Abstract
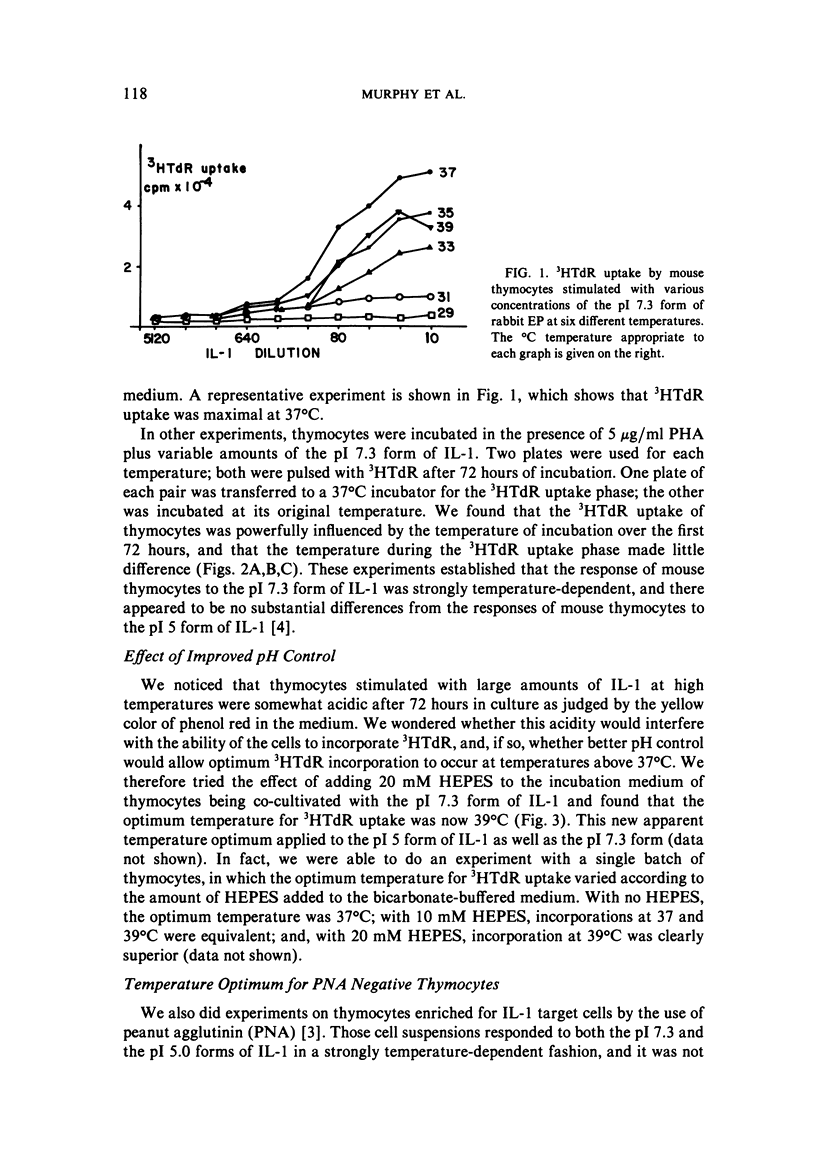
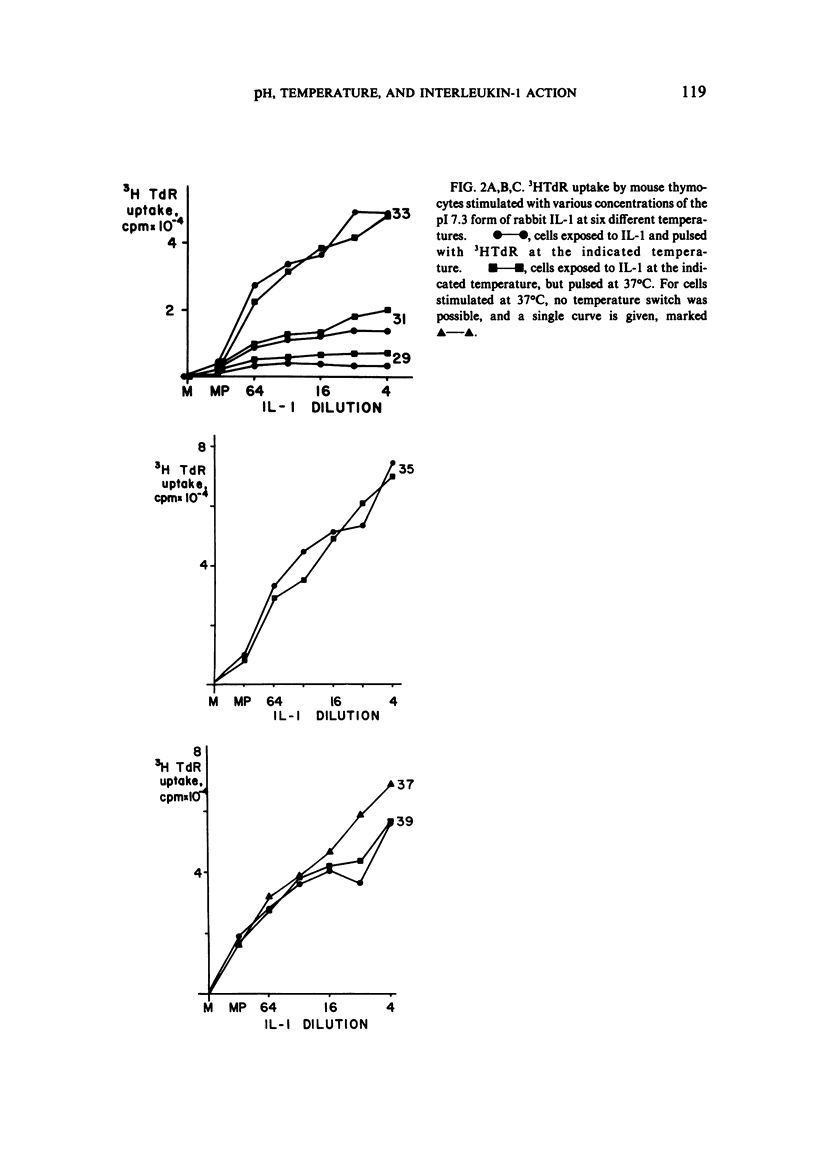
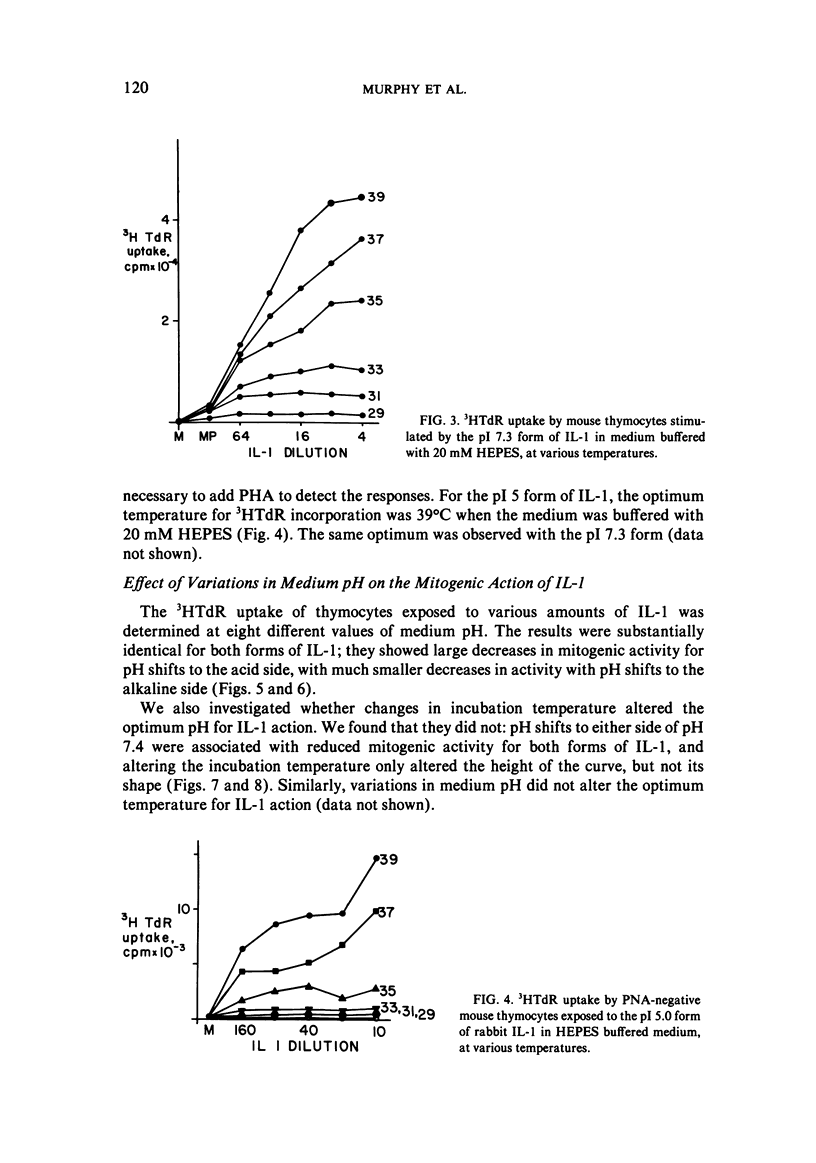
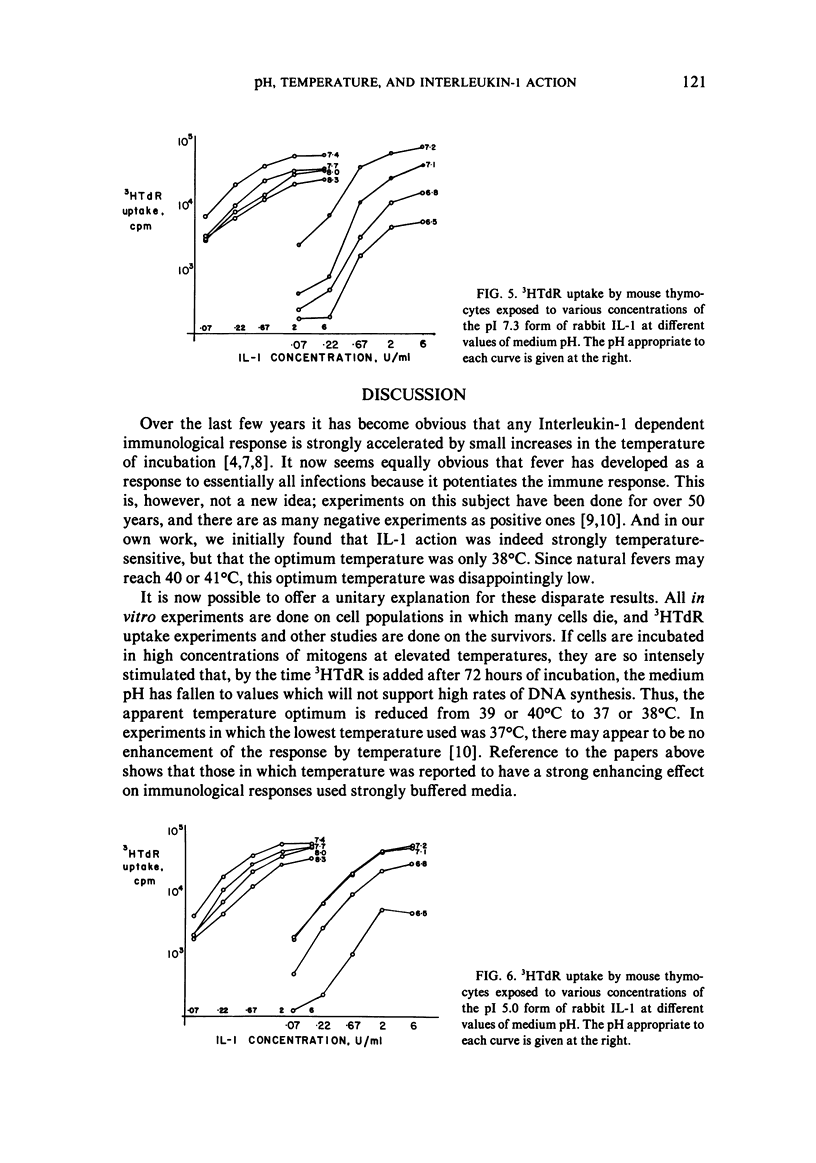
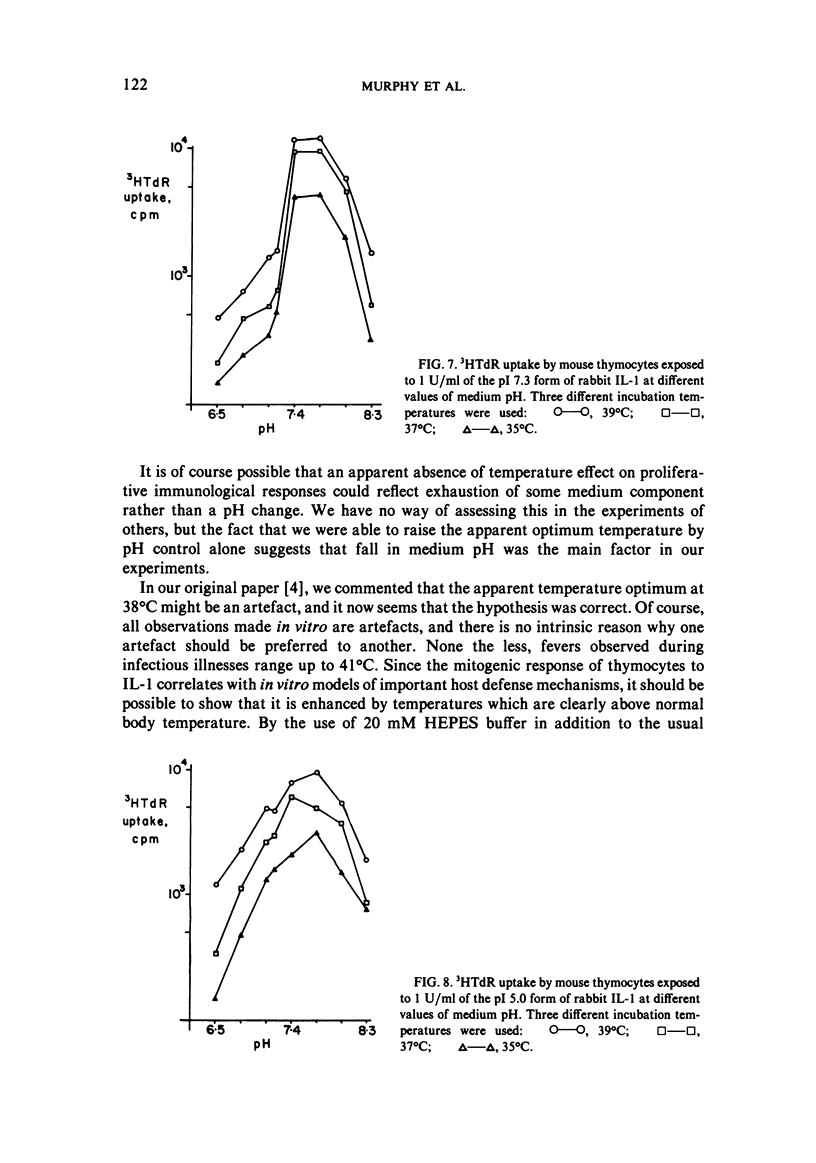
We studied the responses of unpurified mouse thymocytes and of peanut agglutinin-negative mouse thymocytes to the pI 7.3 form of rabbit Interleukin-1. We found that small increases of temperature strongly enhanced the mitogenic effect of this form of IL-1, and that the apparent temperature optimum was 37 degrees C. In both these respects the behavior of the pI 7.3 IL-1 resembled the previously described behavior of pI 5.0 IL-1. We suspected that the low apparent temperature optimum for IL-1 action was due to inadequate pH control by the