Abstract
We hypothesized that there are clinically relevant differences in eosinophil integrin expression and activation in patients with asthma. To evaluate this, surface densities and activation states of integrins on eosinophils in blood and bronchoalveolar lavage (BAL) of 19 asthmatic subjects were studied before and 48 h after segmental Ag challenge. At 48 h, there was increased expression of αD and the N29 epitope of activated β1 integrins on blood eosinophils and of αM, β2, and the mAb24 epitope of activated β2 integrins on airway eosinophils. Changes correlated with the late-phase fall in forced expiratory volume in 1 s (FEV1) after whole-lung inhalation of the Ag that was subsequently used in segmental challenge and were greater in subjects defined as dual responders. Increased surface densities of αM and β2 and activation of β2 on airway eosinophils correlated with the concentration of IL-5 in BAL fluid. Activation of β1 and β2 on airway eosinophils correlated with eosinophil percentage in BAL. Thus, eosinophils respond to an allergic stimulus by activation of integrins in a sequence that likely promotes eosinophilic inflammation of the airway. Before challenge, β1 and β2 integrins of circulating eosinophils are in low-activation conformations, and αDβ2 surface expression is low. After Ag challenge, circulating eosinophils adopt a phenotype with activated β1 integrins and upregulated αDβ2, changes that are predicted to facilitate eosinophil arrest on VCAM-1 in bronchial vessels. Finally, eosinophils present in IL-5-rich airway fluid have a hyperadhesive phenotype associated with increased surface expression of αMβ2 and activation of β2 integrins.
Keywords: adhesion molecules, eosinophils, cell trafficking, inflammation, lung, integrins
Introduction
The contribution of eosinophils to certain aspects of asthma, such as airway hyperreponsiveness, remains controversial; but nevertheless there is evidence that eosinophil recruitment to the airway contributes to asthma exacerbations and the chronic character of asthma, by regulating airway inflammation and remodeling (1–15). Thus, the study of how eosinophils traffic from blood to airway is of considerable importance. Integrins, which are versatile cellular adhesion receptors (16, 17), are likely determinants of how eosinophils roll and arrest on lung endothelium; extravasate and migrate through endothelium, underlying basement membrane and tissue to the airway wall; and traverse the bronchial epithelium to the airway lumen (18–20).
Human eosinophils express seven integrin heterodimers, α4β1 (CD49d/CD29), α6β1 (CD49f/CD29), αLβ2 (CD11a/CD18), αMβ2 (CD11b/CD18), αXβ2 (CD11c/CD18), αDβ2 (CD11d/CD18), and α4β7 (CD49d/β7)(13, 21, 22). Each integrin heterodimer interacts with its own set of ligands, which are counter-receptors on other cells or extracellular matrix (ECM) components (17, 23). The functions of an integrin on a given cell is regulated by expression level and activation state (24–26).
We hypothesized that there are clinically relevant differences in eosinophil integrin expression levels and activation state in patients with asthma. The purpose of the present study was to define changes in the expression levels and activation states of blood and airway eosinophil integrins 48 h following segmental allergen challenge in asthmatic subjects, thus reflecting the development of allergic inflammation. Segmental Ag challenge induces a strong local recruitment of eosinophils (27). We found characteristic integrin changes on blood and airway eosinophils; differences between single and dual responder asthmatics; and associations among integrin changes, magnitude of eosinophil recruitment to airway, and IL-5 concentration in bronchoalveolar lavage (BAL)3 fluid. We propose a scenario in which 1) Ag challenge leads to activation of β1 integrins and increased surface expression of αDβ2 on blood eosinophils, 2) such activated eosinophils are more prone to arrest on VCAM-1 (CD106)-bearing endothelium in challenged segments, and 3) IL-5 and other cytokines trigger activation of β2 integrins, contributing to the hyperadhesive phenotype of airway eosinophils.
Materials and Methods
Subjects and screening
Nineteen subjects with mild asthma as diagnosed by an allergist were studied (Table I). These subjects had a history of asthma exacerbation to aeroallergen, PC20 (provocative concentration of methacholine producing a 20% fall in forced expiratory volume in 1 s [FEV1]) < 8 mg/ml, and/or reversibility to β-agonist > 12%. Subjects were screened as described previously with allergen skin prick tests, determination of airway hyperresponsiveness, and spirometry (28). All subjects had a positive skin prick test to one or more aeroallergens, were nonsmokers, did not have a respiratory infection within 30 days of study, and had not received anti-histamines within seven days or corticosteroids within 30 days of study enrollment. The studies were reviewed and approved by the University of Wisconsin-Madison Health Sciences Human Subjects Committee. Informed written consent was obtained from each subject before participation.
Table I.
Subject characteristics
| Screening
|
Whole-lung challenge
|
Segmental challenge
|
||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| FEV1 falla |
Eosinophils in BAL 48 h after challengeb |
|||||||||
| Subject No. | Age (years) | Sexc | Agd | PC20e (mg/ml) | FEV1f (L;% pred.) | AgPD20g (CBU) | Early (% fall) | Late | (%) | total (× 106) |
| Single respondersh | ||||||||||
| 1 | 22 | M | RW | 9.0 | 3.8 ; 90 | 81 | 37 | 0 | 26 | 9 |
| 2 | 21 | F | HDM | 1.2 | 3.2 ; 85 | 6 | 52 | 4 | 37 | 150 |
| 3 | 26 | F | HDM | 1.8 | 4.0 ; 99 | 1 | 26 | 7 | 21 | 7 |
| 4 | 19 | M | CD | 0.7 | 4.1 ; 85 | 85 | 31 | 4 | ND | ND |
| 5 | 19 | F | HDM | 3.5 | 3.7 ; 105 | 118 | 22 | 0 | 47 | 150 |
| 6 | 21 | M | RW | 0.2 | 4.4 ; 93 | 115 | 23 | 0 | 26 | 24 |
| 7 | 20 | M | CD | 5.5 | 4.3 ; 98 | 68 | 44 | 4 | 60 | 83 |
| 8 | 27 | M | HDM | 20 | 3.7 ; 89 | 52 | 35 | 8 | 38 | 24 |
| 9 | 23 | F | CD | 0.4 | 3.0 ; 91 | 2 | 32 | 11 | 24 | 10 |
| 10 | 26 | F | CD | 2.9 | 4.0 ; 109 | 174 | 40 | 7 | 62 | 88 |
| Median: | 22 | 2.4 | 3.9 ; 92 | 34 | 4 | 37 | 24 | |||
| Dual respondersi,j | ||||||||||
| 11 | 30 | F | HDM | 4.8 | 2.9 ; 95 | 11 | 23 | 25 | 72 | 220 |
| 12 | 19 | M | CD | 3.8 | 4.9 ; 96 | 37 | 22 | 17 | 63 | 230 |
| 13 | 20 | F | RW | 0.3 | 2.9 ; 87 | 1 | 48 | 15 | 34 | 29 |
| 14 | 20 | F | HDM | 1.3 | 3.7 ; 99 | 8 | 31 | 15 | 59 | 410 |
| 15 | 18 | M | HDM | 20 | 5.1 ; 104 | 21 | 21 | 15 | 65 | 120 |
| 16 | 22 | M | HDM | 2.5 | 3.9 ; 87 | 31 | 23 | 32 | 74 | 760 |
| 17 | 22 | M | RW | 20 | 4.7 ; 97 | 48 | 35 | 18 | 66 | 420 |
| 18 | 20 | F | HDM | 0.4 | 3.4 ; 96 | 27 | 31 | 21 | 67 | 280 |
| 19 | 23 | M | RW | 20 | 4.4 ; 106 | 52 | 48 | 24 | 55 | 380 |
| Median: | 20 | 3.8 | 3.9 ; 96 | 31 | 18*** | 65** | 280** | |||
FEV1, forced expiratory volume in 1 s.
BAL, bronchoalveolar lavage.
F, female; M, male.
CD, cat dander; HDM, house dust mite; RW, ragweed.
PC20, provocative concentration of methacholine producing a 20% fall in FEV1.
FEV1 values from the screening visit are expressed in liters and as percentage of predicted value (% pred.).
AgPD20, provocative dose of antigen producing a 20% fall in FEV1, expressed in cumulative breath units (CBU).
Single responder, subject with FEV1 fall 3–8 h after whole-lung Ag challenge (late-phase fall) < 15% (Liu et al. 2004, reference No. 28).
Dual responder, subject with FEV1 fall 3–8 h after whole-lung Ag challenge ≥ 15% (Liu et al. 2004, reference No. 28).
**p ≤ 0.01,
p ≤ 0.001 versus single responders.
Determination of provocative allergen dose and early- and late-phase response to whole-lung allergen challenge
At least four weeks before bronchoscopy, a graded whole-lung inhaled Ag challenge was performed as described (28) to determine the provocative dose of Ag producing a 20% fall in FEV1 (AgPD20) and the magnitude of early- and late-phase responses. Briefly, baseline spirometry was performed and repeated after five breaths of saline diluent. If FEV1 remained within 10% of baseline, five breaths of allergen were inhaled and spirometry was repeated 10 min later. Consecutively greater concentrations of allergen were given until FEV1 fell by ≥ 20% from baseline. The maximum immediate or early-phase (within 15–30 min) fall in FEV1 was determined, and subjects were then monitored every 15 min until FEV1 returned to within 10% of baseline. Thereafter, the subjects were monitored at 1 h intervals for 8 h to determine whether a late-phase response was present (28, 29). Subjects having a FEV1 fall ≥ 15% 3–8 h after the whole-lung Ag challenge were considered to have a dual response phenotype; the other subjects were considered single responders (28).
Segmental bronchoprovocation with allergen and BAL
Bronchoscopy, segmental bronchoprovocation with allergen, and subsequent BAL were performed in two different bronchopulmonary segments as described (27, 28, 30). Baseline BAL at 0 h immediately before segmental bronchoprovocation was performed in two segments. Then, a total dose of 30% of the subject’s AgPD20 was administered incrementally to enhance subject safety: 10% of the AgPD20 in the first segment and, when this dose was well tolerated, 20% in the second segment. Forty-eight hours later, a second bronchoscopy was performed by instilling 160 ml of sterile 0.9% NaCl warmed to 37ºC in each segment. BAL fluid recovered from the two segments was pooled for analysis, and the volume of recovered fluid measured.
Antibodies for flow cytometry and ELISA, and recombinant protein standards
Anti-αD integrin mAb 240I (31) was obtained as a gift from ICOS (Bothell, WA). Activation-sensitive β2 integrin mAb24 (32, 33) was a gift from Nancy Hogg (Cancer Research UK London Research Institute, London, UK). Anti-β1 mAb MAR4; anti-β2 L130, anti-β7 Fib504; anti-α4 9F10; anti-α6 GoH3; anti-αL AI111; anti-αX Bly6; PE-conjugated goat anti-mouse and anti-rat IgG; FITC-conjugated anti-CD14 and anti-CD16; isotype controls mouse IgG1, κ (clone A112-2) and rat IgG2a, κ (A110–2); and unlabeled and biotinylated anti-IL-3, anti-IL-5, and anti-IFN-γ mAbs and corresponding recombinant protein standards for ELISA were from BD Biosciences (San Diego, CA). Anti-αM LM11 and activation-sensitive anti-β1 N29 (34) were from Chemicon (Temecula, CA). Unlabeled and biotinylated anti-GM-CSF mAb and recombinant protein standard for ELISA were from R&D Systems (Minneapolis, MN).
Flow cytometry of blood and BAL cell samples
Because purification of blood eosinophils has been found to cause activation of β1 (35), flow cytometry was done on unfractionated blood and BAL cells. Surface expressions of αM, αL, αX, αD, α4, α6, β1, β2, β7, activated β1, and activated β2 were determined. Blood was drawn routinely into lavender-top standard tubes (giving a final EDTA concentration of 1.8 mg/ml)(BD Vacutainer Systems, Franklin Lake, NJ). For determination of mAb24 reactivity, blood was drawn into green-top tubes (giving a final heparin concentration of 14 USP units/ml). Control experiments revealed that the anticoagulant had no effect on the results, except, as reported, for mAb24, whose epitope is not exposed in the presence of EDTA (32). Not all samples were subjected to complete analysis, due to changes in the mAb panel that were made based on ongoing analysis of results from this study and other studies. Originally, we focused on differences between blood and BAL fluid 48 h after segmental challenge. Blood from before segmental challenge was included as a routine later, when we had indications of interesting differences between blood after versus before challenge. Similarly, we originally focused on subunits of the integrins known to be able to bind VCAM-1, α4β1, α4β7, and αDβ2 (13, 21), and added αM later when we had evidence that αMβ2 is involved in the adhesion of purified airway eosinophils to VCAM-1 and other ligands (22, 36). BAL fluid cells were recovered, cytospun, and stained for differential counts as described (29, 30).
