Abstract
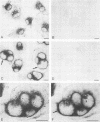
Leishmania amastigotes are intracellular protozoan parasites of mononuclear phagocytes which reside within parasitophorous vacuoles of phagolysosomal origin. The pH of these compartments was studied with the aim of elucidating strategies used by these microorganisms to evade the microbicidal mechanisms of their host cells. For this purpose, rat bone marrow-derived macrophages were infected with L. amazonensis amastigotes. Intracellular acidic compartments were localized by using the weak base 3-(2,4-dinitroanilino)-3'-amino-N-methyldipropylamine as a probe. This indicator, which can be detected by light microscopy by using immunocytochemical methods, mainly accumulated in perinuclear lysosomes of uninfected cells, whereas in infected cells, it was essentially localized in parasitophorous vacuoles, which thus appeared acidified. Phagolysosomal pH was estimated quantitatively in living cells loaded with the pH-sensitive endocytic tracer fluoresceinated dextran. After a 15- to 20-h exposure, the tracer was mainly detected in perinuclear lysosomes and parasitophorous vacuoles of uninfected and infected macrophages, respectively. Fluorescence intensities were determined from digitized video images of single cells after processing and automatic subtraction of background. We found statistically different mean pH values of 5.17 to 5.48 for lysosomes and 4.74 to 5.26 for parasitophorous vacuoles. As for lysosomes of monensin-treated cells, the pH gradient of parasitophorous vacuoles collapsed after monensin was added. This very likely indicates that these vacuoles maintain an acidic internal pH by an active process. These results show that L. amazonensis amastigotes are acidophilic and opportunistic organisms and suggest that these intracellular parasites have evolved means for survival under these harsh conditions and have acquired plasma membrane components compatible with the environment.
Full text
PDF






Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Anderson R. G., Falck J. R., Goldstein J. L., Brown M. S. Visualization of acidic organelles in intact cells by electron microscopy. Proc Natl Acad Sci U S A. 1984 Aug;81(15):4838–4842. doi: 10.1073/pnas.81.15.4838. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Anderson R. G., Orci L. A view of acidic intracellular compartments. J Cell Biol. 1988 Mar;106(3):539–543. doi: 10.1083/jcb.106.3.539. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Antoine J. C., Jouanne C., Ryter A., Benichou J. C. Leishmania amazonensis: acidic organelles in amastigotes. Exp Parasitol. 1988 Dec;67(2):287–300. doi: 10.1016/0014-4894(88)90076-8. [DOI] [PubMed] [Google Scholar]
- Antoine J. C., Jouanne C., Ryter A., Zilberfarb V. Leishmania mexicana: a cytochemical and quantitative study of lysosomal enzymes in infected rat bone marrow-derived macrophages. Exp Parasitol. 1987 Dec;64(3):485–498. doi: 10.1016/0014-4894(87)90063-4. [DOI] [PubMed] [Google Scholar]
- Avrameas S., Ternynck T. Peroxidase labelled antibody and Fab conjugates with enhanced intracellular penetration. Immunochemistry. 1971 Dec;8(12):1175–1179. doi: 10.1016/0019-2791(71)90395-8. [DOI] [PubMed] [Google Scholar]
- Barbieri C. L., Brown K., Rabinovitch M. Depletion of secondary lysosomes in mouse macrophages infected with Leishmania mexicana amazonensis: a cytochemical study. Z Parasitenkd. 1985;71(2):159–168. doi: 10.1007/BF00926266. [DOI] [PubMed] [Google Scholar]
- Black C. M., Paliescheskey M., Beaman B. L., Donovan R. M., Goldstein E. Acidification of phagosomes in murine macrophages: blockage by Nocardia asteroides. J Infect Dis. 1986 Dec;154(6):952–958. doi: 10.1093/infdis/154.6.952. [DOI] [PubMed] [Google Scholar]
- Chang K. P. Cellular and molecular mechanisms of intracellular symbiosis in leishmaniasis. Int Rev Cytol Suppl. 1983;14:267–305. [PubMed] [Google Scholar]
