Abstract
Background
Recent studies suggest dairy consumption and associated nutrients may be protective against some of the components of the metabolic syndrome (MetS).
Objectives
We examined the association between consumption of a variety of dairy products and their related nutrients with obesity, central obesity, and MetS, and attempted to explain some of the ethnic differences in metabolic outcomes through dairy consumption using national data.
Design
Nationally representative indicators of obesity, central obesity, and MetS among US adults were constructed from National Health and Nutrition Examination Survey 1999–2004 data, including direct anthropometric assessments, blood pressure, and laboratory tests. Sample sizes ranged from 4519 for MetS to 14 618 for obesity. Associations between diet (assessed using 24-h recalls) and metabolic and other outcomes were tested using multivariate linear and logistic models and structural equation models.
Results
We found a significant inverse association between intake of whole milk, yogurt, calcium, and magnesium and metabolic disorders. Odds ratios for one more daily serving of yogurt and 100 mg Mg for MetS were 0.40 (95% CI: 0.18, 0.89) and 0.83 (95% CI: 0.72, 0.96), respectively. The opposite was found for intakes of cheese, low-fat milk, and phosphorus. Using structural equation models, ethnic differences in some MetS outcomes, such as body mass index and systolic blood pressure, were partly explained by variations in dairy-related nutrients.
Conclusions
Various dairy products may have differential associations with metabolic disorders, including obesity. Ethnic differences in dairy consumption may explain in part the ethnic disparities in metabolic disorders in the US population.
INTRODUCTION
The continuing rise in prevalence of obesity and metabolic syndrome (MetS) in the United States (1–5) constitutes a serious threat to public health, increasing mortality, disability, and health care costs (6–14). Among the many factors implicated in the rise in obesity and MetS, recent investigations of the intake of dairy products and calcium have yielded inconclusive results (15–17). Although the consumption of some constituents of milk and dairy products, such as saturated fat, point to a deleterious effect on weight status and cardiovascular disease (18–22), other components, such as calcium, may have beneficial effects (23). One hypothesis is that dietary calcium plays a critical role in the regulation of body weight by affecting adipocyte intracellular calcium concentration, decreasing fatty acid synthesis while increasing lipolysis, leading to decreased triacylglycerol (TA) stores (24). Recent prospective cohort- and cross-sectional studies suggest that dairy and calcium consumption may reduce the risk for obesity (25, 26) or central obesity (26), type 2 diabetes (27), hypertension (28, 29), and MetS (28, 30–32). In contrast, other recent studies have not found such beneficial effects (33, 34).
Most of the previous studies, however, are based on non-representative population samples and thus may not have had sufficient diversity in their samples to study either sex, ethnic, and racial differences in dairy consumption or the association between intake and metabolic disorders. Second, the mediating effect of dairy-related nutrients and the types of dairy products that are most beneficial in terms of metabolic outcomes are still largely unknown. We are not aware of any reports in the United States linking the known ethnic differences in dairy consumption (35, 36) with the ethnic disparities in the prevalence of obesity and metabolic disorders (1–5).
The present study assessed the association between consumption of dairy and related nutrients and obesity, central obesity and MetS through the use of the most recent nationally representative data collected from adults in the United States, the 1999–2004 National Health and Nutrition Examination Survey (NHANES). In addition, we examined the ethnic differences in dairy and related nutrient intakes, metabolic disturbances, and the associations between them.
MATERIALS AND METHODS
Database and study design
The NHANES database comprises a series of cross-sectional surveys initiated in the 1960s that provided nationally representative information on the nutrition and health status of the US civilian population. Since 1999, NHANES has been a continuous annual survey. The data were recently made available for the period 1999–2004. All of the NHANES surveys used a stratified, multistage probability cluster sampling design. The NHANES survey consists of an in-home interview for demographic and basic health information as well as a health examination in a mobile examination center (MEC). Household interviews were conducted by trained staff, and the MEC is staffed by physicians, medical and health technicians, and dietary and health interviewers. NHANES MEC examinations included direct anthropometric, blood pressure, and laboratory measurements on all participants, or a subgroup. Detailed descriptions of the study design and data collection have been published elsewhere (2, 37).
Study population
We merged NHANES data for the periods 1999–2000, 2001–2002, and 2003–2004 and focused on adults aged 18 y and over, a sample of 17 061 (8970 women and 8091 men) with complete demographic data. Of these, 15 009 had complete dietary data (7860 women and 7149 men), and 14 618 had both anthropometric and dietary data available (7652 women and 6966 men). Data for these 14 618 subjects were used in our analysis to examine the association between dairy consumption and body weight. Dietary data were available for 14 484 subjects for waist circumference (WC), 14 464 for blood pressure, 7113 for blood glucose, 9411 for HDL cholesterol, and 7030 for plasma TA measurements. Thus, complete data on MetS and dietary intakes were available for 4519 subjects. It is worth noting that fasting blood sample tests collected by Centers for Disease Control and Prevention (eg, fasting blood glucose and TA) were conducted in only half of the eligible adult sample. A subsample excluding pregnant and lactating women (~8% of the total sample) was also considered for sensitivity analysis.
Data collection and key measurements
Dietary intake
NHANES provided one 24-h recall for each of the 1999–2000 and 2001–2002 periods, and 2 for 2003–2004. Only the first day of recall was used in this study to give equal weight to all 3 periods. A sensitivity analysis was conducted to assess differences in estimates of intake using one compared with 2 recalls for 2003–2004, and the correlations between the 2 methods over the whole period of 1999–2004 for dairy-related nutrients (see below) and total energy intake were high, >0.95. The amount of food consumed daily by an individual was estimated by summing the weight of foods belonging to a common group (eg, milk of various fat contents, cheese, yogurt, and all dairy). A database for converting grams of individual foods into servings was created using data available for NHANES 2001–2002, which was then used to convert grams into servings for the whole period of 1999–2004 (38). Dairy consumption-related nutrients [calcium, phosphorus, magnesium, and dairy fat expressed as 14:0 (myristic acid) as percentage of total fat intake] were readily available for all 3 waves and were of particular interest. Correlations between total dairy (expressed as servings) and the related nutrients were 0.88 for calcium, 0.45 for magnesium, 0.69 for phosphorus, and 0.49 for dairy fat (P < 0.05). In most analyses, the nutrients were expressed as per 100 mg. However, it is worth noting the incommensurability between these amounts, given that current Dietary Reference Intakes for healthy adults differ considerably (eg, 1000 mg for calcium, 700 mg for phosphorus, and 420 mg for magnesium) (39).
Anthropometric measures
Anthropometric measurements were recorded for all examinees by a trained examiner in the MEC. In particular, weight was measured for all ages, whereas standing height and waist circumference (WC) was measured for all adults (40). WC was measured using a tape measure starting from the hip bone and wrapping around the waist at the level of the navel in such a way that it is kept parallel to the floor, the person is not holding his or her breath, and it is not wrapped too tight or too loose.
Blood pressure
Three and sometimes 4 blood pressure determinations [systolic (SBP) and diastolic (DBP)] during MEC examinations, and during home examinations for those older than 50 y, were obtained on all eligible individuals using a mercury sphygmomanometer (41). The average of 3 determinations was the main variable of interest, and the fourth one was dropped.
Laboratory tests
Detailed specimen collection and processing instructions are described in the NHANES Laboratory/Medical Technologists Procedures Manual (43). Through use of these instructions, (1) fasting blood glucose was analyzed using a reaction between glucose and ATP catalyzed by the enzyme hexokinase; (2) TAs were measured enzymatically in serum or plasma using a series of coupled reactions in which TAs were hydrolyzed to produce glycerol; and (3) HDL cholesterol was measured using 2 methods. For adults in our sample, a heparin-manganese precipitation method was used, unless limited sample volume was available, in which case a direct immunoassay technique was substituted.
Other main covariates: sociodemographic variables
Age, sex, and ethnicity (categorized as non-Hispanic (NH) whites, non-Hispanic blacks, Mexican Americans, and other groups) were considered as independent variables with both dairy consumption and other health outcomes. Education and family income were chosen as the main indicators of socioeconomic status (SES) (43). Education was measured by years completed, and the participants were then grouped into 0: < high school (HS; 12 y) education, 1: HS education, and 2: >HS education. The poverty income ratio is the ratio of household income and the poverty line published by the Census Bureau for a certain family size in that calendar year. Specifically, we used the poverty income ratio tertiles at each survey to indicate low-, medium-, and high-income groups. Physical activity, self-reported in terms of vigorous activity over the past 30 d that was dichotomized as 1 = yes and 0 = no, was included as dummy variables in models where outcomes were obesity, central obesity, MetS, or separate metabolic outcomes to control for physical activity as a potential confounder.
Classification of key health outcomes: obesity, central obesity, and metabolic syndrome
Obesity was defined as body mass index [BMI (in kg/m2)] ≥30, and central obesity was defined as WC ≥102 cm or 40 inches (men) and ≥88 cm or 35 inches (women) (5).
MetS is a condition defined as occurring when 3 or more of the following conditions exist simultaneously, namely abdominal obesity, insulin resistance, hypertension, and dyslipidemia with high levels of plasma TA and low HDL cholesterol (44), based on the National Cholesterol Education Program Adult Treatment Panel III (2001): 1) central obesity (see above); 2) dyslipidemia: TA ≥1.695 mmol/L (150 mg/dL); 3) dyslipidemia: HDL cholesterol < 40 mg/dL (male), < 50 mg/dL (female); 4) blood pressure ≥130/85 mm Hg; 5) fasting plasma glucose ≥6.1 mmol/L (110 mg/dL) (45). The number of metabolic disturbances was also examined in its continuous form, primarily to test whether rates that varied by sociodemographic factors could be explained by variations in dairy consumption.
Statistical analysis
First, we described the sex and ethnic differences in US adult intakes of dairy and related nutrients as well as differences in obesity and MetS outcomes, taking into account the complex sampling design for variance estimation, and used appropriate weights to obtain nationally representative estimates. Differences in means between groups were tested using analysis of variance and t tests. Associations between categorical variables were tested using chi-square tests.
Next, a series of linear and logistic models were fit to examine sociodemographic factors as potential confounders in their mutual relation with dairy consumption and metabolic outcomes, including obesity, central obesity, and MetS. We fit multivariate logistic regression models to examine the association between intakes of dairy and related nutrients and metabolic outcomes (obesity, central obesity, and MetS). All models controlled for sociodemographic factors, energy intake, and physical activity. In addition, a sensitivity analysis was carried out excluding women who were pregnant or lactating at the time of the survey (~8% of the total sample and 16% of the female sample).
