Abstract
Methicillin-susceptible Staphylococcus aureus (MSSA) can arise from methicillin-resistant S. aureus (MRSA) following partial or complete excision of staphylococcal cassette chromosome mec (SCCmec). This study investigated whether multiresistant MSSA isolates from Irish hospitals, where MRSA has been endemic for decades, harbor SCCmec DNA. Twenty-five multiresistant MSSA isolates recovered between 2002 and 2006 were tested for SCCmec DNA by PCR and were genotyped by multilocus sequence typing and spa typing. All isolates lacked mecA. Three isolates (12%) harbored SCCmec DNA; two of these (genotype ST8/t190) harbored a 26-kb SCCmec IID (II.3.1.2) remnant that lacked part of mecI and all of mecR1, mecA, and IS431; the third isolate (ST8/t3209) harbored the SCCmec region from dcs to orfX. All three isolates were detected as MRSA using the BD GeneOhm and Cepheid's Xpert MRSA real-time PCR assays. Six isolates (ST8/t190, n = 4; ST5/t088, n = 2), including both isolates with the SCCmec IID remnant, harbored ccrAB4 with 100% identity to ccrAB4 from the Staphylococcus epidermidis composite island SCC-CI. This ccrAB4 gene was also identified in 23 MRSA isolates representative of ST8/t190-MRSA with variant SCCmec II subtypes IIA to IIE, which predominated previously in Irish hospitals. ccrAB4 was located 5,549 bp upstream of the left SCCmec junction in both the MRSA and MSSA isolates with SCCmec elements and remnants and 5,549 bp upstream of orfX in the four MSSA isolates with ccrAB4 only on an SCC-CI homologous region. This is the first description of a large SCCmec remnant with ccr and partial mec genes in MSSA and of the S. epidermidis SCC-CI and ccrAB4 genes in S. aureus.
Staphylococcus aureus is a significant human pathogen that can cause a wide variety of diseases, due in part to its ability to acquire and express an extensive array of virulence factors and antimicrobial resistance determinants. Mobile genetic elements are involved in the dissemination of virulence and resistance genes in S. aureus and include plasmids, bacteriophages, pathogenicity islands, transposons, and chromosomal cassettes (3, 4, 18, 24, 26, 30, 38, 45, 63, 64).
Following the introduction of methicillin into clinical use, methicillin-resistant S. aureus (MRSA) has emerged as a major nosocomial problem worldwide. Today MRSA continues to be a significant burden in hospitals but has also emerged as a problem in the community (22). Methicillin resistance in S. aureus is encoded by the mecA gene, which is located within a mobile staphylococcal cassette chromosome (SCC) element known as SCCmec (22). MRSA can emerge from methicillin-susceptible S. aureus (MSSA) upon site-specific integration of SCCmec into the orfX locus in the chromosome of a susceptible isolate. SCCmec consists of three regions: a mec complex carrying mecA and, if present, its regulatory genes mecI and mecR1, a ccr complex carrying cassette chromosome recombinase (ccr) genes, and a series of variable “junkyard” or J regions (30). To date, six types of SCCmec have been recognized in S. aureus, with three additional new types and numerous variants also recently being reported (14, 21, 48, 61). SCCmec has been found in other staphylococcal species, including S. epidermidis, S. haemolyticus, S. hominis, and S. warneri (19, 20, 62, 65). At least five non-mec-containing SCC elements have also been described, two in MSSA (SCCcap1 and SCC476), one in S. epidermidis (SCC-CI with SCCpbp4), and one in S. hominis (SCC12263) (31, 35, 41). The complexity and evolutionary history of SCC demonstrates the versatility with which S. aureus can acquire, disseminate, modify, and delete resistance determinants (14, 34, 43, 44).
