Abstract
To determine the relationship between aortic wall thickness (WT) and distensibility with traditional cardiovascular risk factors in the Multi-Ethnic Study of Atherosclerosis (MESA) cohort; 1053 participants of the MESA study with cardiac MRI were consecutively selected for measurement of aortic wall thickness and distensibility. Double inversion recovery fast spin echo images of the thoracic aorta were obtained to measure average and maximum WT. Aortic distensibility (AD) was measured at the same level using a gradient echo cine sequence. Both average and maximum WT were positively correlated with increasing age, and AD was inversely related to age (p<0.01). Compared to normotensive participants, those with hypertension had significantly greater mean average WT (2.45 mm vs. 2.23 mm, p<0.01), maximum WT (3.61 mm vs. 3.41 mm, p<0.01) and lower AD (0.15 vs. 0.2 mm Hg−1, p<0.01), respectively. In multiple regression analysis, older age and hypertension were significantly associated with higher mean average WT, while older age, male gender, and higher blood pressure were associated with higher mean maximum WT. AD was inversely related to older age, hypertension, current smoker status, African-American ethnicity and lower HDL-cholesterol (HDL-c) levels. In conclusion, in the MESA cohort, older age and higher blood pressure were associated with higher aortic wall thickness and lower aortic distensibilility. Decreased aortic distensibility was further associated with current smoking, African American ethnicity and higher HDL-c levels.
Previous studies have evaluated the relationship between the aortic wall thickness (WT)1 and aortic distensibility (AD)2 with conventional cardiovascular risk factors of cardiovascular disorders. The combination of these 2 in relation to conventional risk factors has not been previously established in a large population-based study. The purpose of this study was to evaluate the relationship between structural and functional measures of the aorta as measured by MRI with conventional cardiovascular disease risk factors in a large population-based study.
Methods
The Multi-Ethnic Study of Atherosclerosis (MESA) is a population-based sample of 6,814 men and women who were 45–84 years old at the time of entry into the study. Participants without clinical cardiovascular disease were included in the study 3. The recruitment centers were located in 6 communities (Baltimore, New York, Minneapolis, Winston-Salem, Los Angeles, and Chicago) in the United States. For the purposes of this study, 1053 consecutive subjects were selected from 3 of the participating field centers (Johns Hopkins University; 255, Wake Forest University; 576, Columbia University; 222). These sites were selected because all used the same manufacturer MRI units (discussed below) with similar software. The institutional review boards at each of the recruiting field centers approved the study and all participants signed informed consent.
MRI was performed using 1.5-T whole body MRI systems: Signa CV/i or Signa LX (General Electric Medical Systems, Waukesha, WI). MRI of the aorta was performed using a double inversion recovery black blood fast spin-echo sequence with electrocardiogram (ECG) gating. Axial images of the descending thoracic aorta were obtained at the level of the right pulmonary artery at mid-diastole. Imaging parameters were TR, 2 R-R intervals; TE, 42; field of view, 36 cm; slice thickness, 6 mm; matrix size 512×256 interpolated to 512×512, echo-train length, 32; and receiver bandwidth, 62.5 kHz. .Gradient echo phase contrast cine MRI with ECG gating was performed to evaluate the distensibility of the aorta. Images of the ascending and descending aorta were obtained in the transverse plane at the level of the right pulmonary artery perpendicular to the vessel lumen. Imaging parameters were TR: 10; TE, 1.9; field of view, 34 cm; slice thickness, 8 mm; matrix 256×224, 2 signal averages, temporal resolution 20 msec, velocity encoding gradient 150 cm/sec; receiver bandwidth +/− 32kHz.
Wall measurements were performed by a single cardiologist using the Magnetic Resonance Analytical Software System (MASS) vessel wall version 5.1 (Medis, Leiden, The Netherlands). Images were magnified to 500 – 1000 %. The gray scale was inverted to suppress periaortic fat. The thickness of the mid thoracic descending aortic wall was measured using electronic calipers at four standard positions: 12, 3, 6, and 9 o'clock. The average value of these four measurements was then calculated. In addition, the maximal wall thickness was measured three times for each slice, and the average of these 3 measurements was used. A preliminary pilot study on 196 cases of the same original sample showed an interclass correlation coefficient of 0.980 for average WT and 0.991 for maximum WT. To determine the aortic distensibility, the minimum and maximum cross-sectional areas of the ascending aorta were determined using an automated contour routine using the software FLOW (Medis, Leiden, The Netherlands). Aortic distensibility (AD) was calculated as ; AD = (Maximum area — Minimum area) / [(Minimum area) ×Δ P] × 1000, where Δ P is the pulse pressure in mmHg. Pulse pressure was the difference between systolic and diastolic measurements of blood pressure. Blood pressure was measured immediately before and after the MRI aortic measurements while the patient was in the supine position on the MRI scanner gantry; the average systolic and diastolic values were then used to calculate pulse pressure. The MRI reader was blinded to all variables of the study subjects except the participant identification number.
