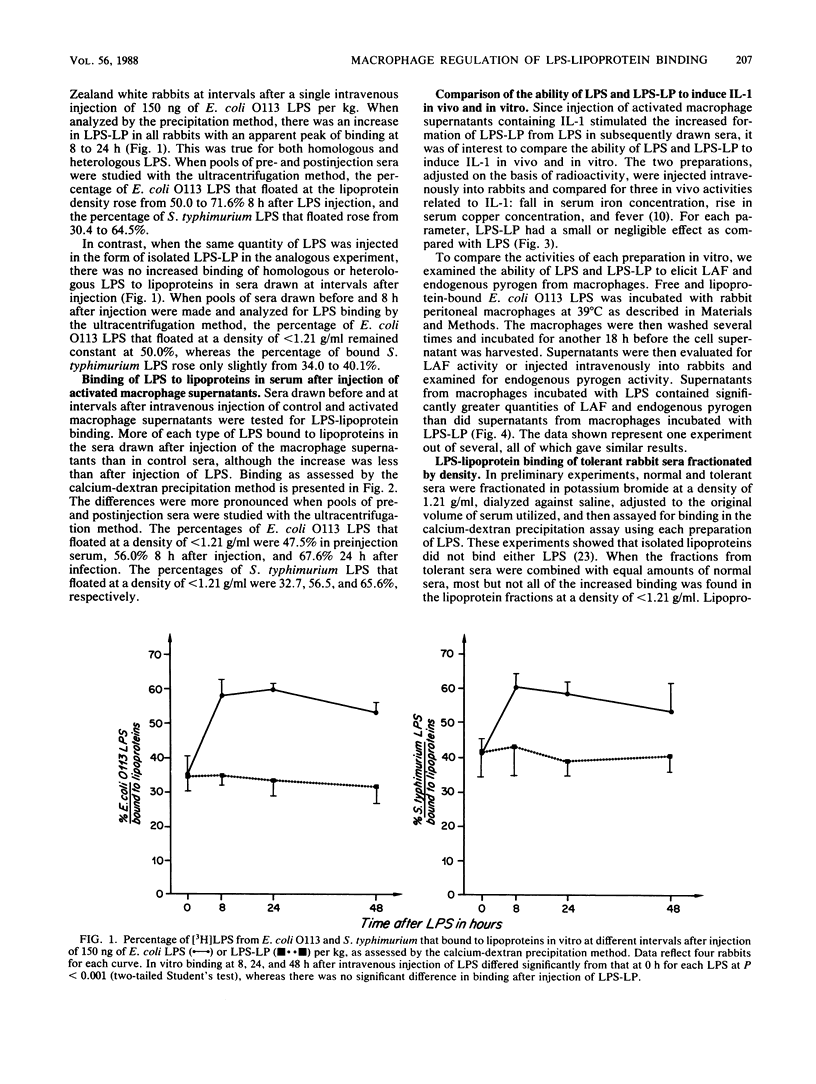
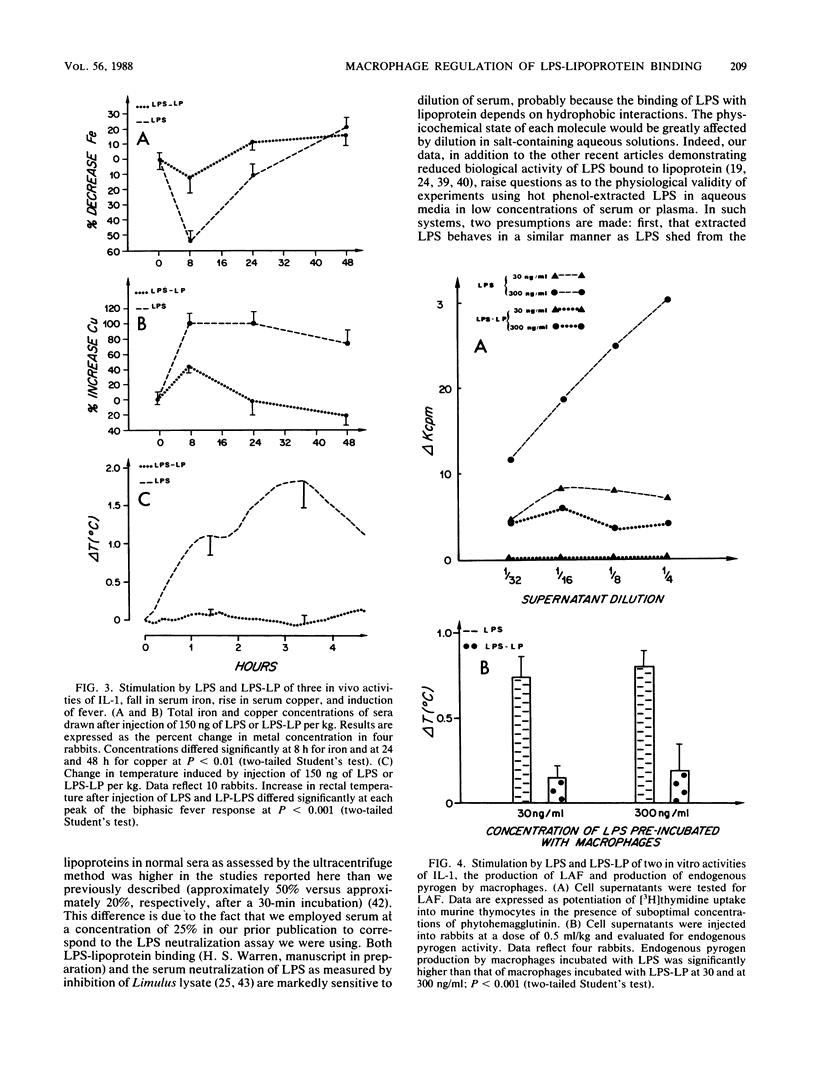
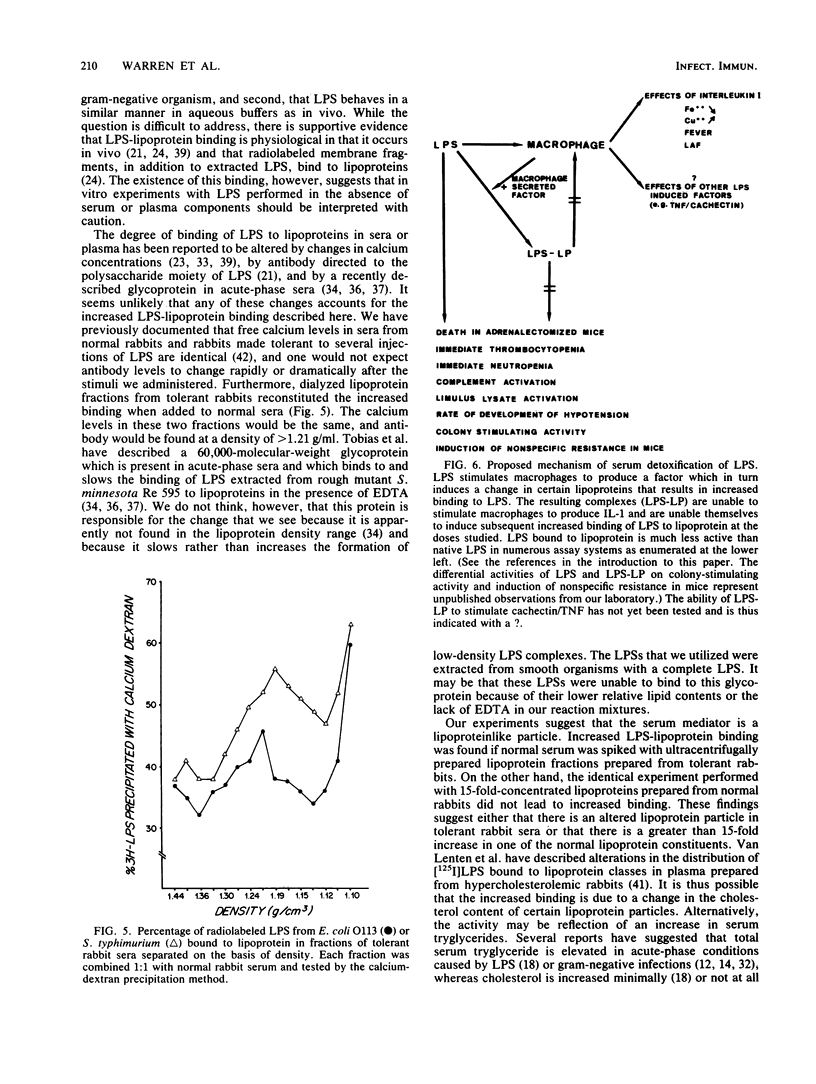
Abstract
Lipopolysaccharides (LPSs) extracted from gram-negative bacteria are much less active when bound to serum lipoproteins. We present evidence here that the binding of radiolabeled LPS extracted from Escherichia coli O113 and Salmonella typhimurium to lipoproteins in rabbit serum is increased 8 to 24 h after a single intravenous injection of homologous or heterologous LPS. Supernatants of activated macrophages containing interleukin-1 also stimulate increased binding. The isolated product of this binding does not induce the production of interleukin-1 by macrophages in vivo or in vitro and is unable itself to stimulate increased binding of LPS to lipoprotein. Normal rabbit sera spiked with lipoprotein fractions prepared from tolerant but not normal rabbit sera bind increased amounts of LPS. These data suggest that there may exist a self-regulated mechanism for decreasing the toxicity of LPS and the production of LPS-induced interleukin-1; this mechanism is controlled by a macrophage factor and functions through altering the binding of LPS to certain serum lipoproteins.