MRSA has been endemic in Irish hospitals for many years. In 2003, the rate of methicillin resistance among S. aureus isolates recovered from blood culture was 42%, placing Ireland among the countries with the highest rates of MRSA in Europe (42). Monitoring of the epidemiological types that comprise the Irish MRSA population by antibiogram-resistogram (AR) typing and chromosomal DNA macrorestriction analysis using pulsed-field gel electrophoresis (PFGE) has shown that the MRSA population changed between 1999 and 2003, with a non-multiantibiotic-resistant strain exhibiting the AR type and PFGE group (AR-PFG) type 06-01 (ST22-MRSA-IV), displacing the previously predominant multiantibiotic-resistant strains AR-PFG type 13-00 or 14-00 (13-00 and 14-00 isolates exhibit the genotype ST8-MRSA-II) (54). In the present study, multiantibiotic resistance is defined as resistance to two or more classes of non-beta-lactam antibiotics (11). Isolates exhibiting AR-PFG 13-00 or 14-00 are resistant to the aminoglycoside antimicrobial agent gentamicin and accounted for 50% of the MRSA population in 1999 (55). Although largely replaced by AR-PFG 06-01 in acutely ill patients with invasive disease (for example, among patients with bacteremia), older strains may persist among chronically ill patients and in long-stay care units. The importance of the long-term persistence of MRSA in patients with osteomyelitis has been noted in both human and veterinary medicine (1, 58).
In some strains of MRSA or in some specific circumstances (e.g., during the absence of antibiotic selective pressure), SCCmec may be unstable and can be excised (8, 11). Furthermore, exposure of MRSA to the glycopeptide antibiotic vancomycin can also lead to mecA excision, and Noto et al. (43) reported that S. aureus may compensate for the fitness cost incurred by developing reduced susceptibility to vancomycin by deleting all or a portion of SCCmec. It has also been reported that when SCCmec is excised from multiantibiotic-resistant MRSA, the resulting MSSA strain may carry a larger number of resistance determinants than would usually be found in MSSA (11).
While there are vast amounts of data in the literature concerning the structure of the numerous SCCmec elements that have been identified in MRSA, there is only very limited information on segments of SCCmec in multiresistant MSSA isolates following partial excision or integration of SCCmec (6, 11, 13, 25, 57). The purpose of the present study was to investigate whether multiresistant MSSA isolates recovered from patients in Irish hospitals carried segments of SCCmec and to compare their genotypes to each other's and to those of multiresistant MRSA isolates previously recovered in Ireland using multilocus sequence typing (MLST) and spa typing. In selecting the MSSA isolates for analysis, gentamicin resistance was used as a surrogate marker for multiantibiotic resistance because evidence from a previous study indicated that in Ireland, multiantibiotic-resistant MRSA strains were also gentamicin resistant (53). In the present study, isolates carrying SCCmec- and/or SCC-specific DNA were subsequently tested using two commercially available real-time PCR assays for the rapid detection of MRSA from clinical specimens. The assays chosen were the BD GeneOhm MRSA and Cepheid's Xpert MRSA assays, both of which target the region from SCCmec to orfX.
MATERIALS AND METHODS
Isolates.
All gentamicin-resistant MSSA (GrMSSA) isolates recovered between 2004 and 2006 (n = 19) from patients attending a large (936-bed) tertiary-referral Dublin hospital (hospital 1), together with GrMSSA isolates (n = 6) from five other Irish hospitals, referred to the Irish National MRSA Reference Laboratory for investigation of methicillin resistance, were included in the study (Table 1). One isolate per patient was investigated except in one instance, where two isolates with different antibiograms were recovered from one patient (Table 1). All isolates were identified as MSSA, typed by AR typing, and screened for the presence or absence of the mecA gene by PCR as described previously (56). Isolates were stored at −80°C in Protect bacterial preserver vials (Technical Services Consultants Ltd., Heywood, United Kingdom) and cultured on tryptone soya agar (Oxoid Ltd., Basingstoke, United Kingdom) prior to incubation overnight at 37°C.
TABLE 1.