Finally, racial/ethnic differences in metabolic outcomes were examined using structural equation models (SMs) (46, 47). Direct, indirect, and total effects of ethnicity as well as mediation proportions (MPs) by dairy and related nutrient intakes were estimated as indicated in Figure 1 (47). MPs >10% were considered significant mediation whereas MPs between 5% and 10% were considered borderline significant. We focused on those total effects of ethnicity on MetS outcomes that were statistically significant. In all analyses, except for SMs, in which data were fitted using the SAS CALIS procedure, version 9.1 (48), design complexity [primary sampling units (PSUs), strata, and sampling weights] was taken into account using STATA release 9.0 (49) survey-related commands.
FIGURE 1.
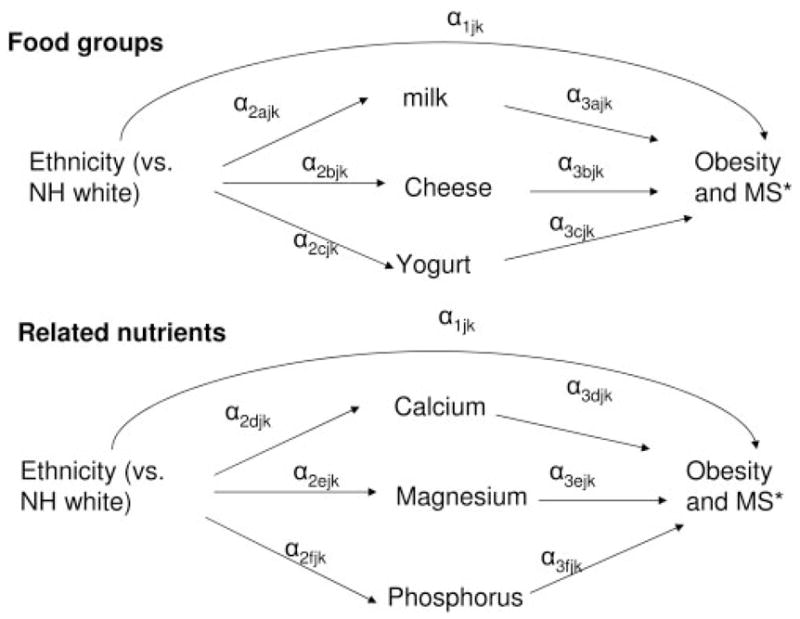
Path diagram for direct and indirect effects of ethnicity on obesity and metabolic disturbance outcomes through intakes of dairy and related nutrients. NH, non-Hispanic; MS, metabolic syndrome. *Modeled using the continuous measures of body mass index, waist circumference, systolic blood pressure, fasting blood glucose, and total number of disturbances as the outcome variables. α: path coefficient between measured variables in model j; j: model notation specific to metabolic outcome. Direct effect: α1; indirect effect: Σα2× α3 for k = 1 and 2, separately. k: 1 for food groups and 2 for related nutrients. Mediation proportion = Indirect× 100/(Indirect + Direct effects). Exogenous variables included in the model were age, education, sex, and income. Energy was included as an additional mediator or endogenous variable.
RESULTS
Ethnic differences in intakes of dairy products and related nutrients as well as the prevalence of obesity and MetS
The distribution of daily dairy consumption, related nutrients, and metabolic outcomes by sex and race/ethnicity is presented in Table 1. On average, US adults consumed 1.54 servings of dairy, (0.90 in fluid milk and 0.60 in cheese) per day. Mean dairy intake was 247.3 g, with men consuming a larger amount. Only 18% of men and 10% of women consumed the recommended 3 servings/d or more of dairy products. NH blacks had fewer mean servings per day compared with NH whites and other minorities. These ethnic disparities in dairy consumption were also reflected in the dairy-related nutrients, with NH blacks having the lowest daily intakes of calcium, magnesium, phosphorus, and dairy fat.
TABLE 1.
Gender and ethnic differences in intakes of dairy foods and related nutrients, obesity, and metabolic outcomes: NHANES, 1999–20041
| Men and women (n = 17 061)
|
Men (n = 8091)
|
Women (n = 8970)
|
NH white (n = 8302)
|
NH black (n = 3458)
|
Mex Am (n = 4013)
|
Other (n = 1288)
|
||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Mean | SE | Mean | SE | Mean | SE | Mean | SE | Mean | SE | Mean | SE | Mean | SE | |
| Dairy and related nutrients | ||||||||||||||
| All dairy (g) | 247.3 | 5.0 | 272.8 | 6.6 | 223.9 | 5.42 | 273.4 | 5.8 | 138.8 | 5.0 | 224.2 | 9.6 | 192.9 | 10.02 |
| Fluid milk (g) | 165.7 | 4.3 | 187.2 | 6.0 | 146.0 | 4.92 | 182.4 | 5.0 | 89.1 | 4.6 | 164.8 | 8.7 | 128.6 | 8.62 |
| Whole milk (g) | 44.8 | 1.8 | 54.2 | 3.0 | 36.2 | 1.92 | 39.8 | 2.2 | 45.6 | 3.7 | 83.4 | 5.2 | 52.3 | 5.92 |
| Low fat milk (g) | 76.8 | 2.9 | 91.8 | 4.5 | 63.0 | 2.72 | 88.9 | 3.3 | 31.8 | 2.7 | 58.4 | 5.0 | 51.5 | 7.02 |
| Skim milk (g) | 36.8 | 3.1 | 33.7 | 3.0 | 39.7 | 3.82 | 46.7 | 4.0 | 6.7 | 1.6 | 12.9 | 2.0 | 15.7 | 4.32 |
| Yogurt (g) | 6.2 | 0.5 | 5.2 | 0.6 | 7.0 | 0.62 | 7.1 | 0.7 | 2.1 | 0.5 | 2.5 | 0.4 | 6.7 | 1.42 |
| Cheese (g) | 19.7 | 0.5 | 22.1 | 0.7 | 17.6 | 0.52 | 22.2 | 0.7 | 11.7 | 0.6 | 17.3 | 1.1 | 12.7 | 1.22 |
| All dairy (servings) | 1.54 | 0.02 | 1.75 | 0.03 | 1.34 | 0.032 | 1.70 | 0.03 | 0.97 | 0.03 | 1.39 | 0.05 | 1.20 | 0.052 |
| All dairy, ≥3 servings/d (%) | 14.0 | 0.5 | 18.3 | 0.7 | 10.0 | 0.52 | 16.0 | 0.6 | 6.5 | 0.5 | 11.4 | 1.0 | 9.2 | 0.92 |
| Fluid milk (servings) | 0.90 | 0.02 | 1.01 | 0.02 | 0.80 | 0.022 | 0.99 | 0.02 | 0.52 | 0.02 | 0.84 | 0.04 | 0.70 | 0.042 |
| Yogurt (servings) | 0.03 | 0.00 | 0.03 | 0.00 | 0.03 | 0.002 | 0.03 | 0.00 | 0.01 | 0.00 | 0.01 | 0.00 | 0.03 | 0.012 |
| Cheese (servings) | 0.60 | 0.01 | 0.72 | 0.02 | 0.50 | 0.012 | 0.65 | 0.02 | 0.43 | 0.02 | 0.53 | 0.02 | 0.46 | 0.022 |
| Calcium (mg/d) | 864.6 | 9.1 | 986.3 | 12.0 | 752.4 | 9.92 | 916.4 | 11.7 | 661.2 | 11.6 | 856.8 | 17.6 | 714.5 | 19.12 |
| Calcium, >recommended mg/d (%) | 31.4 | 0.6 | 38.4 | 0.7 | 25.0 | 0.82 | 33.7 | 0.8 | 22.4 | 0.1 | 34.1 | 0.9 | 22.8 | 1.42 |
| Magnesium (mg/d) | 283.2 | 2.6 | 328.1 | 2.9 | 241.7 | 2.92 | 292.0 | 3.5 | 235.6 | 3.3 | 293.8 | 3.6 | 262.7 | 5.02 |
| Phosphorus (mg/d) | 1331.8 | 9.3 | 1560.6 | 12.2 | 1120.8 | 10.22 | 1372.9 | 11.7 | 1146.8 | 14.3 | 1394.4 | 17.2 | 1186.7 | 22.12 |
| Dairy fatty acids (g/100 g fat) | 2.71 | 0.02 | 2.68 | 0.02 | 2.74 | 0.03 | 2.81 | 0.03 | 2.30 | 0.03 | 2.60 | 0.04 | 2.53 | 0.052 |
| Metabolic outcomes3 | ||||||||||||||
| BMI (kg/m2) | 27.9 | 0.1 | 27.8 | 0.1 | 28.1 | 0.12 | 27.8 | 0.1 | 29.6 | 0.2 | 28.3 | 0.2 | 27 | 0.32 |
| Waist circumference (cm) | 95.9 | 0.3 | 99.0 | 0.2 | 93.1 | 0.32 | 96.4 | 0.3 | 96.7 | 0.4 | 95.1 | 0.5 | 92.1 | 0.82 |
| SBP (mm Hg) | 122.5 | 0.3 | 123.4 | 0.4 | 121.7 | 0.42 | 122.6 | 0.4 | 125.7 | 0.5 | 118.7 | 0.6 | 121.5 | 0.82 |
| DBP (mm Hg) | 71.5 | 0.2 | 73.1 | 0.2 | 69.9 | 0.32 | 71.3 | 0.3 | 72.9 | 0.4 | 69.7 | 0.4 | 72.2 | 0.62 |
| HDL-C (mg/dL) | 50.8 | 0.4 | 45.4 | 0.4 | 55.9 | 0.52 | 51.0 | 0.5 | 53.8 | 0.6 | 48.3 | 0.4 | 48.7 | 0.92 |
| TA (mg/dL) | 146.0 | 2.7 | 159.5 | 4.7 | 133.4 | 2.32 | 150.6 | 3.4 | 107.5 | 2.7 | 155.8 | 6.2 | 147 | 6.22 |
| Fasting blood glucose (mg/dL) | 100.5 | 0.5 | 103.7 | 0.6 | 97.5 | 0.62 | 99.9 | 0.5 | 100.6 | 1.4 | 103 | 1.3 | 102.9 | 2.12 |
| Obesity (%, BMI ≥30) | 30.4 | 0.7 | 27.7 | 0.7 | 32.8 | 0.92 | 29.6 | 0.8 | 40.0 | 1.0 | 32.0 | 1.4 | 23.3 | 1.82 |
| Central obesity (%)4 | 48.6 | 0.8 | 38.1 | 0.8 | 58.4 | 1.02 | 49.6 | 1.0 | 52.3 | 0.8 | 45.5 | 1.7 | 39.3 | 2.22 |
| MetS (%)5 | 25.8 | 0.7 | 24.4 | 1.3 | 27.2 | 0.8 | 26.5 | 0.8 | 20.6 | 1.5 | 24.4 | 2.1 | 27.6 | 0.4 |
| Number of metabolic disturbances6 | 1.62 | 0.03 | 1.56 | 0.04 | 1.68 | 0.032 | 1.63 | 0.03 | 1.47 | 0.06 | 1.58 | 0.06 | 1.68 | 0.102 |
NHANES, National Health and Nutrition Examination Survey; SBP, systolic blood pressure; DBP, diastolic blood pressure; TA, triacylglycerols; MetS, metabolic syndrome, NH, non-Hispanic; Mex Am, Mexican American.
