Abstract
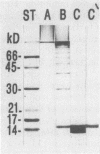
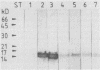
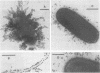
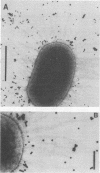
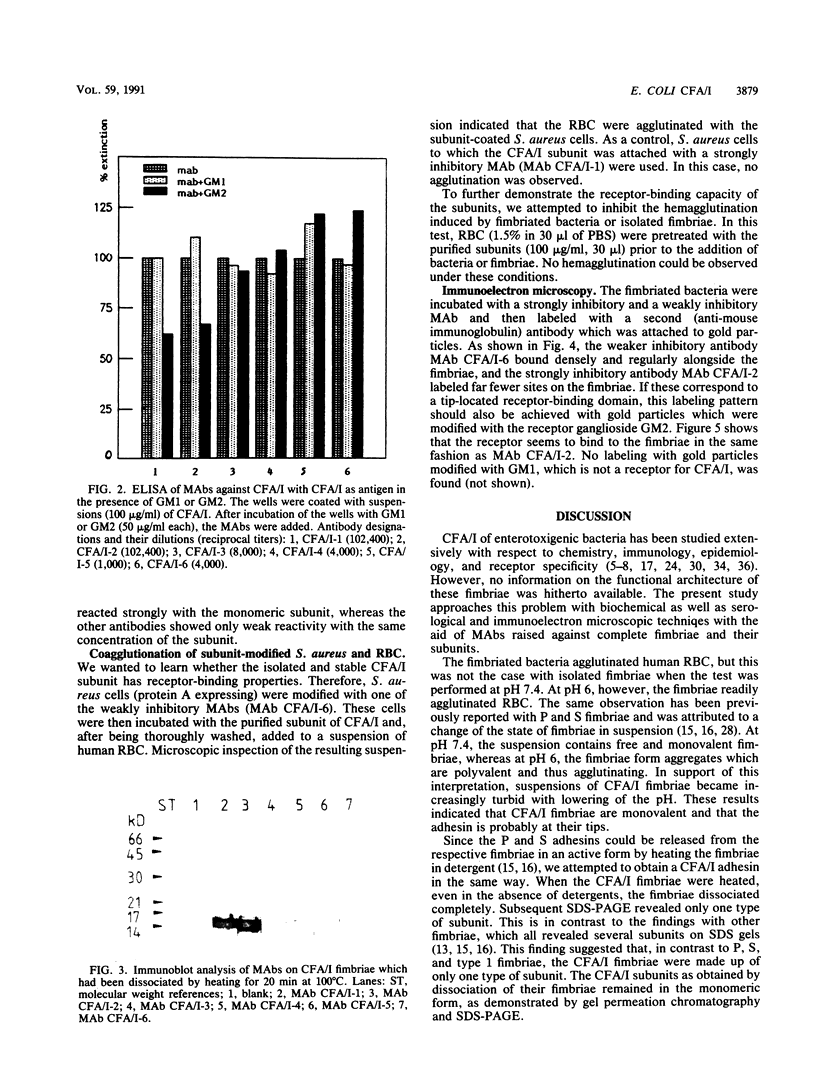
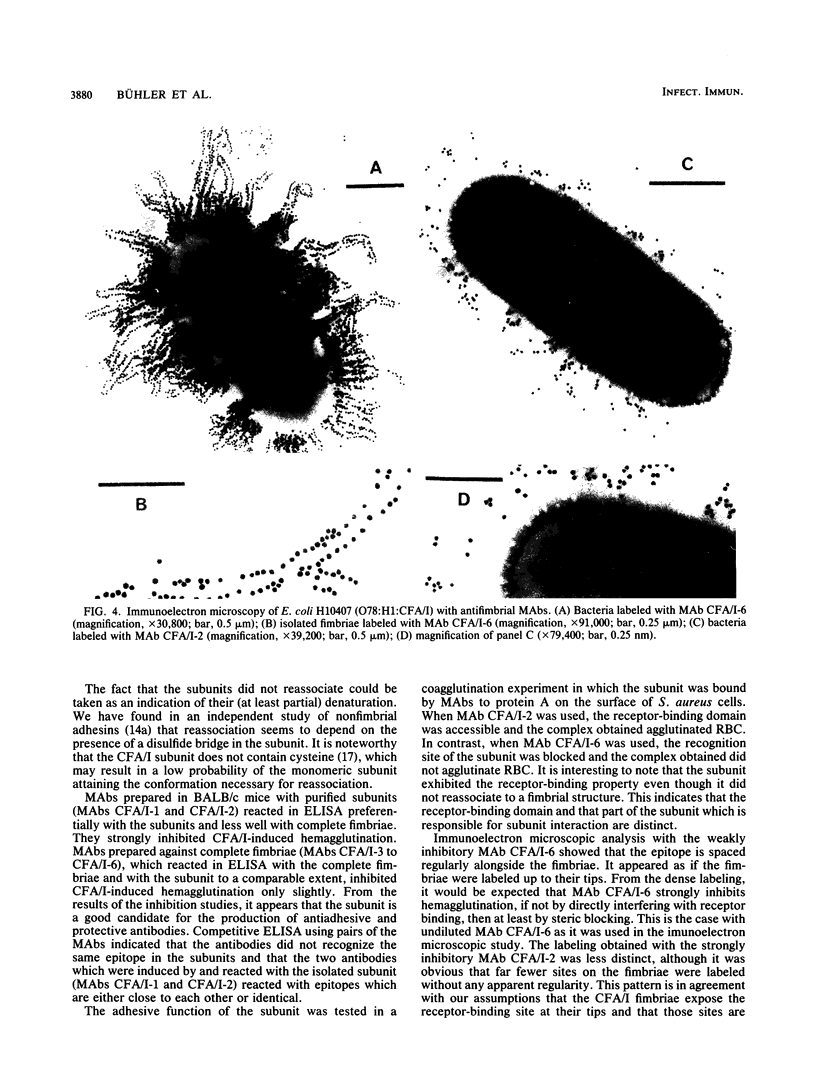
Colonization factor antigen I (CFA/I) of enterotoxigenic Escherichia coli was dissociated into one type of subunit (15 kDa). The dissociation was achieved either by heating CFA/I in sodium dodecyl sulfate at 100 degrees C or by heating it for 20 min in water. Heating in water to 100 degrees C yielded only in the 15-kDa subunit, but heating to 85 degree C yielded small amounts of oligomers in addition. The monomeric subunits obtained after heating in water are stable, as demonstrated by gel permeation chromatography and sodium dodecyl sulfate-polyacrylamide gel electrophoresis without heating prior to the electrophoretic run. These subunits inhibited CFA/I-induced hemagglutination, indicating that they had maintained their receptor-binding properties. When the hybridoma technique was used, two types of monoclonal anti-CFA/I antibodies were obtained. Antibodies obtained by immunization with the purified subunits were more reactive with subunits than with fimbriae, as shown by enzyme-linked immunosorbent assay. These antibodies strongly inhibited CFA/I-induced hemagglutination. When examined by immunoelectron microscopy, these antibodies seemed to label the fimbrial tips. A similar labeling pattern was obtained with gold particles modified with the receptor ganglioside GM2. Antibodies obtained by immunization with fimbriae reacted in enzyme-linked immunosorbent assays equally well with fimbriae and subunits. They inhibited CFA/I-induced hemagglutination only slightly. Immunoelectron microscopy revealed that these antibodies labeled the fimbriae densely and regularly over their entire lengths. In a coagglutination experiment with Staphylococcus aureus and monoclonal antibodies, the subunits retained their receptor-binding properties. From these results, we conclude that CFA/I fimbriae consist entirely of one type of adhesive subunit, of which only the one at the tip is accessible to the receptor.