Full text
PDF





Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Abehsira-Amar O., Damais C., Parant M., Chedid L. Strain dependence of muramyl dipeptide-induced LAF(IL 1) release by murine-adherent peritoneal cells. J Immunol. 1985 Jan;134(1):365–368. [PubMed] [Google Scholar]
- Beutler B. A., Cerami A. Recombinant interleukin 1 suppresses lipoprotein lipase activity in 3T3-L1 cells. J Immunol. 1985 Dec;135(6):3969–3971. [PubMed] [Google Scholar]
- Beutler B. A., Milsark I. W., Cerami A. Cachectin/tumor necrosis factor: production, distribution, and metabolic fate in vivo. J Immunol. 1985 Dec;135(6):3972–3977. [PubMed] [Google Scholar]
- Beutler B., Milsark I. W., Cerami A. C. Passive immunization against cachectin/tumor necrosis factor protects mice from lethal effect of endotoxin. Science. 1985 Aug 30;229(4716):869–871. doi: 10.1126/science.3895437. [DOI] [PubMed] [Google Scholar]
- Burstein M., Scholnick H. R., Morfin R. Rapid method for the isolation of lipoproteins from human serum by precipitation with polyanions. J Lipid Res. 1970 Nov;11(6):583–595. [PubMed] [Google Scholar]
- Bøg-Hansen T. C., Krog H. H., Back U. Plasma lipoprotein-associated arylesterase is induced by bacterial lipopolysaccharide. FEBS Lett. 1978 Sep 1;93(1):86–90. doi: 10.1016/0014-5793(78)80811-4. [DOI] [PubMed] [Google Scholar]
- Cabana V. G., Gewurz H., Siegel J. N. Inflammation-induced changes in rabbit CRP and plasma lipoproteins. J Immunol. 1983 Apr;130(4):1736–1742. [PubMed] [Google Scholar]
- Cham B. E., Knowles B. R. A solvent system for delipidation of plasma or serum without protein precipitation. J Lipid Res. 1976 Mar;17(2):176–181. [PubMed] [Google Scholar]
- Damais C., Riveau G., Parant M., Gerota J., Chedid L. Production of lymphocyte activating factor in the absence of endogenous pyrogen by rabbit or human leukocytes stimulated by a muramyl dipeptide derivative. Int J Immunopharmacol. 1982;4(5):451–462. doi: 10.1016/0192-0561(82)90020-0. [DOI] [PubMed] [Google Scholar]
- Dinarello C. A., Cannon J. G., Wolff S. M., Bernheim H. A., Beutler B., Cerami A., Figari I. S., Palladino M. A., Jr, O'Connor J. V. Tumor necrosis factor (cachectin) is an endogenous pyrogen and induces production of interleukin 1. J Exp Med. 1986 Jun 1;163(6):1433–1450. doi: 10.1084/jem.163.6.1433. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dinarello C. A. Interleukin-1. Rev Infect Dis. 1984 Jan-Feb;6(1):51–95. doi: 10.1093/clinids/6.1.51. [DOI] [PubMed] [Google Scholar]
- Estavoyer J. M., Stimmesse B., Ottignon Y., Cardot N., Boillot A., Dupont M. J. Serum lipids in patients with leptospirosis. J Infect Dis. 1984 Dec;150(6):963–964. doi: 10.1093/infdis/150.6.963-a. [DOI] [PubMed] [Google Scholar]
- FREEDMAN H. H. Passive transfer of protection against lethality of homologous heterologous endotoxins. Proc Soc Exp Biol Med. 1959 Nov;102:504–506. doi: 10.3181/00379727-102-25296. [DOI] [PubMed] [Google Scholar]
- Gallin J. I., Kaye D., O'Leary W. M. Serum lipids in infection. N Engl J Med. 1969 Nov 13;281(20):1081–1086. doi: 10.1056/NEJM196911132812001. [DOI] [PubMed] [Google Scholar]
- Greisman S. E., Young E. J., Carozza F. A., Jr Mechanisms of endotoxin tolerance. V. Specificity of the early and late phases of pyrogenic tolerance. J Immunol. 1969 Dec;103(6):1223–1236. [PubMed] [Google Scholar]
- Greisman S. E., Young E. J., DuBuy B. Mechanisms of endotoxin tolerance. 8. Specificity of serum transfer. J Immunol. 1973 Nov;111(5):1349–1360. [PubMed] [Google Scholar]
