Semiconductor nanocrystals (quantum dots, QDs) are usually described as fluorophores having remarkable photostability, large absorption cross sections, and tunable emission peaks. Equally important, QDs also serve as versatile nanoscale objects of precisely tunable size and morphology, having exceptionally narrow size distributions.1, 2 Size is an especially important parameter in the design of nanomaterials for applications in biology, for both in vitro and in vivo applications. In cell labeling applications, for example, size can affect endocytosis or limit access to receptors of interest, such as those in the neuronal synapse.3 In live animals, particle size can dramatically affect biodistribution and pharmacokinetics.4, 5 The optimal size depends on the application. For example, we have previously shown that Type II CdTe(CdSe) core(shell) QDs with a hydrodynamic diameter (HD) of ∼19 nm can be used to selectively map sentinel lymph nodes.6 More recently, we have also shown that 8.7 nm HD Type I InAs(ZnSe) QDs allowed the mapping of multiple lymph nodes and also showed the potential extravasation of QDs from the vasculature.7 There has been a strong interest in reducing the HD of QDs for in vivo applications as this could increase bioavailability and lead to an improved understanding of clearance mechanisms. In this communication, we demonstrate bio-compatible fluorescent QDs with an exceptionally small HD of ∼6 nm using a CdSe(ZnCdS) core(shell) structure coated with DL-cysteine and show renal clearance of these QDs in rat models.
Cysteine-coated nanocrystals have been demonstrated previously with CdS QDs synthesized directly from aqueous solution in the presence of L-cysteine hydrochloride.8 However, these QDs suffered from low QY (6−8%) and broad fluorescence (FWHM >100 nm) due to emission from surface trap sites. We report the synthesis of high-quality cysteine-coated CdSe(ZnCdS) core(shell) QDs using well-developed nanocrystal synthetic procedures, in which cores are formed through the rapid injection of metal and chalcogenide precursors into hot solvent and then overcoated with a thin shell of a higher band gap material. Following overcoating, the native ligands are exchanged with DL-cysteine to yield water dispersible QDs (QD-Cys).
Ligand exchange with cysteine was achieved using a biphasic exchange method in which QDs dispersed in chloroform were mixed with a solution of DL-cysteine in phosphate buffered saline (PBS). This biphasic mixture was stirred vigorously at room temperature, and phase transfer of QDs from the organic to the aqueous phase occurred over ∼2 h, leaving a colorless chloroform layer (Figure 1a). The QDs were precipitated twice with ethanol and re-dispersed in PBS at pH 7.4 for analysis. QD-Cys as synthesized formed macroscopic aggregates upon standing at room temperature overnight. Storing the samples in the dark at 4 °C only extended their stability to ∼24 h. QD-Cys precipitation was attributed to dissociation of the ligand from the QD followed by spontaneous oxidation to form cysteine dimers (cystine), which do not bind to the QD surface, resulting in QD aggregation. By introducing a reducing agent, such as sodium borohydride or dithiothreitol (DTT), to prevent cystine formation, the stability of QD-Cys increased to ∼1 week under ambient conditions (Figure S1) and to at least three months when stored in the dark at 4 °C.
Figure 1.