- Chang K. P. Human cutaneous lieshmania in a mouse macrophage line: propagation and isolation of intracellular parasites. Science. 1980 Sep 12;209(4462):1240–1242. doi: 10.1126/science.7403880. [DOI] [PubMed] [Google Scholar]
- Collot M., Louvard D., Singer S. J. Lysosomes are associated with microtubules and not with intermediate filaments in cultured fibroblasts. Proc Natl Acad Sci U S A. 1984 Feb;81(3):788–792. doi: 10.1073/pnas.81.3.788. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Coombs G. H. Proteinases of Leishmania mexicana and other flagellate protozoa. Parasitology. 1982 Feb;84(1):149–155. doi: 10.1017/s003118200005174x. [DOI] [PubMed] [Google Scholar]
- Evans M. J., Levy L. Ultrastructural changes in cells of the mouse footpad infected with Mycobacterium leprae. Infect Immun. 1972 Feb;5(2):238–247. doi: 10.1128/iai.5.2.238-247.1972. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ferris A. L., Brown J. C., Park R. D., Storrie B. Chinese hamster ovary cell lysosomes rapidly exchange contents. J Cell Biol. 1987 Dec;105(6 Pt 1):2703–2712. doi: 10.1083/jcb.105.6.2703. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Frehel C., de Chastellier C., Lang T., Rastogi N. Evidence for inhibition of fusion of lysosomal and prelysosomal compartments with phagosomes in macrophages infected with pathogenic Mycobacterium avium. Infect Immun. 1986 Apr;52(1):252–262. doi: 10.1128/iai.52.1.252-262.1986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- GRAHAM R. C., Jr, LUNDHOLM U., KARNOVSKY M. J. CYTOCHEMICAL DEMONSTRATION OF PEROXIDASE ACTIVITY WITH 3-AMINO-9-ETHYLCARBAZOLE. J Histochem Cytochem. 1965 Feb;13:150–152. doi: 10.1177/13.2.150. [DOI] [PubMed] [Google Scholar]
- Germain R. N. Immunology. The ins and outs of antigen processing and presentation. Nature. 1986 Aug 21;322(6081):687–689. doi: 10.1038/322687a0. [DOI] [PubMed] [Google Scholar]
- Glaser T. A., Baatz J. E., Kreishman G. P., Mukkada A. J. pH homeostasis in Leishmania donovani amastigotes and promastigotes. Proc Natl Acad Sci U S A. 1988 Oct;85(20):7602–7606. doi: 10.1073/pnas.85.20.7602. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Goren M. B., Swendsen C. L., Fiscus J., Miranti C. Fluorescent markers for studying phagosome-lysosome fusion. J Leukoc Biol. 1984 Sep;36(3):273–292. doi: 10.1002/jlb.36.3.273. [DOI] [PubMed] [Google Scholar]
- Graham R. C., Jr, Karnovsky M. J. The early stages of absorption of injected horseradish peroxidase in the proximal tubules of mouse kidney: ultrastructural cytochemistry by a new technique. J Histochem Cytochem. 1966 Apr;14(4):291–302. doi: 10.1177/14.4.291. [DOI] [PubMed] [Google Scholar]
- Hackstadt T., Williams J. C. Biochemical stratagem for obligate parasitism of eukaryotic cells by Coxiella burnetii. Proc Natl Acad Sci U S A. 1981 May;78(5):3240–3244. doi: 10.1073/pnas.78.5.3240. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Handman E., Mitchell G. F., Goding J. W. Identification and characterization of protein antigens of Leishmania tropica isolates. J Immunol. 1981 Feb;126(2):508–512. [PubMed] [Google Scholar]
- Hart P. D., Armstrong J. A., Brown C. A., Draper P. Ultrastructural study of the behavior of macrophages toward parasitic mycobacteria. Infect Immun. 1972 May;5(5):803–807. doi: 10.1128/iai.5.5.803-807.1972. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Horwitz M. A., Maxfield F. R. Legionella pneumophila inhibits acidification of its phagosome in human monocytes. J Cell Biol. 1984 Dec;99(6):1936–1943. doi: 10.1083/jcb.99.6.1936. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kahl L. P., McMahon-Pratt D. Structural and antigenic characterization of a species- and promastigote-specific Leishmania mexicana amazonensis membrane protein. J Immunol. 1987 Mar 1;138(5):1587–1595. [PubMed] [Google Scholar]