EDTA-treated blood (100 μl) was incubated with 0.5 μg primary antibody or isotype control in 100 μl FACS buffer (PBS with 2% BSA and 0.2% NaN3) for 30 min. For mAb24 (and its isotype control), heparin-treated blood was used, since the mAb24 epitope is not exposed in EDTA (32), and was incubated with primary antibody in RPMI 1640 with 10% FBS at 37ºC following the protocol especially designed for mAb24 (37). After primary antibody incubation, samples were washed with 1 ml PBS, washed with 250 μl FACS buffer, and resuspended in 250 μl FACS buffer containing PE-conjugated goat anti-mouse or anti-rat IgG (2 μg/ml). After incubation for 30 min, samples were washed again with PBS, resuspended in 100 μl FACS buffer with a mixture of FITC-conjugated anti-CD14 (0.125 μg) and anti-CD16 (0.625 μg) and incubated for 30 min. Red blood cells were lysed by incubation with 2 ml FACS lysing solution (BD Biosciences) for 10 min, followed by centrifugation. Incubations were at room temperature until after red blood cell lysis and then at 4ºC. Samples were washed with 500 μl FACS buffer, resuspended in 250 μl FACS fixative (1% paraformaldehyde, 67.5 mM sodium cacodylate, 113 mM NaCl, pH 7.2), stored at 4ºC in the dark, and washed with 1 ml PBS and resuspended in 250 μl FACS buffer just prior to data collection. Fixation did not decrease signals (not shown). Data were collected from 30,000 - 170,000 events, using a FACS Calibur (BD Biosciences; available through the Flow Cytometry Facility, Comprehensive Cancer Center, University of Wisconsin-Madison) and Cellquest software (BD Biosciences). Rainbow calibration fluorescent beads (Spherotech, Libertyville, IL) were run each day to calibrate and check the performance of the instrument and channels. Data were analyzed using Cellquest and FlowJo (TreeStar, Ashland, OR). Eosinophils were gated based both on scattering (high side scatter) and lack of staining with anti-CD14 and anti-CD16 (30, 35). Thus, the cells that were analyzed for PE signal fit two criteria for eosinophils by being gated inside both characteristic regions in a plot of side scatter versus FITC staining and a plot of side versus forward scatter. When cells gated on side versus forward scatter were collected by cell sorting followed by cytospin, ≥96% stained for the eosinophil marker eosinophil major basic protein. To analyze circulating neutrophils and monocytes in the same data sets, these leukocyte types were gated inside characteristic regions in the two plots, neutrophils having intermediate side scatter and FITC positivity and monocytes having relatively low side scatter and FITC positivity. Data are expressed as specific geometric mean channel fluorescence (gMCF; specific gMCF = gMCF with a specific integrin mAb – gMCF with isotype control) and as percentage of positive cells (isotype control set with a marker to 2% positive cells) as before (35). For unfractionated BAL cells, flow cytometry was performed with 2 × 105 cells (in 100 μl) and the protocol was as described for blood samples, except that all incubations were at 4ºC and the erythrocyte lysis step was omitted and replaced by a wash with 1 ml PBS.
ELISA for cytokines in BAL fluid
To measure cytokine concentrations, BAL fluid was concentrated ten-fold at 4ºC using a low protein-binding Centriprep® centrifugal filter unit (Millipore, Billerica, MA) with a molecular weight cut-off limit of 3 kDa. A sensitive two-step sandwich ELISA was used as described (38). The assay sensitivities were below 3 pg/ml for IL-5 and GM-CSF, 12 pg/ml for IL-3, and 25 pg/ml for IFN-γ. Values are presented as the concentration in recovered BAL fluid prior to the concentration step.
Statistics
The Mann-Whitney U test was used to compare data between groups. The Spearman rank correlation test was used to analyze correlations. A level of p ≤ 0.05 was considered significant. Analyses were performed using Prism 3.0 (GraphPad, San Diego, CA).
Results
Response to whole-lung allergen challenge
All subjects responded to whole-lung inhaled Ag challenge with a drop in FEV1 within 15–30 min to a median value of 31% early FEV1 fall (Table I). Nine of the 19 subjects had a late-phase response (39), as defined by a FEV1 fall ≥ 15% from baseline 3–8 h after challenge (Table I). Using this criterion (28), the nine subjects were classified as having a dual response phenotype. The other ten subjects were considered single responders (Table I). There were no significant differences between the single and dual responders in regards to age, sex, airway responsiveness to methacholine, FEV1 at the screening visit, or early-phase fall in response to whole-lung Ag challenge (Table I). FEV1 48 h after segmental Ag challenge was decreased minimally compared to before segmental Ag challenge and was not significantly different between single and dual responders (medians 98% and 96% of the value before segmental challenge in single and dual responders, respectively).
Median number of BAL eosinophils after segmental challenge was > tenfold higher in the dual responders than in the single responders (Table I). Median numbers of BAL neutrophils and macrophages, in contrast, were about twofold higher and not significantly different in dual versus single responders (medians in single and dual responders were 3.1 × 106 and 7.5 × 106 neutrophils, and 43 × 106 and 93 × 106 macrophages; respectively). Neutrophil percentage was not significantly different and macrophage percentage was significantly lower in dual responders, due to the increased proportion of eosinophils (medians in single and dual responders were 2.3% and 3.0% neutrophils, and 47% and 22% macrophages; respectively).
Integrin expression on blood and BAL eosinophils after segmental allergen challenge
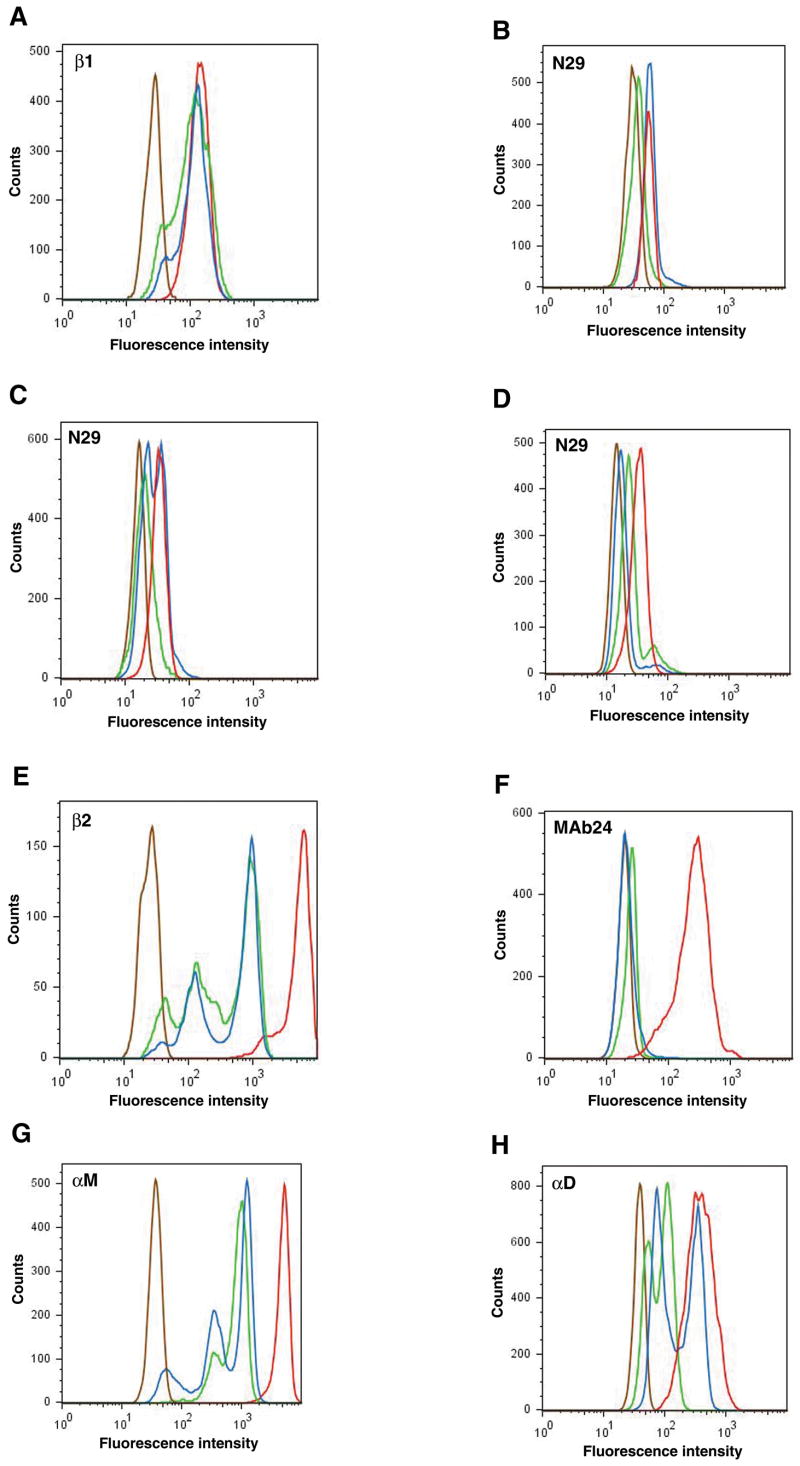
Sample flow cytometry histograms for eosinophil expression of total β1 (A), the activation-sensitive β1 epitope for mAb N29 (B–D), total β2 (E), the activation-sensitive β2 epitope for mAb24 (F), αM (G), and αD (H) are shown in Fig. 1. Expression distributions were homogeneous in some samples (Fig. 1A,B,F) and heterogeneous and asymmetric with one or more “shoulders” in others (Fig. 1C,D,G,H). β1 distributions were mostly heterogeneous on blood eosinophils and uniformly homogeneous on BAL eosinophils (Fig. 1A, Table II). N29 reactivity of blood eosinophils was variable (Fig. 1B–D, Table II). Some samples had two peaks (Fig. 1C) or a “shoulder” of reactivity (Fig. 1D). N29 reactivity of BAL eosinophils was uniformly homogeneous (Fig. 1B–D, Table II). β2 was heterogeneous on blood eosinophils, often with two or several distinct populations; whereas the peak of BAL eosinophils was mostly homogeneous and shifted to the right with higher fluorescence intensity compared to the most positive blood eosinophil population (Fig. 1E, Table II). MAb24 reactivity was homogeneous and low on blood eosinophils and mostly homogeneous and shifted to the right on BAL eosinophils (Fig. 1F, Table II). αM was mostly heterogeneous on blood eosinophils and homogeneous and shifted to the right on BAL eosinophils (Fig. 1G, Table II). αD was mostly heterogeneous on blood eosinophils and mostly homogeneous on BAL eosinophils (Fig. 1H, Table II). Thus, distributions on BAL eosinophils were typically homogeneous; whereas distributions of β1, β2, αM, and αD on blood eosinophils were mostly heterogeneous.
FIGURE 1. Integrin expression on blood and BAL eosinophils before and after segmental Ag challenge.
Representative flow cytometry histograms of expression of total β1 integrin (A), activation-sensitive β1 integrin mAb N29 epitope (B–D), total β2 integrin (E), activation-sensitive β2 integrin mAb24 epitope (F), αM integrin (G), or αD integrin (H) on blood eosinophils before segmental Ag challenge (green), blood eosinophils 48 h after segmental Ag challenge (blue), or BAL eosinophils 48 h after segmental Ag challenge (red); or isotype control (brown). For N29 epitope expression three examples are shown: B; one homogeneous, symmetric peak on blood eosinophils before and after challenge, and higher mean expression level after challenge than before. C; one peak on blood eosinophils before challenge and two peaks after challenge, and higher mean expression level after challenge. D; an asymmetric peak with a “shoulder” to the right on blood eosinophils before and after challenge, and decrease in the size of the “shoulder” and lower mean expression level after challenge. Numbers of individuals with the various patterns are summarized in Table II.
Table II.
Integrin expression on blood and BAL eosinophils as distribution patterns and percentage of positive cells before and 48 h after segmental Ag challengea
| Blood eosinophils before | Blood eosinophils 48 h afterb | BAL eosinophils 48 h afterb,c | ||||||
|---|---|---|---|---|---|---|---|---|
| segmental Ag challenge | ||||||||
| Integrin (mAb) | H/Td | %pose | H/T | %pos. | H/T | %pos. | ||
| All tested subjects | ||||||||
| Total β1 (MAR4) | 2/9 | 82±4 | 6/16 | 80±5 | 17/17 | 85±6 | ||
| Activated β1 (N29) | 8/9 | 54±7 | 11/16 | 57±7 | 15/15 | 55±8 | ||
| Total β2 (L130) | 1/10 | 91±2 | 1/17 | 93±2 | 12/14 | 97±1*,† | ||
| Activated β2 (mAb24) | 3/3 | 23±17 | 3/3 | 18±11 | 10/13 | 65±11 | ||
| αM | 1/4 | 92±5 | 2/12 | 95±3 | 8/9 | 96±3 | ||
| αD | 2/9 | 79 ±2 | 4/15 | 87±6** | 13/16 | 81±8 | ||
| Single respondersf | ||||||||
| Total β1 (MAR4) | 2/5 | 85±7 | 5/9 | 79±8 | 9/9 | 80±9 | ||
| Activated β1 (N29) | 4/5 | 53±11 | 5/9 | 48±10 | 8/8 | 49±10 | ||
| Total β2 (L130) | 1/5 | 94±2 | 0/9 | 92±4 | 6/7 | 95±3 | ||
| Activated β2(mAb24)g | 2/2 | 30±27 | 2/2 | 26±14 | 6/6 | 42±19 | ||
| αM h | 1/2 | 100±0 | 1/7 | 93±4 | 4/5 | 95±5 | ||
| αD | 2/5 | 82±4 | 3/8 | 80±12 | 7/8 | 77±11 | ||
| Dual respondersh,i | ||||||||
| Total β1 (MAR4) | 0/4 | 78±3 | 1/7 | 83±5 | 8/8 | 90±7* | ||
| Activated β1 (N29) | 4/4 | 56±8 | 6/7 | 70±7 | 7/7 | 61±14 | ||
| Total β2 (L130) | 0/5 | 88±4 | 1/8 | 94±2 | 6/7 | 99±0* | ||
| Activated β2(mAb24)g | 1/1 | 8 | 1/1 | 3 | 4/7 | 85±4 | ||
| αM g | 0/2 | 84±4 | 1/5 | 97±1 | 4/4 | 98±2 | ||
| αD | 0/4 | 76±1‡ | 1/7 | 95±2** | 6/8 | 85±11* | ||
BAL, bronchoalveolar lavage.