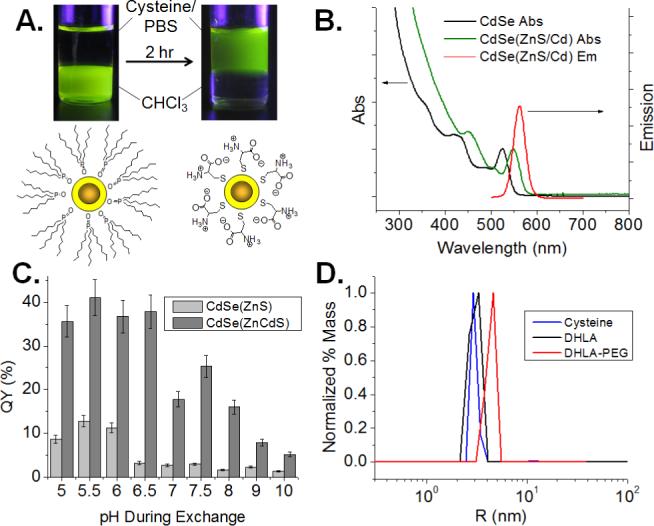
(A) Schematic and image of QDs before and after ligand exchange with DL-cysteine. (B) QD absorption and emission spectra before and after overcoating with ZnCdS alloy shell. (C) QY as a function of pH for ZnS and ZnCdS alloy shell structures. (D) DLS data for QD565 coated with the indicated ligands, showing HDs in PBS of 5.95 nm (cysteine), 6.02 nm (DHLA), and 8.60 nm (DHLA-PEG).
Following phase transfer to water, the QDs exhibited decreased quantum yield (QY), consistent with previous reports of ligand exchange with monothiol ligands.9 The final QY was highly sensitive to both the quality of the inorganic shell and the pH of the aqueous solution during ligand exchange. The most robust inorganic shells were obtained by the formation of a ZnxCd(1-x)S alloy, as we previously reported.10 The large red-shift of the absorbance maximum upon overcoating results from the presence of an alloy shell (Figure 1b). To illustrate the dramatic difference a robust shell makes on the QY, ligand exchange with cysteine was performed with CdSe QDs bearing 3 monolayers of either a pure ZnS or an alloy ZnCdS shell (30% Cd by mass). While the QY of the CdSe(ZnS) QDs dropped from ∼68% in growth solution to ∼13% in water, the QY of the CdSe(ZnCdS) QDs dropped from ∼66% in growth solution to ∼40% in water after ligand exchange under identical conditions. To test the effect of pH during ligand exchange on QY, exchange with cysteine was performed in aqueous buffer ranging from pH 5 to pH 10. The resulting QD-Cys were precipitated with ethanol and re-dispersed in PBS at pH 7.4 for analysis. The maximum QY was achieved at pH 5.5, with QY dropping drastically above pH 6.0 (Figure 1c), which was correlated with a blue-shift in the absorbance spectrum, indicative of shell degradation. This degradation may be due to the formation of the reactive thiolate ion on cysteine at more basic pH, which can etch the shell during exchange. The more robust CdSe(ZnCdS) QDs showed the same pH dependent blue-shift in the absorbance spectrum as CdSe(ZnS) QDs, but exhibited >2-fold higher QY across the pH range tested, suggesting that the alloyed shell is not necessarily more resistant to etching, but is better at maintaining the passivation of surface trap sites.
The HD of QD-Cys was 5.9 nm by dynamic light scattering (DLS) (Figure 1d). Transmission electron microscopy (TEM) showed an inorganic particle size of 3.6 nm (Figure S2), indicating that the cysteine coating contributes ∼1.2 nm to the overall radius, which is reasonable given the small extended size of cysteine (∼0.6 nm) plus a hydration shell. By comparison, the same QD cores exchanged with dihydrolipoic acid (DHLA) and DHLA conjugated to a short polyethylene glycol (n=8; DHLA-PEG) showed HDs of 6.0 and 8.6 nm, respectively (Figure 1d). Incubation of DHLA-coated QDs in fetal bovine serum (FBS) for 4 h at 37 °C resulted in a large increase in HD to >15 nm (data not shown) due to non-specific protein binding, consistent with previous reports of proteins binding to QDs bearing charged ligands.7, 11 Incubation of QD-Cys in FBS under the same conditions did not alter the size of the QD, indicating that QD-Cys does not aggregate or bind proteins in serum (Figure 2b), which is attributed to the zwitterionic nature of the ligand at pH 7.4, rendering the QD surface effectively charge neutral. Indeed, changing the pH of the FBS to favor positively or negatively charged forms of cysteine at low and high pH, respectively, induced protein binding as seen by a large increase in size after serum incubation (Figure 2b). QDs ligand exchanged with L-cysteine exhibited similar solution properties and serum binding behavior as those coated with DL-cysteine (Figure S3).
