Abstract
Recent basic science studies on discogenic low back pain have provided new knowledge about this condition. This paper reviews some of these results and presents an overview of the following findings. The rat lumbar intervertebral disk may be innervated non-segmentally through the paravertebral sympathetic nerve and segmentally through the sinuvertebral nerves, and also by dichotomizing sensory fibers. The exposure of the nucleus pulposus (NP) to the outer annulus fibrosus (AF) may induce nerve injury and ingrowth into the disk. Nerve growth factor (NGF)-sensitive neurons are predominant in the rat intervertebral disk, which indicates that hyperalgesic responses can be induced by inflammation. NGF in the NP may promote axonal growth. Lumbar fusion may inhibit nerve ingrowth into the degenerated disk and reduce the percentage of calcitonin gene related peptide (CGRP)-positive neurons.
Keywords: Low back pain, Lumbar spine, Intervertebral disk, Pathophysiology
Low back pain is a common clinical problem and a significant socio-economic problem. According to Nachemson [12], in Europeans, a one-time occurrence of low back pain is found in 15–30% of the population; the 1-month prevalence is 19–43% of the population; and the lifetime prevalence is up to 60–80% of the population. Low back pain can be divided into two categories: specific or nonspecific. Specific low back pain can be induced by apparent causes, such as spinal tumors or infection. However, 80% or 90% of low back pain is classified as nonspecific, without identifiable causes, and is often chronic and difficult to treat.
Intervertebral disk (IVD) pathology is thought to be one of the causative factors of low back pain. However, the pathophysiology of discogenic low back pain is not well established. Recent studies on discogenic low back pain have revealed new knowledge in this area. This paper reviews some of the results of basic research on discogenic low back pain done at the authors’ institution (Chiba University).
In 1970, Shinohara [15] reported the presence of nerve fibers in the deep layers of the annulus fibrosus of degenerated lumbar disks obtained during anterior decompression and interbody fusion surgery for patients with lumbar disk pain. Because the report was published in Japanese, these findings did not receive international attention. Twenty years later, several reports published in international journals demonstrated the presence of nerve fibers in the inner layers of the annulus fibrosus and nucleus pulposus in patients with low back pain [3, 4, 6]. These studies revealed that innervation of the inner disk was observed only in painful disks, not in normal control disks. Based on these observations, nerve ingrowth into the inner disk may be a cause of nonspecific discogenic low back pain.
It had been widely believed that the lumbar IVD is segmentally innervated by dorsal root ganglion (DRG) neurons through the sinuvertebral nerves. However, Takahashi et al. [16] demonstrated that the L5–L6 disk was innervated by upper (L2) DRG neurons in rats. They applied capsaicin, a C-fiber stimulator, into L5–L6 IVDs after Evans blue stain was intravenously injected into rats. Lumbar disk stimulation by capsaicin caused extravasation of the dye in the groin skin, which is innervated by L2, but not L5. This was the first paper to report a relationship between lumbar disks and the L2 nerve in rats.
Following the study of Takahashi et al., Morinaga et al. [11], using a retrograde tracing method, investigated the distribution of DRG neurons innervating the anterior portion of the rat lumbar IVD. Horseradish peroxidase crystals were placed on the anterior portion of the rat L5–L6 disk and the labeling of L1 and L2 DRG neurons was observed. This study showed the anterior portion of the L5–L6 disk may be innervated from L1 or L2 spinal nerves in rats.
Motivated by these findings, Nakamura et al. [13] sought to uncover the distribution of nerves in the posterior portion of lumbar IVDs in rats using serial sympathectomy. After removal of the dura, the anterior portion of the spinal canal was observed from the posterior side before and after the serial sympathectomy. Prior to the procedure, numerous nerves were observed to be present on the posterior portion of the disk. Following sympathectomy, the amount of nerve staining decreased, indicating a relationship between the posterior disk nerves and the sympathetic trunks.
In humans, disk lesions usually occur in the posterior portion of the IVD. To investigate innervation of the posterior portion of the disk, Ohtori et al. [14] inserted a 21 gauge needle into a rat disk from the anterior side, stopping prior to penetration of the posterior annulus, and the neurotracer Fluoro-Gold (FG), was injected. Their results showed that sensory fibers from T13, L1, and L2 DRGs innervate the dorsal portion of the L5–L6 disk through the paravertebral sympathetic trunks, whereas those from L3–L5 DRGs may innervate the disk through the sinuvertebral nerves.