P < 0.05 for testing the null hypothesis that means or proportions are the same between groups.
Data are available for a subset of the sample: n = 15 331 and 15 078 for BMI and waist circumference; n = 15 040 for blood pressure; n = 9823 for HDL-C; n = 7257 for TA and n = 7345 for fasting blood glucose.
Defined as waist circumference >102 cm for men and >88 cm for women.
Defined based on NCEP ATP III described in Methods.
Three or more metabolic disturbances as listed above represent MetS. Metabolic disturbances may range between 0 and 5.
In terms of metabolic profile, men had worse metabolic outcomes than did women, with the exception of BMI, prevalence of obesity, central obesity, and the mean number of metabolic disturbances, all of which were higher among women. BMI, WC, SBP, DBP, and HDL cholesterol were highest, whereas plasma TA was lowest, among NH blacks, whereas fasting blood glucose was highest among Mexican Americans, closely followed by the “other” category. Whereas obesity and central obesity prevalence rates were highest among NH blacks, mean number of metabolic disturbances was highest among the “other” race/ethnicity category, and lowest among NH blacks, whereas no significant ethnic differences were noted in terms of MetS.
Multivariate linear regression analyses suggested that dairy consumption is significantly higher among subjects with more than a HS education, and significantly lower among women and minority groups (Table 2). Only obesity and central obesity were significantly higher among women. NH blacks were more likely than NH whites to be obese or centrally obese [odds ratio (OR) 1.55; 95% CI: 1.42, 1.70 and 1.16; 95% CI: 1.04, 1.29, respectively] but were less likely to have MetS. These associations were more pronounced among women. In fact, among men, NH blacks were less likely than NH whites to have central obesity or MetS. For both genders combined, income (poverty income ratio: high compared with low) was inversely related to obesity and central obesity, whereas education (>HS compared with < HS) was inversely related to central obesity (OR: 0.84; 95% CI: 0.73, 0.96) and MetS (OR: 0.63; 95% CI: 0.45, 0.89).
TABLE 2.
Associations between sociodemographic characteristics and dairy consumption, obesity, central obesity, and metabolic syndrome: NHANES, 1999–20041
| Dairy consumption (servings) (n = 14 980)
|
Obesity (n = 5306)
|
Central obesity (n = 15 054)
|
Metabolic syndrome (n = 4575)
|
||||||
|---|---|---|---|---|---|---|---|---|---|
| β | SE | P value | OR | (95% CI) | OR | (95% CI) | OR | (95% CI) | |
| Female (vs male) | −0.38 | 0.03 | < 0.001 | 1.27 | (1.18, 1.37)2 | 2.31 | (2.13, 2.49)2 | 1.08 | (0.90, 1.29) |
| All subjects | |||||||||
| Ethnicity | |||||||||
| NH white | Ref | 1.00 | 1.00 | 1.00 | |||||
| NH black | −0.71 | 0.04 | < 0.001 | 1.55 | (1.42, 1.70)2 | 1.16 | (1.04, 1.29)2 | 0.75 | (0.57, 0.99)2 |
| Mex Am | −0.34 | 0.06 | < 0.001 | 1.15 | (0.98, 1.35) | 1.03 | (0.88, 1.21) | 1.04 | (0.75, 1.44) |
| Other | −0.49 | 0.06 | < 0.001 | 0.71 | (0.59, 0.86)2 | 0.67 | (0.55, 0.80)2 | 1.10 | (0.73, 1.68) |
| Income (poverty income ratio) | |||||||||
| Low | ref | 1.00 | 1.00 | 1.00 | |||||
| Medium | 0.02 | 0.08 | 0.800 | 0.99 | (0.83, 1.18) | 0.98 | (0.83, 1.17)2 | 0.74 | (0.53, 1.05) |
| High | 0.08 | 0.05 | 0.131 | 0.87 | (0.77, 0.99)2 | 0.85 | (0.75, 0.97)2 | 0.69 | (0.52, 0.91)2 |
| Education | |||||||||
| < High school | ref | 1.00 | 1.00 | 1.00 | |||||
| High school | 0.11 | 0.05 | 0.038 | 1.07 | (0.92, 1.24) | 1.03 | (0.91, 1.17) | 0.98 | (0.69, 1.39) |
| >High school | 0.18 | 0.04 | < 0.001 | 0.89 | (0.77, 1.04) | 0.84 | (0.73, 0.96)2 | 0.63 | (0.45, 0.89)2 |
| Men | |||||||||
| Ethnicity | |||||||||
| NH white | ref | 1.00 | 1.00 | 1.00 | |||||
| NH black | −0.83 | 0.06 | < 0.001 | 1.04 | (0.88, 1.22) | 0.64 | (0.55, 0.74)2 | 0.57 | (0.41, 0.80)2 |
| Mex Am | −0.56 | 0.03 | < 0.001 | 1.07 | (0.87, 1.32) | 0.79 | (0.64, 0.98)2 | 0.97 | (0.65, 1.44) |
| Other | −0.71 | 0.10 | < 0.001 | 0.65 | (0.51, 0.82)2 | 0.46 | (0.34, 0.62)2 | 0.92 | (0.57, 1.48) |
| Income (poverty income ratio) | |||||||||
| Low | ref | 1.00 | 1.00 | 1.00 | |||||
| Medium | −0.09 | 0.14 | 0.390 | 1.22 | (0.98, 1.51) | 1.07 | (0.8, 1.40) | 1.13 | (0.74, 1.73) |
| High | 0.02 | 0.09 | 0.148 | 1.02 | (0.85, 1.23) | 1.14 | (0.93, 1.38) | 1.06 | (0.78, 1.60) |
| Education | |||||||||
| < High school | ref | 1.00 | 1.00 | 1.00 | |||||
| High school | 0.09 | 0.10 | 0.514 | 1.09 | (0.85, 1.40) | 1.17 | (0.93, 1.45) | 1.15 | (0.73, 1.80) |
| >High school | 0.12 | 0.08 | 0.812 | 1.10 | (0.90, 1.35) | 0.94 | (0.79, 1.12) | 0.77 | (0.53, 1.11) |
| Women | |||||||||
| Ethnicity | |||||||||
| NH white | ref | 1.00 | 1.00 | 1.00 | |||||
| NH black | −0.61 | 0.05 | < 0.001 | 2.08 | (1.84, 2.37)2 | 1.91 | (1.62, 2.26)2 | 0.94 | (0.64, 1.39) |
| Mex Am | −0.14 | 0.06 | 0.025 | 1.25 | (1.03, 1.52)2 | 1.36 | (1.08, 1.72)2 | 1.16 | (0.74, 1.80) |
| Other | −0.32 | 0.07 | < 0.001 | 0.76 | (0.59, 0.98)2 | 0.87 | (0.69, 1.10) | 1.23 | (0.77, 1.98) |
| Income (poverty income ratio) | |||||||||
| Low | ref | 1.00 | 1.00 | 1.00 | |||||
| Medium | 0.11 | 0.06 | 0.111 | 0.97 | (0.79, 1.18) | 1.00 | (0.81, 1.22) | 0.60 | (0.35, 1.03) |
| High | 0.10 | 0.05 | 0.050 | 0.76 | (0.65, 0.89)2 | 0.72 | (0.60, 0.85)2 | 0.55 | (0.36, 0.82)2 |
| Education | |||||||||
| < High school | ref | 1.00 | 1.00 | 1.00 | |||||
| High school | 0.11 | 0.06 | 0.060 | 0.95 | (0.79, 1.12) | 0.88 | (0.75, 1.03) | 0.85 | (0.53, 1.35) |
| >High school | 0.23 | 0.05 | < 0.01 | 0.78 | (0.65, 0.94)2 | 0.72 | (0.60, 0.87)2 | 0.55 | (0.35, 0.86)2 |
See Table 1 for definitions of obesity, central obesity, and metabolic syndrome. These are based on multivariate regression analyses. Linear regression was conducted for dairy consumption, and logistic regression was conducted for obesity, central obesity, and metabolic syndrome. All models were controlled for age. NHANES, National Health and Nutrition Examination Survey; OR, odds ratio; Ref, reference; NH, non-Hispanic; Mex Am, Mexican American.
P < 0.05 for the null hypothesis that the regression coefficient β =0 or that the regression coefficient corresponding to OR is equal to zero.
The association between intakes of dairy and related nutrients and obesity and MetS outcomes
Multivariate logistic regression models suggested that there was overall a net increase of 5% in prevalence of central obesity for each dairy serving among men (borderline significant likelihood ratio test for interaction between sex and dairy servings; P < 0.10) (Table 3). The same pattern of association was found among NH whites and other ethnic groups. On average, each serving of dairy products increased the risk of MetS by 8% among men. This association remained significant after controlling for other major food groups, such as grains, meat, fruits and vegetables, alcohol, caffeine, and added sugars.
TABLE 3.
