1.0 regardless of symptom duration. A positive combination resulted in a Sens of 1.0. This finding corroborates those of other studies, which have offered MRI as a useful tool for early detection of spine disorders [5, 6, 31]. The dependence of the result on the hyperextension test may be a reflection of its use in helping to discern the presence or absence of a pars defect. Nonetheless, the predictive use of MRI in distinguishing between MBP and organic causes of back pain not routinely detected on SPECT (eg, degenerative disc disease, herniated nucleus pulposus, spinal tumor, and possibly infection) offer further support for its consideration as a first-line modality. Considerably increased costs compared with SPECT, however, must be considered.
The decision to pursue an imaging test or a particular guideline is multifactorial. One important factor in decision-making is cost-effectiveness. Multiple reports have highlighted the astronomical increases in Medicare spending for imaging services; spending for MRI and positron emission tomography scans have increased 25.8% and 12.6%, respectively, from the previous year for the last 2 years [7, 8, 29]. Increases in the volume and complexity of imaging services were twice as high as that for all other Medicare services between 1999 and 2002 [29]. Overall, Medicare spending on imaging services is approaching $100 billion per year [7]. Clearly, the benefits from improved technology and imaging services must be weighed against the economic cost to society and skyrocketing national healthcare bill.
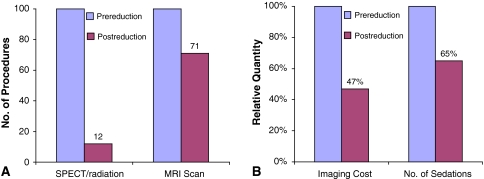
Using the two most reliable studies only (NPV 1.0 and Sens 1.0) before 6 weeks’ pain duration, SPECT, and more than 6 weeks, MRI, we could have reduced the number of SPECT scans (and exposure to unnecessary radiation) by 88% and MRI scans by 29% (Fig. 3A) and not have missed a single diagnosis. At our institution in 2006, that would translate to a savings of $195,782 ($1511 for SPECT and $2166 for MRI) applied over 100 patients evaluated by both imaging modalities (or a 47% reduction in cost) (Fig. 3B).
Fig. 3A–B.
The graphs show the potential reduction of imaging, cost, and sedation (among 100 patients evaluated) that would have been achieved using SPECT during the first 6 weeks of symptom duration and MRI after 6 weeks of symptoms. Relative reduction in (A) imaging numbers and (B) costs and sedation numbers are shown. The blue bars represent the actual total in our study population and maroon bars represent the potential total had our study results been used as guidelines (SPECT scan for symptom duration within 6 weeks and MRI for longer duration).
Considerable adverse events in the hospital setting and after discharge home have been associated with sedation and should not be taken lightly when ordering imaging studies. There are published reports showing intraprocedural and postprocedural complications of sedation in children [17, 18]. Sedation for SPECT was much less likely than for MRI at our institution during the same period as our study, and applying our 6-week bone scan criteria would have resulted in a 35% reduction in the actual number of sedations (Fig. 3B) and an additional cost savings of $27,020 ($772 per sedation performed). Ultimately, the decision to order one test or another, or both, will depend on not only the clinical efficacy of the studies, but also the clinical/parental urgency; SPECT scans usually can be obtained up to five times faster at our institution; aversion to radiation (none for MRI) or sedation (less likely for SPECT); and other situational factors. A conscious decision to systematically use available data and evidence in the decision-making process will have implications for the health of a patient, family, and society at large.
Limitations notwithstanding, we offer the following conclusions and recommendations. In our study, the most cost-effective and useful first-line imaging modality for evaluation of low back pain in children (best able to differentiate between MBP and OBP and make a diagnosis) presenting with pain for less than 6 weeks is a SPECT scan. In children presenting after 6 weeks, MRI seems to be superior but dependent on the hyperextension test. Ultimately, the decision to pursue a particular study or an additional study should be contextual and include diagnostic accuracy, urgency, and potential side effects or complications. The inclusion of evidence involving cost-effectiveness and relative efficacy between diagnostic studies should improve the overall efficiency of making the correct diagnoses in the evaluation of low back pain in children.
Footnotes
Each author certifies that he or she has no commercial associations (eg, consultancies, stock ownership, equity interest, patent/licensing arrangements, etc) that might pose a conflict of interest in connection with the submitted article.
Each author certifies that his or her institution has approved the human protocol for this investigation, that all investigations were conducted in conformity with ethical principles of research, and that informed consent was obtained.
References
- 1.Balague F, Dutoit G, Waldburger M. Low back pain in schoolchildren: an epidemiological study. Scand J Rehabil Med. 1988;20:175–179. [PubMed]
- 2.Bellah RD, Summerville DA, Treves ST, Micheli LJ. Low-back pain in adolescent athletes: detection of stress injury to the pars interarticularis with SPECT. Radiology. 1991;180:509–512. [DOI] [PubMed]
- 3.Bodner RJ, Heyman S, Drummond DS, Gregg JR. The use of single photon emission computed tomography (SPECT) in the diagnosis of low-back pain in young patients. Spine. 1988;13:1155–1160. [DOI] [PubMed]
- 4.Bono CM. Low-back pain in athletes. J Bone Joint Surg Am. 2004;86:382–396. [DOI] [PubMed]
- 5.Campbell RS, Grainger AJ, Hide IG, Papastefanou S, Greenough CG. Juvenile spondylolysis: a comparative analysis of CT, SPECT and MRI. Skeletal Radiol. 2005;34:63–73. [DOI] [PubMed]
- 6.Cohen E, Stuecker RD. Magnetic resonance imaging in diagnosis and follow-up of impending spondylolysis in children and adolescents: early treatment may prevent pars defects. J Pediatr Orthop B. 2005;14:63–67. [DOI] [PubMed]
- 7.Colliver V. Curbing costs of medical scans: insurers seek to rein in fast-growing use of pricey high-tech MRIs and CTs. San Francisco Chronicle. 2005:E1.