Patients’ MRSA histories and phenotypic and genotypic characteristics of the 25 MSSA isolates investigated
| Isolatea | MRSA historyb | Resistance pattern
|
spa clusterf | spa type | MLST CC | ST | Amplimer(s) obtained with SCCmec typing PCRg
|
|||
|---|---|---|---|---|---|---|---|---|---|---|
| Aminoglycosides/ aminocyclitolc | Other antimicrobialsd | PCR scheme 1 (mec) | PCR scheme 2 (ccr) | PCR scheme 3i | ||||||
| M06/0075 | GrMRSA | GEN, KAN, NEO, SPC, STR, TOB | AMP, CAD, CIP, ERY, ETBR, LIN, MC, PMA | 1 | t190 | 8 | ST8 | Negative | ccrAB4 and ccrAB2 | mecI and dcs |
| M06/0179 | GrMRSA | GEN, KAN, NEO, SPC, STR, TOB | CIP, ERY, LIN, MUPe | 1 | t190 | 8 | ST8 | Negative | ccrAB4 and ccrAB2 | mecI and dcs |
| M05/0146 | GrMRSA | GEN, KAN, NEO, SPC, STR, TOB | AMP, CAD, CIP, ERY, ETBR, MC, MUP,e PMA, TMP | 1 | t190 | 8 | ST8 | Negative | ccrAB4 | Negative |
| M02/0021 | GrMRSA | AMI,e GEN, KAN, NEO, SPC, STR, TOB | AMP, CAD, CIP, ERY, ETBR, MC, MUP,e PMA | 1 | t190 | 8 | ST8 | Negative | ccrAB4 | Negative |
| M04/0269 | GrMRSA | GEN, KAN, NEO, SPC, STR, TOB | AMP, CAD, CIP, ERY, ETBR, MC, PMA | 1 | t3209 | 8 | ST8 | Negative | Negative | dcs |
| M06/0324 | None | GEN, KAN, TOB | AMP, CIP | 2 | t2658 | 8 | ST8 | Negative | Negative | Negative |
| M06/0329 | None | GEN, KAN, TOB | AMP, CIP | 2 | t2658 | 8 | ST8 | Negative | Negative | Negative |
| M05/0065 | None | GEN, KAN, TOB | AMP, CAD, CIP,e MC, TMP | 2 | t2658 | 8 | ST8 | Negative | Negative | Negative |
| M05/0330 | None | GEN, KAN, NEO, STR, TOB | AMP, SUL, TET, TMP | 2 | t064 | 8 | ST8 | Negative | Negative | Negative |
| M06/0004 | GsMRSA | GEN, KAN, TOB | AMP, ERY, FUS, LIN, MUP | Singleton | t088 | 5 | ST5 | Negative | ccrAB4 | Negative |
| M06/0288 | Not known | GEN, KAN, NEO, TOB | ERY, FUS, LIN, MUP | Singleton | t088 | 5 | ST5 | Negative | ccrAB4 | Negative |
| M05/0083 | Not known | GEN, KAN, TOB | AMP, CAD, CIP, ERY, LIN, MUP | 3 | t379 | 22 | ST22 | Negative | Negative | Negative |
| M05/0077 | None | GEN, KAN, TOB | AMP | 3 | t005 | 22 | ST22 | Negative | Negative | Negative |
| M04/0261 | None | GEN, KAN, TOB | AMP, TMP | 3 | t005 | 22 | ST22 | Negative | Negative | Negative |
| M05/0232 | None | GEN, KAN, TOB | AMP, CAD, TMP | 3 | t891 | 22 | ST854 | Negative | Negative | Negative |
| M06/0378 | None | GEN, KAN, TOB | AMP, ERY, LIN, MUP, TET | 4 | t127 | 1 | ST1 | Negative | Negative | Negative |
| M04/0262 | None | GEN, KAN, TOB | AMP, CAD,e TET | 4 | t1383 | 1 | ST1135h | Negative | Negative | Negative |
| M05/0084 | GsMRSA | AMI,e GEN, KAN, NEO, TOB | AMP, CAD, LIN, MUP | Singleton | t3500h | 45 | ST1096h | Negative | Negative | Negative |
| M05/0167 | GsMRSA | GEN, KAN, TOB | AMP, ETBR, LIN, MUP | Singleton | t3500h | 45 | ST1096h | Negative | Negative | Negative |
| M05/0045 | GsMRSA | GEN, KAN, TOB | AMP | Singleton | t2078 | 101 | ST101 | Negative | Negative | Negative |
| M05/0080 | GsMRSA | GEN, KAN, TOB | AMP TMP | Singleton | t2078 | 101 | ST101 | Negative | Negative | Negative |
| M05/0223 | None | GEN, KAN, TOB | AMP, CAD, FUS, MUP | Singleton | t021 | 30 | ST30 | Negative | Negative | Negative |
| M04/0260 | None | GEN, KAN, TOB | ERY | 5 | t084 | 15 | ST582 | Negative | Negative | Negative |
| M05/0133 | None | GEN, KAN, TOB | ERY | 5 | t491 | 15 | ST582 | Negative | Negative | Negative |
| M06/0392 | None | GEN, KAN, TOB | AMP, CAD, TET | Singleton | t164 | 20 | ST1134h | Negative | Negative | Negative |
One isolate per patient was included except in one instance, where two isolates with different antibiograms (M06/0179 and M06/0075) were recovered from one patient. Isolates shown with bold and underlined typeface yielded amplimers by SCCmec typing PCR.