- Hoffman J. S., Benditt E. P. Changes in high density lipoprotein content following endotoxin administration in the mouse. Formation of serum amyloid protein-rich subfractions. J Biol Chem. 1982 Sep 10;257(17):10510–10517. [PubMed] [Google Scholar]
- Kaufmann R. L., Matson C. F., Beisel W. R. Hypertriglyceridemia produced by endotoxin: role of impaired triglyceride disposal mechanisms. J Infect Dis. 1976 May;133(5):548–555. doi: 10.1093/infdis/133.5.548. [DOI] [PubMed] [Google Scholar]
- Mathison J. C., Ulevitch R. J. In vivo interaction of bacterial lipopolysaccharide (LPS) with rabbit platelets: modulation by C3 and high density lipoproteins. J Immunol. 1981 Apr;126(4):1575–1580. [PubMed] [Google Scholar]
- Munford R. S., Dietschy J. M. Effects of specific antibodies, hormones, and lipoproteins on bacterial lipopolysaccharides injected into the rat. J Infect Dis. 1985 Jul;152(1):177–184. doi: 10.1093/infdis/152.1.177. [DOI] [PubMed] [Google Scholar]
- Munford R. S., Hall C. L., Dietschy J. M. Binding of Salmonella typhimurium lipopolysaccharides to rat high-density lipoproteins. Infect Immun. 1981 Dec;34(3):835–843. doi: 10.1128/iai.34.3.835-843.1981. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Munford R. S., Hall C. L., Lipton J. M., Dietschy J. M. Biological activity, lipoprotein-binding behavior, and in vivo disposition of extracted and native forms of Salmonella typhimurium lipopolysaccharides. J Clin Invest. 1982 Oct;70(4):877–888. doi: 10.1172/JCI110684. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Munford R. S., Hall C. L. Radioimmunoassay for Gram-negative bacterial lipopolysaccharide O antigens: influence of antigen solubility. Infect Immun. 1979 Oct;26(1):42–48. doi: 10.1128/iai.26.1.42-48.1979. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Novitsky T. J., Roslansky P. F., Siber G. R., Warren H. S. Turbidimetric method for quantifying serum inhibition of Limulus amoebocyte lysate. J Clin Microbiol. 1985 Feb;21(2):211–216. doi: 10.1128/jcm.21.2.211-216.1985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- OSBORN M. J., ROSEN S. M., ROTHFIELD L., HORECKER B. L. Biosynthesis of bacterial lipopolysaccharide. I. Enzymatic incorporation of galactose in a mutant strain of Salmonella. Proc Natl Acad Sci U S A. 1962 Oct 15;48:1831–1838. doi: 10.1073/pnas.48.10.1831. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pontet M., Ayrault-Jarrier M., Burdin J., Gelin M., Engler R. Préparation et forme sérique de la protéine réactive C de lapin. Biochimie. 1979;61(11-12):1293–1299. [PubMed] [Google Scholar]
- Riveau G. R., Novitsky T. J., Roslansky P. F., Dinarello C. A., Warren H. S. Role of interleukin-1 in augmenting serum neutralization of bacterial lipopolysaccharide. J Clin Microbiol. 1987 May;25(5):889–892. doi: 10.1128/jcm.25.5.889-892.1987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Riveau G., Parant M., Damais C., Parant F., Chedid L. Dissociation between muramyl dipeptide-induced fever and changes in plasma metal levels. Am J Physiol. 1986 Apr;250(4 Pt 1):C572–C577. doi: 10.1152/ajpcell.1986.250.4.C572. [DOI] [PubMed] [Google Scholar]
- Romeo D., Girard A., Rothfield L. Reconstitution of a functional membrane enzyme system in a monomolecular film. I. Formation of a mixed monolayer of lipopolysaccharide and phospholipid. J Mol Biol. 1970 Nov 14;53(3):475–490. doi: 10.1016/0022-2836(70)90078-1. [DOI] [PubMed] [Google Scholar]
- Rudbach J. A., Akiya F. I., Elin R. J., Hochstein H. D., Luoma M. K., Milner E. C., Milner K. C., Thomas K. R. Preparation and properties of a national reference endotoxin. J Clin Microbiol. 1976 Jan;3(1):21–25. doi: 10.1128/jcm.3.1.21-25.1976. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Segura F., Force L., Espejo R., Soriano E., San Miguel J. G. Hypertriglyceridemia in patients with typhoid fever. J Infect Dis. 1983 Mar;147(3):606–606. doi: 10.1093/infdis/147.3.606. [DOI] [PubMed] [Google Scholar]