Hypertension was defined as diastolic blood pressure (DBP) ≥90 mm Hg, systolic blood pressure (SBP) ≥140 mm Hg, or treatment for hypertension. Total cholesterol ≥240 mg/dl (6.2 mmol/l), LDL cholesterol (LDL-c) ≥160 mg/dl (4.1 mmol/l), and triglycerides ≥150 mg/dl (1.7 mmol/l) were considered as elevated. HDL-c <40 mg/dl (1.0 mmol/l) was defined as low. Hyperlipidemic patients were defined as either participants with abnormal lipid levels or receiving lipid-lowering therapy. The lipid levels was defined according to third report of the National Cholesterol Education Program (NCEP) (Adult Treatment Panel III) . Diabetic individuals were defined as either having fasting plasma glucose ≥126 mg/dl (7.0 mmol/l) or if the participant was receiving treatment for diabetes. Impaired fasting glucose was defined as fasting glucose of 100–125 mg/dL and not taking oral diabetic medication or insulin. Smoking status was defined as current smoking, former smoking, or never smoked.
Unpaired student t-test and analysis of variance (ANOVA) were used to compare means of aortic average and maximum WT among different levels of binary and categorical variables, respectively. A significant p value in analysis of variance would lead to an appropriate multiple comparisons test. Linear regression models were used to determine the association of average and maximum WT and distensibility with continuous and categorical variables. Unless stated otherwise, all results of the linear regression models are reported as regression coefficients for 1 standard deviation (SD) increase in the continuous variables. For categorical variables, regression coefficients are defined relative to the reference category. . Multivariable linear regression models were used to assess the independent association of risk factors with aortic wall thickness and distensibility. The variables included in the multiple regression model were age, gender, race (Caucasian, African-American, or Hispanic), body mass index (BMI), serum LDL-c level, serum HDL-c level, treatment for hyperlipidemia, glucose tolerance (in 4 categories: normal, impaired glucose tolerance, untreated DM, and treated DM), systolic blood pressure, antihypertensive medication, and cigarette smoking (3 categories: non-smoker, former smoker, current smoker). All models were checked for the assumptions and for the effect of highly influential observations. Statistical analyses were performed using Stata version 8.2 for Windows (StataCorp, College Station, Texas, USA). A p value < 0.05 was considered statistically significant.
Results
The age range for1053 consecutive participants, was 45–84, mean [standard deviation (SD)]: 62.09 (9.7) (Table 1). 503 participants were men. Average WT was measured in 1053 cases, maximum WT was measured in 782 participants, and AD was measured in 989 cases. The reason for incomplete measurement on all samples was limited image quality (including lack of blood suppression or motion). Most of the participants were of non-Hispanic White background (51 %). Table 1 shows the demographic information of the studied sample. The mean (SD) of average and maximum WT were 2.35 (0.50) and 3.51 (0.84) mm, respectively. The mean (SD) for AD was 1.7 (1.2) mm Hg−1.
Table 1. Participant characteristics.
General characteristic of this study population in comparison with all MESA participants. Data are presented as number, mean ± SD, or range. Percentages are in parentheses. BMI = body mass index; DBP = diastolic blood pressure; HDL = high-density lipoprotein; LDL = low-density lipoprotein; SBP = systolic blood pressure, IFG = impaired fasting glucose.