Figure 2.
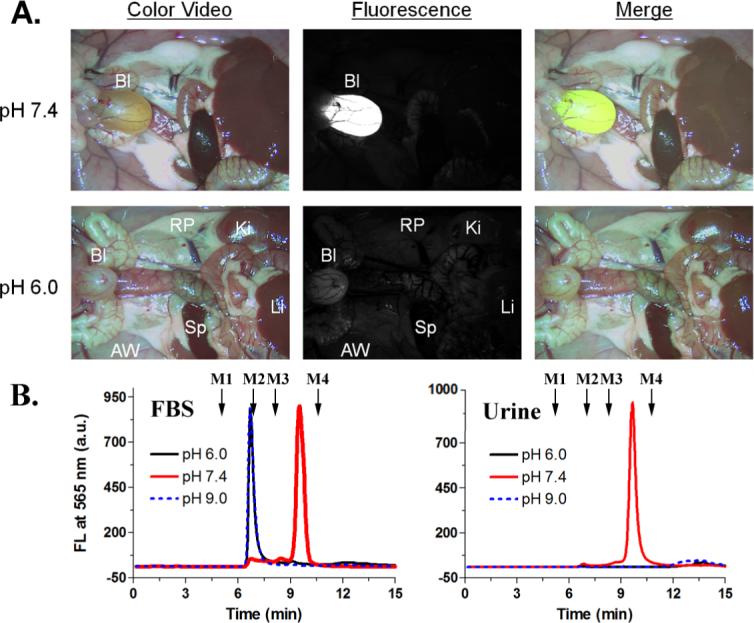
(A) Fluorescence images of rats injected with QD-Cys incubated in FBS at the indicated pH, 4 h postinjection (Bl, bladder; Ki, kidneys; Li, liver; RP, retroperitoneum; AW, abdominal wall, and Sp, spleen.): color video (left), 565 nm fluorescence (middle), merged (right). (B) GFC analysis of QD-Cys incubated in FBS at various pH (left), and in urine (right) 4 hours postinjection (fluorescence detection at 565 nm). Mw markers M1 (thyroglobulin, 670 kDa), M2 (γ-globulin, 158 kDa), M3 (ovalbumin, 44 kDa), and M4 (myoglobin, 17 kDa) are shown by arrows.
The exceptionally small size of QD-Cys and their stability in serum enabled us to observe interesting, new in vivo behavior. When injected into rats intravenously, QD-Cys accumulated mainly in the bladder 4 h postinjection, indicating that the QDs were near or below the size threshold for renal clearance (Figure 2a). Gel filtration chromatography (GFC) analysis of the urine confirmed the presence of QDs in the bladder (Figure 2b). Injecting protein-bound QD-Cys (incubated with FBS at pH 6.0 or 9.0 for 4 h) showed no renal clearance 4 h postinjection, suggesting both that protein-QD complexes are stable over this time scale and that renal clearance is largely dependent on the size of the QD. By systematically altering the core(shell) size of QD-Cys, it is possible to precisely determine the size threshold for renal filtration, at which 50% of QD-Cys are cleared from the body within 4 h.12
To demonstrate the ability to potentially form active constructs for in-vivo targeting and sensing we show that it is possible to covalently conjugate to QD-Cys through the C-terminus of the cysteine residue. We conjugated 515 nm emitting QD-Cys to Alexa fluor 555 through carbodiimide coupling chemistry and purified the construct with three cycles of spin filtration through a 10 kDa Mw cutoff filter. By altering the initial ratio of dye to QD, the average number of dye molecules ultimately attached to each QD was tuned from <1 to 9, as determined by UV-Vis absorption (Table S1). Excitation of the QD at a wavelength with minimal dye absorption resulted largely in dye fluorescence and quenched QD emission, suggesting efficient fluorescence resonance energy transfer (FRET) between QD and dye (Figure S4).