*p ≤ 0.05,
p ≤ 0.01 versus blood eosinophils before segmental challenge.
†p ≤ 0.05 versus blood eosinophils after segmental challenge.
H, number of subjects with homogeneous expression distribution; T, total number of subjects.
Expression as percentage of positive cells (mean±SEM).
Single responder, subject with FEV1 (forced expiratory volume in 1 s) fall 3–8 h after whole-lung antigen challenge < 15% (Liu et al. 2004, reference No. 28).
Note regarding data with n < 3: These data are within the ranges of our overall data on blood eosinophils from unchallenged donors.
Dual responder, subject with FEV1 fall 3–8 h after whole-lung antigen challenge ≥ 15% (Liu et al. 2004, reference No. 28).
‡p ≤ 0.05 versus single responders.
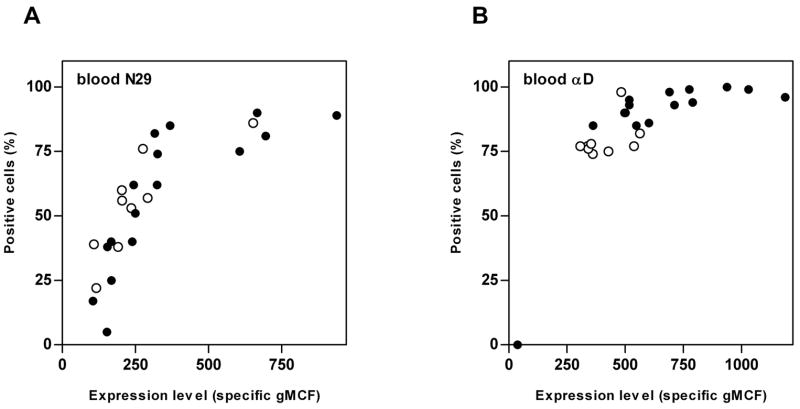
Expression data were scored either as percentage positive cells or as expression level (specific geometric mean channel fluorescence [gMCF])(Table II, Fig. 2). Fig. 3 shows a comparison between the results with these two scoring methods on blood eosinophils for N29 (Fig. 3A) and αD (Fig. 3B). At lower fluorescence intensity, percentage positive cells and expression level correlate; whereas at higher intensity the percentage positive cells plateaus (Fig. 3A,B). Because of its greater dynamic range, specific gMCF was chosen as the more informative measure of integrin expression and activation (Fig. 2). It should be emphasized that for the samples displaying heterogeneous bimodal or multimodal distributions of the β2 integrin subunits, neither gMCF nor percentage positive cells fully captures the complexity of the distributions.
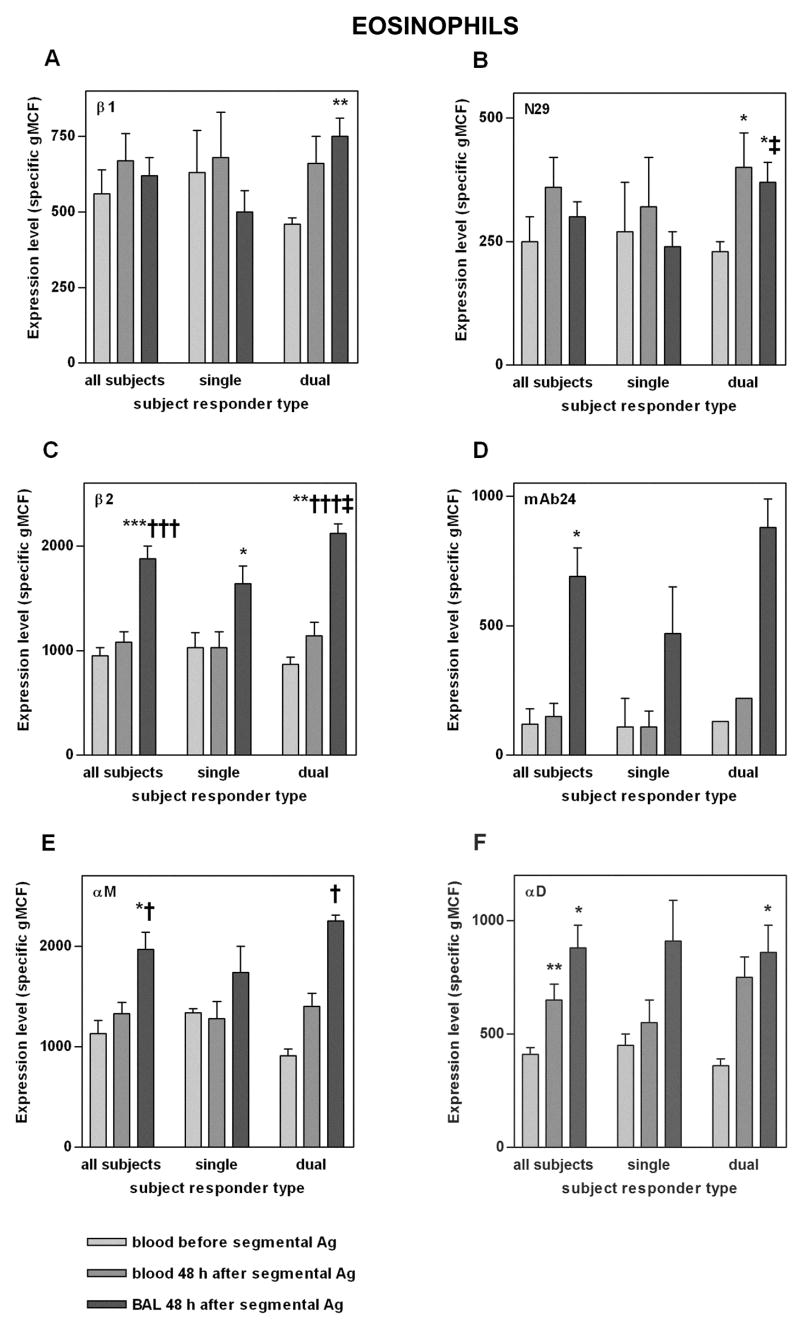
FIGURE 2. Integrin expression on blood and BAL eosinophils before and 48 h after segmental Ag challenge.
Expression of total β1 integrin (A), activation-sensitive β1 integrin mAb N29 epitope (B), total β2 integrin (C), activation-sensitive β2 integrin mAb24 epitope (D), αM integrin (E), and αD integrin (F) on blood eosinophils before segmental Ag challenge (light gray), blood eosinophils 48 h after segmental Ag challenge (medium gray), and BAL eosinophils 48 h after segmental Ag challenge (dark gray) in all tested subjects, single responders, and dual responders. Values shown are specific mean channel fluorescence (gMCF)(mean ± SEM). For n values and expression given as percentage of positive cells, see Table II). *p ≤ 0.05, **p ≤ 0.01, ***p ≤ 0.001 versus blood eosinophils before segmental challenge; †p ≤ 0.05, †††p ≤ 0.001 versus blood eosinophils after segmental challenge; ‡p ≤ 0.05 versus single responders. BAL, bronchoalveolar lavage.
FIGURE 3. Integrin expression on blood eosinophils as percentage of positive cells and expression level.
Blood eosinophil expression of activation-sensitive β1 integrin mAb N29 epitope (A) or αD integrin (B) before (empty symbols) and 48 h after (filled symbols) segmental Ag challenge, expressed as percentage of positive cells (y axes) or as expression level (specific geometric mean channel fluorescence [gMCF])(x axes).
At 48 h after segmental Ag challenge, on circulating eosinophils of dual responders the expression level of the epitope for activation-sensitive anti-β1 mAb N29 (34, 40, 41) was higher 48 h after segmental challenge as compared to before challenge (Fig. 2B). Circulating eosinophils also had significantly higher expression of the αD integrin subunit compared to before segmental challenge (Fig. 2F). αD and N29 epitope expression on blood eosinophils 48 h after segmental challenge correlated with each other (rs [Spearman rank correlation coefficient] = 0.73, p = 0.002). Thus, the results demonstrate that segmental Ag challenge causes significantly increased surface expression of the αD subunit of β2 integrins and, in dual responders, activation of β1 integrins at 48 h on blood eosinophils.
Eosinophils in BAL 48 h after segmental Ag challenge had significantly higher β2 and αM subunit expression than eosinophils in blood obtained before or 48 h after segmental challenge (Fig. 2C,E). The expression of the epitope for the activation-sensitive anti-β2 mAb24 (32, 33) was significantly increased on BAL eosinophils; binding of mAb24 to blood eosinophils was low in blood both before and after segmental challenge (Fig. 2D). αD expression was significantly increased on BAL eosinophils compared to blood eosinophils before challenge but not compared to blood also sampled at 48 h (Fig. 2F). In dual responders, BAL eosinophils had significantly more total and activated β1 (as ascertained by N29) than blood eosinophils before challenge (Fig. 2A,B). α6 expression was lower on BAL eosinophils than on blood eosinophils after challenge (specific gMCF = 560 ± 140 [mean ± SEM] versus 920 ± 110, p ≤ 0.05)(not shown). No differences were found in expression of αL, αX, α4, or β7 (not shown). Overall, the results indicate that αMβ2 is upregulated on BAL eosinophils and BAL eosinophil β2 integrins are in a conformationally altered and activated state compared to on blood eosinophils.
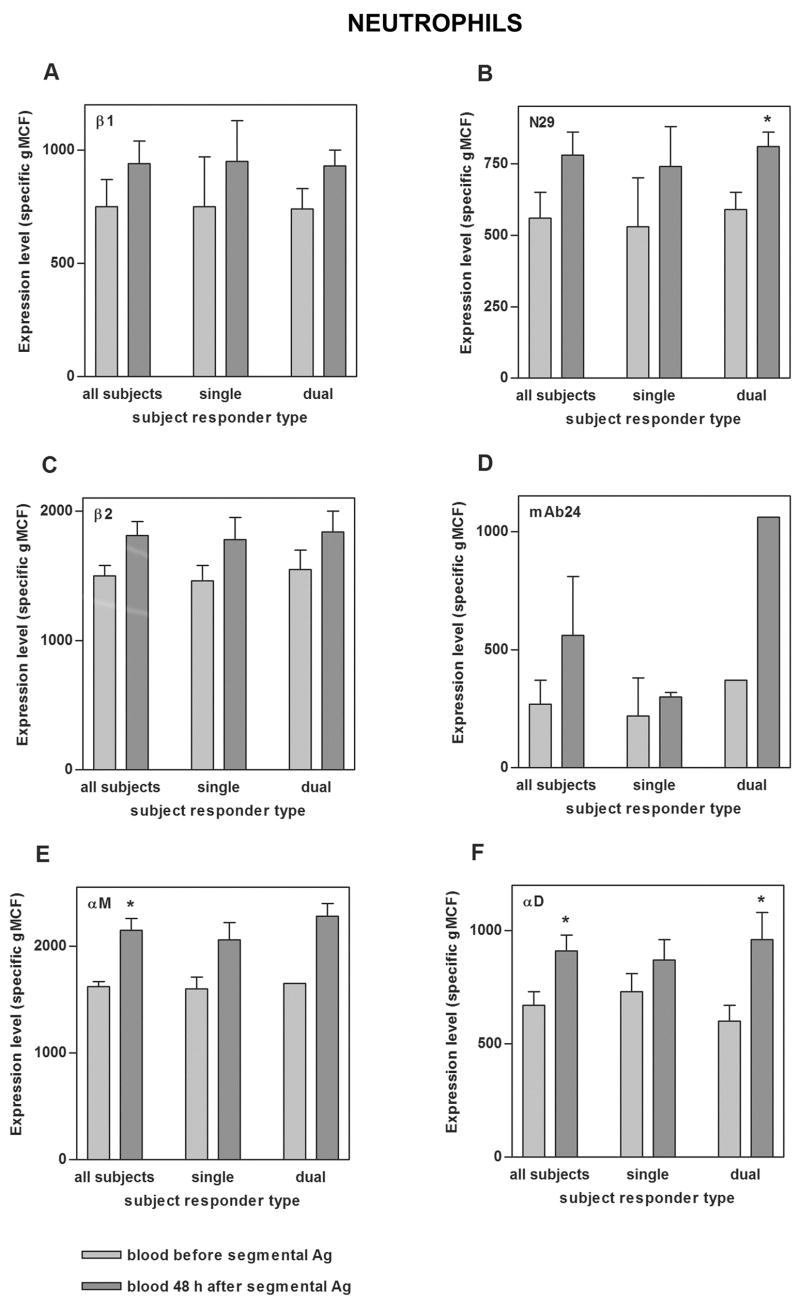
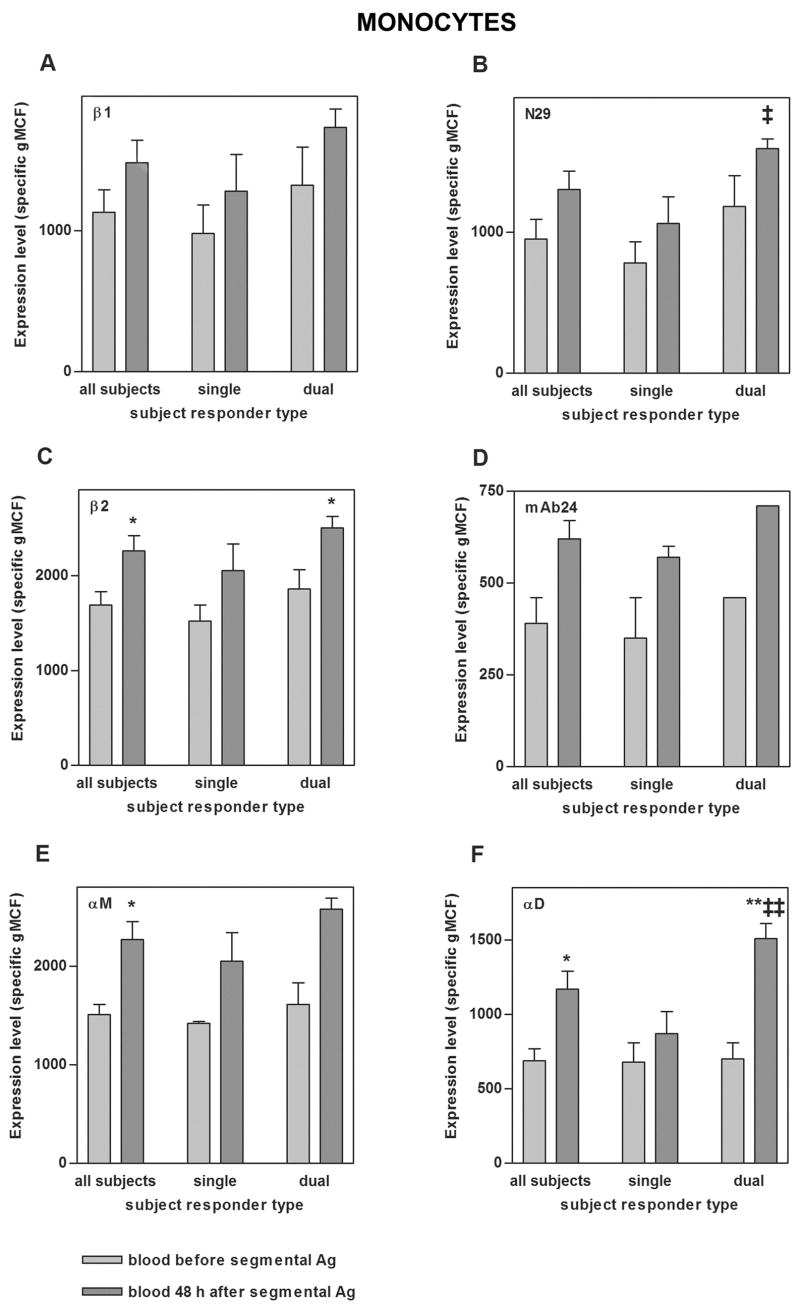
To examine the specificity of the changes on blood eosinophils, circulating neutrophils and monocytes were also analyzed. Before segmental challenge, neutrophils had higher N29 reactivity than eosinophils and monocytes even higher (Figs. 4B and 5B, compare to Fig. 2B). At 48 h after segmental Ag challenge, circulating neutrophils and monocytes, like eosinophils, had significantly higher αD expression compared to before challenge (Figs. 4F and 5F). Monocytes from dual responders had higher αD after segmental challenge than those from single responders (Fig. 5F). N29 reactivity of neutrophils (as with eosinophils) increased significantly in dual responders upon segmental challenge, although to a lesser degree (1.4-fold for neutrophils, 1.7-fold for eosinophils)(Fig. 4B, compare to Fig. 2B); whereas N29 reactivity of monocytes did not increase significantly (Fig. 5B). Further, αM on blood neutrophils and monocytes and β2 on blood monocytes increased significantly upon segmental challenge (Figs. 4E and 5C,E); in contrast to blood eosinophil αM and β2, which did not (Fig. 2C,E).