| Variable | n (%) | All MESA Participants |
|---|---|---|
| Men | 503 (47.8 %) | 3213 (47.2 %) |
| Women | 550 (52.2 %) | 3601 (52.8 %) |
| Total | 1,053 (100 %) | 6814 (100 %) |
| Non-Hispanic White | 537 (51.0 %) | 2624 (38.5 %) |
| African-American | 414 (39.3 %) | 1895 (27.8 %) |
| Hispanic | 102 (9.7 %) | 1492 (21.9 %) |
| Chinese | 0 | 803 (11.8 %) |
| Age (years) | 62.09± 9.7 | 62.1 ± 10.2 |
| Body mass index (kg/m2) | 28.2 ± 5.0 | 28.3 ± 5.5 |
| Systolic blood pressure (mm Hg) | 130.0 ± 21.4 | 126.6 ± 21.5 |
| Diastolic blood pressure (mm Hg) | 73.4 ± 10.4 | 71.9±10.3 |
| Treatment for hypertension | 432 (41.0 %) | 2455 (36.0 %) |
| Hypertension | 569 (54.0 %) | 3251 (47.7 %) |
| Total cholesterol (mg/dl) | 191.1 ± 34.1 | 194.2± 35.7 |
| LDL cholesterol (mg/dl) | 115.6 ± 30.7 | 117.2 ± 31.5 |
| HDL cholesterol (mg/dl) | 51.2 ± 14.9 | 51.0 ± 14.8 |
| Triglycerides (mg/dl) | 123.2 ± 87.0 | 131.6 ± 88.8 |
| Treatment for hyperlipidemia | 187 (17.8 %) | 1069 (15.7 %) |
| Never smoked | 474 (45.4 %) | 3426 (50.6 %) |
| Former smoker | 420 (40.2 %) | 2464 (36.4 %) |
| Current smoker | 150 (14.4 %) | 885 (13.06 %) |
| Plasma glucose (mg/dl) | 101.4 ± 26.4 | 104.5 ± 30.9 |
| Impaired fasting glucose | 266 (25.4 %) | 1895 (27.9 %) |
| Untreated diabetes mellitus | 36 (3.4 %) | 293 (4.3 %) |
| Treated diabetes mellitus | 86 (8.2 %) | 676 (10 %) |
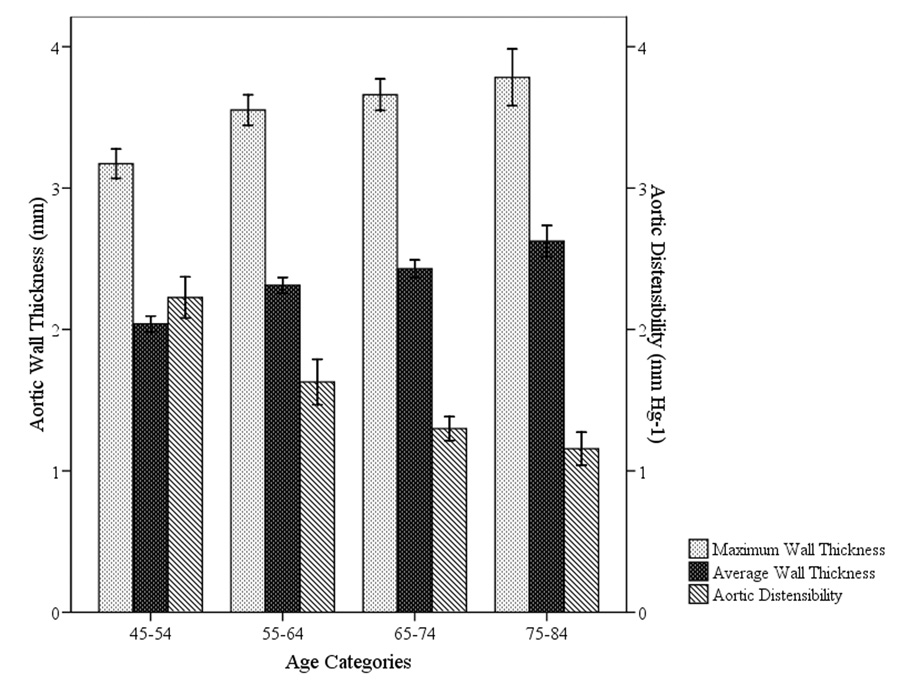
There was a dose-response relationship between average WT, maximum WT and AD and age group (p <0.01). Each 10 year increase in age was associated with 0.2 mm increase in average and maximum WT (both p <0.01). The dose response relationship for AD was −0.04 mm Hg−1 decrease in AD each year (p <0.01) (Figure 1). Men had higher maximum WT (3.60 vs. 3.43 mm, p<0.01) than women while average WT (2.38 vs. 2.33 mm, p = 0.10) was similar. AD was similar for women and men (1.7 (1.2) mm Hg−1, 1.7 (1.0) mm Hg−1, respectively). In multiple regression analysis, the difference in maximum WT between genders (0.19 mm) remained significant (p < 0.01).
Figure 1.
Average and maximum aortic wall thickness (scale on the left) and aortic distensibility (scale on the right) cross different age categories (p <0.01).
African-Americans and Whites had similar average and maximum WT, but Hispanics had lower average WT than Whites (2.21 vs. 2.36 mm, p<0.01, respectively) and maximum WT (3.31 vs. 3.54 mm, p:0.02, respectively). The mean AD for Whites, African-Americans and Hispanics was 1.7 (1.0), 1.5 (0.9) and 1.7 (1.0) mm Hg−1, respectively. AD was significantly lower in African-Americans compared to other ethnicities in multiple regression analysis. In participants with AD measurements, the number (%) of participants with high blood pressure across different ethnic groups were; 217 (43.5) out of 498 for non-Hispanic Whites, 244 (62.1) out of 393 for African American, and 35 (37.2) out of 94 for Hispanics. Participants with hypertension had a greater average WT (2.45 vs. 2.23 mm, p<0.01) and maximum WT than non-hypertensive individuals (3.61 vs. 3.41 mm, p < 0.01, respectively). Hypertensive participants also had a more rigid aorta (AD 0.15 vs. 0.21 mm Hg−1 for reference group, p < 0.01, respectively) than participants with normal blood pressure. In multiple regression analysis, both systolic and diastolic blood pressure were positively associated with average and maximum WT and negatively associated with AD, but in the backward regression modeling, the slightly stronger association of systolic blood pressure with the outcome variables led to it being retained in the models and diastolic blood pressure being dropped.