To further investigate the innervation pattern of the lumbar spine, Kurokawa et al. [10] employed double fluorescent-labeling using two types of neurotracers in rat lumbar disks. The rats were divided into four groups, and the neurotracer, DiI was applied equally to the L5–L6 disks in all groups. A second neurotracer, FG, was then selectively applied to the L2–L3, L3–L4, L4–L5, or L6–S IVDs. For example, in group 1, DiI was applied to the L5–L6 disk, and FG was applied to the L2–L3 disk; while in group 2, DiI was applied to L5–L6 and FG was applied to L3–L4. The number of double-labeled neurons stained with both FG and DiI was then counted. The results indicate that 7–12% of the DRG neurons innervating the L5–L6 disk also innervate other lumbar IVDs in rats. These findings suggest the existence of dichotomizing sensory fibers innervating multiple levels of lumbar disks. This may also explain why a patient often cannot specify the location of his or her painful IVD.
The majority of patients with lumbar disk herniation can be successfully treated conservatively without residual low back pain. However, some patients may experience chronic low back pain. We speculate that neural ingrowth into the disk is an important factors in discogenic low back pain. To investigate the mechanism of nerve ingrowth into the IVD, Inoue et al. [8] exposed the nucleus pulposus to the outer surface of the annulus fibrosus in the rat. FG was first applied to the disk to identify the DRG neurons innervating the disk. Ten days later a marker for nerve injury (ATF3) and a marker for axonal growth (GAP43) were examined on the labeled DRGs. The DRG cells that were positive for FG, ATF3, and GAP43 were thought to have nerve injury and axonal growth. Exposure of the nucleus pulposus induced ATF3-positive cells in 17% and GAP43-positive cells in 30.8% of the FG-positive cells. In contrast, ATF3-positive DRG neurons were only 1.8% and GAP43-positive were 6.3% of the FG-positive cells of the control group where only the outer annulus was scratched. Exposure of the nucleus pulposus to the outer annulus fibrosus appears to induce nerve injury and ingrowth into the disk. Aoki et al. [2] reported that histological examination in a rabbit disk degeneration model revealed the extension of nerve fibers into scar tissue formed following prolapse of the nucleus pulposus. This nerve growth may have been initiated by disk herniation, and may be a clue to the etiology of chronic low back pain following lumbar disk herniation.
DRG neurons are divided into small and large neurons. Large neurons are thought to be involved in proprioception, while most small neurons are involved in nociception. Small DRG neurons are further subclassified into nerve growth factor (NGF)-sensitive neurons and glial cell line-derived neurotrophic factor (GDNF)-sensitive neurons. NGF-sensitive neurons contain neuropeptides, such as substance P (SP) and calcitonin gene related peptide (CGRP), whereas GDNF-sensitive neurons lack neuropeptides. Previous studies indicate that NGF-sensitive neurons are critical to hyperalgesic responses induced by inflammation, while GDNF-sensitive neurons are important in neuropathic pain. Aoki et al. [1] reported that NGF-sensitive neurons predominate in the rat disk, and very few GDNF-sensitive neurons (0.7%) innervate the lumbar IVD in rats when compared with skin or bladder. NGF is synthesized in inflamed tissue and is related to sensitization of NGF-sensitive neurons [17]. As reviewed by Edgar [5], peripheral or central sensitization of the nervous system is thought to be involved in the generation of discogenic pain. Freemont et al. [7] reported that the levels of NGF are higher in painful disks than in asymptomatic disks. Based on these observations, NGF may play a role in sensitizing the painful disk (Fig. 1).
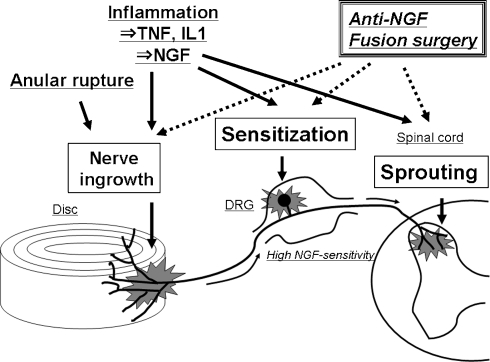
Fig. 1.