Associations between dairy consumption, related nutrients, obesity, central obesity, and metabolic syndrome: NHANES, 1999–20041
| Obesity (BMI ≥ 30)
|
Central obesity2 |
Metabolic syndrome3 |
||||
|---|---|---|---|---|---|---|
| Dairy consumption | OR | 95% CI | OR | 95% CI | OR | 95% CI |
| Model 1: all dairy (servings) | ||||||
| Among all subjects | 1.02 | (0.98, 1.06) | 1.03 | (1.00, 1.07) | 1.05 | (0.97, 1.14) |
| Stratified analysis | ||||||
| Men | 1.04 | (0.99, 1.09) | 1.05 | (1.01, 1.09)4,5 | 1.08 | (1.00, 1.17)4 |
| Women | 0.98 | (0.93, 1.03) | 1.01 | (0.97, 1.06) | 0.99 | (0.86, 1.14) |
| NH white | 1.02 | (0.98, 1.07) | 1.04 | (1.00, 1.08)4 | 1.05 | (0.95, 1.16) |
| NH black | 0.99 | (0.91, 1.07) | 1.01 | (0.94, 1.08) | 1.02 | (0.78, 1.33) |
| Mex Am | 0.95 | (0.88, 1.01) | 1.02 | (0.96, 1.08) | 0.99 | (0.90, 1.09) |
| Other ethnicity | 1.02 | (0.98, 1.06) | 1.05 | (1.01, 1.08)4 | 1.05 | (0.97, 1.14) |
| Model 2: dairy food groups | ||||||
| Among all subjects | ||||||
| Whole milk (per 100 g) | 0.97 | (0.94, 1.00) | 0.96 | (0.94, 0.99)4 | 0.98 | (0.90, 1.07) |
| Low fat milk (per 100 g) | 1.01 | (0.98, 1.03) | 1.03 | (1.00, 1.06)4 | 1.02 | (0.97, 1.06) |
| Skim milk (per 100 g) | 0.97 | (0.93, 1.02) | 1.00 | (0.97, 1.03) | 1.01 | (0.94, 1.10) |
| Cheese (servings) | 1.14 | (1.08, 1.21)4 | 1.11 | (1.05, 1.17)4 | 1.16 | (1.04, 1.29)4 |
| Yogurt (servings) | 0.51 | (0.36, 0.73)4 | 0.51 | (0.37, 0.70)4 | 0.40 | (0.18, 0.89)4 |
| NH blacks | ||||||
| Whole milk (per 100 g) | 0.95 | (0.89, 1.01) | 0.95 | (0.90, 1.00) | 0.99 | (0.87, 1.12) |
| Low fat milk (per 100 g) | 1.00 | (0.95, 1.05) | 1.01 | (0.97, 1.05) | 0.91 | (0.75, 1.10) |
| Skim milk (per 100 g) | 0.95 | (0.85, 1.09) | 0.96 | (0.85, 1.08) | 1.05 | (0.88, 1.26) |
| Cheese (servings) | 1.08 | (0.93, 1.25) | 1.11 | (0.98, 1.25) | 1.13 | (0.72, 1.79) |
| Yogurt (servings) | 0.89 | (0.35, 2.23) | 1.00 | (0.39, 2.56) | 0.17 | (0.00, 7.88) |
| Dairy-related nutrients | ||||||
| Model 3 | ||||||
| Among all subjects | ||||||
| Calcium (per 100 mg) | 0.97 | (0.95, 0.99)4 | 0.99 | (0.97, 1.00) | 0.99 | (0.95, 1.02) |
| Phosphorus (per 100 mg) | 1.07 | (1.05, 1.10)4 | 1.06 | (1.03, 1.08)4 | 1.05 | (1.01, 1.10)4 |
| Magnesium (per 100 mg) | 0.78 | (0.72, 0.85)4 | 0.80 | (0.76, 0.85)4 | 0.83 | (0.72, 0.96)4 |
| Dairy fatty acids (g/100 g fat) | 1.02 | (0.98, 1.05) | 1.00 | (0.96, 1.04) | 1.03 | (0.96, 1.11) |
| NH blacks | ||||||
| Calcium (per 100 mg) | 1.01 | (0.98, 1.05) | 1.01 | (0.98, 1.04) | 1.07 | (0.93, 1.23) |
| Phosphorus (per 100 mg) | 1.00 | (0.96, 1.04) | 1.02 | (0.97, 1.06) | 0.88 | (0.79, 0.98)4 |
| Magnesium (per 100 mg) | 0.87 | (0.76, 0.97)4 | 0.83 | (0.73, 0.94)4 | 0.89 | (0.64, 1.24) |
| Dairy fatty acids (g/100 g fat) | 0.95 | (0.89, 1.02) | 0.96 | (0.89, 1.03) | 0.96 | (0.80, 1.15) |
| Testing mediation effects by nutrients among all subjects | ||||||
| Model 4: Compared to model 2 | ||||||
| Whole milk (per 100 g) | 0.95 | (0.92, 0.99)4 | 0.95 | (0.92, 0.99)4 | 0.97 | (0.88, 1.07) |
| Low fat milk (per 100 g) | 0.99 | (0.96, 1.02) | 1.02 | (0.99, 1.06) | 1.01 | (0.95, 1.07) |
| Skim milk (per 100 g) | 0.96 | (0.92, 0.99)4 | 0.99 | (0.96, 1.02) | 1.01 | (0.89, 1.15) |
| Cheese (servings) | 1.07 | (0.99, 1.15) | 1.05 | (0.96, 1.15) | 1.10 | (0.89, 1.35) |
| Yogurt (servings) | 0.51 | (0.36, 0.72)4 | 0.51 | (0.37, 0.71)4 | 0.41 | (0.17, 1.01) |
| Calcium (per 100 mg) | 0.98 | (0.96, 1.00) | 0.99 | (0.96, 1.01) | 0.98 | (0.93, 1.03) |
| Phosphorus (per 100 mg) | 1.08 | (1.05, 1.10)4 | 1.06 | (1.03, 1.08)4 | 1.05 | (1.00, 1.10)4 |
| Magnesium (per 100 mg) | 0.80 | (0.74, 0.86)4 | 0.82 | (0.77, 0.86)4 | 0.85 | (0.74, 0.99)4 |
| Dairy fatty acids (g/100 g fat) | 1.00 | (0.96, 1.04) | 0.99 | (0.95, 1.03) | 1.02 | (0.94, 1.12) |
| Testing confounding by related dietary patterns among all subjects6 | ||||||
| Model 5: Compared to model 2 | ||||||
| Whole milk (per 100 g) | 0.97 | (0.93, 1.00) | 0.96 | (0.93, 0.98)4 | 0.98 | (0.89, 1.08) |
| Low fat milk (per 100 g) | 1.01 | (0.98, 1.04) | 1.04 | (1.01, 1.07)4 | 1.01 | (0.96, 1.06) |
| Skim milk (per 100 g) | 0.98 | (0.94, 1.03) | 1.02 | (0.99, 1.05) | 1.01 | (0.94, 1.09) |
| Cheese (servings) | 1.12 | (1.05, 1.19)4 | 1.06 | (0.99, 1.12) | 1.17 | (1.06, 1.29)4 |
| Yogurt (servings) | 0.57 | (0.40, 0.82)4 | 0.58 | (0.42, 0.81)4 | 0.42 | (0.18, 1.00) |
NHANES, National Health and Nutrition Examination Survey; OR, odds ratio; NH, non-Hispanic; Mex Am, Mexican American. Each model controls for age, sex, ethnicity, socioeconomic status (education and poverty income ratio), energy intake, and physical activity. Additional models derived from model 4 showed that adding only calcium in the model explained the effect of yogurt on metabolic syndrome. Adding only magnesium or phosphorus retained the significance of both the effects of cheese and yogurt. The effect of cheese was explained away by phosphorus and magnesium. Those two nutrients did not explain the effect of yogurt on their own. The effect of low-fat milk on central obesity is explained away by magnesium. The effects of low-fat and whole milk on central obesity were not explained away by other food groups.
Defined as waist circumference > 102 for men and > 88 for women.
Based on NCEP ATP III described in Methods.
P < 0.05 for the null hypothesis that the regression coefficient corresponding to the OR is equal to zero.
P < 0.10 for interaction between subgroup variable and total dairy intake (servings) in affecting outcome.
Model 5 is model 2 for all subjects after control for selected major food group servings: total fruit, dark green vegetables, deep yellow vegetables, whole grains, nonwhole grains, legumes, nuts/seeds, soy, total meat/poultry/fish, eggs, grams of discretionary solid fat, grams of discretionary oils, added sugars (teaspoons), alcoholic beverages (servings), and mg of caffeine.
In model 2, a distinction between high-fat and low-fat dairy products was drawn, and milk consumption was expressed in 100-g increments. Using this model, whole milk was weakly and negatively associated with the prevalence of central obesity, whereas low-fat milk had the opposite effect. Each serving of yogurt was associated on average with a 2- to 2.5-fold reduction in the prevalence odds of obesity, central obesity, and MetS. Cheese seemed to be associated with a higher prevalence of our main outcomes, on average by 11–16%. None of the significant associations between dairy consumption and metabolic outcomes that were found in the total population were observed among NH blacks.
In terms of dairy-related nutrients, model 3 suggested that magnesium and, to a much lesser extent, calcium were inversely related, and phosphorus was positively related, to poor metabolic profiles. An increase in calcium intake by 100 mg was associated with a 3% reduction in the prevalence odds of obesity, whereas the same increment in magnesium was associated with a greater magnitude of reduction in the prevalence of obesity (OR: 0.78; 95% CI: 0.72, 0.85), central obesity (OR: 0.80; 95% CI: 0.76, 0.85), and MetS (OR: 0.83; 95% CI: 0.72, 0.96). In contrast, each 100-mg daily intake of phosphorus was associated with a 5–7% increase in prevalence of all 3 outcomes. Among NH blacks, magnesium was inversely associated with obesity and central obesity, whereas phosphorus was inversely associated with MetS (OR: 0.88; 95% CI: 0.79, 0.98).
Furthermore, in model 5, we adjusted for additional food groups (eg, total fruits, dark green vegetables, whole grains, and added sugars) to assess confounding of the relation between dairy consumption and binary metabolic outcomes by associated dietary patterns. A major portion of the association between yogurt and MetS appeared to be explained by other dietary patterns, mainly lower intake of eggs and higher intake of dark green vegetables and caffeine. Similarly, the positive association between cheese and central obesity was partly explained by reduced intake of fruits and increased intake of animal-source foods, discretionary solid fat and oils. All other associations remained significant.
When dairy consumption and related nutrients were examined in relation to individual metabolic outcomes as continuous variables (Table 4), we found that among all subjects, and among men in particular, fluid milk (servings) was inversely related to blood pressure (SBP and DBP) but was not associated with the other metabolic outcomes. Yogurt was associated with better metabolic outcomes, ie, reduced BMI, WC, SBP, and fasting glucose for both genders, reduced total number of disturbances in the whole sample, and increased HDL cholesterol level among women. In contrast, cheese was positively associated with BMI, WC, SBP, and fasting glucose level in the whole sample, and with total number of disturbances among men, but none of the associations with cheese were significant among women. Dietary phosphorus was associated with increased BMI, WC, fasting glucose, reduced HDL cholesterol, and increased number of metabolic disturbances for both sexes combined. In contrast, magnesium was associated with an overall better metabolic profile, including reduced BMI, WC, DBP, fasting glucose and TAs, increased HDL cholesterol, and reduced number of metabolic disturbances, with few sex differences.
TABLE 4.