Maximum WT was significantly greater in untreated diabetic participants compared to non-diabetics (difference: 0.3 mm, p < 0.01). In univariable analysis (Table 2, AD was lower in treated diabetics compare to non-diabetics (−0.3 mm Hg−1, p = 0.02); however this relationship was not significant in multiple regression analysis. Regarding smoking, there was decreased AD in current smokers (−0.3 mm Hg−1, p = 0.004) compared to former and nonsmokers in multivariable regression analysis; all other comparisons for average and maximum WT showed no differences. There were no significant associations between family history of myocardial infarction, serum cholesterol or LDL-c, for average and maximum WT and AD. However, lower HDL-c was associated with increased distensibility in multiple regression analysis (regression coefficient, 0.07 mm Hg−1 decrease in AD for every 14.9 mg/dl increase in HDL-c value, p = 0.04).
Table 2. Univariate analysis.
Simple linear regression coefficients relating aortic wall parameters (p value) and traditional cardiovascular risk factors. Data are presented as univariable coefficients (p value).
Using medications for hypertension and hyperlipidemia were included in the univariable models for blood pressure or serum lipids, respectively, as independent variables.
| Variable | Univariable analysis coefficients* (p-values) | ||
|---|---|---|---|
| Maximum WT (mm) | Average WT | AD ** (mm Hg−)1 | |
| Age (per 10.2 years) | 0.21 (<0.01) | 0.21 (<0.01) | −0.4 (<0.01) |
| Male vs. Female | 0.17 (<0.01) | 0.05 (0.10) | 0.02 (0.72) |
| Systolic blood pressure (per 21.5 mm Hg) | 0.15 (<0.01) | 0.12 (<0.01) | −0.3 (<0.01) |
| Diastolic blood pressure (per 10.3 mm Hg) | 0.08 (<0.01) | 0.04 (0.01) | −0.1 (<0.01) |
| Normal glucose (<100 mg/dl) | Reference | Reference | Reference |
| Impaired fasting glucose (≥100 and <126 | 0.10 (0.18) | 0.07 (0.06) | −0.06 (0.4) |
| Untreated diabetes mellitus | 0.29(0.07) | 0.31 (<0.01) | −0.3 (0.07) |
| Treated diabetes mellitus | 0.15 (0.19) | 0.02 (0.68) | −0.3 (0.02) |
| Total Cholesterol (per 34.1 mg/dl) | 0.013 (0.6) | 0.02 (0.2) | −0.01 (0.7) |
| HDL (per 14.9 mg/dl ) | −0.06 (0.05) | 0.009 (0.5) | −0.05 (<0.01) |
| LDL (per 30.7 mg/dl) | 0.016 (0.5) | 0.015 (0.3) | 0.003 (0.8) |
| Never smoker | Reference | Reference | Reference |
| Former smoker | 0.17(<0.01) | 0.06 (0.05) | −0.1 (0.15) |
| Current smoker | −0.05 (0.59) | −0.03 (0.53) | −0.1 (0.24) |
| Body mass index (kg/m2) | 0.05 (0.10) | −0.006 (0.71) | 0.03 (0.41) |
| <25 | Reference | Reference | Reference |
| 25–29.99 | 0.14 (0.07) | 0.05 (0.169) | 0.07 (0.39) |
| ≥30 | 0.14 (0.07) | −0.006 (0.89) | 0.1 (0.19) |
| Non-Hispanic White | Reference | Reference | Reference |
| African-American | −0.001 (0.99) | 0.01 (0.67) | −0.2 (0.002) |
| Hispanic | −0.23 (0.02) | −0.15 (0.01) | 0.02 (0.84) |
All results are reported as regression coefficient for 1 standard deviation (SD) increase in the continuous variables or with respect to the defined reference category for categorical variables.
BMI = body mass index; DBP = diastolic blood pressure; HDL = high-density lipoprotein; LDL = low-density lipoprotein; SBP = systolic blood pressure; WT= wall thickness.
In multiple regression analysis (Table 3), age, male gender and higher systolic blood pressure were associated with higher maximum WT. Higher average WT was also associated with age and higher systolic blood pressure. Decreased AD was associated with age, higher systolic blood pressure, current smoking, higher HDL-c and African-American ethnic background.
Table 3. Multiple regression analysis.
Relationship of aortic wall parameters (p value) to traditional cardiovascular risk factors from the multiple regression analysis.
| Variable | Regression coefficients * (p-values) | ||
|---|---|---|---|
| Maximum WT (mm) | Average WT (mm) | AD (mm Hg−1) | |
| Age (per 10.2 years) | 0.18 (<0.01) | 0.18 (<0.01) | −0.3 (<0.01) |
| Male vs. Female | 0.15 (0.02) | 0.03 (0.35) | −0.035 (0.6) |
| Systolic blood pressure (per 21.5 mm Hg) | 0.09 (<0.01) | 0.055 (<0.01) | −0.2 (<0.01) |
| Never smoker | Reference | Reference | Reference |
| Former smoker | 0.11 (0.1) | 0.02 (0.42) | −0.03 (0.61) |
| Current smoker | 0.05 (0.6) | 0.04 (0.33) | −0.3 (0.004) |
| Non-Hispanic White | Reference | Reference | Reference |
| African-American | −0.03 (0.64) | 0.02 (0.5) | −0.18 (0.01) |
| Hispanic | −0.02 (0.05) | −0.07 (0.15) | −0.16 (0.15) |
| HDL per (14.9 mg/dl) | −0.03 (0.3) | −0.02 (0.2) | −0.07 (0.04) |
The variables included in multiple regression model were age, gender, race, body mass index categories, serum LDL level, serum HDL level, treatment for hyperlipidemia, diabetes mellitus categories, systolic blood pressure, antihypertensive medication, and cigarette smoking.