FIGURE 4. Integrin expression on blood neutrophils before and 48 h after segmental Ag challenge.
Expression of total β1 integrin (A), activation-sensitive β1 integrin mAb N29 epitope (B), total β2 integrin (C), activation-sensitive β2 integrin mAb24 epitope (D), αM integrin (E), and αD integrin (F) on blood neutrophils before segmental Ag challenge (light gray) and 48 h after segmental Ag challenge (medium gray) in all tested subjects, single responders, and dual responders. Values shown are specific mean channel fluorescence (gMCF)(mean ± SEM). *p ≤ 0.05 versus before segmental challenge.
FIGURE 5. Integrin expression on blood monocytes before and 48 h after segmental Ag challenge.
Expression of total β1 integrin (A), activation-sensitive β1 integrin mAb N29 epitope (B), total β2 integrin (C), activation-sensitive β2 integrin mAb24 epitope (D), αM integrin (E), and αD integrin (F) on blood monocytes before segmental Ag challenge (light gray) and 48 h after segmental Ag challenge (medium gray) in all tested subjects, single responders, and dual responders. Values shown are specific mean channel fluorescence (gMCF)(mean ± SEM). *p ≤ 0.05, **p ≤ 0.01 versus before segmental challenge; ‡p ≤ 0.05, ‡‡p ≤ 0.01 versus single responders.
Thus, 1) αD is upregulated on all three leukocyte populations, not just eosiniophils; 2) β1 at baseline is more activated on neutrophils and monocytes compared to eosinophils; 3) in dual responders we found increased activation state of β1 on neutrophils despite the high baseline activation state; and 4) surface expression of αM is upregulated on monocytes and neutrophils, but not on blood eosinophils.
Correlations between integrin expression and eosinophil numbers in BAL after segmental allergen challenge
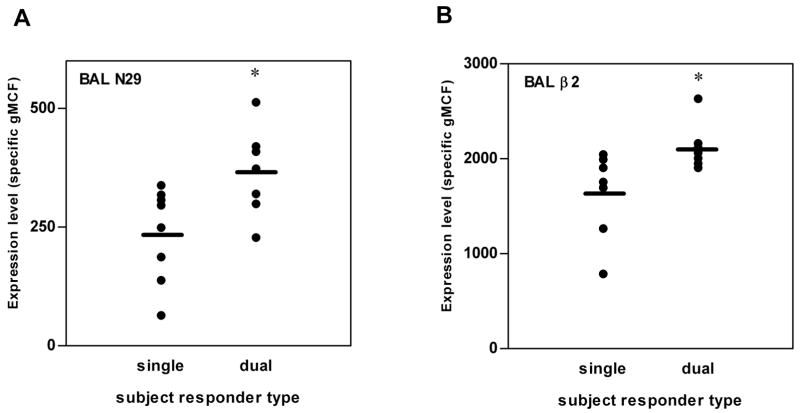
As with the variability in eosinophil numbers in BAL after segmental Ag challenge in the 19 subjects (Table I), there was considerable variability in integrin activation or expression on BAL eosinophils (Fig. 2). BAL eosinophils from dual responders had significantly higher N29 epitope and total α2 expression than BAL eosinophils from single responders (Figs. 2 and 6). Grouping all subjects together, reactivity of BAL eosinophils with the activation-sensitive mAbs, N29 against β1 and mAb24 against β2, correlated significantly with percentage of eosinophils in BAL (Fig. 7). Surface expression of other integrin subunits on BAL eosinophils did not correlate with BAL eosinophil percentage (not shown). These results indicate that activation of both β1 and β2 integrins are associated with eosinophil recruitment to the airway.
FIGURE 6. Differences in integrin activation and expression on BAL eosinophils between single and dual responders.
BAL eosinophil expression 48 h after segmental Ag challenge of activation-sensitive β1 integrin mAb N29 epitope (A) or total β2 integrin (B) in single and dual responders. Bar = mean, *p ≤ 0.05 versus single responders. BAL, bronchoalveolar lavage; gMCF, geometric mean channel fluorescence.
FIGURE 7. Correlations between integrin activation on BAL eosinophils and BAL eosinophil percentage.
Correlations between BAL eosinophil expression of activation-sensitive β1 integrin mAb N29 epitope (A) or activation-sensitive β2 integrin mAb24 epitope (B) and percentage of eosinophils in BAL 48 h after segmental Ag challenge. BAL, bronchoalveolar lavage; gMCF, geometric mean channel fluorescence. rs, Spearman rank correlation coefficient.
Correlations between integrin expression after segmental allergen challenge and the magnitude of the late-phase fall in FEV1 after whole-lung allergen challenge
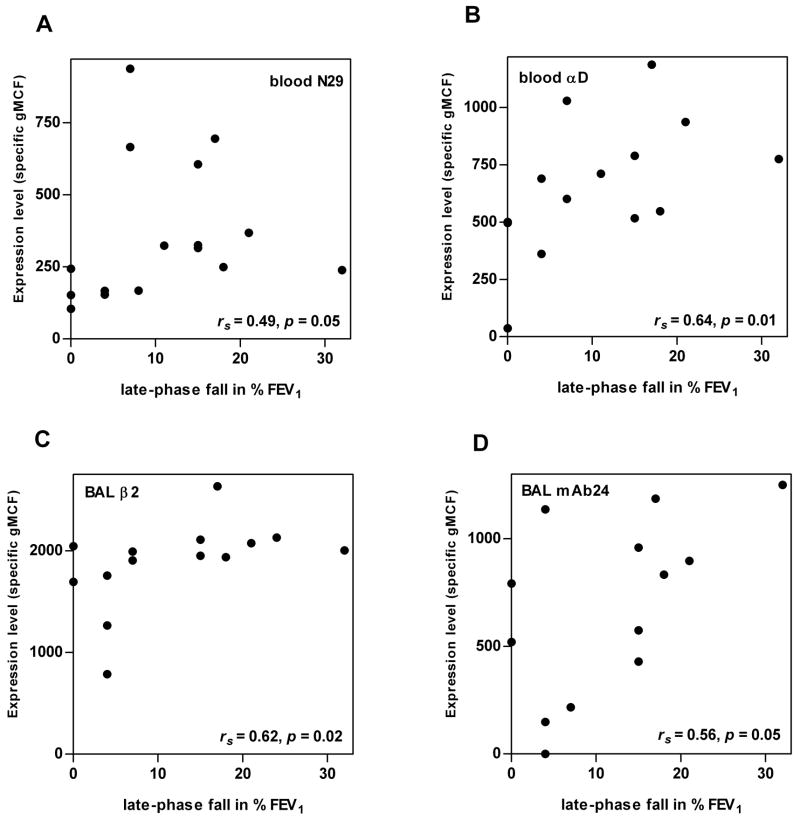
We also analyzed for possible correlations between eosinophil integrins after segmental challenge and the magnitude of the maximum fall in FEV1 3–8 h after whole-lung allergen challenge (late-phase fall). αD expression and reactivity with N29 of blood eosinophils 48 h after segmental challenge each correlated with the magnitude of the late-phase fall in FEV1 after whole-lung challenge (Fig. 8A,B). β2 expression and reactivity with mAb24 of BAL eosinophils 48 h after segmental challenge also correlated with the magnitude of the late-phase FEV1 fall (Fig. 8C,D), as did αM expression of BAL eosinophils (rs = 0.83, p = 0.008)(not shown). In addition, the late-phase FEV1 fall correlated with the percentage of eosinophils in BAL 48 h after segmental challenge (rs = 0.70, p = 0.001)(not shown). Thus, the results indicate that greater αD upregulation and β1 activation on blood eosinophils and greater αMβ2 upregulation and β2 activation on BAL eosinophils after segmental challenge in a subject are associated with a greater late-phase FEV1 fall in response to whole-lung challenge.
FIGURE 8. Correlations between integrin activation and expression on blood and BAL eosinophils and the magnitude of the fall in FEV1 3-8 h after whole-lung Ag challenge.
Correlations between blood eosinophil expression of activation-sensitive β1 integrin mAb N29 epitope (A) or αD integrin (B) or BAL eosinophil expression of total β2 integrin (C) or activation-sensitive β2 integrin mAb24 epitope (D) 48 h after segmental Ag challenge and the magnitude of the late-phase fall in FEV1 3–8 h after whole-lung Ag challenge. BAL, bronchoalveolar lavage; FEV1, forced expiratory volume in 1 s; gMCF, geometric mean channel fluorescence. rs, Spearman rank correlation coefficient.
Correlations between integrin expression and cytokine concentrations in BAL fluid after segmental allergen challenge
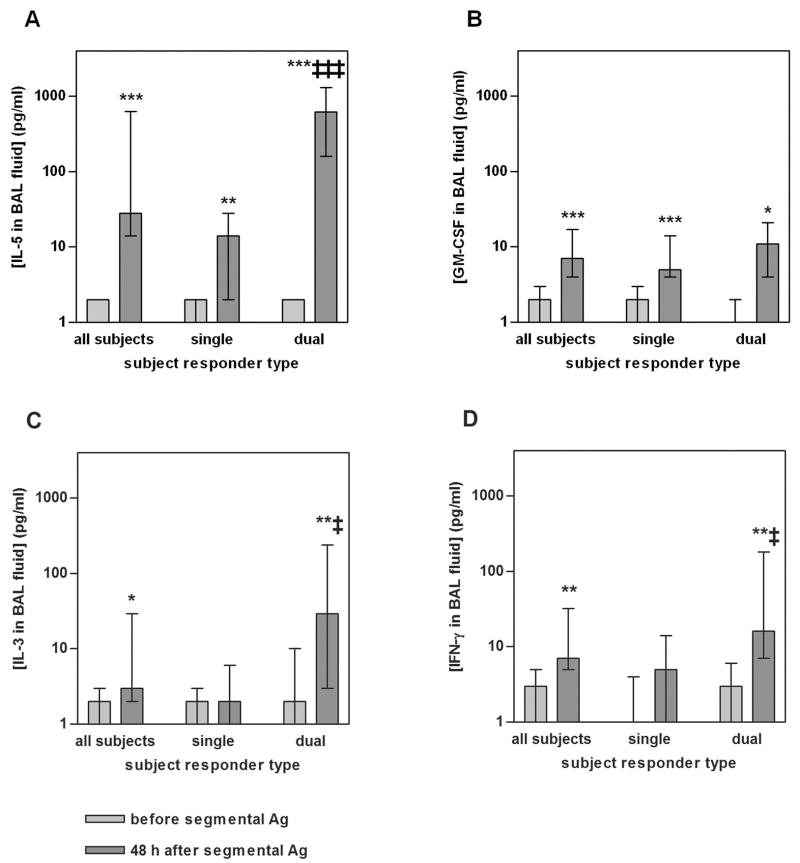
To identify factor(s) possibly responsible for the changes in integrins observed following segmental Ag challenge, the concentrations of IL-5, GM-CSF, IL-3, and IFN-γ in BAL fluid were measured (Fig. 9). The concentration of all four cytokines was significantly higher 48 h after segmental challenge than in samples obtained immediately before segmental challenge (Fig. 9). There was a highly significant difference in BAL fluid IL-5 after segmental challenge between single and dual responders; median IL-5 concentration in BAL fluid from dual responders was about 40-fold greater than from single responders (Fig. 9). Also IL-3 and IFN-γ were significantly different between single and dual responders; medians from dual responders were about ten- and three-fold those from single responders for IL-3 and IFN-γ, respectively (Fig. 9). GM-CSF was not different between single and dual responders (Fig. 9).