In summary, we have developed a versatile nanoparticle construct using a compact cysteine coating on a highly robust CdSe(ZnCdS) core(shell) structure to form aqueous QDs that are biologically compatible, extraordinarily compact, highly fluorescent, and easily functionalized. These properties, combined with the observation of renal clearance, open up the possibility of functionalized nanocrystals for in vivo targeted imaging, in which small molecules can be conjugated to QD-Cys for targeting, and unbound QDs can be rapidly cleared to achieve high signal/noise ratios and to reduce background toxicity.
Acknowledgments
This work was supported by NIH Grants R33-EB-000673 and R01-CA-115296, a CIMIT Application Development Award, the NSF-MRSEC program (DMR-9808941), the Biophysical Instrumentation Facility (NSF-0070319 and NIH GM68762), and the MIT-Harvard NIH Center for Cancer Nanotechnology Excellence (1U54-CA119349).
Footnotes
Supporting Information Available: Experimental procedures, transmission electron microscopy data, and additional optical characterization. This material is available free of charge via the Internet at http://pubs.acs.org.
Supplementary Material
References
- 1.Murray CB, Norris DJ, Bawendi MG. J. Am. Chem. Soc. 1993;115:8706–8715. [Google Scholar]
- 2.Wu X, Liu H, Liu J, Haley KN, Treadway JA, Larson JP, Ge N, Peale F, Bruchez MP. Nature Biotechnol. 2003;21:41–46. doi: 10.1038/nbt764. [DOI] [PubMed] [Google Scholar]
- 3.Howarth M, Takao K, Hayashi Y, Ting AY. PNAS. 2005;102:7583–7588. doi: 10.1073/pnas.0503125102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Caliceti P, V. F. M. Adv. Drug Delivery Rev. 55:1261–1277. doi: 10.1016/s0169-409x(03)00108-x. [DOI] [PubMed] [Google Scholar]
- 5.DeNardo SJ, Yao Z, Lam KS, Song A, Burke PA, Mirick GR, Lamborn KR, O'Donnell RT, DeNardo GL. Clin. Cancer Res. 2003;9:3854S–3864. [PubMed] [Google Scholar]
- 6.Kim S, Lim YT, Soltesz EG, De Grand AM, Lee J, Nakayama A, Parker JA, Mihaljevic T, Laurence RG, Dor DM, Cohn LH, Bawendi MG, Frangioni JV. Nat. Biotech. 2004;22:93–97. doi: 10.1038/nbt920. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Zimmer JP, Kim S-W, Ohnishi S, Tanaka E, Frangioni JV, Bawendi MG. J. Am. Chem. Soc. 2006;128:2526–2527. doi: 10.1021/ja0579816. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Chen JL, Zhu CQ. Analytica Chimica Acta. 2005;546:147–153. [Google Scholar]
- 9.Smith AM, Duan H, Rhyner MN, Ruan G, Nie S. Phys. Chem. Chem. Phys. 2006;8:3895–3903. doi: 10.1039/b606572b. [DOI] [PubMed] [Google Scholar]
- 10.Snee PT, Chan Y, Nocera DG, Bawendi MG. Adv. Mater. 2005;17:1131–1136. [Google Scholar]
- 11.Bentzen EL, Tomlinson ID, Mason J, Gresch P, Warnement MR, Wright D, Sanders-Bush E, Blakely R, Rosenthal SJ. Bioconjug. Chem. 2005;16:1488–1494. doi: 10.1021/bc0502006. [DOI] [PubMed] [Google Scholar]
- 12.Choi HS, Liu W, Misra P, Tanaka E, Zimmer JP, Kandapallil B, Bawendi MG, Frangioni JV. Nat. Biotech. 2007 doi: 10.1038/nbt1340. In press. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.