Not known, data on patient's MRSA history were not available.
Aminoglycosides/aminocyclitols tested were as follows: AMI, amikacin; GEN, gentamicin; KAN, kanamycin; NEO, neomycin; SPC, spectinomycin; STR, streptomycin; TOB, tobramycin.
Other antimicrobials tested included the following: AMP, ampicillin; CAD, cadmium acetate; CHL, chloramphenicol; CIP, ciprofloxacin; ERY, erythromycin; ETBR, ethidium bromide; FUS, fusidic acid; LIN, lincomycin; MC, mercuric chloride; MUP, mupirocin; PMA, phenyl mercuric acetate; RIF, rifampin; SUL, sulfonamide; TET, tetracycline; TMP, trimethoprim; VAN, vancomycin.
Intermediately susceptible to the antimicrobial indicated.
The BURP algorithm was applied to all isolates for the determination of clusters of related isolates based on the spa types identified (40).
Sequencing was performed to confirm the identities of all amplimers obtained by SCCmec typing PCR. Negative, no PCR amplimers were obtained.
New STs or spa types identified in the present study.
See reference 47.
During the course of the present study, PCR used for SCCmec typing and nucleotide sequence analysis of resulting amplimers showed that some GrMSSA isolates harbored segments of DNA with sequences similar to regions of SCCmec elements in MRSA isolates recovered in Ireland between 1989 and 2002. All 54 of these earlier isolates exhibited MLST sequence type 8 (ST8) and variant subtypes of SCCmec II (59). Isolates representative of each of the variant SCCmec II subtypes identified during that study (n = 23) were spa typed, and all exhibited the same spa type (t190). These 23 MRSA isolates recovered between 1989 and 2002 were subsequently investigated for carriage of ccrAB4 because the earlier study was undertaken before amplification of ccrAB4 formed part of any SCCmec typing scheme.
Chemicals, enzymes, and oligonucleotides.
All chemicals used were of analytical grade or molecular biology grade and were purchased from the Sigma-Aldrich Chemical Co. (Tallaght, Dublin, Ireland). Enzymes were purchased from the Promega Corporation (Madison, WI) or Roche Diagnostics Ltd. (Lewes, East Sussex, United Kingdom) and were used according to the manufacturer's instructions. DNA molecular weight markers were purchased from Promega. Oligonucleotide primers were custom synthesized by the Sigma-Aldrich Company Ltd. (Haverhill, United Kingdom).
Molecular characterization.
All isolates were investigated by SCCmec typing PCR, spa typing, and MLST. Genomic DNA was extracted using the DNeasy kit (Qiagen, Crawley, United Kingdom) according to the manufacturer's instructions. PCRs were performed using GoTaq Flexi DNA polymerase (Promega) according to the manufacturer's instructions using the published protocols for each method described below. Amplifications were performed in a Thermo-Hybaid Multiblock system thermal cycler (Thermo-Hybaid, Ashford, Middlesex, United Kingdom). PCR products were visualized by conventional agarose gel electrophoresis and purified with the GenElute PCR cleanup kit unless otherwise indicated in the appropriate sections below (Sigma-Aldrich). Sequencing was performed commercially by Cogenics (Essex, United Kingdom) using an ABI 3730xl DNA analyzer (Applied Biosystems, Foster City, CA).