HDL = high-density lipoprotein; WT = wall thickness.
Discussion
We evaluated the relationship of both aortic wall thickness and distensibility, measured by MRI, in relation to traditional cardiovascular risk factors. This study is the largest population-based study to assess both aortic wall thickness and distensibility in a population free of established cardiovascular disease. In this population, the primary factors related to increased aortic wall thickness as well as decreased aortic distensibility were older age and hypertension. Maximum aortic wall thickness was greater in men than women. Decreased distensibility (a stiffer aorta) was associated with older age, hypertension, and African-American ethnicity. Additionally, decreased aortic distensibility was present in current smokers and in individuals with higher HDL-c levels.
The associations between age and aortic wall thickness 4,5, and aortic distensibility by pulse wave velocity (PWV)6 and MRI 7 have been reported in previous studies. Pathologic studies in animal models have shown that these changes can be attributed to structural modification of the arterial wall with increase in medial thickness, collagen content, and collagen/elastin ratio but decreases in elastin density. This is probably due to disintegration of orderly arrangement of elastic fibers and laminae with an increase in collagenous material and calcium deposition 8,9. Since increased aortic stiffness10,11 and increased aortic wall thickness 12 13 have been proposed to be associated with increased incidence of cardiovascular events in rapidly growing number of older population, therefore burden of disease related to aortic stiffness and atherosclerosis would be expected to rise significantly in the future.
Increased blood pressure was associated with increased average and maximum WT (Figure 2), and decreased AD. The association of aortic atherosclerotic plaque and systolic blood pressure has been demonstrated in previous asymptomatic cohorts 14. and MRI studies of patients with advanced disease 4. Also previous studies have examined the relationship of hypertension and aortic compliance by MRI 15. Hypertension may predispose to aortic atherosclerosis, as hypothesized for other atherosclerotic manifestations. Alternatively, aortic atherosclerosis may be associated with increased stiffness of the ascending aorta, resulting in secondary elevation of systemic blood pressure 16.
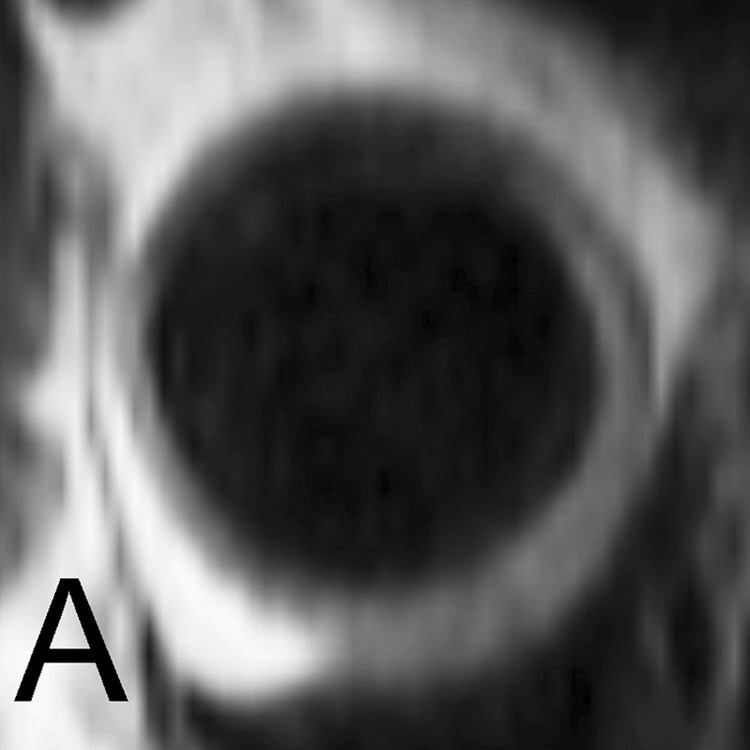
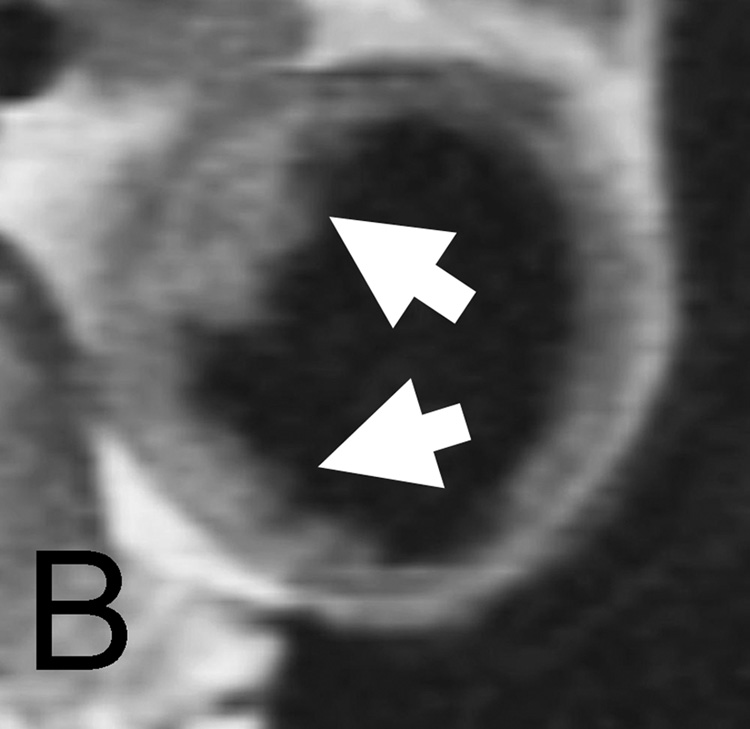
Figure 2.