Associations between dairy consumption, related nutrients, and metabolic disturbances: NHANES, 1999–20041
| All subjects | BMI | Waist circumference | Systolic blood pressure | Diastolic blood pressure | Fasting glucose | Fasting tricaylglycerols | HDL-C | No. of disturbances2 |
|---|---|---|---|---|---|---|---|---|
| Model 1 | ||||||||
| All fluid milk (servings) | −0.03 | 0.01 | −0.383 | −0.223 | −0.31 | 0.24 | −0.21 | −0.01 |
| Cheese (servings) | 0.383 | 0.923 | 0.603 | 0.363 | 1.043 | 1.19 | −0.14 | 0.05 |
| Yogurt (servings) | −1.543 | −4.053 | −1.873 | −0.79 | −4.293 | −21.403 | 1.843 | −0.473 |
| Model 2 | ||||||||
| Calcium (per 100 mg) | −0.05 | −0.09 | 0.02 | 0.00 | −0.13 | −0.43 | −0.05 | −0.01 |
| Phosphorus (per 100 mg) | 0.203 | 0.453 | −0.04 | −0.03 | 0.343 | 0.46 | −0.133 | 0.023 |
| Magnesium (per 100 mg) | −0.643 | −1.523 | −0.453 | −0.493 | −0.823 | −5.273 | 1.173 | −0.123 |
| Dairy fat (g/100 g fat) | −0.03 | −0.02 | −0.04 | 0.11 | 0.02 | 2.03 | −0.13 | 0.01 |
| Men | ||||||||
| Model 1 | ||||||||
| All fluid milk (servings) | −0.01 | 0.01 | −0.403 | −0.17 | −0.43 | 1.00 | −0.21 | −0.01 |
| Cheese (servings) | 0.413 | 1.063 | 0.493 | 0.39 | 1.39 | 4.13 | −0.26 | 0.073 |
| Yogurt (servings) | −0.813 | −2.983 | −2.053 | −0.90 | −7.393 | −42.373 | 0.22 | −0.493 |
| Model 2 | ||||||||
| Calcium (per 100 mg) | −0.03 | −0.06 | −0.01 | 0.03 | −0.10 | −0.05 | −0.06 | 0.00 |
| Phosphorus (per 100 mg) | 0.163 | 0.393 | −0.04 | −0.03 | 0.26 | 0.82 | −0.183 | 0.02 |
| Magnesium (per 100 mg) | −0.413 | −1.193 | −0.39 | −0.623 | −0.54 | −8.353 | 1.013 | −0.123 |
| Dairy fat (pg/100 g fat) | −0.04 | 0.07 | 0.07 | 0.06 | 0.12 | 2.15 | −0.04 | −0.02 |
| Women | ||||||||
| Model 1 | ||||||||
| All fluid milk (servings) | 0.09 | −0.03 | 0.22 | −0.28 | −0.18 | −0.35 | −0.18 | −0.01 |
| Cheese (servings) | 0.31 | 0.75 | 0.50 | 0.28 | 0.49 | −4.42 | 0.02 | 0.00 |
| Yogurt (servings) | −2.213 | −5.013 | 2.00 | −0.78 | −2.92 | −11.33 | 3.643 | −0.453 |
| Model 2 | ||||||||
| Calcium (per 100 mg) | −0.103 | −0.173 | 0.00 | −0.06 | −0.21 | −0.81 | −0.07 | −0.02 |
| Phosphorus (per 100 mg) | 0.283 | 0.543 | −0.02 | −0.03 | 0.48 | −0.43 | −0.02 | 0.03 |
| Magnesium (per 100 mg) | −1.023 | −2.073 | −0.31 | −0.29 | −1.36 | 0.71 | 1.493 | −0.123 |
| Dairy fat (pg/100 g fat) | −0.03 | −0.01 | −0.04 | 0.18 | 0.03 | 2.37 | −0.21 | 0.04 |
NHANES, National Health and Nutrition Examination Survey; HDL-C, HDL cholesterol. Each model controlled for age, sex, ethnicity, socioeconomic status (education and poverty income ratio), energy intake, and physical activity.
Based on NCEP ATP III criteria described in Methods.
P < 0.05 for null hypothesis that β = 0.
We also tested whether the associations between dairy consumption and metabolic outcomes would hold when adjusted for overall dietary quality resulting from intakes of food groups and nutrients other than dairy foods based on the dietary intake guidelines for Americans (using the revised US Department of Agriculture Healthy Eating Index components) (50). After control for the total score of the nondairy components of the new Healthy Eating Index in addition to energy intake and demographic factors, our models indicated that the most of the associations shown in Table 4 did not change, although it seemed that some of those between dairy products and HDL cholesterol or TAs were mainly explained by associated healthy dietary patterns (Appendix A).
Are the associations of dairy products with obesity and related metabolic outcomes mainly because of their nutrient content?
We assessed whether the associations between total or individual dairy products and various metabolic outcomes were mediated by their nutrient content (Table 3, model 4). Some dairy-related nutrients appeared to mediate the association between dairy consumption on some metabolic outcomes. In fact, additional analysis suggested that the weak positive association between low-fat milk and central obesity was apparently mediated by magnesium intake. Moreover, whereas the effect of yogurt remained significant after introducing related nutrients into the model, in the case of obesity and central obesity, the effect became nonsignificant for MetS. Additional analyses suggested that calcium was sufficient to explain away the effect of yogurt on the risk of MetS. In contrast, the effect of cheese on MetS may be mediated through variations in magnesium and phosphorus intakes. All 3 nutrients were needed to explain the association between cheese and central obesity. All analyses were conducted again after excluding women who were pregnant or lactating at the time of the survey and results were similar, leading to the same overall conclusions.
Are ethnic disparities in the prevalence of obesity and metabolic disorders mediated by differences in dairy consumption?
Our SMs reveal that among total effects that were statistically significant, differences in BMI between the NH black and NH whites (β = 1.70; P < 0.05) were explained in small but significant part by dairy-related nutrients, particularly higher intakes of calcium and magnesium, and lower phosphorus intake (MP = 5.29). In terms of SBP, differences between NH blacks and NH whites (β = 5.5; P < 0.05) were also partly explained by dairy-related nutrients with a borderline significant MP = 5.60. A similar pattern was observed for SBP when comparing Mexican Americans with NH whites (β = 1.81; P < 0.05) with an even more significant MP = −11.0. None of the other statistically significant ethnic contrasts were appreciably explained by either dairy products or dairy-related nutrients (Table 5).
TABLE 5.
Total, direct, and indirect effect of race/ethnicity on selected metabolic outcomes through dairy consumption and related nutrients based on structural equation models, NHANES, 1999–20041
| Race/ethnicity (NH black compared with NH white)
|
Race/ethnicity (Mex Am compared with NH white)
|
Race/ethnicity (others compared with NH white)
|
||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Total α1+ Σ (α2 × α3) | Direct α1 | Indirect α2 × α3 | MP Σ indirect × 100/total | Total α1 + Σ (α2 × α3) | Direct α1 | Indirect α2 × α3 | MP Σ indirect × 100/total | Total α1 + Σ (α2 × α3) | Direct α1 | Indirect α2 × α3 | MP Σ indirect × 100/total | |
| BMI | ||||||||||||
| Model 1a | ||||||||||||
| All fluid milk (servings) | 1.71 | 1.66 | 0.06 | 2.92 | 0.81 | 0.80 | 0.02 | 1.21 | −0.44 | −0.44 | 0.03 | 0.00 |
| Cheese (servings) | —2 | —2 | −0.04 | —2 | —2 | −0.03 | —2 | —2 | −0.03 | |||
| Yogurt (servings) | 0.03 | 0.02 | 0.00 | |||||||||
| Model 2a | ||||||||||||
| Calcium (per 100 mg) | 1.70 | 1.61 | 0.16 | 5.29 | 0.80 | 1.81 | 0.03 | −1.25 | −0.39 | −0.40 | 0.09 | 2.56 |
| Phosphorus (per 100 mg) | —2 | —2 | −0.26 | —3 | —2 | —2 | 0.08 | —2 | —2 | −0.09 | ||
| Magnesium (per 100 mg) | 0.19 | −0.12 | 0.01 | |||||||||
| Waist circumference | ||||||||||||
| Model 1b | ||||||||||||
| All fluid milk (servings) | 0.49 | 0.45 | 0.05 | 8.16 | 0.074 | 0.070 | 0.017 | 5.40 | −2.86 | −2.85 | 0.03 | 0.35 |
| Cheese (servings) | NS | NS | −0.09 | —3 | NS | NS | −0.069 | —3 | —2 | —2 | −0.08 | |
| Yogurt (servings) | 0.08 | 0.056 | 0.04 | |||||||||
| Model 2b | ||||||||||||
| Calcium (per 100 mg) | 0.47 | 0.30 | 0.28 | 36.17 | 0.070 | 0.12 | 0.05 | −71.43 | −2.88 | −2.75 | 0.17 | 4.51 |
| Phosphorus (per 100 mg) | NS | NS | −0.59 | —4 | NS | NS | 0.19 | —4 | —2 | —2 | −0.21 | |
| Magnesium (per 100 mg) | 0.48 | −0.29 | −0.09 | |||||||||
| Systolic blood pressure | ||||||||||||
| Model 1c | ||||||||||||
| All fluid milk (servings) | 5.51 | 5.42 | 0.16 | 1.63 | 1.81 | 1.79 | 0.05 | 1.10 | 2.00 | 1.97 | 0.08 | 1.50 |
| Cheese (servings) | —2 | —2 | −0.13 | —2 | —2 | −0.07 | —2 | —2 | −0.08 | |||
| Yogurt (servings) | 0.06 | 0.04 | 0.03 | |||||||||
| Model 2c | ||||||||||||
| Calcium (per 100 mg) | 5.53 | 5.22 | −0.08 | 5.60 | 1.81 | 2.01 | −0.01 | −11.05 | 1.99 | 2.01 | −0.05 | −1.00 |
| Phosphorus (per 100 mg) | —2 | —2 | 0.18 | —3 | —2 | —2 | −0.06 | —4 | —2 | —2 | 0.07 | |
| Magnesium (per 100 mg) | 0.21 | −0.13 | −0.04 | |||||||||
| Fasting blood glucose | ||||||||||||
| Model 1d | ||||||||||||
| All fluid milk (servings) | 4.19 | 4.25 | 0.01 | −1.43 | 6.72 | 6.83 | 0.00 | −1.64 | 4.87 | 5.00 | 0.00 | −2.67 |
| Cheese (servings) | —2 | —2 | −0.20 | —2 | —2 | −0.18 | —2 | —2 | −0.19 | |||
| Yogurt (servings) | 0.13 | 0.07 | 0.06 | |||||||||
| Model 2d | ||||||||||||
| Calcium (per 100 mg) | 4.21 | 4.34 | 0.60 | −3.09 | 6.31 | 6.50 | −0.17 | −3.01 | 4.87 | 4.91 | 0.38 | 0.82 |
| Phosphorus (per 100 mg) | —2 | —2 | −1.08 | —2 | —2 | 0.25 | —2 | —2 | −0.40 | |||
| Magnesium (per 100 mg) | 0.35 | −0.27 | −0.02 | |||||||||
| Number of metabolic disturbances | ||||||||||||
| Model 1e | ||||||||||||
| All fluid milk (servings) | −0.08 | −0.09 | 0.01 | −12.50 | 0.19 | 0.19 | 0.00 | 0.00 | 0.44 | 0.16 | 0.00 | 63.6 |
| Cheese (servings) | NS | NS | −0.01 | —4 | NS | NS | −0.01 | NS | NS | −0.00 | —4 | |
| Yogurt (servings) | 0.01 | 0.01 | 0.28 | |||||||||
| Model 2e | ||||||||||||
| Calcium (per 100 mg) | −0.07 | −0.10 | 0.03 | −42.86 | 0.19 | 0.19 | 0.01 | 0.00 | 0.16 | 0.16 | 0.02 | 0.00 |
| Phosphorus (per 100 mg) | NS | NS | −0.04 | —4 | NS | NS | 0.01 | NS | −0.02 | 0.00 | ||
| Magnesium (per 100 mg) | 0.04 | −0.02 | 0.00 | |||||||||
All models were adjusted for age, sex, education, and poverty income ratio as exogenous variables and for energy intake as an endogenous variable. NHANES, National Health and Nutrition Examination Survey; MP, mediation proportion; NH, non-Hispanic; Mex Am, Mexican American.