FIGURE 9. Cytokine concentrations in BAL fluid before and 48 h after segmental Ag challenge.
Concentrations of IL-5 (A), GM-CSF (B), IL-3 (C), and IFN-γ (D) in BAL fluid before (light gray bars) and 48 h after (medium gray bars) segmental Ag challenge from all tested subjects (n = 19), single responders (n = 10), or dual responders (n = 9). Values shown are medians with 25th and 75th percentiles of concentrations in recovered, unconcentrated BAL fluid. *p ≤ 0.05, **p ≤ 0.01, ***p ≤ 0.001 versus before segmental challenge; ‡p ≤ 0.05, ‡‡‡p ≤ 0.001 versus single responders. BAL, bronchoalveolar lavage.
The concentration of BAL fluid IL-5, but not that of the other cytokines, correlated significantly and strongly with the percentage of eosinophils in BAL 48 h after segmental Ag challenge (rs = 0.69, p = 0.001 for IL-5; rs = 0.23, p = 0.36 for GM-CSF; rs = 0.32, p = 0.19 for IL-3; and rs = 0.39, p = 0.11 for IFN-γ). It is possible that one or both of the other IL-5 family cytokines would correlate with BAL eosinophil percentage if a higher number of subjects were studied. IL-5 also correlated inversely with the percentage of macrophages (not shown), reflecting the decreased macrophage percentage as eosinophil percentage increases. Further, BAL fluid IL-5 after segmental challenge correlated strongly with the magnitude of the late-phase fall in FEV1 after whole-lung challenge (rs = 0.79, p < 0.0001). IL-3 also correlated with late-phase FEV1 fall but less well (rs = 0.51, p = 0.03). GM-CSF (rs = 0.37, p = 0.12) and IFN-γ (rs = 0.36, p = 0.13) did not correlate with late-phase FEV1 fall.
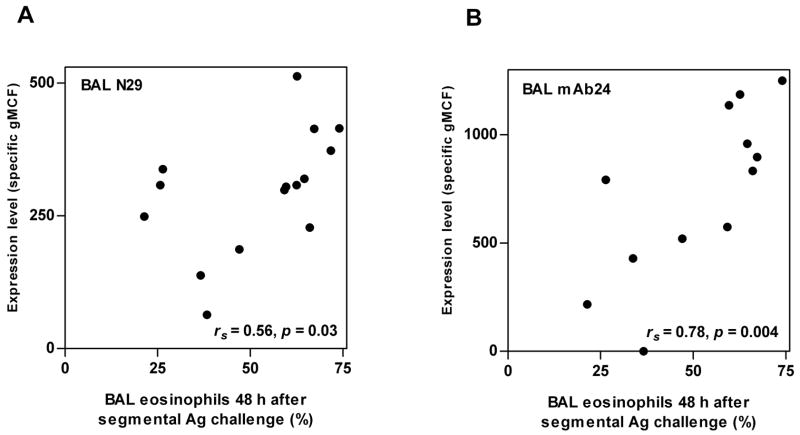
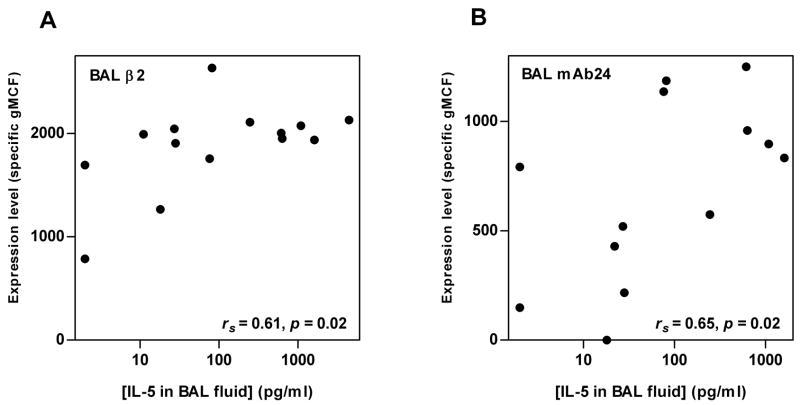
Levels of β2 and reactivity with mAb24 of BAL eosinophils correlated significantly with the concentration of IL-5 in BAL fluid 48 h after segmental Ag challenge (Fig. 10), as did level of αM (rs = 0.70, p = 0.04)(not shown). These integrins did not correlate with concentrations of the other cytokines (not shown). There was no correlation with BAL fluid IL-5 before segmental challenge (not shown). Integrins of blood eosinophils did not correlate with cytokine concentrations in BAL fluid 48 h after segmental Ag challenge (not shown). Thus, a greater IL-5 concentration in BAL fluid in a subject is associated with greater αMβ2 upregulation and β2 activation on BAL eosinophils after segmental challenge.
FIGURE 10. Correlations between integrin activation and expression on BAL eosinophils and IL-5 in BAL fluid.
Correlations between blood eosinophil expression of total β2 integrin (A) or activation-sensitive β2 integrin mAb24 epitope (B) and the concentration of IL-5 in BAL fluid 48 h after segmental Ag challenge. BAL, bronchoalveolar lavage; gMCF, geometric mean channel fluorescence. rs, Spearman rank correlation coefficient.
Discussion
We observed changes in integrins on eosinophils in blood and airway of 19 subjects with mild allergic asthma after segmental bronchial allergen challenge that were related to the reactivities of subjects in the whole Ag challenge protocol. Eosinophils in blood 48 h after challenge had increased expression of the αD integrin subunit and, in asthmatics with a dual response phenotype, increased reactivity with activation-sensitive anti-β1 integrin mAb N29 compared to before challenge. Eosinophils in BAL obtained 48 h after challenge had increased expression of β2 and αM integrin subunits and increased reactivity of activation-sensitive anti-β2 integrin mAb24 compared to eosinophils in blood. β2, αM, and mAb24 epitope expression on BAL eosinophils correlated with the concentration of IL-5 in BAL fluid obtained 48 h after segmental Ag challenge. In addition, αD expression and, in dual responders, N29 reactivity were higher on BAL eosinophils than on blood eosinophils before challenge. Integrin expression patterns on BAL eosinophils were more homogeneous and symmetric than on blood eosinophils. The observations of N29 epitope and αD expression are compatible with a scenario whereby extravasated eosinophils appearing in the airway are derived from a subpopulation of the total, more heterogeneous population of circulating eosinophils. However, the observations of αMβ2 expression, β2 activation state, and IL-5 levels in BAL fluid indicate that after leaving the circulation eosinophils undergo IL-5-triggered activation of β2 and upregulation of αMβ2.
Analysis of neutrophils and monocytes in the same blood samples revealed that αD expression increased on all three leukocyte types upon segmental challenge; thus, αD upregulation may occur through a common mechanism. At baseline, N29 reactivity was higher on neutrophils than on eosinophils and even higher on monocytes. N29 reactivity increased significantly on neutrophils in dual responders upon challenge, but to a relatively lesser degree than on eosinophils; whereas it did not change significantly on monocytes. The mechanisms responsible for maintaining the baseline β1 activation levels on the different leukocytes and responsible for the elevation of activation state upon challenge are unknown. Finally, neutrophil and monocyte αM and monocyte β2 were upregulated upon challenge, in contrast to eosinophil αM and β2, which were upregulated in BAL but not blood. Thus, circulating eosinophils are less sensitive than neutrophils or monocytes to increased surface expression of αMβ2.
β1 activation on blood eosinophils, assessed by reactivity with N29, has been shown to correlate inversely with FEV1 in an inhaled corticosteroid (ICS) withdrawal study (35). The present results complement the steroid withdrawal study by demonstrating persistent β1 activation in dual responders with more eosinophilic inflammation in the airway. Blood eosinophils from dual responders, but not from single responders, have also been shown to have enhanced activation of FcγRII (CD32), as assessed with the phage mAb A17, 6 h after challenge (42). Whether activation of β1 integrin and CD32 on circulating eosinophils are triggered by the same or different stimuli and signaling pathways is an interesting question that remains to be investigated.
The changes observed here in BAL eosinophils reproduce observations in the literature, mostly on isolated eosinophils, that BAL eosinophils have higher αM and β2 than blood eosinophils (43–46); activated αMβ2, as monitored by anti-active αM mAb CBRM1/5 (22, 36); and increased αMβ2-inhibitable adhesion to diverse ligands (22, 36). Our finding that dual responders have higher total numbers of cells and eosinophils and higher proportion of eosinophils in BAL after segmental Ag challenge than single responders also is in accord with prior reports (47–50). We also found that β1 activation state and β2 surface expression were significantly higher on BAL eosinophils from dual responders than on those from single responders. Activation of β1 and β2 integrins, as assessed with the activation-sensitive mAbs N29 and mAb24, respectively, on BAL eosinophils correlated with eosinophil percentage in BAL, indicating that activation of both these subfamilies of integrins is important for eosinophil recruitment. This idea is consistent with in vivo studies supporting the involvement of α4β1 in eosinophil appearance in the airway and in vitro studies showing involvement of both β1 and β2 integrins in eosinophil transendothelial migration (22, 51–56). The evidence for involvement of both α4β1 and β2 integrins in eosinophil migration in vivo is further strengthened by a recent report that eosinophil recruitment to airway after Ag challenge was severely attenuated in both a conditional α4 integrin knockout mouse and a β2-deficient mouse (57). Finally, αD expression and β1 activation on blood eosinophils as well as β2 and αM expression and β2 activation on BAL eosinophils after segmental Ag challenge all correlated with the magnitude of the late-phase fall in FEV1 in response to whole-lung Ag challenge.
A number of mediators are increased in airway after challenge (28, 29, 49, 58, 59). Of these, the most likely candidates to account for upregulation and activation of αMβ2 are the IL-5 family cytokines (36, 60, 61). Concentrations of IL-5, GM-CSF, IL-3, and IFN-γ in BAL fluid recovered 48 h after segmental Ag challenge were all increased significantly compared to in BAL fluid recovered before challenge. IL-5 correlated with BAL eosinophil αM and β2 expression and β2 activation 48 h after segmental challenge. The median concentration of IL-5 in BAL fluid from dual responders after segmental challenge was about 600 pg/ml, which was more than 20-fold the median concentration of the other cytokines. Further, the IL-5 concentration in the five BAL samples containing eosinophils with the most highly activated β2 was 100 to > 1000 pg/ml. During recovery of BAL the volume of the fluid lining the airway epithelium in vivo is estimated to become diluted 100-fold (62). Thus, the levels of IL-5 in the epithelial lining fluid of these subjects (all of whom were dual responders) is estimated to be about 10–100 ng/ml. Treatment of blood eosinophils in vitro with IL-5 at concentrations in this range is known to saturate the IL-5 receptor (63–68) and lead to αMβ2 upregulation and activation and induction of adhesion to ICAM-1 and other substrates (36, 60, 61), priming and enhanced response to chemoattractants (69), enhanced viability (70), degranulation and granule protein release (30), IL-5 receptor α downregulation (71), and enhanced expression of certain genes (72). In contrast, median concentrations of the other IL-5 family cytokines prior to dilution are estimated to be about 1–3 ng/ml. Thus, the concentration of IL-5 in the lining fluid in vivo, assuming that a significant portion of the IL-5 measured represents active IL-5, is estimated to be sufficient to cause αMβ2 upregulation and activation of β2 integrins. These results are in accord with the earlier observation that the IL-5 receptor is downregulated specifically compared to the GM-CSF receptor on eosinophils recovered by BAL 48 h after segmental Ag challenge (30). We also found that BAL fluid IL-5 correlated with the percentage of eosinophils in BAL and with the magnitude of the late-phase fall in FEV1. A correlation between BAL fluid IL-5 and eosinophil recruitment to the airway after segmental Ag challenge has been reported before (38, 73–76) and is consistent with the observation that anti-IL-5 therapy causes a significant decrease in sputum eosinophils (1).