Normal cross-section of the descending aortic wall in an 81-year old male without history of diabetes and hypertension, and no indication of atherosclerotic plaque (A). Descending aortic wall of a 67-year old male with history of hypertension (B) the area of increased wall thickness is shown by arrow.
There was significantly lower distensibility of the aorta in African-Americans compared to Whites and Hispanics. These results are consistent with previous studies of pulse wave velocity analysis 17,18. This has been postulated to be related to differences in beta adrenergic sensitivity 18,19 and nicotine metabolism 20,21. Interestingly the number of hypertensive participants in African American population was higher compare to other ethnic groups (62.1% compare to 43.5% of Whites and 37.2% of Hispanics). The presence of both findings in African American participants can be indicative of a cause and effect relationship between aortic distensibility and hypertension. However, this hypothesis should be validated by assessment of longitudinally ascertained aortic distensibility and blood pressure measures in this population.
Current smoking status was related to decreased aortic distensibility. In a pathologic study, McGill et al. 22, assessed the aorta from 2 cm proximal to the ligamentum arteriosum to a point 2 cm distal from the iliac bifurcation in a postmortem study of 1079 and 364 young men and women respectively. McGill et al. and others 4 concluded that smoking is associated with fatty streaks and raised lesions in the abdominal aorta. However, these studies did not report a significant relationship between morphological atherosclerotic changes in the thoracic aorta and smoking 4,22; similarly, our results did not show increased wall thickness in smokers. This relationship to current smoking status is suggesting that aortic function is a more sensitive indicator of disease compared to wall thickness measured by MRI.
Somewhat paradoxically, there was increased aortic distensibility in individuals with lower HDL-c levels. This observation was also reported in a small sample (55 subject) of the healthy subjects by using MRI 23 and a study of 680 hypertensive, age 65 –84 years measured by echocardiography 24. Previous pathologic studies of induced atheromatosis have shown that during early stages of atherosclerosis development, accumulation of cholesterol and foam cells may lead to increased distensibility 25,26. Stiffening may occur later, as collagen deposition and calcification occurs. Other studies, however, have shown lower HDL-c is related to decreased distensibility in large vessels 27 or no association has been detected 28,29. These variable results may be related to differences in the population that were studied or the methods used to assess aortic function and will need further exploration.
One of the limitations of this study is that in this study we measured aortic wall thickness on a single cross section of the descending aorta, at the level of right pulmonary artery. This level was selected in order to be highly reproducible in our pilot studies between different MRI technologists at different MRI centers. Due to the irregular nature of atherosclerotic changes in aortic wall, aortic wall thickness at a single level is unlikely to reflect plaque burden. We did not include participants with Chinese ethnicity in this study; therefore the sample is not ethnically an exact representative of the MESA study, and perhaps general population
Acknowledgement
This research was supported by contracts N01-HC-95159 through N01-HC-95169 from the National Heart, Lung, and Blood Institute. The authors thank the other investigators, the staff, and the participants of the MESA study for their valuable contributions. A full list of participating MESA investigators and institutions can be found at http://www.mesa-nhlbi.org.
Grant Support: This research was supported by contracts N01-HC-95159 through N01-HC-95169 from the National Heart, Lung, and Blood Institute. Bethesda, MD, USA.