Schematic representation of a hypothetical mechanism for discogenic low back pain. An anular rupture causes exposure of the nucleus pulposus to the outer annulus fibrosus, which may induce nerve injury and nerve ingrowth into the disc. Under inflammatory conditions, nerve growth factor (NGF) is induced in the disc and acts on the dorsal root ganglion (DRG) neurons through the peripheral terminals in the disc. Disc-innervating DRG neurons have a high sensitivity to NGF. Therefore, NGF may promote nerve ingrowth into the disc, sensitize DRG neurons, and cause neuronal sprouting into the dorsal horn. Our recent studies demonstrated the possibility that anti-NGF treatment or fusion surgery can resolve these pathological conditions related to the generation of discogenic pain
Yamauchi et al. [18] co-cultured DRG cells from newborn Wistar rats with conditioned medium from human nucleus pulposus or annulus fibrosus cells with or without anti-NGFβ, and the average length of the axons from DRG neurons was measured. Their results show that axonal growth of DRG cells co-cultured with conditioned medium of nucleus pulposus cells from painful human disks was inhibited by anti-NGFβ. These results demonstrated that NGF in the nucleus pulposus promoted axonal growth.
Patients with low back pain can be treated clinically with spinal fusion surgery, although the optimal method for fusion has not been determined. Koshi et al. [9] studied the effect of posterolateral fusion (PLF) on DRG neurons. Posterolateral fusion was performed in rats using autografts from the iliac bone. Fusion was confirmed manually at necropsy. They then measured the percentage of FG-labeled neurons that were GAP43- and CGRP-positive. Rats with disk degeneration or disk degeneration with nonunion of the PLF had higher percentages of GAP43 and CGRP neurons, indicating the possibility of nociceptive nerve ingrowth in these disks. Although the group with disk degeneration and solid bony union had a slightly higher percentages of GAP43 and CGRP neurons than the normal disk with PLF or sham surgery groups, it was a much lower percentage than that shown in the disk degeneration group or disk degeneration with nonunion group. The results demonstrated that the percentages of GAP43- and CGRP-positive neurons were significantly lower in the PLF union group than in the disk degeneration group or PLF nonunion group.
These results indicate that spinal fusion surgery may lessen the neuropathological changes that take place in degenerating disks (Fig. 1). Anti-NGF therapy may have some value in reducing neuropathological changes and may become a candidate for the treatment of discogenic low back pain. While nonspecific low back pain remains uncharacterized, IVD-related research shows potential for elucidating some of its etiology.
Conflict of interest statement
None of the authors has any potential conflict of interest.
References
- 1.Aoki Y, Ohtori S, Takahashi K, Ino H, Takahashi Y, Chiba T, Moriya H. Innervation of the lumbar intervertebral disc by nerve growth factor-dependent neurons related to inflammatory pain. Spine. 2004;29:1077–1081. doi: 10.1097/00007632-200405150-00005. [DOI] [PubMed] [Google Scholar]
- 2.Aoki Y, Akeda K, An H, Muehleman C, Takahashi K, Moriya H, Masuda K. Nerve fiber ingrowth into scar tissue formed following nucleus pulposus extrusion in the rabbit anular-puncture disc degeneration model: effects of depth of puncture. Spine. 2006;31:E774–E780. doi: 10.1097/01.brs.0000238681.71537.41. [DOI] [PubMed] [Google Scholar]
- 3.Coppes MH, Marani E, Thomeer RT, Oudega M, Groen GJ. Innervation of annulus fibrosus in low back pain. Lancet. 1990;336:189–190. doi: 10.1016/0140-6736(90)91723-N. [DOI] [PubMed] [Google Scholar]