Our study has several limitations. The data sets were incomplete in that all integrins were not assayed from all samples. Sampling of blood and BAL were performed only at one time point (48 h) after segmental Ag challenge. Future experiments with blood sampling at various time points after segmental and/or whole-lung Ag challenge are required to record the time-course of αD and N29 expression on blood eosinophils in dual and single responders and learn when the values diverge. Further, the importance of the integrin expression heterogeneity observed on blood eosinophils, particularly regarding β2 and αM, is not known. Such heterogeneity, to our knowledge, has not been described or discussed before. Of note, we subjected whole blood to primary antibody incubation without previous cell isolation or centrifugation. Earlier studies on blood eosinophil integrin expression have been performed on a buffy coat preparation or purified eosinophils (43–45). Dextran sedimentation during buffy coat preparation has been shown to cause upregulation of αM on blood eosinophils (and neutrophils) compared to in whole, unfractionated blood (77). Eosinophils are likely similar to neutrophils, which are known to store a large proportion of αM in granules, wherefrom it is translocated to the plasma membrane after cell stimulation (78). Remarkably, eosinophils in BAL had strong homogeneous labeling for β2 and αM. One possibility is that higher reacting subpopulations of circulating leukocytes represent cells that have undergone “retrograde” migration back to the blood from tissues, as has been demonstrated for zebrafish neutrophils in vivo (79) and human neutrophils in vitro (80). Blood sampling at various time points after Ag challenge, including times beyond 48 h, may shed further light on circulating eosinophil subpopulations.
Our results and the literature are compatible with the schematic of eosinophil recruitment after segmental Ag challenge that is depicted in Fig. 11. At baseline, β1 and β2 integrins on a circulating eosinophil are in a low activation state, the level of eosinophil surface αD is low, and VCAM-1 is absent from endothelial surfaces of the bronchial circulation. Yet-to-be-identified stimuli resulting from Ag challenge cause enhanced activation of β1 integrins and increased surface expression of αD on circulating eosinophils. Because increased expression of the β1 activation epitope occurs on most eosinophils, the activation likely takes place in the pulmonary circulation through which the eosinophils constantly pass or in response to release of an activating substance into the circulation (Fig. 11). Such activation contrasts with the model for recruitment of leukocytes (16), including eosinophils (20), in which integrin activation is assumed to occur locally and concurrently with rolling and tethering on endothelium. Interestingly, bone marrow-derived progenitor cells or circulating eosinophils of asthmatics have been shown to be activated upon Ag challenge also as measured by upregulation of IL-5 receptor α and CCR3, activation of CD32, and greater responsiveness to chemoattractants (42, 81–83). Further, eosinophils from allergic asthmatics have been shown to have a greater capacity to adhere to and transmigrate through endothelium than eosinophils from normal donors (51, 84).
FIGURE 11. Sequential upregulation and activation of eosinophil integrins during recruitment to airway after exposure to allergen.
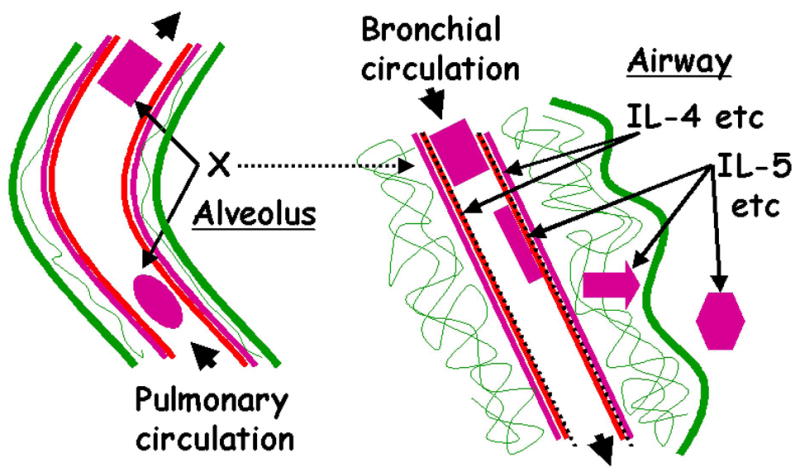
Schematic relating activation of β1 and β2 integrins to eosinophil trafficking during and after segmental Ag challenge. Left, An eosinophil (oval) with unactivated β1 integrins and low surface expression of αD is shown entering the pulmonary circulation of a segment subjected to Ag challenge. A yet-to-be-identified stimulus or stimuli (X) cause(s) activation of β1 integrins and increased surface expression of αD during transit of the eosinophil (rectangle) through the vessel. Right, Concurrently, IL-4 and other mediators are released and specifically induce surface expression of VCAM-1 (discontinuous black line) on the endothelial cells of a bronchial blood vessel in the challenged segment. An eosinophil with activated β1 and upregulated αD (rectangle) is shown entering the bronchial circulation. The eosinophil arrests and adheres (elongated rectangle) to VCAM-1. After exposure to IL-5 the eosinophil becomes responsive to chemotactic factors, migrates (arrow) and assumes a hyperadhesive airway phenotype (hexagon) in the lumen. The airway eosinophil displays activated β1, upregulated αD, activated β2, ανδ υπρεγυλατεδ αM and β2.
The question then arises as to how activated circulating eosinophils localize to the parts of the lung subjected to Ag challenge in preference to neutrophils and monocytes. As a parallel response to Ag challenge, mediators, including IL-4 and IL-13, are elaborated and activate bronchial endothelium to specifically synthesize and express VCAM-1. VCAM-1 has been shown to be preferentially expressed in the asthmatic lung or after Ag challenge (85, 86). Further, VCAM-1 can support adhesion of eosinophils but not neutrophils (9, 18, 22, 87–91). Circulating eosinophils with activated β1 and upregulated αD are presumed to have a higher probability of arresting on VCAM-1 on activated endothelium of the bronchial circulation (Fig. 11), since adhesion of purified blood eosinophils to VCAM-1 is mediated by α4β1 (22, 92), with a possible contribution by αDβ2 (22, 31).
αM and β2 are shown as being upregulated and β2 integrins as being activated by IL-5, also elaborated in response to allergen. Eosinophils appearing in the airway lumen have a hyperadhesive phenotype that is marked by activated β1 integrins, upregulated αDβ2, activated β2 integrins, and upregulated αMβ2 (Fig. 11). Eosinophils, particularly after priming by IL-5 (69), responds to chemoattractants, such as eotaxin (9, 13)(Fig. 11). αMβ2 is believed to be important for eosinophil migration (22, 51, 53, 93–95). Thus, the scenario has migrating eosinophils using activated αMβ2 and possibly other integrins to interact with multiple substrates (22, 36, 92), including VCAM-1, ICAM-1 and ECM proteins, on endothelial cells, in connective tissue, and on epithelium of the bronchial wall.
Acknowledgments
We thank Mary Jo Jackson and Erin Billmeyer for patient recruitment, screening, and assistance with bronchoscopy; Lin-Ying Liu, Sarah Panzer, Rebecca Lawniczak, and Rose DeGrauw for processing BAL samples and providing blood samples; Melissa Heim and Lisa Skoggs for assistance with ELISA; Nancy Hogg for providing mAb24; ICOS for providing anti-αD mAb 240I; Kathleen Schell, Joan Batchelder, and Joel Puchalski for help with flow cytometry data collection and analysis; Michael Evans for advice on statistics; and Julie Sedgwick and Lin-Ying Liu for advice.
Footnotes
This work was supported by Specialized Center of Research grant HL56396, Program Project grant HL88594, and General Clinical Research Center grant M01 RR03186 from the National Institutes of Health.
Abbreviations used in this paper: AgPD20, provocative dose of Ag producing a 20% fall in FEV1; BAL, bronchoalveolar lavage; CBU, cumulative breath unit; CD, cat dander; ECM, extracellular matrix; F, female; FEV1, forced expiratory volume in 1 s; gMCF, geometric mean channel fluorescence; H, homogeneous (distribution); HDM, house dust mite; ICS, inhaled corticosteroid; M, male; PC20, provocative concentration of methacholine producing a 20% fall in FEV1; rs, Spearman rank correlation coefficient; RW, ragweed; T, total; % pos., percentage of positive cells; % pred., percentage of predicted value.
This is an author-produced version of a manuscript accepted for publication in The Journal of Immunology (The JI). The American Association of Immunologists, Inc. (AAI), publisher of The JI, holds copyright to this manuscript. This version of the manuscript has not yet been copyedited or subjected to editorial proofreading by The JI; hence, it may differ from the final version published in The JI (online and in print). The AAI (The JI) is not liable for errors or omissions in this author-produced version of the manuscript or in any version derived from it by the U.S. National Institutes of Health or any other third party. The final, citable version of record can be found at www.jimmunol.org
Disclosures
M.W.J., W.W.B, and D.F.M. have, with the help of the Wisconsin Alumni Research Foundation (WARF), a patent application pending on “Beta1 integrin activation as a marker for asthma”, based on the associations between N29 reactivity and FEV1 reported by Johansson et al. 2006 (reference No. 35). W.W.B. has or has had research support from Novartis, Dynavax, Wyeth, Centocor, GlaxoSmithKline, Medicinova, Pfizer, and Biowa/MedImmune. E.A.B.K. and N.N.J. have no financial conflict of interest.
References
- 1.Leckie MJ, ten Brinke A, Khan J, Diamant Z, O'Connor BJ, Walls CM, Mathur AK, Cowley HC, Chung KF, Djukanovic R, Hansel TT, Holgate ST, Sterk PJ, Barnes PJ. Effects of an interleukin-5 blocking monoclonal antibody on eosinophils, airway hyper-responsiveness, and the late asthmatic response. Lancet. 2000;356:2144–2148. doi: 10.1016/s0140-6736(00)03496-6. [DOI] [PubMed] [Google Scholar]
- 2.Busse WW, Lemanske RF., Jr Asthma. N Engl J Med. 2001;344:350–362. doi: 10.1056/NEJM200102013440507. [DOI] [PubMed] [Google Scholar]
- 3.Green RH, Brightling CE, McKenna S, Hargadon B, Parker D, Bradding P, Wardlaw AJ, Pavord ID. Asthma exacerbations and sputum eosinophil counts: a randomised controlled trial. Lancet. 2002;360:1715–1721. doi: 10.1016/S0140-6736(02)11679-5. [DOI] [PubMed] [Google Scholar]
- 4.Mattes J, Yang M, Mahalingam S, Kuehr J, Webb DC, Simson L, Hogan SP, Koskinen A, McKenzie AN, Dent LA, Rothenberg ME, Matthaei KI, Young IG, Foster PS. Intrinsic defect in T cell production of interleukin (IL)-13 in the absence of both IL-5 and eotaxin precludes the development of eosinophilia and airways hyperreactivity in experimental asthma. J Exp Med. 2002;195:1433–1444. doi: 10.1084/jem.20020009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Flood-Page PT, Menzies-Gow AN, Kay AB, Robinson DS. Eosinophil's role remains uncertain as anti-interleukin-5 only partially depletes numbers in asthmatic airway. Am J Respir Crit Care Med. 2003;167:199–204. doi: 10.1164/rccm.200208-789OC. [DOI] [PubMed] [Google Scholar]
- 6.Flood-Page P, Menzies-Gow A, Phipps S, Ying S, Wangoo A, Ludwig MS, Barnes N, Robinson D, Kay AB. Anti-IL-5 treatment reduces deposition of ECM proteins in the bronchial subepithelial basement membrane of mild atopic asthmatics. J Clin Invest. 2003;112:1029–1036. doi: 10.1172/JCI17974. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Justice JP, Borchers MT, Crosby JR, Hines EM, Shen HH, Ochkur SI, McGarry MP, Lee NA, Lee JJ. Ablation of eosinophils leads to a reduction of allergen-induced pulmonary pathology. Am J Physiol Lung Cell Mol Physiol. 2003;284:L169–L178. doi: 10.1152/ajplung.00260.2002. [DOI] [PubMed] [Google Scholar]
- 8.Shen HH, Ochkur SI, McGarry MP, Crosby JR, Hines EM, Borchers MT, Wang H, Biechelle TL, O'Neill KR, Ansay TL, Colbert DC, Cormier SA, Justice JP, Lee NA, Lee JJ. A causative relationship exists between eosinophils and the development of allergic pulmonary pathologies in the mouse. J Immunol. 2003;170:3296–3305. doi: 10.4049/jimmunol.170.6.3296. [DOI] [PubMed] [Google Scholar]
- 9.Wills-Karp M, Karp CL. Eosinophils in asthma: remodeling a tangled tale. Science. 2004;305:1726–1729. doi: 10.1126/science.1104134. [DOI] [PubMed] [Google Scholar]
- 10.Bochner BS, Busse WW. Allergy and asthma. J Allergy Clin Immunol. 2005;115:953–959. doi: 10.1016/j.jaci.2005.02.032. [DOI] [PubMed] [Google Scholar]