SCCmec typing PCRs.
Three multiplex SCCmec typing PCRs were performed with template DNA from each isolate to determine if they harbored segments of SCCmec elements I to VI. Two of these methods were described previously by Kondo et al. (32), where they were named MPCR-1 and MPCR-2, respectively, and the third method was described previously by Oliveira and de Lencastre (47). The first multiplex PCR (MPCR-2), referred to here as PCR scheme 1, amplifies the class A, B, and C mec complexes (32). The second multiplex SCCmec typing method (MPCR-1), referred to here as PCR scheme 2, detects the presence of the ccr complex genes ccrAB1, ccrAB2, ccrAB3, ccrAB4, and ccrC (32). The third multiplex SCCmec typing method, referred to here as PCR scheme 3, amplifies partial nucleotide sequences in the mec complex (mecI gene only) and in the junkyard regions of SCCmec I to IV, including subtypes IA (I.1.1.3), IIIA (III.1), and IIIB (III.1.1.3) (47). In each of these multiplex PCR schemes, amplification of the mecA gene was used as an internal control.
The following S. aureus control strains and clinical isolates were used as positive controls for SCCmec typing PCR as indicated: phenotype II 43.2 (SCCmec I, ccrAB1) (59), CA05 (SCCmec IV, class B mec, ccrAB2) (36), WIS (class C mec) (29), 07.4/0237 (SCCmec II) (59), JCSC 4744 (IVA) (47), M00/0005.2 (ccrAB4), and E0898 (SCCmec III, class A mec, ccrAB3 ccrC).
All SCCmec typing amplimers were sequenced to confirm their identities using the PCR primers yielding those amplimers. Analysis of chromatograms and sequences was carried out using the TraceViewer software program (version 1.1.3; CodonCode Corporation, Massachusetts) and DNA Strider 1.3f11 software (CEA/Saclay, Gif-sur-Yvette, France), respectively. Homology searches were performed using BLAST software (http://ncbi.nih.gov/BLAST).
MLST.
MLST was performed as described previously (15). PCR products were purified using the Qiaquick 96 PCR purification kit (Qiagen). Analysis of chromatograms and sequences was performed using the BioNumerics software package (version 5.0; Applied Maths, Ghent, Belgium). The alleles at each of the seven housekeeping loci were identified by comparison with sequences held in the MLST database (http://saureus.mlst.net). The allelic profile and hence the ST of each isolate was also determined using this database. The stringent group definition for clonal complex (CC) determination was used, where isolates with specific STs are assigned to a particular CC if they are related to at least one other ST in that CC at six out of the seven MLST loci used (16). Isolates that do not share alleles at six of the seven MLST loci with any other ST in the MLST database are deemed singletons (16).
spa typing.
The primers and thermal cycling conditions recommended by the European Network of Laboratories for Sequence Based Typing of Microbial Pathogens (SeqNet) were used for spa typing (http://www.seqnet.org/). The Ridom StaphType software program, version 1.3 (Ridom Gmbh, Wurzburg, Germany), was used for spa sequence analysis, assignment of spa types, and BURP (based upon related patterns) spa clonal complex determination. The default parameters for cluster definition were used for BURP. These include the exclusion of spa types that are shorter than five repeats because they are deemed too short for the deduction of evolutionary history and the clustering of spa types only if the cost (i.e., the steps of evolution between two different spa types) is less than or equal to four (40).
Nucleotide sequencing of SCCmec remnant in isolate M06/0075.