Full text
PDF




Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Ansorge W. Fast and sensitive detection of protein and DNA bands by treatment with potassium permanganate. J Biochem Biophys Methods. 1985 May;11(1):13–20. doi: 10.1016/0165-022x(85)90037-5. [DOI] [PubMed] [Google Scholar]
- Duguid J. P., Clegg S., Wilson M. I. The fimbrial and non-fimbrial haemagglutinins of Escherichia coli. J Med Microbiol. 1979 May;12(2):213–227. doi: 10.1099/00222615-12-2-213. [DOI] [PubMed] [Google Scholar]
- Evans D. G., Evans D. J., Jr, Clegg S., Pauley J. A. Purification and characterization of the CFA/I antigen of enterotoxigenic Escherichia coli. Infect Immun. 1979 Aug;25(2):738–748. doi: 10.1128/iai.25.2.738-748.1979. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Evans D. G., Evans D. J., Jr, Tjoa W. Hemagglutination of human group A erythrocytes by enterotoxigenic Escherichia coli isolated from adults with diarrhea: correlation with colonization factor. Infect Immun. 1977 Nov;18(2):330–337. doi: 10.1128/iai.18.2.330-337.1977. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Evans D. G., Silver R. P., Evans D. J., Jr, Chase D. G., Gorbach S. L. Plasmid-controlled colonization factor associated with virulence in Esherichia coli enterotoxigenic for humans. Infect Immun. 1975 Sep;12(3):656–667. doi: 10.1128/iai.12.3.656-667.1975. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Evans D. J., Jr, Evans D. G. Colonization factor antigens of human pathogens. Curr Top Microbiol Immunol. 1990;151:129–145. doi: 10.1007/978-3-642-74703-8_7. [DOI] [PubMed] [Google Scholar]
- Friguet B., Djavadi-Ohaniance L., Pages J., Bussard A., Goldberg M. A convenient enzyme-linked immunosorbent assay for testing whether monoclonal antibodies recognize the same antigenic site. Application to hybridomas specific for the beta 2-subunit of Escherichia coli tryptophan synthase. J Immunol Methods. 1983 Jun 10;60(3):351–358. doi: 10.1016/0022-1759(83)90292-2. [DOI] [PubMed] [Google Scholar]
- Goldhar J., Perry R., Golecki J. R., Hoschutzky H., Jann B., Jann K. Nonfimbrial, mannose-resistant adhesins from uropathogenic Escherichia coli O83:K1:H4 and O14:K?:H11. Infect Immun. 1987 Aug;55(8):1837–1842. doi: 10.1128/iai.55.8.1837-1842.1987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hacker J. Genetic determinants coding for fimbriae and adhesins of extraintestinal Escherichia coli. Curr Top Microbiol Immunol. 1990;151:1–27. doi: 10.1007/978-3-642-74703-8_1. [DOI] [PubMed] [Google Scholar]
- Hanson M. S., Brinton C. C., Jr Identification and characterization of E. coli type-1 pilus tip adhesion protein. Nature. 1988 Mar 17;332(6161):265–268. doi: 10.1038/332265a0. [DOI] [PubMed] [Google Scholar]
- Jann K., Hoschützky H. Nature and organization of adhesins. Curr Top Microbiol Immunol. 1990;151:55–70. doi: 10.1007/978-3-642-74703-8_3. [DOI] [PubMed] [Google Scholar]
- Klemm P. Primary structure of the CFA1 fimbrial protein from human enterotoxigenic Escherichia coli strains. Eur J Biochem. 1982 May 17;124(2):339–348. doi: 10.1111/j.1432-1033.1982.tb06597.x. [DOI] [PubMed] [Google Scholar]
- Kolb-Bachofen V. Carbohydrate receptor binding using colloidal gold. Methods Enzymol. 1989;179:111–121. doi: 10.1016/0076-6879(89)79119-9. [DOI] [PubMed] [Google Scholar]
- Kramer M. D., Schaible U. E., Wallich R., Moter S. E., Petzoldt D., Simon M. M. Characterization of Borrelia burgdorferi associated antigens by monoclonal antibodies. Immunobiology. 1990 Nov;181(4-5):357–366. doi: 10.1016/S0171-2985(11)80504-8. [DOI] [PubMed] [Google Scholar]
- Köhler G., Milstein C. Derivation of specific antibody-producing tissue culture and tumor lines by cell fusion. Eur J Immunol. 1976 Jul;6(7):511–519. doi: 10.1002/eji.1830060713. [DOI] [PubMed] [Google Scholar]