- 11.Kay AB. The role of eosinophils in the pathogenesis of asthma. Trends Mol Med. 2005;11:148–152. doi: 10.1016/j.molmed.2005.02.002. [DOI] [PubMed] [Google Scholar]
- 12.Liu LY, Mathur SK, Sedgwick JB, Jarjour NN, Busse WW, Kelly EA. Human airway and peripheral blood eosinophils enhance Th1 and Th2 cytokine secretion. Allergy. 2006;61:589–597. doi: 10.1111/j.1398-9995.2006.01060.x. [DOI] [PubMed] [Google Scholar]
- 13.Rothenberg ME, Hogan SP. The eosinophil. Annu Rev Immunol. 2006;24:147–174. doi: 10.1146/annurev.immunol.24.021605.090720. [DOI] [PubMed] [Google Scholar]
- 14.Kariyawasam HH, Robinson DS. The role of eosinophils in airway tissue remodelling in asthma. Curr Opin Immunol. 2007;19:681–686. doi: 10.1016/j.coi.2007.07.021. [DOI] [PubMed] [Google Scholar]
- 15.Liu LY, Bates ME, Jarjour NN, Busse WW, Bertics PJ, Kelly EA. Generation of Th1 and Th2 chemokines by human eosinophils: evidence for a critical role of TNF-alpha. J Immunol. 2007;179:4840–4848. doi: 10.4049/jimmunol.179.7.4840. [DOI] [PubMed] [Google Scholar]
- 16.Springer TA. Traffic signals for lymphocyte recirculation and leukocyte emigration: the multistep paradigm. Cell. 1994;76:301–314. doi: 10.1016/0092-8674(94)90337-9. [DOI] [PubMed] [Google Scholar]
- 17.Hynes RO. Integrins: bidirectional, allosteric signaling machines. Cell. 2002;110:673. doi: 10.1016/s0092-8674(02)00971-6. [DOI] [PubMed] [Google Scholar]
- 18.Broide D, Sriramarao P. Eosinophil trafficking to sites of allergic inflammation. Immunol Rev. 2001;179:163–172. doi: 10.1034/j.1600-065x.2001.790116.x. [DOI] [PubMed] [Google Scholar]
- 19.Wardlaw AJ. Eosinophil trafficking in asthma. Clin Med. 2001;1:214–218. doi: 10.7861/clinmedicine.1-3-214. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Rosenberg HF, Phipps S, Foster PS. Eosinophil trafficking in allergy and asthma. J Allergy Clin Immunol. 2007;119:1303–1310. doi: 10.1016/j.jaci.2007.03.048. [DOI] [PubMed] [Google Scholar]
- 21.Tachimoto H, Bochner BS. The surface phenotype of human eosinophils. Chem Immunol. 2000;76:45–62. doi: 10.1159/000058780. [DOI] [PubMed] [Google Scholar]
- 22.Barthel SR, Johansson MW, McNamee DM, Mosher DF. Roles of integrin activation in eosinophil function and the eosinophilic inflammation of asthma. J Leukoc Biol. 2008;83:1–12. doi: 10.1189/jlb.0607344. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Humphries JD, Byron A, Humphries MJ. Integrin ligands at a glance. J Cell Sci. 2006;119:3901–3903. doi: 10.1242/jcs.03098. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Palecek SP, Loftus JC, Ginsberg MH, Lauffenburger DA, Horwitz AF. Integrin-ligand binding properties govern cell migration speed through cell-substratum adhesiveness. Nature. 1997;385:537–540. doi: 10.1038/385537a0. [DOI] [PubMed] [Google Scholar]
- 25.Humphries MJ. Monoclonal antibodies as probes of integrin priming and activation. Biochem Soc Trans. 2004;32:407–411. doi: 10.1042/BST0320407. [DOI] [PubMed] [Google Scholar]
- 26.Xiao T, Takagi J, Coller BS, Wang JH, Springer TA. Structural basis for allostery in integrins and binding to fibrinogen-mimetic therapeutics. Nature. 2004;432:59–67. doi: 10.1038/nature02976. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Calhoun WJ, Jarjour NN, Gleich GJ, Stevens CA, Busse WW. Increased airway inflammation with segmental versus aerosol antigen challenge. Am Rev Respir Dis. 1993;147:1465–1471. doi: 10.1164/ajrccm/147.6_Pt_1.1465. [DOI] [PubMed] [Google Scholar]
- 28.Liu L, Jarjour NN, Busse WW, Kelly EA. Enhanced generation of helper T type 1 and 2 chemokines in allergen-induced asthma. Am J Respir Crit Care Med. 2004;169:1118–1124. doi: 10.1164/rccm.200312-1659OC. [DOI] [PubMed] [Google Scholar]
- 29.Kelly EA, Busse WW, Jarjour NN. Inhaled budesonide decreases airway inflammatory response to allergen. Am J Respir Crit Care Med. 2000;162:883–890. doi: 10.1164/ajrccm.162.3.9910077. [DOI] [PubMed] [Google Scholar]
- 30.Liu LY, Sedgwick JB, Bates ME, Vrtis RF, Gern JE, Kita H, Jarjour NN, Busse WW, Kelly EA. Decreased expression of membrane IL-5 receptor alpha on human eosinophils: I. Loss of membrane IL-5 receptor alpha on airway eosinophils and increased soluble IL-5 receptor alpha in the airway after allergen challenge. J Immunol. 2002;169:6452–6458. doi: 10.4049/jimmunol.169.11.6452. [DOI] [PubMed] [Google Scholar]
- 31.Grayson MH, Van der Vieren M, Sterbinsky SA, Gallatin MW, Hoffman PA, Staunton DE, Bochner BS. alphadbeta2 integrin is expressed on human eosinophils and functions as an alternative ligand for vascular cell adhesion molecule 1 (VCAM-1) J Exp Med. 1998;188:2187–2191. doi: 10.1084/jem.188.11.2187. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Dransfield I, Hogg N. Regulated expression of Mg2+ binding epitope on leukocyte integrin alpha subunits. EMBO J. 1989;8:3759–3765. doi: 10.1002/j.1460-2075.1989.tb08552.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Lu C, Shimaoka M, Zang Q, Takagi J, Springer TA. Locking in alternate conformations of the integrin alphaLbeta2 I domain with disulfide bonds reveals functional relationships among integrin domains. Proc Natl Acad Sci USA. 2001;98:2393–2398. doi: 10.1073/pnas.041618598. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Wilkins JA, Li A, Ni H, Stupack DG, Shen C. Control of beta1 integrin function. Localization of stimulatory epitopes. J Biol Chem. 1996;271:3046–3051. [PubMed] [Google Scholar]
- 35.Johansson MW, Barthel SR, Swenson CA, Evans MD, Jarjour NN, Mosher DF, Busse WW. Eosinophil beta(1) integrin activation state correlates with asthma activity in a blind study of inhaled corticosteroid withdrawal. J Allergy Clin Immunol. 2006;117:1502–1504. doi: 10.1016/j.jaci.2006.02.032. [DOI] [PubMed] [Google Scholar]
- 36.Barthel SR, Jarjour NN, Mosher DF, Johansson MW. Dissection of the hyperadhesive phenotype of airway eosinophils in asthma. Am J Respir Cell Mol Biol. 2006;35:378–386. doi: 10.1165/rcmb.2006-0027OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Leitinger B, Hogg N. Effects of I domain deletion on the function of the beta2 integrin lymphocyte function-associated antigen-1. Mol Biol Cell. 2000;11:677–690. doi: 10.1091/mbc.11.2.677. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Kelly EA, Rodriguez RR, Busse WW, Jarjour NN. The effect of segmental bronchoprovocation with allergen on airway lymphocyte function. Am J Respir Crit Care Med. 1997;156:1421–1428. doi: 10.1164/ajrccm.156.5.9703054. [DOI] [PubMed] [Google Scholar]
- 39.Lemanske RF., Jr The late phase response: clinical implications. Adv Intern Med. 1991;36:171–193. [PubMed] [Google Scholar]
- 40.Ni H, Li A, Simonsen N, Wilkins JA. Integrin activation by dithiothreitol or Mn2+ induces a ligand-occupied conformation and exposure of a novel NH2-terminal regulatory site on the beta1 integrin chain. J Biol Chem. 1998;273:7981–7987. doi: 10.1074/jbc.273.14.7981. [DOI] [PubMed] [Google Scholar]
- 41.Mould AP, Travis MA, Barton SJ, Hamilton JA, Askari JA, Craig SE, Macdonald PR, Kammerer RA, Buckley PA, Humphries MJ. Evidence that monoclonal antibodies directed against the integrin beta subunit plexin/semaphorin/integrin domain stimulate function by inducing receptor extension. J Biol Chem. 2005;280:4238–4246. doi: 10.1074/jbc.M412240200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Kanters D, ten Hove W, Luijk B, van Aalst C, Schweizer RC, Lammers JW, Leufkens HG, Raaijmakers JA, Bracke M, Koenderman L. Expression of activated Fc gamma RII discriminates between multiple granulocyte-priming phenotypes in peripheral blood of allergic asthmatic subjects. J Allergy Clin Immunol. 2007;120:1073–1081. doi: 10.1016/j.jaci.2007.06.021. [DOI] [PubMed] [Google Scholar]
- 43.Georas SN, Liu MC, Newman W, Beall LD, Stealey BA, Bochner BS. Altered adhesion molecule expression and endothelial cell activation accompany the recruitment of human granulocytes to the lung after segmental antigen challenge. Am J Respir Cell Mol Biol. 1992;7:261–269. doi: 10.1165/ajrcmb/7.3.261. [DOI] [PubMed] [Google Scholar]
- 44.Sedgwick JB, Calhoun WJ, Vrtis RF, Bates ME, McAllister PK, Busse WW. Comparison of airway and blood eosinophil function after in vivo antigen challenge. J Immunol. 1992;149:3710–3718. [PubMed] [Google Scholar]
- 45.Kroegel C, Liu MC, Hubbard WC, Lichtenstein LM, Bochner BS. Blood and bronchoalveolar eosinophils in allergic subjects after segmental antigen challenge: surface phenotype, density heterogeneity, and prostanoid production. J Allergy Clin Immunol. 1994;93:725–734. doi: 10.1016/0091-6749(94)90252-6. [DOI] [PubMed] [Google Scholar]
- 46.Mengelers HJ, Maikoe T, Brinkman L, Hooibrink B, Lammers JW, Koenderman L. Immunophenotyping of eosinophils recovered from blood and BAL of allergic asthmatics. Am J Respir Crit Care Med. 1994;149:345–351. doi: 10.1164/ajrccm.149.2.8306028. [DOI] [PubMed] [Google Scholar]
- 47.Aalbers R, Kauffman HF, Vrugt B, Smith M, Koeter GH, Timens W, de Monchy JG. Bronchial lavage and bronchoalveolar lavage in allergen-induced single early and dual asthmatic responders. Am Rev Respir Dis. 1993;147:76–81. doi: 10.1164/ajrccm/147.1.76. [DOI] [PubMed] [Google Scholar]
- 48.Shaver JR, O'Connor JJ, Pollice M, Cho SK, Kane GC, Fish JE, Peters SP. Pulmonary inflammation after segmental ragweed challenge in allergic asthmatic and nonasthmatic subjects. Am J Respir Crit Care Med. 1995;152:1189–1191. doi: 10.1164/ajrccm.152.4.7551369. [DOI] [PubMed] [Google Scholar]
- 49.Gauvreau GM, Watson RM, O'Byrne PM. Kinetics of allergen-induced airway eosinophilic cytokine production and airway inflammation. Am J Respir Crit Care Med. 1999;160:640–647. doi: 10.1164/ajrccm.160.2.9809130. [DOI] [PubMed] [Google Scholar]
- 50.Wood LJ, Sehmi R, Dorman S, Hamid Q, Tulic MK, Watson RM, Foley R, Wasi P, Denburg JA, Gauvreau G, O'Byrne PM. Allergen-induced increases in bone marrow T lymphocytes and interleukin-5 expression in subjects with asthma. Am J Respir Crit Care Med. 2002;166:883–889. doi: 10.1164/rccm.2108015. [DOI] [PubMed] [Google Scholar]
- 51.Moser R, Fehr J, Bruijnzeel PLB. Il-4 controls the selective endothelium-driven transmigration of eosinophils from allergic individuals. J Immunol. 1992;149:1432–1438. [PubMed] [Google Scholar]
- 52.Ebisawa M, Yamada T, Bickel C, Klunk D, Schleimer RP. Eosinophil transendothelial migration induced by cytokines. III. Effect of the chemokine RANTES. J Immunol. 1994;153:2153–2160. [PubMed] [Google Scholar]
- 53.Jia GQ, Gonzalo JA, Hidalgo A, Wagner D, Cybulsky M, Gutierrez-Ramos JC. Selective eosinophil transendothelial migration triggered by eotaxin via modulation of Mac-1/ICAM-1 and VLA-4/VCAM-1 interactions. Int Immunol. 1999;11:1–10. doi: 10.1093/intimm/11.1.1. [DOI] [PubMed] [Google Scholar]
- 54.Nagata M, Yamamoto H, Tabe K, Sakamoto Y. Eosinophil transmigration across VCAM-1-expressing endothelial cells is upregulated by antigen-stimulated mononuclear cells. Int Arch Allergy Immunol. 2001;125(Suppl 1):7–11. doi: 10.1159/000053844. [DOI] [PubMed] [Google Scholar]
- 55.Norris V, Choong L, Tran D, Corden Z, Boyce M, Arshad H, Holgate S, O'Connor B, Millet S, Miller B, Rohatagi S, Kirkesseli S. Effect of IVL745, a VLA-4 antagonist, on allergen-induced bronchoconstriction in patients with asthma. J Allergy Clin Immunol. 2005;116:761–767. doi: 10.1016/j.jaci.2005.04.045. [DOI] [PubMed] [Google Scholar]
- 56.Yamamoto H, Nagata M, Sakamoto Y. CC chemokines and transmigration of eosinophils in the presence of vascular cell adhesion molecule 1. Ann Allergy Asthma Immunol. 2005;94:292–300. doi: 10.1016/S1081-1206(10)61311-7. [DOI] [PubMed] [Google Scholar]
- 57.Banerjee ER, Jiang Y, Henderson WR, Jr, Scott LM, Papayannopoulou T. Alpha4 and beta2 integrins have nonredundant roles for asthma development, but for optimal allergen sensitization only alpha4 is critical. Exp Hematol. 2007;35:605–617. doi: 10.1016/j.exphem.2007.01.052. [DOI] [PubMed] [Google Scholar]