- Kaye P. M. Antigen presentation and the response to parasitic infection. Parasitol Today. 1987 Oct;3(10):293–299. doi: 10.1016/0169-4758(87)90186-4. [DOI] [PubMed] [Google Scholar]
- Klebanoff S. J., Locksley R. M., Jong E. C., Rosen H. Oxidative response of phagocytes to parasite invasion. Ciba Found Symp. 1983;99:92–112. doi: 10.1002/9780470720806.ch6. [DOI] [PubMed] [Google Scholar]
- Kress Y., Bloom B. R., Wittner M., Rowen A., Tanowitz H. Resistance of Trypanosoma cruzi to killing by macrophages. Nature. 1975 Oct 2;257(5525):394–396. doi: 10.1038/257394a0. [DOI] [PubMed] [Google Scholar]
- Long E. O., Jacobson S. Pathways of viral antigen processing and presentation to CTL: defined by the mode of virus entry? Immunol Today. 1989 Feb;10(2):45–48. doi: 10.1016/0167-5699(89)90303-4. [DOI] [PubMed] [Google Scholar]
- Matteoni R., Kreis T. E. Translocation and clustering of endosomes and lysosomes depends on microtubules. J Cell Biol. 1987 Sep;105(3):1253–1265. doi: 10.1083/jcb.105.3.1253. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mukkada A. J., Meade J. C., Glaser T. A., Bonventre P. F. Enhanced metabolism of Leishmania donovani amastigotes at acid pH: an adaptation for intracellular growth. Science. 1985 Sep 13;229(4718):1099–1101. doi: 10.1126/science.4035350. [DOI] [PubMed] [Google Scholar]
- Nogueira N., Cohn Z. Trypanosoma cruzi: mechanism of entry and intracellular fate in mammalian cells. J Exp Med. 1976 Jun 1;143(6):1402–1420. doi: 10.1084/jem.143.6.1402. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ohkuma S., Poole B. Fluorescence probe measurement of the intralysosomal pH in living cells and the perturbation of pH by various agents. Proc Natl Acad Sci U S A. 1978 Jul;75(7):3327–3331. doi: 10.1073/pnas.75.7.3327. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rikihisa Y., Ito S. Intracellular localization of Rickettsia tsutsugamushi in polymorphonuclear leukocytes. J Exp Med. 1979 Sep 19;150(3):703–708. doi: 10.1084/jem.150.3.703. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sansonetti P. J., Ryter A., Clerc P., Maurelli A. T., Mounier J. Multiplication of Shigella flexneri within HeLa cells: lysis of the phagocytic vacuole and plasmid-mediated contact hemolysis. Infect Immun. 1986 Feb;51(2):461–469. doi: 10.1128/iai.51.2.461-469.1986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sibley L. D., Weidner E., Krahenbuhl J. L. Phagosome acidification blocked by intracellular Toxoplasma gondii. 1985 May 30-Jun 5Nature. 315(6018):416–419. doi: 10.1038/315416a0. [DOI] [PubMed] [Google Scholar]
- Ternynck T., Avrameas S. Polyacrylamide-protein immunoadsorbents prepared with glutaraldehyde. FEBS Lett. 1972 Jun 1;23(1):24–28. doi: 10.1016/0014-5793(72)80274-6. [DOI] [PubMed] [Google Scholar]
- Walker T. S., Winkler H. H. Interactions between Rickettsia prowazekii and rabbit polymorphonuclear leukocytes: rickettsiacidal and leukotoxic activities. Infect Immun. 1981 Jan;31(1):289–296. doi: 10.1128/iai.31.1.289-296.1981. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Weisman L. S., Wickner W. Intervacuole exchange in the yeast zygote: a new pathway in organelle communication. Science. 1988 Jul 29;241(4865):589–591. doi: 10.1126/science.3041591. [DOI] [PubMed] [Google Scholar]
- Yamashiro D. J., Maxfield F. R. Acidification of morphologically distinct endosomes in mutant and wild-type Chinese hamster ovary cells. J Cell Biol. 1987 Dec;105(6 Pt 1):2723–2733. doi: 10.1083/jcb.105.6.2723. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zilberstein D., Dwyer D. M. Identification of a surface membrane proton-translocating ATPase in promastigotes of the parasitic protozoan Leishmania donovani. Biochem J. 1988 Nov 15;256(1):13–21. doi: 10.1042/bj2560013. [DOI] [PMC free article] [PubMed] [Google Scholar]