bicarbonate-buffered medium. Experiments showed that small decreases in medium pH strongly inhibited the mitogenic action of both forms of IL-1. Furthermore, if thymocytes were stimulated with either form of IL-1 in strongly buffered media, the temperature optimum was at least 39 degrees C. The pI 7.3 and pI 5.0 forms of IL-1 are known to differ both biochemically and immunologically. Our experimental discovery that their temperature sensitivities are much the same suggests that temperature sensitivity is a property of the T cell rather than of the IL-1 molecules themselves.
Full text
PDF



Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Eagle H. Buffer combinations for mammalian cell culture. Science. 1971 Oct 29;174(4008):500–503. doi: 10.1126/science.174.4008.500. [DOI] [PubMed] [Google Scholar]
- Hanson D. F., Murphy P. A. Demonstration of interleukin 1 activity in apparently homogeneous specimens of the pI 5 form of rabbit endogenous pyrogen. Infect Immun. 1984 Aug;45(2):483–490. doi: 10.1128/iai.45.2.483-490.1984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hanson D. F., Murphy P. A., Silicano R., Shin H. S. The effect of temperature on the activation of thymocytes by interleukins I and II. J Immunol. 1983 Jan;130(1):216–221. [PubMed] [Google Scholar]
- Hanson D. F., Murphy P. A., Windle B. E. Failure of rabbit neutrophils to secrete endogenous pyrogen when stimulated with staphylococci. J Exp Med. 1980 Jun 1;151(6):1360–1371. doi: 10.1084/jem.151.6.1360. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hirsch R. L., Jeffries B. D., Gray I. Temperature effects on lymphocyte transformation invitro. Immunol Commun. 1977;6(1):63–80. doi: 10.3109/08820137709055804. [DOI] [PubMed] [Google Scholar]
- Jampel H. D., Duff G. W., Gershon R. K., Atkins E., Durum S. K. Fever and immunoregulation. III. Hyperthermia augments the primary in vitro humoral immune response. J Exp Med. 1983 Apr 1;157(4):1229–1238. doi: 10.1084/jem.157.4.1229. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Murphy P. A., Cebula T. A., Levin J., Windle B. E. Rabbit macrophages secrete two biochemically and immunologically distinct endogenous pyrogens. Infect Immun. 1981 Oct;34(1):177–183. doi: 10.1128/iai.34.1.177-183.1981. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Murphy P. A., Simon P. L., Willoughby W. F. Endogenous pyrogens made by rabbit peritoneal exudate cells are identical with lymphocyte-activating factors made by rabbit alveolar macrophages. J Immunol. 1980 May;124(5):2498–2501. [PubMed] [Google Scholar]
- Roberts N. J., Jr, Steigbigel R. T. Hyperthermia and human leukocyte functions: effects on response of lymphocytes to mitogen and antigen and bactericidal capacity of monocytes and neutrophils. Infect Immun. 1977 Dec;18(3):673–679. doi: 10.1128/iai.18.3.673-679.1977. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Smith J. B., Knowlton R. P., Agarwal S. S. Human lymphocyte responses are enhanced by culture at 40 degrees C. J Immunol. 1978 Aug;121(2):691–694. [PubMed] [Google Scholar]