- 4.Coppes MH, Marani E, Raph TWM, Thomeer RT, Groen GJ. Innervation of “painful” lumbar discs. Spine. 1997;22(20):2342–2349. doi: 10.1097/00007632-199710150-00005. [DOI] [PubMed] [Google Scholar]
- 5.Edgar MA. Nerve supply of the lumbar intervertebral disc. J Bone Joint Surg (Br) 2007;89-B:1135–1139. doi: 10.1302/0301-620X.89B9.18939. [DOI] [PubMed] [Google Scholar]
- 6.Freemont AJ, Peacock TE, Goupille P, Hoyland JA, O’Brien J, Jayson MIV. Nerve ingrowth into diseased intervertebral disc in chronic back pain. Lancet. 1997;350:178–181. doi: 10.1016/S0140-6736(97)02135-1. [DOI] [PubMed] [Google Scholar]
- 7.Freemont AJ, Watkins A, Maitre CL, Baird P, Jeziorska M, Knight MTN, Ross ERS, O’Brien JP, Hoyland JA. Nerve growth factor expression and innervation of the painful intervertebral disc. J Pathol. 2002;197:286–292. doi: 10.1002/path.1108. [DOI] [PubMed] [Google Scholar]
- 8.Inoue G, Ohtori S, Aoki Y, Ozawa T, Doya H, Saito T, Ito T, Akazawa T, Moriya H, Takahashi K. Exposure of the nucleus pulposus to the outside of the annulus fibrosus induces nerve injury and regeneration of the afferent fibers innervating the lumbar intervertebral discs in rats. Spine. 2006;31:1433–1438. doi: 10.1097/01.brs.0000219946.25103.db. [DOI] [PubMed] [Google Scholar]
- 9.Koshi T, Ohtori S, Yamashita M, Yamauchi K, Suzuki M, Takahashi K. Lumbar posterior lateral fusion inhibits sensory nerve ingrowth into degenerated lumbar intervertebral discs and discogenic low back pain in rats. J Jpn Orthop Assoc. 2007;81:S1000. [Google Scholar]
- 10.Kurokawa M, Takahashi Y, Takahashi Ka, Chiba T, Yamagata M, Sameda H, Aoki Y, Ozawa T, Moriya H (2003) DRG neurons innervating three levels of lumbar intervertebral discs in rats. Abstracts of 30th annual meeting of International Society for the Study of the Lumbar Spine 2003, p. 3
- 11.Morinaga T, Takahashi K, Yamagata M, Chiba T, Tanaka K, Takahashi Y, Nakamura S, Suseki K, Moriya H. Sensory innervation to the anterior portion of lumbar intervertebral disc. Spine. 1996;21:1848–1851. doi: 10.1097/00007632-199608150-00002. [DOI] [PubMed] [Google Scholar]
- 12.Nachemson A. Epidemiology and the economics of low back pain. In: Herkowits HN, Dvorak J, Bell G, Nordin M, Grob D, editors. The lumbar spine. 3. Philadelphia: Lippincott; 2004. pp. 3–10. [Google Scholar]
- 13.Nakamura S, Takahashi K, Takahashi Y, Morinaga T, Shimada Y, Moriya H. Origin of nerves supplying the posterior portion of lumbar intervertebral discs in rats. Spine. 1996;21:917–924. doi: 10.1097/00007632-199604150-00003. [DOI] [PubMed] [Google Scholar]
- 14.Ohtori S, Takahashi Y, Takahashi K, Chiba T, Tanaka K, Hirayama J, Moriya H. Sensory innervation of the dorsal portion of the lumbar intervertebral disc in rats. Spine. 1999;24:2295–2299. doi: 10.1097/00007632-199911150-00002. [DOI] [PubMed] [Google Scholar]
- 15.Shinohara H. A study on lumbar disc lesion significance of histology of free nerve endings in lumbar disc. J Jpn Orthop Assoc. 1970;44:553–570. [PubMed] [Google Scholar]
- 16.Takahashi Y, Nakajima Y, Sakamoto T, Moriya H, Takahashi K. Capsaicin applied to rat lumbar intervertebral disc causes extravasation in the groin skin: a possible mechanism of referred pain of the intervertebral disc. Neurosci Lett. 1993;161:1–3. doi: 10.1016/0304-3940(93)90125-5. [DOI] [PubMed] [Google Scholar]
- 17.Woolf CJ, Allchorne A, Safieh-Garabedian B, Poole S. Cytokines, nerve growth factor and inflammatory hyperalgesia: the contribution of tumor necrosis factor alpha. Br J Pharmacol. 1997;121(3):417–424. doi: 10.1038/sj.bjp.0701148. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Yamauchi K, Ohtori S, Endo M, Nakagawa K, Takahashi K, Yamashita T. Axonal outgrowth of Substance P immunoreactive DRG fibers in cultured human degenerative intervertebral discs related with pain transmission pathways in vitro. J Jpn Orthop Assoc. 2007;81:S999. [Google Scholar]