P < 0.05 for null hypothesis that total effect β = 0. NS, P >0.05 for null hypothesis that total effect β = 0.
MP >5%.
MP >10%.
DISCUSSION
Our findings, based on the most recent nationally representative US data, revealed a significant inverse association between consumption of dairy products and their related nutrients, particularly milk, yogurt, calcium, and magnesium, and health outcomes such as obesity, central obesity, and MetS. However, there was a positive association with cheese consumption and phosphorus intake. For example, the ORs between cheese consumption (servings) and obesity, central obesity, and MetS were 1.14 (1.08, 1.21), 1.11 (1.05, 1.17), and 1.16 (1.04, 1.29), respectively; whereas the corresponding ORs for yogurt consumption (servings) were 0.51 (0.36, 0.73), 0.51 (0.37, 0.70), and 0.40 (0.18, 0.89), respectively. The risks of these conditions were ~20% lower with a 100 mg/d increase in magnesium intake. Compared with magnesium, which appeared to mediate the association between low-fat milk intake and central obesity, calcium had a weak inverse association with obesity but may be mediating the association between yogurt and MetS. The combined variation of magnesium and phosphorus mediated the effect of cheese on MetS. When considering individual metabolic components of MetS, milk was significantly associated with reduced SBP and DBP. All other types of dairy products and related nutrients yielded similar results to those observed with binary outcomes, particularly cheese, yogurt, phosphorus, and magnesium.
Our study confirmed some previous findings regarding the beneficial effects of dairy and calcium intake. Our results also highlight the complex relation between dairy foods and obesity and metabolic outcomes. For example, the beneficial effect was not detected when all types of dairy products were pooled together. In contrast, an adverse effect was detected in some population groups. Among men, dairy increased the risk of central obesity by 5% (per serving) and of MetS by 8%. Consumption of cheese, low-fat milk, and phosphorus were all positively associated with some of these adverse health outcomes as well. Some previous studies reported an inverse association between total dairy intake and adiposity (25, 26, 30, 51, 52) or obesity co-morbidities (27–32, 53), whereas others did not (33, 34). The differences in findings were likely mainly attributable to variations in study samples (including heterogeneity in their ethnic/genetic background, age groups, and dietary consumption, including the types of dairy products), study design, outcome assessment, and data analysis methods. Our study systematically examined the various types of dairy products and related nutrients and included all adult age groups.
Whereas many studies have focused on the calcium component of dairy (25, 26, 28, 34, 51–56), fewer have assessed the effects of dairy fat (26) and magnesium (53, 57–59), and none have examined the effects of phosphorus intake. Our study examined all these conjointly. Unlike our findings, however, one study reported low plasma phosphorus levels in subjects with MetS compared with healthy individuals (60). In general, most studies observed weakly inverse or null associations between calcium and magnesium and obesity, central obesity, and MetS. Whereas calcium was shown to mainly alter adipocyte metabolism, as discussed earlier (51), as well as modulate blood pressure (61), magnesium’s biological pathway involves regulating insulin sensitivity, vascular tone, and blood pressure homeostasis. In addition, intracellular or serum magnesium deficiency is a common feature of both diabetic and hypertensive states, as well as various other cardiovascular and metabolic processes, and aging (62). This result is in line with our findings regarding magnesium’s inverse association with elevated blood pressure and glucose. Its inverse association with obesity and central obesity found in our present study may be attributable to dietary magnesium’s ability to form soaps with fatty acids in the intestine and thus reduce the digestible energy content of the diet (63). Finally, the inverse relation between magnesium and plasma TAs, and its direct positive association with HDL cholesterol can be explained by magnesium’s ability to increase lipoprotein lipase activity, which is involved in the conversion of TAs to HDL cholesterol (64).
A few previous studies made a distinction between various types of dairy products, including 2 recent large prospective cohort studies that found a general pattern of low-fat dairy being protective against type 2 diabetes and MetS (27, 31). Similar to our findings, a recent cross-sectional study among older adults in the Netherlands found that whole milk was associated with lower BMI and WC, whereas low-fat milk had the opposite effect(29). We suspect that in both this study and ours, obese people, when consuming milk, might have modified their eating behaviors and been more likely to choose low-fat milk, whereas thinner subjects were less likely to alter their behavior in response to perceived fat content.
The protective effect of yogurt (23, 24, 65) and adverse effect of cheese (29) have been shown in other selective study samples. The deleterious effect of cheese may be the result of its higher energy density compared with other dairy products (eg, 336 kcal/per 100 g compared with 110 kcal for yogurt and 35–61 kcal for milk of various fat content, skim to whole milk), its elevated content in saturated fat, and possibly its higher phosphorus content compared with other dairy products. Another possible explanation is its high level of sodium compared with other dairy products, which may also trigger excessive intake of high-calorie beverages. Yogurt, a fermented dairy product, was shown to reduce absorption of cholesterol and therefore prevent dyslipidemia (24) and is thought to increase calcium bioavailability through its high acidity (24).
Another main finding of our analysis is that large ethnic disparities exist for intakes of dairy and calcium, and for all metabolic outcomes. In particular, NH blacks had lower dairy intake levels, and the highest prevalence of obesity and central obesity, whereas the “others” ethnic group had the highest mean number of metabolic disturbances. Interestingly, NH blacks had the fewest metabolic disturbances compared with other ethnic groups. The differences appear to be mainly the result of their high HDL cholesterol and low TAs.
Our analyses suggest that ethnic differences in metabolic outcomes, particularly higher BMI among NH blacks and SBP among NH blacks and Mexican Americans compared with NH whites, may be at least in part explained by variations in dairy-related nutrients. Inadequate dairy consumption (< 3 servings) was found in over 80% of the study population, and mean dairy servings were particularly low among minority groups. Only 6.5% of NH blacks and 11.4% of Mexican Americans reported consuming 3 servings or more per day, as recommended. Greater efforts are needed to help all Americans consume more dairy products, in particular the low-fat items, to have adequate calcium and vitamin D intake. The lower intake of dairy products by minority groups, particularly NH blacks, may be the result of their higher prevalence of lactose intolerance compared with NH whites (66). However, this concept has been recently challenged by studies suggesting that NH blacks can consume at least one cup (8 oz) of milk without experiencing symptoms, and that tolerance can be improved by consuming the milk with a meal, choosing yogurt or hard cheeses, or using products that aid in the digestion of lactose (such as lactase supplements or lactose-reduced milks) (67, 68). Our lack of ability to detect significant associations between dairy consumption and binary metabolic outcomes (obesity, central obesity, and MetS) within the NH black group may be partly because of their smaller sample size.
The present study has a number of strengths. First, it is based on recent, nationally representative data of adults in the United States. Our analysis accounted for design complexity and yielded national estimates. Second, this study is one of very few that examined both dairy consumption of varying types and dairy-related nutrients in relation to adiposity and various metabolic outcomes of interest. Third, to our knowledge, this attempt is the first to examine the role of dairy consumption in explaining ethnic disparities in adiposity and metabolic outcomes, through use of structural equations modeling techniques.
A main limitation of this study is its cross-sectional design, which precludes inferences on causation. Residual confounding caused by measurement error in covariates included, or noninclusion of important confounders, as well as possible eating behavior changes as a result of health conditions are possible explanations for some of the associations, including the positive association of low-fat milk with metabolic outcomes. In addition, some recent studies suggest that MetS may not be a clinically useful endpoint to study and that the clustering of disturbances may not be more predictive of cardiovascular disease and mortality than its separate components (69, 70). Many studies, however, still consider MetS an important endpoint that can be targeted and prevented by lifestyle and environmental changes.
In summary, our results suggest that the health effects of dairy products and related nutrients are complex and may not be uniform across the population, at least for obesity and related metabolic disorders. The data also indicate that variations in consumption of dairy products and dairy-related nutrients appear to be factors that account for some of the disparities in risk of obesity and its co-morbidities between major ethnic groups in the US.