- 8.Costs of inpatient drugs rise rapidly than other hospital services. Available at: www.solucient.com/news_press/news20040817.shtml. Accessed August 20, 2004.
- 9.Duggleby T, Kumar S. Epidemiology of juvenile low back pain: a review. Disabil Rehabil. 1997;19:505–512. [DOI] [PubMed]
- 10.Dutton JA, Hughes SP, Peters AM. SPECT in the management of patients with back pain and spondylolysis. Clin Nucl Med. 2000;25:93–96. [DOI] [PubMed]
- 11.Feldman DS, Hedden DM, Wright JG. The use of bone scan to investigate back pain in children and adolescents. J Pediatr Orthop. 2000;20:790–795. [DOI] [PubMed]
- 12.Ginsburg GM, Bassett GS. Back pain in children and adolescents: evaluation and differential diagnosis. J Am Acad Orthop Surg. 1997;5:67–78. [DOI] [PubMed]
- 13.Harvey CJ, Richenberg JL, Saifuddin A, Wolman RL. The radiological investigation of lumbar spondylolysis. Clin Radiol. 1998;53:723–728. [DOI] [PubMed]
- 14.Hensinger RN. Back pain in children. In: Bradford DS, Hensinger RN, eds. The Pediatric Spine. New York, NY: Thieme Inc; 1985:41–60.
- 15.King HA. Back pain in children. Pediatr Clin North Am. 1984;31:1083–1095. [DOI] [PubMed]
- 16.King HA. Back pain in children. In: Weinstein SL, ed. The Pediatric Spine: Principles, Practice. Philadelphia, PA: Lippincott Williams & Wilkins; 2001:123–132.
- 17.Malviya S, Voepel-Lewis T, Prochaska G, Tait AR. Prolonged recovery and delayed side effects of sedation for diagnostic imaging studies in children. Pediatrics. 2000;105:E42. [DOI] [PubMed]
- 18.Malviya S, Voepel-Lewis T, Tait AR. Adverse events and risk factors associated with the sedation of children by nonanesthesiologists. Anesth Analg. 1997;85:1207–1213. [DOI] [PubMed]
- 19.Mandell GA. Nuclear medicine in pediatric musculoskeletal imaging. Semin Musculoskelet Radiol. 1999;3:289–316. [DOI] [PubMed]
- 20.Olsen TL, Anderson RL, Dearwater SR, Kriska AM, Cauley JA, Aaron DJ, LaPorte RE. The epidemiology of low back pain in an adolescent population. Am J Public Health. 1992;82:606–608. [DOI] [PMC free article] [PubMed]
- 21.Papanicolaou N, Wilkinson RH, Emans JB, Treves S, Micheli LJ. Bone scintigraphy and radiography in young athletes with low back pain. AJR Am J Roentgenol. 1985;145:1039–1044. [DOI] [PubMed]
- 22.Ralston S, Weir M. Suspecting lumbar spondylolysis in adolescent low back pain. Clin Pediatr (Phila). 1998;37:287–293. [DOI] [PubMed]
- 23.Roberts FF, Kishore PR, Cunningham ME. Routine oblique radiography of the pediatric lumbar spine: is it necessary? AJR Am J Roentgenol. 1978;131:297–298. [DOI] [PubMed]
- 24.Ryan PJ, Evans PA, Gibson T, Fogelman I. Chronic low back pain: comparison of bone SPECT with radiography and CT. Radiology. 1992;182:849–854. [DOI] [PubMed]
- 25.Saifuddin A, Burnett SJ. The value of lumbar spine MRI in the assessment of the pars interarticularis. Clin Radiol. 1997;52:666–671. [DOI] [PubMed]
- 26.Salminen JJ, Pentti J, Terho P. Low back pain and disability in 14-year-old schoolchildren. Acta Paediatr. 1992;81:1035–1039. [DOI] [PubMed]
- 27.Selbst SM, Lavelle JM, Soyupak SK, Markowitz RI. Back pain in children who present to the emergency department. Clin Pediatr (Phila). 1999;38:401–406. [DOI] [PubMed]
- 28.Sward L, Hellstrom M, Jacobsson B, Nyman R, Peterson L. Disc degeneration and associated abnormalities of the spine in elite gymnasts: a magnetic resonance imaging study. Spine. 1991;16:437–443. [DOI] [PubMed]
- 29.The Committee on Ways and Means. Johnson announces hearing on managing the use of imaging services. Available at: http://waysandmeans.house.gov/hearings.asp?formmode=printfriendly&id=2538. Accessed March 10, 2005.
- 30.Turner PG, Green JH, Galasko CS. Back pain in childhood. Spine. 1989;14:812–814. [DOI] [PubMed]
- 31.Udeshi UL, Reeves D. Routine thin slice MRI effectively demonstrates the lumbar pars interarticularis. Clin Radiol. 1999;54:615–619. [DOI] [PubMed]
- 32.Yamane T, Yoshida T, Mimatsu K. Early diagnosis of lumbar spondylolysis by MRI. J Bone Joint Surg Br. 1993;75:764–768. [DOI] [PubMed]