The entire nucleotide sequence of the SCCmec remnant from one MSSA isolate, M06/0075, was determined. This isolate was selected because, along with another isolate recovered from the same patient (M06/0179), it yielded the greatest number of SCCmec typing amplimers. Sequencing was undertaken to determine if these amplimers were part of a larger remnant of SCCmec. The element was amplified and sequenced from the left chromosomal SCCmec junction to orfX using a combination of previously described and newly designed overlapping primers based on the published SCCmec II nucleotide sequence (http://www.ncbi.nlm.nih.gov/sites/entrez; accession number D86934). The SCCmec II nucleotide sequence was chosen because three out of the four amplimers produced by GrMSSA isolate M06/0075 indicated the presence of an SCCmec remnant with closest similarity to the type II SCCmec element (i.e., dcs, mecI, and ccrAB2). The primers used are listed in Table 2.
TABLE 2.
Primers used in the present study
| Primer application | Primer pair(s) | Nucleotide sequence (5′-3′) | Nucleotide coordinates | SCCmec region amplified | Reference |
|---|---|---|---|---|---|
| Amplification and sequencing of the SCCmec IID remnant | IRLII F | CTCTGCGTATCAGTTAATGA | 4684-4703a | Left chromosomal/SCCmec junction to ccrA2 | 59 |
| ccrA R | GCTTCGATAGCCTGTTTCTG | 25490-25471a | 59 | ||
| ccrA2 F5 | AACTTATCGAGATATTAGCC | 25265-25284a | ccrA2 to Tn554 | This study | |
| Tn554 R | AAGCTATCCACGTTCAATCTCAAC | 32442-32419a | 59 | ||
| N044 F | AAATAGTATAATGCTCGGTC | 30481-30500a | N044 to Tn554 | This study | |
| TN554 R6 | TGGAGACATATTAGACACAA | 36440-36421a | This study | ||
| Tn554 F7 | GCGATAAAGGACAGTGACTT | 36191-36210a | Tn554 to mecI | This study | |
| mecI R2 | AGGAAACAATCAAGTCGTTG | 42507-42488a | This study | ||
| mecI P2 | ATCAAGACTTGCATTCAGGC | 42428-42447a | mecI to dcs | 47 | |
| dcs R1 | AGACGAAGATAAGAAAGAAC | 56433-56414a | This study | ||
| dcs F | GTCAATGAGATCATCTACAT | 56109-56128a | dcs to right chromosomal/SCCmec junction | 59 | |
| orfX R | CCCAAGGGCAAAGCGAC | 57826-57810a | 59 | ||
| Amplification of ccrAB4 from SCC-CI in S. epidermidis from historic Irish nosocomial ST8-t190 MRSA isolates | α4.3 | AGCGTATGAATCAAAA | 27155-27170b | ccrA4 to ccrB4 of S. epidermidis SCC-CI | This study |
| β4.3 | CGATGACAAATTAAAAT | 27907-27891b | This study | ||
| Determination of the location of ccrAB4 in GrMSSA isolates with the SCCmec IID remnant and in MRSA isolates | ccrB4 F | TTTCGTCCATTACCTACATC | 27075-27056b | ccrB4 to left chromosomal/SCCmec IID remnant junction | This study |
| LCIVb R | TGAGGAGTTTAACAAGTTAT | 160-141c | This study | ||
| Determination of the location of ccrAB4 in GrMSSA isolates with ccrAB4 only | ccrB4 F | TTTCGTCCATTACCTACATC | 27075-27056b | ccrB4 to orfX | This study |
| orfX R | CCCAAGGGCAAGCGAC | 57826-57810a | 59 | ||
| Determination of the location of dcs in the GrMSSA isolate with dcs only | dcs F | GTCAATGAGATCATCTACAT | 56109-56128a | dcs to orfX | 59 |
| orfX R | CCCAAGGGCAAGCGAC | 57826-57810a | 59 |
DNA fragments for sequencing were obtained by PCR amplification of chromosomal DNA using either the Expand high-fidelity PCR system (Roche) for the primer pair dcs F and orfX R or the Expand long-template PCR system (Roche) for all other primer sets according to the manufacturer's instructions. Amplified DNA was separated by agarose gel electrophoresis using at least 5-μl volumes of each PCR product. Amplimers were purified using either the GenElute PCR cleanup kit (Sigma-Aldrich) or the Qiaex II gel extraction kit (Qiagen) prior to direct sequencing using primer walking. Analysis of chromatograms and sequences and homology searches were performed as described above for sequencing of the SCCmec typing amplimers.