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
References
- 1.Amarenco P, Cohen A, Tzourio C, Bertrand B, Hommel M, Besson G, Chauvel C, Touboul PJ, Bousser MG. Atherosclerotic disease of the aortic arch and the risk of ischemic stroke. N Engl J Med. 1994;331:1474–1479. doi: 10.1056/NEJM199412013312202. [DOI] [PubMed] [Google Scholar]
- 2.Laurent S, Boutouyrie P, Asmar R, Gautier I, Laloux B, Guize L, Ducimetiere P, Benetos A. Aortic stiffness is an independent predictor of all-cause and cardiovascular mortality in hypertensive patients. Hypertension. 2001;37:1236–1241. doi: 10.1161/01.hyp.37.5.1236. [DOI] [PubMed] [Google Scholar]
- 3.Bild DE, Bluemke DA, Burke GL, Detrano R, Diez Roux AV, Folsom AR, Greenland P, Jacob DR, Jr, Kronmal R, Liu K, Nelson JC, O'Leary D, Saad MF, Shea S, Szklo M, Tracy RP. Multi-ethnic study of atherosclerosis: objectives and design. Am J Epidemiol. 2002;156:871–881. doi: 10.1093/aje/kwf113. [DOI] [PubMed] [Google Scholar]
- 4.Taniguchi H, Momiyama Y, Fayad ZA, Ohmori R, Ashida K, Kihara T, Hara A, Arakawa K, Kameyama A, Noya K, Nagata M, Nakamura H, Ohsuzu F. In vivo magnetic resonance evaluation of associations between aortic atherosclerosis and both risk factors and coronary artery disease in patients referred for coronary angiography. Am Heart J. 2004;148:137–143. doi: 10.1016/j.ahj.2004.03.008. [DOI] [PubMed] [Google Scholar]
- 5.Li AE, Kamel I, Rando F, Anderson M, Kumbasar B, Lima JA, Bluemke DA. Using MRI to assess aortic wall thickness in the multiethnic study of atherosclerosis: distribution by race, sex, and age. AJR Am J Roentgenol. 2004;182:593–597. doi: 10.2214/ajr.182.3.1820593. [DOI] [PubMed] [Google Scholar]
- 6.Mitchell GF, Guo CY, Benjamin EJ, Larson MG, Keyes MJ, Vita JA, Vasan RS, Levy D. Cross-sectional correlates of increased aortic stiffness in the community: the Framingham Heart Study. Circulation. 2007;115:2628–2636. doi: 10.1161/CIRCULATIONAHA.106.667733. [DOI] [PubMed] [Google Scholar]
- 7.Rerkpattanapipat P, Hundley WG, Link KM, Brubaker PH, Hamilton CA, Darty SN, Morgan TM, Kitzman DW. Relation of aortic distensibility determined by magnetic resonance imaging in patients > or =60 years of age to systolic heart failure and exercise capacity. Am J Cardiol. 2002;90:1221–1225. doi: 10.1016/s0002-9149(02)02838-2. [DOI] [PubMed] [Google Scholar]
- 8.Gaballa MA, Jacob CT, Raya TE, Liu J, Simon B, Goldman S. Large artery remodeling during aging: biaxial passive and active stiffness. Hypertension. 1998;32:437–443. doi: 10.1161/01.hyp.32.3.437. [DOI] [PubMed] [Google Scholar]
- 9.Marque V, Kieffer P, Atkinson J, Lartaud-Idjouadiene I. Elastic properties and composition of the aortic wall in old spontaneously hypertensive rats. Hypertension. 1999;34:415–422. doi: 10.1161/01.hyp.34.3.415. [DOI] [PubMed] [Google Scholar]
- 10.Sutton-Tyrrell K, Najjar SS, Boudreau RM, Venkitachalam L, Kupelian V, Simonsick EM, Havlik R, Lakatta EG, Spurgeon H, Kritchevsky S, Pahor M, Bauer D, Newman A. Elevated aortic pulse wave velocity, a marker of arterial stiffness, predicts cardiovascular events in well-functioning older adults. Circulation. 2005;111:3384–3390. doi: 10.1161/CIRCULATIONAHA.104.483628. [DOI] [PubMed] [Google Scholar]
- 11.Mattace-Raso FU, van der Cammen TJ, Hofman A, van Popele NM, Bos ML, Schalekamp MA, Asmar R, Reneman RS, Hoeks AP, Breteler MM, Witteman JC. Arterial stiffness and risk of coronary heart disease and stroke: the Rotterdam Study. Circulation. 2006;113:657–663. doi: 10.1161/CIRCULATIONAHA.105.555235. [DOI] [PubMed] [Google Scholar]
- 12.Fazio GP, Redberg RF, Winslow T, Schiller NB. Transesophageal echocardiographically detected atherosclerotic aortic plaque is a marker for coronary artery disease. J Am Coll Cardiol. 1993;21:144–150. doi: 10.1016/0735-1097(93)90729-k. [DOI] [PubMed] [Google Scholar]
- 13.Matsumura Y, Takata J, Yabe T, Furuno T, Chikamori T, Doi YL. Atherosclerotic aortic plaque detected by transesophageal echocardiography: its significance and limitation as a marker for coronary artery disease in the elderly. Chest. 1997;112:81–86. doi: 10.1378/chest.112.1.81. [DOI] [PubMed] [Google Scholar]
- 14.Agmon Y, Khandheria BK, Meissner I, Schwartz GL, Petterson TM, O'Fallon WM, Gentile F, Whisnant JP, Wiebers DO, Seward JB. Independent association of high blood pressure and aortic atherosclerosis: A population-based study. Circulation. 2000;102:2087–2093. doi: 10.1161/01.cir.102.17.2087. [DOI] [PubMed] [Google Scholar]