APPENDIX A
Associations between dairy consumption, related nutrients, and metabolic outcomes among all subjects, controlling for nondairy components of the Healthy Eating Index: NHANES, 1999–20041
| BMI | WC | SBP | DBP | Fasting glucose |
Fasting triacylglycerols |
HDL-C | No. of disturbances |
|
|---|---|---|---|---|---|---|---|---|
| Model 1 | ||||||||
| All fluid milk (servings) | −0.04 | 0.02 | −0.382 | −0.232 | −0.30 | 0.20 | −0.19 | −0.01 |
| Cheese (servings) | 0.332 | 0.792 | 0.582 | 0.22 | 1.102 | 0.85 | −0.01 | 0.05 |
| Yogurt (servings) | −1.482 | −3.852 | −1.842 | −0.59 | −4.392 | −20.9 | 1.63 | −0.462 |
| Model 2 | ||||||||
| Calcium (per 100 mg) | −0.05 | −0.10 | 0.02 | −0.00 | −0.12 | −0.39 | −0.05 | −0.01 |
| Phosphorous (per 100 mg) | 0.202 | 0.452 | −0.04 | −0.02 | 0.332 | 0.43 | −0.13 | 0.022 |
| Magnesium (per 100 mg) | −0.582 | −1.312 | −0.462 | −0.322 | −0.922 | −6.252 | 1.192 | −0.122 |
| Dairy fat (pg/100 fat) | −0.07 | −0.16 | −0.03 | 0.01 | 0.09 | 2.71 | −0.14 | 0.01 |
NHANES, National Health and Nutrition Examination Survey; WC, waist circumference; SBP, systolic blood pressure; DBP, diastolic blood pressure; HDL-C, HDL cholesterol. Each model controls for those variables controlled in models in Table 4 (ie, age, sex, ethnicity, education, poverty income ratio, energy intake, and physical activity) plus a measure of dietary quality, ie, the new USDA 2005 Healthy Eating Index score excluding the dairy component. Metabolic disturbances were defined based on NCEP ATP III criteria described in Methods.
P < 0.05 for null hypothesis that β = 0.
Footnotes
Supported in part by the Johns Hopkins Center for a Livable Future, the US Department of Agriculture (2044–05322), the National Institute of Diabetes and Digestive and Kidney Diseases/NIH (R01 DK63383), and National Institute of Child Health and Human Development/NIH 1R03HD056073.
The authors’ responsibilities were as follows—MAB: data management; MAB and YW: conceptualization, statistical analysis, literature review, write-up of the manuscript; MAB, TLG, BC, RL, LJC, and YW: interpretation of results; TLG, BC, RL, and LJC: revision of the manuscript. None of the authors had a conflict of interest.
References
- 1.Loucks EB, Magnusson KT, Cook S, Rehkopf DH, Ford ES, Berkman LF. Socioeconomic position and the metabolic syndrome in early, middle, and late life: evidence from NHANES 1999–2002. Ann Epidemiol. 2007;17:782–90. doi: 10.1016/j.annepidem.2007.05.003. [DOI] [PubMed] [Google Scholar]
- 2.Loucks EB, Rehkopf DH, Thurston RC, Kawachi I. Socioeconomic disparities in metabolic syndrome differ by gender: evidence from NHANES III. Ann Epidemiol. 2007;17:19–26. doi: 10.1016/j.annepidem.2006.07.002. [DOI] [PubMed] [Google Scholar]
- 3.Ogden CL, Carroll MD, Curtin LR, McDowell MA, Tabak CJ, Flegal KM. Prevalence of overweight and obesity in the United States, 1999–2004. JAMA. 2006;295:1549–55. doi: 10.1001/jama.295.13.1549. [DOI] [PubMed] [Google Scholar]
- 4.Park YW, Zhu S, Palaniappan L, Heshka S, Carnethon MR, Heymsfield SB. The metabolic syndrome: prevalence and associated risk factor findings in the US population from the Third National Health and Nutrition Examination Survey, 1988–1994. Arch Intern Med. 2003;163:427–36. doi: 10.1001/archinte.163.4.427. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Wang Y, Beydoun MA. The obesity epidemic in the United States–gender, age, socioeconomic, racial/ethnic, and geographic characteristics: a systematic review and meta-regression analysis. Epidemiol Rev. 2007;29:6–28. doi: 10.1093/epirev/mxm007. [DOI] [PubMed] [Google Scholar]
- 6.Appels CW, Vandenbroucke JP. Overweight, obesity, and mortality. N Engl J Med. 2006;355:2699. doi: 10.1056/NEJMc062590. author reply 2700–1. [DOI] [PubMed] [Google Scholar]
- 7.Bender R, Zeeb H, Schwarz M, Jockel KH, Berger M. Causes of death in obesity: relevant increase in cardiovascular but not in all-cancer mortality. J Clin Epidemiol. 2006;59:1064–71. doi: 10.1016/j.jclinepi.2006.01.006. [DOI] [PubMed] [Google Scholar]
- 8.Doig GS. Obesity-related excess mortality: What should we do now? Crit Care Med. 2004;32:1084–5. doi: 10.1097/01.ccm.0000119934.01548.af. [DOI] [PubMed] [Google Scholar]
- 9.Ferrucci L, Alley D. Obesity, disability, and mortality: a puzzling link. Arch Intern Med. 2007;167:750–1. doi: 10.1001/archinte.167.8.750. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Solomon CG, Manson JE. Obesity and mortality: a review of the epidemiologic data. Am J Clin Nutr. 1997;66(suppl):S1044–50. doi: 10.1093/ajcn/66.4.1044S. [DOI] [PubMed] [Google Scholar]
- 11.Stevens J. Obesity and mortality in Africans-Americans. Nutr Rev. 2000;58:346–53. doi: 10.1111/j.1753-4887.2000.tb01832.x. [DOI] [PubMed] [Google Scholar]
- 12.Colditz GA. Economic costs of obesity and inactivity. Med Sci Sports Exerc. 1999;31:S663–7. doi: 10.1097/00005768-199911001-00026. [DOI] [PubMed] [Google Scholar]
- 13.Hill JO, Sallis JF, Peters JC. Economic analysis of eating and physical activity: a next step for research and policy change. Am J Prev Med. 2004;27:111–6. doi: 10.1016/j.amepre.2004.06.010. [DOI] [PubMed] [Google Scholar]
- 14.Wolf AM, Colditz GA. Current estimates of the economic cost of obesity in the United States. Obes Res. 1998;6:97–106. doi: 10.1002/j.1550-8528.1998.tb00322.x. [DOI] [PubMed] [Google Scholar]
- 15.Barr SI. Increased dairy product or calcium intake: is body weight or composition affected in humans? J Nutr. 2003;133(suppl):S245–8. doi: 10.1093/jn/133.1.245S. [DOI] [PubMed] [Google Scholar]
- 16.Bucher HC, Cook RJ, Guyatt GH, et al. Effects of dietary calcium supplementation on blood pressure. A meta-analysis of randomized controlled trials. JAMA. 1996;275:1016–22. doi: 10.1001/jama.1996.03530370054031. [DOI] [PubMed] [Google Scholar]
- 17.Pittas AG, Lau J, Hu FB, Dawson-Hughes B. The role of vitamin D and calcium in type 2 diabetes. A systematic review and meta-analysis. J Clin Endocrinol Metab. 2007;92:2017–29. doi: 10.1210/jc.2007-0298. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Bray GA, Popkin BM. Dietary fat intake does affect obesity! Am J Clin Nutr. 1998;68:1157–73. doi: 10.1093/ajcn/68.6.1157. [DOI] [PubMed] [Google Scholar]
- 19.Heitmann BL, Lissner L, Sorensen TI, Bengtsson C. Dietary fat intake and weight gain in women genetically predisposed for obesity. Am J Clin Nutr. 1995;61:1213–7. doi: 10.1093/ajcn/61.6.1213. [DOI] [PubMed] [Google Scholar]
- 20.Kant AK, Graubard BI, Schatzkin A, Ballard-Barbash R. Proportion of energy intake from fat and subsequent weight change in the NHANES I Epidemiologic Follow-up Study. Am J Clin Nutr. 1995;61:11–7. doi: 10.1093/ajcn/61.1.11. [DOI] [PubMed] [Google Scholar]
- 21.Maron DJ, Fair JM, Haskell WL. Saturated fat intake and insulin resistance in men with coronary artery disease. The Stanford Coronary Risk Intervention Project Investigators and Staff. Circulation. 1991;84:2020–7. doi: 10.1161/01.cir.84.5.2020. [DOI] [PubMed] [Google Scholar]
- 22.Miller WC, Lindeman AK, Wallace J, Niederpruem M. Diet composition, energy intake, and exercise in relation to body fat in men and women. Am J Clin Nutr. 1990;52:426–30. doi: 10.1093/ajcn/52.3.426. [DOI] [PubMed] [Google Scholar]
- 23.Pfeuffer M, Schrezenmeir J. Bioactive substances in milk with properties decreasing risk of cardiovascular diseases. Br J Nutr. 2000;84(suppl 1):S155–9. doi: 10.1017/s0007114500002385. [DOI] [PubMed] [Google Scholar]
- 24.Pfeuffer M, Schrezenmeir J. Milk and the metabolic syndrome. Obes Rev. 2007;8:109–18. doi: 10.1111/j.1467-789X.2006.00265.x. [DOI] [PubMed] [Google Scholar]
- 25.Marques-Vidal P, Goncalves A, Dias CM. Milk intake is inversely related to obesity in men and in young women: data from the Portuguese Health Interview Survey 1998–1999. Int J Obes (Lond) 2006;30:88–93. doi: 10.1038/sj.ijo.0803045. [DOI] [PubMed] [Google Scholar]
- 26.Rosell M, Johansson G, Berglund L, Vessby B, de Faire U, Hellenius ML. Associations between the intake of dairy fat and calcium and abdominal obesity. Int J Obes Relat Metab Disord. 2004;28:1427–34. doi: 10.1038/sj.ijo.0802769. [DOI] [PubMed] [Google Scholar]
- 27.Choi HK, Willett WC, Stampfer MJ, Rimm E, Hu FB. Dairy consumption and risk of type 2 diabetes mellitus in men: a prospective study. Arch Intern Med. 2005;165:997–1003. doi: 10.1001/archinte.165.9.997. [DOI] [PubMed] [Google Scholar]
- 28.Azadbakht L, Mirmiran P, Esmaillzadeh A, Azizi F. Dairy consumption is inversely associated with the prevalence of the metabolic syndrome in Tehranian adults. Am J Clin Nutr. 2005;82:523–30. doi: 10.1093/ajcn.82.3.523. [DOI] [PubMed] [Google Scholar]
- 29.Snijder MB, van der Heijden AA, van Dam RM, et al. Is higher dairy consumption associated with lower body weight and fewer metabolic disturbances? The Hoorn Study. Am J Clin Nutr. 2007;85:989–95. doi: 10.1093/ajcn/85.4.989. [DOI] [PubMed] [Google Scholar]
- 30.Elwood PC, Pickering JE, Fehily AM. Milk and dairy consumption, diabetes and the metabolic syndrome: the Caerphilly prospective study. J Epidemiol Community Health. 2007;61:695–8. doi: 10.1136/jech.2006.053157. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Liu S, Song Y, Ford ES, Manson JE, Buring JE, Ridker PM. Dietary calcium, vitamin D, and the prevalence of metabolic syndrome in middle-aged and older U.S. women. Diabetes Care. 2005;28:2926–32. doi: 10.2337/diacare.28.12.2926. [DOI] [PubMed] [Google Scholar]
- 32.Pereira MA, Jacobs DR, Jr, Van Horn L, Slattery ML, Kartashov AI, Ludwig DS. Dairy consumption, obesity, and the insulin resistance syndrome in young adults: the CARDIA Study. JAMA. 2002;287:2081–9. doi: 10.1001/jama.287.16.2081. [DOI] [PubMed] [Google Scholar]
- 33.Steffen LM, Kroenke CH, Yu X, et al. Associations of plant food, dairy product, and meat intakes with 15-y incidence of elevated blood pressure in young black and white adults: the Coronary Artery Risk Development in Young Adults (CARDIA) Study. Am J Clin Nutr. 2005;82:1169–77. doi: 10.1093/ajcn/82.6.1169. quiz 1363–4. [DOI] [PubMed] [Google Scholar]
- 34.Rajpathak SN, Rimm EB, Rosner B, Willett WC, Hu FB. Calcium and dairy intakes in relation to long-term weight gain in US men. Am J Clin Nutr. 2006;83:559–66. doi: 10.1093/ajcn.83.3.559. [DOI] [PubMed] [Google Scholar]
- 35.Looker AC, Loria CM, Carroll MD, McDowell MA, Johnson CL. Calcium intakes of Mexican Americans, Cubans, Puerto Ricans, non-Hispanic whites, and non-Hispanic blacks in the United States. J Am Diet Assoc. 1993;93:1274–9. doi: 10.1016/0002-8223(93)91954-o. [DOI] [PubMed] [Google Scholar]
- 36.Beydoun MA, Wang Y. How do socio-economic status, perceived economic barriers and nutritional benefits affect quality of dietary intake among US adults? Eur J Clin Nutr. 2008;62:303–13. doi: 10.1038/sj.ejcn.1602700. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Centers for Disease Control and Prevention (CDC) The Third National Health and Nutrition Examination Survey (NHANES III 1988–94) Reference Manuals and Reports (CD-ROM) Bethesda, MD: Centers for Disease Control and Prevention; 1996. [Google Scholar]
- 38.United States Department of Agriculture (USDA), Agriculture Research Service. MyPyramid Equivalents Database for USDA Survey Food Codes Version 1.0. USDA: 2007. [accessed July 2007]. Internet: http://www.ars.usda.gov/Services/docs.htm?docid=8503. [Google Scholar]
- 39.National Academies. Dietary Reference Intakes for Calcium, Phosphorous, Magnesium, Vitamin D, and Fluoride 1997. 1997 Internet: www.nap.edu. [PubMed]
- 40.Centers for Disease Control (CDC) NHANES III anthropometric procedures video, 1988–1994. [accessed September 2007]; Internet: http://www.cdc.gov/nchs/about/major/nhanes/avideo.htm.