- Skarnes R. C. The inactivation of endotoxin after interaction with certain proteins of normal serum. Ann N Y Acad Sci. 1966 Jun 30;133(2):644–662. doi: 10.1111/j.1749-6632.1966.tb52395.x. [DOI] [PubMed] [Google Scholar]
- Tobias P. S., McAdam K. P., Soldau K., Ulevitch R. J. Control of lipopolysaccharide-high-density lipoprotein interactions by an acute-phase reactant in human serum. Infect Immun. 1985 Oct;50(1):73–76. doi: 10.1128/iai.50.1.73-76.1985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tobias P. S., McAdam K. P., Ulevitch R. J. Interactions of bacterial lipopolysaccharide with acute-phase rabbit serum and isolation of two forms of rabbit serum amyloid A. J Immunol. 1982 Mar;128(3):1420–1427. [PubMed] [Google Scholar]
- Tobias P. S., Soldau K., Ulevitch R. J. Isolation of a lipopolysaccharide-binding acute phase reactant from rabbit serum. J Exp Med. 1986 Sep 1;164(3):777–793. doi: 10.1084/jem.164.3.777. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tobias P. S., Ulevitch R. J. Control of lipopolysaccharide-high density lipoprotein binding by acute phase protein(s). J Immunol. 1983 Oct;131(4):1913–1916. [PubMed] [Google Scholar]
- Tracey K. J., Beutler B., Lowry S. F., Merryweather J., Wolpe S., Milsark I. W., Hariri R. J., Fahey T. J., 3rd, Zentella A., Albert J. D. Shock and tissue injury induced by recombinant human cachectin. Science. 1986 Oct 24;234(4775):470–474. doi: 10.1126/science.3764421. [DOI] [PubMed] [Google Scholar]
- Ulevitch R. J., Johnston A. R. The modification of biophysical and endotoxic properties of bacterial lipopolysaccharides by serum. J Clin Invest. 1978 Dec;62(6):1313–1324. doi: 10.1172/JCI109252. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ulevitch R. J., Johnston A. R., Weinstein D. B. New function for high density lipoproteins. Their participation in intravascular reactions of bacterial lipopolysaccharides. J Clin Invest. 1979 Nov;64(5):1516–1524. doi: 10.1172/JCI109610. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Van Lenten B. J., Fogelman A. M., Haberland M. E., Edwards P. A. The role of lipoproteins and receptor-mediated endocytosis in the transport of bacterial lipopolysaccharide. Proc Natl Acad Sci U S A. 1986 Apr;83(8):2704–2708. doi: 10.1073/pnas.83.8.2704. [DOI] [PMC free article] [PubMed] [Google Scholar]
- WOLFF S. M., MULHOLLAND J. H., WARD S. B., RUBENSTEIN M., MOTT P. D. EFFECT OF 6-MERCAPTOPURINE ON ENDOTOXIN TOLERANCE. J Clin Invest. 1965 Aug;44:1402–1409. doi: 10.1172/JCI105245. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Warren H. S., Knights C. V., Siber G. R. Neutralization and lipoprotein binding of lipopolysaccharides in tolerant rabbit serum. J Infect Dis. 1986 Nov;154(5):784–791. doi: 10.1093/infdis/154.5.784. [DOI] [PubMed] [Google Scholar]
- Webster C. J. Principles of a quantitative assay for bacterial endotoxins in blood that uses Limulus lysate and a chromogenic substrate. J Clin Microbiol. 1980 Nov;12(5):644–650. doi: 10.1128/jcm.12.5.644-650.1980. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Woloski B. M., Fuller G. M. Identification and partial characterization of hepatocyte-stimulating factor from leukemia cell lines: comparison with interleukin 1. Proc Natl Acad Sci U S A. 1985 Mar;82(5):1443–1447. doi: 10.1073/pnas.82.5.1443. [DOI] [PMC free article] [PubMed] [Google Scholar]