- 15.Resnick LM, Militianu D, Cunnings AJ, Pipe JG, Evelhoch JL, Soulen RL. Direct magnetic resonance determination of aortic distensibility in essential hypertension: relation to age, abdominal visceral fat, and in situ intracellular free magnesium. Hypertension. 1997;30:654–659. doi: 10.1161/01.hyp.30.3.654. [DOI] [PubMed] [Google Scholar]
- 16.Smulyan H, Safar ME. Systolic blood pressure revisited. J Am Coll Cardiol. 1997;29:1407–1413. doi: 10.1016/s0735-1097(97)00081-8. [DOI] [PubMed] [Google Scholar]
- 17.Ferreira AV, Viana MC, Mill JG, Asmar RG, Cunha RS. Racial differences in aortic stiffness in normotensive and hypertensive adults. J Hypertens. 1999;17:631–637. doi: 10.1097/00004872-199917050-00006. [DOI] [PubMed] [Google Scholar]
- 18.Zion AS, Bond V, Adams RG, Williams D, Fullilove RE, Sloan RP, Bartels MN, Downey JA, De Meersman RE. Low arterial compliance in young African-American males. Am J Physiol Heart Circ Physiol. 2003;285:H457–H462. doi: 10.1152/ajpheart.00497.2002. [DOI] [PubMed] [Google Scholar]
- 19.Lemogoum D, Van Bortel L, Van den Abeele W, Ciarka A, Degaute JP, van de Borne P, Leeman M. Effect of beta-adrenergic stimulation on pulse wave velocity in black and white subjects. J Hypertens. 2004;22:2349–2353. doi: 10.1097/00004872-200412000-00017. [DOI] [PubMed] [Google Scholar]
- 20.Lemogoum D, Van Bortel L, Leeman M, Degaute JP, van de Borne P. Ethnic differences in arterial stiffness and wave reflections after cigarette smoking. J Hypertens. 2006;24:683–689. doi: 10.1097/01.hjh.0000217850.87960.16. [DOI] [PubMed] [Google Scholar]
- 21.Benowitz NL, Perez-Stable EJ, Fong I, Modin G, Herrera B, Jacob P., 3rd Ethnic differences in N-glucuronidation of nicotine and cotinine. J Pharmacol Exp Ther. 1999;291:1196–1203. [PubMed] [Google Scholar]
- 22.McGill HC, Jr, McMahan CA, Malcom GT, Oalmann MC, Strong JP. Effects of serum lipoproteins and smoking on atherosclerosis in young men and women. The PDAY Research Group. Pathobiological Determinants of Atherosclerosis in Youth. Arterioscler Thromb Vasc Biol. 1997;17:95–106. doi: 10.1161/01.atv.17.1.95. [DOI] [PubMed] [Google Scholar]
- 23.Kupari M, Hekali P, Keto P, Poutanen VP, Tikkanen MJ, Standerstkjold-Nordenstam CG. Relation of aortic stiffness to factors modifying the risk of atherosclerosis in healthy people. Arterioscler Thromb. 1994;14:386–394. doi: 10.1161/01.atv.14.3.386. [DOI] [PubMed] [Google Scholar]
- 24.Adams GJ, Greene J, Vick GW, 3rd, Harrist R, Kimball KT, Karmonik C, Ballantyne CM, Insull W, Jr, Morrisett JD. Tracking regression and progression of atherosclerosis in human carotid arteries using high-resolution magnetic resonance imaging. Magn Reson Imaging. 2004;22:1249–1258. doi: 10.1016/j.mri.2004.08.020. [DOI] [PubMed] [Google Scholar]
- 25.Newman DL, Gosling RG, Bowden NL. Changes in aortic distensibility and area ratio with the development of atherosclerosis. Atherosclerosis. 1971;14:231–240. doi: 10.1016/0021-9150(71)90052-9. [DOI] [PubMed] [Google Scholar]
- 26.Farrar DJ, Bond MG, Riley WA, Sawyer JK. Anatomic correlates of aortic pulse wave velocity and carotid artery elasticity during atherosclerosis progression and regression in monkeys. Circulation. 1991;83:1754–1763. doi: 10.1161/01.cir.83.5.1754. [DOI] [PubMed] [Google Scholar]
- 27.van Popele NM, Westendorp IC, Bots ML, Reneman RS, Hoeks AP, Hofman A, Grobbee DE, Witteman JC. Variables of the insulin resistance syndrome are associated with reduced arterial distensibility in healthy non-diabetic middle-aged women. Diabetologia. 2000;43:665–672. doi: 10.1007/s001250051356. [DOI] [PubMed] [Google Scholar]
- 28.Amar J, Ruidavets JB, Chamontin B, Drouet L, Ferrieres J. Arterial stiffness and cardiovascular risk factors in a population-based study. J Hypertens. 2001;19:381–387. doi: 10.1097/00004872-200103000-00005. [DOI] [PubMed] [Google Scholar]
- 29.Dart AM, Gatzka CD, Cameron JD, Kingwell BA, Liang YL, Berry KL, Reid CM, Jennings GL. Large artery stiffness is not related to plasma cholesterol in older subjects with hypertension. Arterioscler Thromb Vasc Biol. 2004;24:962–968. doi: 10.1161/01.ATV.0000126371.14332.ab. [DOI] [PubMed] [Google Scholar]