- 41.Perloff D, Grim C, Flack J, et al. Human blood pressure determination by sphygmomanometry. Circulation. 1993;88:2460–70. doi: 10.1161/01.cir.88.5.2460. [DOI] [PubMed] [Google Scholar]
- 42.Centers for Disease Control and Prevention (CDC) National Health and Nutrition Examination Survey 2006. Internet: http://www.cdc.gov/nchs/nhanes.htm.
- 43.Williams DE, Collins C. US socioeconomic and racial differences in health: patterns and explanations. Annu Rev Sociol. 1995;21:349–386. [Google Scholar]
- 44.Grundy SM. Hypertriglyceridemia, insulin resistance, and the metabolic syndrome. Am J Cardiol. 1999;83:25F–9F. doi: 10.1016/s0002-9149(99)00211-8. [DOI] [PubMed] [Google Scholar]
- 45.Executive Summary of the Third Report of the National Cholesterol Education Program (NCEP) Expert Panel on Detection, Evaluation, and Treatment of High Blood Cholesterol in Adults (Adult Treatment Panel III) JAMA. 2001;285:2486–97. doi: 10.1001/jama.285.19.2486. [DOI] [PubMed] [Google Scholar]
- 46.Bollen K. Structural equations with latent variables. New York: John Wiley and Sons; 1989. [Google Scholar]
- 47.Ditlevsen S, Christensen U, Lynch J, Damsgaard MT, Keiding N. The mediation proportion: a structural equation approach for estimating the proportion of exposure effect on outcome explained by an intermediate variable. Epidemiology. 2005;16:114–20. doi: 10.1097/01.ede.0000147107.76079.07. [DOI] [PubMed] [Google Scholar]
- 48.Hatcher L. A step-by-step approach to using the SAS system for factor analysis ad structural equation modeling. Cary, NC: SAS Institute; 1994. [Google Scholar]
- 49.STATA. Statistics/data analysis: release 9. 9.0. College Station, TX: Stata Corporation; 2005. [Google Scholar]
- 50.US Department of Agriculture. Healthy Eating Index 2005. Internet: http://www.cnpp.usda.gov/Publications/HEI/healthyeatingindex2005factsheet.pdf.
- 51.Zemel MB, Shi H, Greer B, Dirienzo D, Zemel PC. Regulation of adiposity by dietary calcium. FASEB J. 2000;14:1132–8. [PubMed] [Google Scholar]
- 52.Jacqmain M, Doucet E, Despres JP, Bouchard C, Tremblay A. Calcium intake, body composition, and lipoprotein-lipid concentrations in adults. Am J Clin Nutr. 2003;77:1448–52. doi: 10.1093/ajcn/77.6.1448. [DOI] [PubMed] [Google Scholar]
- 53.He K, Liu K, Daviglus ML, et al. Magnesium intake and incidence of metabolic syndrome among young adults. Circulation. 2006;113:1675–82. doi: 10.1161/CIRCULATIONAHA.105.588327. [DOI] [PubMed] [Google Scholar]
- 54.Boon N, Koppes LL, Saris WH, Van Mechelen W. The relation between calcium intake and body composition in a Dutch population: The Amsterdam Growth and Health Longitudinal Study. Am J Epidemiol. 2005;162:27–32. doi: 10.1093/aje/kwi161. [DOI] [PubMed] [Google Scholar]
- 55.Bowen J, Noakes M, Clifton PM. Effect of calcium and dairy foods in high protein, energy-restricted diets on weight loss and metabolic parameters in overweight adults. Int J Obes (Lond) 2005;29:957–65. doi: 10.1038/sj.ijo.0802895. [DOI] [PubMed] [Google Scholar]
- 56.Gunther CW, Legowski PA, Lyle RM, et al. Dairy products do not lead to alterations in body weight or fat mass in young women in a 1-y intervention. Am J Clin Nutr. 2005;81:751–6. doi: 10.1093/ajcn/81.4.754. [DOI] [PubMed] [Google Scholar]
- 57.Ford ES, Li C, McGuire LC, Mokdad AH, Liu S. Intake of dietary magnesium and the prevalence of the metabolic syndrome among U.S. adults. Obesity (Silver Spring) 2007;15:1139–46. doi: 10.1038/oby.2007.628. [DOI] [PubMed] [Google Scholar]
- 58.Guerrero-Romero F, Rodriguez-Moran M. Low serum magnesium levels and metabolic syndrome. Acta Diabetol. 2002;39:209–13. doi: 10.1007/s005920200036. [DOI] [PubMed] [Google Scholar]
- 59.Song Y, Ridker PM, Manson JE, Cook NR, Buring JE, Liu S. Magnesium intake, C-reactive protein, and the prevalence of metabolic syndrome in middle-aged and older U.S. women. Diabetes Care. 2005;28:1438–44. doi: 10.2337/diacare.28.6.1438. [DOI] [PubMed] [Google Scholar]
- 60.Kalaitzidis R, Tsimihodimos V, Bairaktari E, Siamopoulos KC, Elisaf M. Disturbances of phosphate metabolism: another feature of metabolic syndrome. Am J Kidney Dis. 2005;45:851–8. doi: 10.1053/j.ajkd.2005.01.005. [DOI] [PubMed] [Google Scholar]
- 61.Resnick LM. Cellular calcium and magnesium metabolism in the pathophysiology and treatment of hypertension and related metabolic disorders. Am J Med. 1992;93(suppl):S11–20. doi: 10.1016/0002-9343(92)90290-r. [DOI] [PubMed] [Google Scholar]
- 62.Barbagallo M, Dominguez LJ, Resnick LM. Magnesium metabolism in hypertension and type 2 diabetes mellitus. Am J Ther. 2007;14:375–85. doi: 10.1097/01.mjt.0000209676.91582.46. [DOI] [PubMed] [Google Scholar]
- 63.Drenick EJ. The influence of ingestion of calcium and other soap-forming substances on fecal fat. Gastroenterology. 1961;41:242–4. [PubMed] [Google Scholar]
- 64.Rayssiguier Y, Noe L, Etienne J, Gueux E, Cardot P, Mazur A. Effect of magnesium deficiency on post-heparin lipase activity and tissue lipoprotein lipase in the rat. Lipids. 1991;26:182–6. doi: 10.1007/BF02543968. [DOI] [PubMed] [Google Scholar]
- 65.Zemel MB, Richards J, Mathis S, Milstead A, Gebhardt L, Silva E. Dairy augmentation of total and central fat loss in obese subjects. Int J Obes (Lond) 2005;29:391–7. doi: 10.1038/sj.ijo.0802880. [DOI] [PubMed] [Google Scholar]
- 66.Jackson KA, Savaiano DA. Lactose maldigestion, calcium intake and osteoporosis in African-, Asian-, and Hispanic-Americans. J Am Coll Nutr. 2001;20(suppl):S198–207. doi: 10.1080/07315724.2001.10719032. [DOI] [PubMed] [Google Scholar]
- 67.Jarvis JK, Miller GD. Overcoming the barrier of lactose intolerance to reduce health disparities. J Natl Med Assoc. 2002;94:55–66. [PMC free article] [PubMed] [Google Scholar]
- 68.Byers KG, Savaiano DA. The myth of increased lactose intolerance in African-Americans. J Am Coll Nutr. 2005;24(suppl):S569–73. doi: 10.1080/07315724.2005.10719505. [DOI] [PubMed] [Google Scholar]
- 69.Darsow T, Kendall D, Maggs D. Is the metabolic syndrome a real clinical entity and should it receive drug treatment? Curr Diab Rep. 2006;6:357–64. doi: 10.1007/s11892-006-0006-y. [DOI] [PubMed] [Google Scholar]
- 70.Kahn R. Metabolic syndrome: is it a syndrome? Does it matter? Circulation. 2007;115:1806–10. doi: 10.1161/CIRCULATIONAHA.106.658336. discussion 1811. [DOI] [PubMed] [Google Scholar]