- Labigne-Roussel A. F., Lark D., Schoolnik G., Falkow S. Cloning and expression of an afimbrial adhesin (AFA-I) responsible for P blood group-independent, mannose-resistant hemagglutination from a pyelonephritic Escherichia coli strain. Infect Immun. 1984 Oct;46(1):251–259. doi: 10.1128/iai.46.1.251-259.1984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Laemmli U. K. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature. 1970 Aug 15;227(5259):680–685. doi: 10.1038/227680a0. [DOI] [PubMed] [Google Scholar]
- Lindberg F., Lund B., Johansson L., Normark S. Localization of the receptor-binding protein adhesin at the tip of the bacterial pilus. Nature. 1987 Jul 2;328(6125):84–87. doi: 10.1038/328084a0. [DOI] [PubMed] [Google Scholar]
- Lopez-Vidal Y., Klemm P., Svennerholm A. M. Monoclonal antibodies against different epitopes on colonization factor antigen I of enterotoxin-producing Escherichia coli. J Clin Microbiol. 1988 Oct;26(10):1967–1972. doi: 10.1128/jcm.26.10.1967-1972.1988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lund B., Lindberg F., Marklund B. I., Normark S. The PapG protein is the alpha-D-galactopyranosyl-(1----4)-beta-D-galactopyranose-binding adhesin of uropathogenic Escherichia coli. Proc Natl Acad Sci U S A. 1987 Aug;84(16):5898–5902. doi: 10.1073/pnas.84.16.5898. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Minion F. C., Abraham S. N., Beachey E. H., Goguen J. D. The genetic determinant of adhesive function in type 1 fimbriae of Escherichia coli is distinct from the gene encoding the fimbrial subunit. J Bacteriol. 1986 Mar;165(3):1033–1036. doi: 10.1128/jb.165.3.1033-1036.1986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Moch T., Hoschützky H., Hacker J., Kröncke K. D., Jann K. Isolation and characterization of the alpha-sialyl-beta-2,3-galactosyl-specific adhesin from fimbriated Escherichia coli. Proc Natl Acad Sci U S A. 1987 May;84(10):3462–3466. doi: 10.1073/pnas.84.10.3462. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ofek I., Sharon N. Adhesins as lectins: specificity and role in infection. Curr Top Microbiol Immunol. 1990;151:91–113. doi: 10.1007/978-3-642-74703-8_5. [DOI] [PubMed] [Google Scholar]
- Pieroni P., Worobec E. A., Paranchych W., Armstrong G. D. Identification of a human erythrocyte receptor for colonization factor antigen I pili expressed by H10407 enterotoxigenic Escherichia coli. Infect Immun. 1988 May;56(5):1334–1340. doi: 10.1128/iai.56.5.1334-1340.1988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Riegman N., Hoschützky H., van Die I., Hoekstra W., Jann K., Bergmans H. Immunocytochemical analysis of P-fimbrial structure: localization of minor subunits and the influence of the minor subunit FsoE on the biogenesis of the adhesin. Mol Microbiol. 1990 Jul;4(7):1193–1198. doi: 10.1111/j.1365-2958.1990.tb00694.x. [DOI] [PubMed] [Google Scholar]
- Smith P. K., Krohn R. I., Hermanson G. T., Mallia A. K., Gartner F. H., Provenzano M. D., Fujimoto E. K., Goeke N. M., Olson B. J., Klenk D. C. Measurement of protein using bicinchoninic acid. Anal Biochem. 1985 Oct;150(1):76–85. doi: 10.1016/0003-2697(85)90442-7. [DOI] [PubMed] [Google Scholar]
- Svennerholm A. M., Gothefors L., Sack D. A., Bardhan P. K., Holmgren J. Local and systemic antibody responses and immunological memory in humans after immunization with cholera B subunit by different routes. Bull World Health Organ. 1984;62(6):909–918. [PMC free article] [PubMed] [Google Scholar]
- Worobec E. A., Shastry P., Smart W., Bradley R., Singh B., Paranchych W. Monoclonal antibodies against colonization factor antigen I pili from enterotoxigenic Escherichia coli. Infect Immun. 1983 Sep;41(3):1296–1301. doi: 10.1128/iai.41.3.1296-1301.1983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- de Graaf F. K. Genetics of adhesive fimbriae of intestinal Escherichia coli. Curr Top Microbiol Immunol. 1990;151:29–53. doi: 10.1007/978-3-642-74703-8_2. [DOI] [PubMed] [Google Scholar]