- 58.Liu LY, Swensen CA, Kelly EA, Kita H, Busse WW. The relationship of sputum eosinophilia and sputum cell generation of IL-5. J Allergy Clin Immunol. 2000;106:1063–1069. doi: 10.1067/mai.2000.110792. [DOI] [PubMed] [Google Scholar]
- 59.Kelly EAB, Busse WW, Jarjour NN. A comparison of the airway response to segmental antigen bronchoprovocation in atopic asthma and allergic rhinitis. J Allergy Clin Immunol. 2003;111:79–86. doi: 10.1067/mai.2003.28. [DOI] [PubMed] [Google Scholar]
- 60.Zhu X, Munoz NM, Kim KP, Sano H, Cho W, Leff AR. Cytosolic phospholipase A2 activation is essential for beta 1 and beta 2 integrin-dependent adhesion of human eosinophils. J Immunol. 1999;163:3423–3249. [PubMed] [Google Scholar]
- 61.Wong CK, Ip WK, Lam CW. Interleukin-3, -5, and granulocyte macrophage colony-stimulating factor-induced adhesion molecule expression on eosinophils by p38 mitogen-activated protein kinase and nuclear factor-[kappa] B. Am J Respir Cell Mol Biol. 2003;29:133–147. doi: 10.1165/rcmb.2002-0289OC. [DOI] [PubMed] [Google Scholar]
- 62.Rennard SI, Basset G, Lecossier D, O'Donnell KM, Pinkston P, Martin PG, Crystal RG. Estimation of volume of epithelial lining fluid recovered by lavage using urea as marker of dilution. J Appl Physiol. 1986;60:532–538. doi: 10.1152/jappl.1986.60.2.532. [DOI] [PubMed] [Google Scholar]
- 63.Chihara J, Plumas J, Gruart V, Tavernier J, Prin L, Capron A, Capron M. Characterization of a receptor for interleukin 5 on human eosinophils: variable expression and induction by granulocyte/macrophage colony-stimulating factor. J Exp Med. 1990;172:1347–1351. doi: 10.1084/jem.172.5.1347. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Plaetinck G, Van der Heyden J, Tavernier J, Fache I, Tuypens T, Fischkoff S, Fiers W, Devos R. Characterization of interleukin 5 receptors on eosinophilic sublines from human promyelocytic leukemia (HL-60) cells. J Exp Med. 1990;172:683–691. doi: 10.1084/jem.172.3.683. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Ingley E, I, Young G. Characterization of a receptor for interleukin-5 on human eosinophils and the myeloid leukemia line HL-60. Blood. 1991;78:339–344. [PubMed] [Google Scholar]
- 66.Migita M, Yamaguchi N, Mita S, Higuchi S, Hitoshi Y, Yoshida Y, Tomonaga M, Matsuda I, Tominaga A, Takatsu K. Characterization of the human IL-5 receptors on eosinophils. Cell Immunol. 1991;133:484–497. doi: 10.1016/0008-8749(91)90120-z. [DOI] [PubMed] [Google Scholar]
- 67.Tavernier J, Devos R, Cornelis S, Tuypens T, Van der Heyden J, Fiers W, Plaetinck G. A human high affinity interleukin-5 receptor (IL5R) is composed of an IL5-specific alpha chain and a beta chain shared with the receptor for GM-CSF. Cell. 1991;66:1175–1184. doi: 10.1016/0092-8674(91)90040-6. [DOI] [PubMed] [Google Scholar]
- 68.Zhang J, Wu P, Kuvelkar R, Schwartz JL, Egan RW, Billah MM, Wang P. A scintillation proximity assay for human interleukin-5 (hIL-5) high-affinity binding in insect cells coexpressing hIL-5 receptor alpha and beta subunits. Anal Biochem. 1999;268:134–142. doi: 10.1006/abio.1998.3058. [DOI] [PubMed] [Google Scholar]
- 69.Bates ME, V, Green L, Bertics PJ. ERK1 and ERK2 activation by chemotactic factors in human eosinophils is interleukin 5-dependent and contributes to leukotriene C(4) biosynthesis. J Biol Chem. 2000;275:10968–10975. doi: 10.1074/jbc.275.15.10968. [DOI] [PubMed] [Google Scholar]
- 70.Hall DJ, Cui J, Bates ME, Stout BA, Koenderman L, Coffer PJ, Bertics PJ. Transduction of a dominant-negative H-Ras into human eosinophils attenuates extracellular signal-regulated kinase activation and interleukin-5-mediated cell viability. Blood. 2001;98:2014–2021. doi: 10.1182/blood.v98.7.2014. [DOI] [PubMed] [Google Scholar]
- 71.Liu LY, Sedgwick JB, Bates ME, Vrtis RF, Gern JE, Kita H, Jarjour NN, Busse WW, Kelly EA. Decreased expression of membrane IL-5 receptor alpha on human eosinophils: II. IL-5 down-modulates its receptor via a proteinase-mediated process. J Immunol. 2002;169:6459–6466. doi: 10.4049/jimmunol.169.11.6459. [DOI] [PubMed] [Google Scholar]
- 72.Bates ME, Liu LY, Esnault S, Stout BA, Fonkem E, Kung V, Sedgwick JB, Kelly EA, Bates DM, Malter JS, Busse WW, Bertics PJ. Expression of interleukin-5- and granulocyte macrophage-colony-stimulating factor-responsive genes in blood and airway eosinophils. Am J Respir Cell Mol Biol. 2004;30:736–743. doi: 10.1165/rcmb.2003-0234OC. [DOI] [PubMed] [Google Scholar]
- 73.Zangrilli JG, Shaver JR, Cirelli RA, Cho SK, Garlisi CG, Falcone A, Cuss FM, Fish JE, Peters SP. sVCAM-1 levels after segmental antigen challenge correlate with eosinophil influx, IL-4 and IL-5 production, and the late phase response. Am J Respir Crit Care Med. 1995;151:1346–1353. doi: 10.1164/ajrccm.151.5.7537586. [DOI] [PubMed] [Google Scholar]
- 74.Sur S, Kita H, Gleich GJ, Chenier TC, Hunt LW. Eosinophil recruitment is associated with IL-5, but not with RANTES, twenty-four hours after allergen challenge. J Allergy Clin Immunol. 1996;97:1272–1278. doi: 10.1016/s0091-6749(96)70195-1. [DOI] [PubMed] [Google Scholar]
- 75.Jarjour NN, Calhoun WJ, Kelly EA, Gleich GJ, Schwartz LB, Busse WW. The immediate and late allergic response to segmental bronchopulmonary provocation in asthma. Am J Respir Crit Care Med. 1997;155:1515–1521. doi: 10.1164/ajrccm.155.5.9154851. [DOI] [PubMed] [Google Scholar]
- 76.Sedgwick JB, Calhoun WJ, Gleich GJ, Kita H, Abrams JS, Schwartz LB, Volovitz B, Ben-Yaakov M, Busse WW. Immediate and late airway response of allergic rhinitis patients to segmental antigen challenge. Characterization of eosinophil and mast cell mediators. Am Rev Resp Dis. 1991;144:1274–1281. doi: 10.1164/ajrccm/144.6.1274. [DOI] [PubMed] [Google Scholar]
- 77.Berends C, Dijkhuizen B, de Monchy JG, Gerritsen J, Kauffman HF. Induction of low density and up-regulation of CD11b expression of neutrophils and eosinophils by dextran sedimentation and centrifugation. J Immunol Methods. 1994;167:183–193. doi: 10.1016/0022-1759(94)90087-6. [DOI] [PubMed] [Google Scholar]
- 78.Todd RF, 3rd, Arnaout MA, Rosin RE, Crowley CA, Peters WA, Babior BM. Subcellular localization of the large subunit of Mo1 (Mo1 alpha; formerly gp 110), a surface glycoprotein associated with neutrophil adhesion. J Clin Invest. 1984;74:1280–1290. doi: 10.1172/JCI111538. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Mathias JR, Perrin BJ, Liu TX, Kanki J, Look AT, Huttenlocher A. Resolution of inflammation by retrograde chemotaxis of neutrophils in transgenic zebrafish. J Leukoc Biol. 2006;80:1281–1288. doi: 10.1189/jlb.0506346. [DOI] [PubMed] [Google Scholar]
- 80.Buckley CD, Ross EA, McGettrick HM, Osborne CE, Haworth O, Schmutz C, Stone PC, Salmon M, Matharu NM, Vohra RK, Nash GB, Rainger GE. Identification of a phenotypically and functionally distinct population of long-lived neutrophils in a model of reverse endothelial migration. J Leukoc Biol. 2006;79:303–311. doi: 10.1189/jlb.0905496. [DOI] [PubMed] [Google Scholar]
- 81.Warringa RA, Mengelers HJ, Raaijmakers JA, Bruijnzeel PL, Koenderman L. Upregulation of formyl-peptide and interleukin-8-induced eosinophil chemotaxis in patients with allergic asthma. J Allergy Clin Immunol. 1993;91:1198–1205. doi: 10.1016/0091-6749(93)90323-8. [DOI] [PubMed] [Google Scholar]
- 82.Sehmi R, Wood LJ, Watson R, Foley R, Hamid Q, O'Byrne PM, Denburg JA. Allergen-induced increases in IL-5 receptor alpha-subunit expression on bone marrow-derived CD34+ cells from asthmatic subjects. A novel marker of progenitor cell commitment towards eosinophilic differentiation. J Clin Invest. 1997;100:2466–2475. doi: 10.1172/JCI119789. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Sehmi R, Dorman S, Baatjes A, Watson R, Foley R, Ying S, Robinson DS, Kay AB, O'Byrne PM, Denburg JA. Allergen-induced fluctuation in CC chemokine receptor 3 expression on bone marrow CD34+ cells from asthmatic subjects: significance for mobilization of haemopoietic progenitor cells in allergic inflammation. Immunology. 2003;109:536–546. doi: 10.1046/j.1365-2567.2003.01686.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Moser R, Fehr J, Olgiati L, Bruijnzeel PL. Migration of primed human eosinophils across cytokine-activated endothelial cell monolayers. Blood. 1992;79:2937–2945. [PubMed] [Google Scholar]
- 85.Nakajima H, Sano H, Nishimura T, Yoshida S, Iwamoto I. Role of vascular cell adhesion molecule 1/very late activation antigen 4 and intercellular adhesion molecule 1/lymphoycte function-associated antigen 1 interactions in antigen-induced eosinophil and T cell recruitment into the tissue. J Exp Med. 1994;179:1145–1154. doi: 10.1084/jem.179.4.1145. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.ten Hacken NH, Postma DS, Bosma F, Drok G, Rutgers B, Kraan J, Timens W. Vascular adhesion molecules in nocturnal asthma: a possible role for VCAM-1 in ongoing airway wall inflammation. Clin Exp Allergy. 1998;28:1518–1525. doi: 10.1046/j.1365-2222.1998.00426.x. [DOI] [PubMed] [Google Scholar]
- 87.Masinovsky B, Urdal D, Gallatin WM. IL-4 acts synergistically with IL-1 beta to promote lymphocyte adhesion to microvascular endothelium by induction of vascular cell adhesion molecule-1. J Immunol. 1990;145:2886–2895. [PubMed] [Google Scholar]
- 88.Thornhill MH, Haskard DO. IL-4 regulates endothelial cell activation by IL-1, tumor necrosis factor, or IFN-gamma. J Immunol. 1990;145:865–872. [PubMed] [Google Scholar]
- 89.Thornhill MH, Wellicome SM, Mahiouz DL, Lanchbury JS, Kyan-Aung U, Haskard DO. Tumor necrosis factor combines with IL-4 or IFN-gamma to selectively enhance endothelial cell adhesiveness for T cells. The contribution of vascular cell adhesion molecule-1-dependent and -independent binding mechanisms. J Immunol. 1991;146:592–598. [PubMed] [Google Scholar]
- 90.Weller PF, Rand TH, Goelz SE, Chi-Rosso G, Lobb RR. Human eosinophil adherence to vascular endothelium mediated by binding to vascular cell adhesion molecule 1 and endothelial leukocyte adhesion molecule 1. Proc Natl Acad Sci USA. 1991;88:7430–7433. doi: 10.1073/pnas.88.16.7430. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Schleimer RP, Sterbinsky SA, Kaiser J, Bickel CA, Klunk DA, Tomioka K, Newman W, Luscinskas FW, Gimbrone MA, Jr, McIntyre BW, Bochner BS. IL-4 induces adherence of human eosinophils and basophils but not neutrophils to endothelium. Association with expression of VCAM-1. J Immunol. 1992;148:1086–1092. [PubMed] [Google Scholar]
- 92.Barthel SR, Annis DS, Mosher DF, Johansson MW. Differential engagement of modules 1 and 4 of vascular cell adhesion molecule-1 (CD106) by integrins alpha4beta1 (CD49d/29) and alphaMbeta2 (CD11b/18) of eosinophils. J Biol Chem. 2006;281:32175–32187. doi: 10.1074/jbc.M600943200. [DOI] [PubMed] [Google Scholar]
- 93.Ebisawa M, Bochner BS, Georas SN, Schleimer RP. Eosinophil transendothelial migration induced by cytokines. I. Role of endothelial and eosinophil adhesion molecules in IL-1 beta-induced transendothelial migration. J Immunol. 1992;149:4021–4028. [PubMed] [Google Scholar]
- 94.Liu L, Hakansson L, Ridefelt P, Garcia RC, Venge P. Priming of eosinophil migration across lung epithelial cell monolayers and upregulation of CD11b/CD18 are elicited by extracellular Ca2+ Am J Respir Cell Mol Biol. 2003;28:713–721. doi: 10.1165/rcmb.4771. [DOI] [PubMed] [Google Scholar]
- 95.Ulfman LH, Alblas J, van Aalst CW, Zwaginga JJ, Koenderman L. Differences in potency of CXC chemokine ligand 8-, CC chemokine ligand 11-, and C5a-induced modulation of integrin function on human eosinophils. J Immunol. 2005;175:6092–6099. doi: 10.4049/jimmunol.175.9.6092. [DOI] [PubMed] [Google Scholar]