Investigation of MRSA isolates for presence of ccrAB4.
Twenty-three MRSA isolates with the same MLST/spa type genetic background and similar SCCmec regions to those of the GrMSSA isolate M06/0075 were investigated to see whether they harbored ccrAB4. The ccrAB4 gene was amplified using the primers α4.3 and β4.3 (Table 2). This primer pair was designed to be specific for the ccrAB4 gene from S. epidermidis because the ccrAB4 gene identified in the GrMSSA isolates in this study had 100% homology with ccrAB4 from the S. epidermidis composite island SCC-CI. PCRs were performed using GoTaq Flexi DNA polymerase (Promega) according to the manufacturer's instructions. Amplifications were performed in a Thermo-Hybaid Multiblock system thermal cycler (Thermo-Hybaid, Ashford, Middlesex, United Kingdom) with an initial denaturation step at 94°C for 2 min, followed by 30 cycles of denaturation at 94°C for 2 min, annealing at 48°C for 1 min, and elongation at 72°C for 2 min. A final elongation step was performed for 2 min at 72°C. The MRSA isolate HDE288, which harbors SCCmec VI with a different ccrAB4 allotype from that found in S. epidermidis, and S. epidermidis ATCC 12228, harboring SCC-CI with ccrAB4, were used as negative and positive controls, respectively, for this PCR assay (41, 48). Isolates harboring ccrAB4 genes identical to those in S. epidermidis yield an 800-bp product using this primer pair, and isolates harboring ccrAB4 from S. aureus should yield no product. PCR products were visualized by agarose gel electrophoresis and purified using the GenElute PCR cleanup kit (Sigma-Aldrich). The identities of amplimers from isolates representative of each variant SCCmec II subtype (IIA [II.3.1.1], IIB [II.3.2.1], IIC [II.3.3.1], IID [II.3.1.2], and IIE [II.3.3.2]) among these MRSA isolates were confirmed by sequencing.
Determination of location of ccrAB4.
Two primers, ccrB4 F and LCIVb R, based on the S. epidermidis ccrAB4 gene and the left extremity of the SCCmec IID remnant, respectively, were designed to investigate the location of ccrAB4 in the two GrMSSA isolates with the SCCmec IID remnant (M06/0075 and M06/0179) recognized in the present study and in five MRSA isolates representative of each SCCmec II variant subtype IIA to IIE that were also found to harbor ccrAB4. A second primer pair, ccrB4 F and orfX R, based on the S. epidermidis ccrAB4 gene sequence and orfX, respectively, was selected to locate ccrAB4 in the four GrMSSA isolates that yielded ccrAB4 only (M02/0021, M05/0146, M06/0004, and M06/0288). Primer sequences are shown in Table 2. DNA fragments were obtained by PCR amplification of chromosomal DNA using the Expand long-template PCR system (Roche) according to the manufacturer's instructions. Amplified DNA was separated by agarose gel electrophoresis using 5-μl volumes of each PCR product.
Determination of location of dcs.
The Primer pair dcs F and orfX R (Table 2) was used to determine the location of dcs in the GrMSSA isolate (M04/0269) that yielded the dcs amplimer only by SCCmec typing PCR. DNA fragments were obtained by PCR amplification of chromosomal DNA as described above.
Investigation of GrMSSA isolates with SCCmec/SCC-associated DNA using real-time PCR MRSA rapid detection assays.
The seven GrMSSA isolates harboring SCCmec/SCC-associated DNA identified in the present study were tested with two commercially available real-time PCR assays for the rapid detection of MRSA in clinical specimens to determine if such isolates would be detected as MRSA. Isolates were tested with the BD GeneOhm MRSA assay (BD Diagnostics, Ste Foy, Quebec, Canada) using a Smart Cycler II thermal cycler (Cepheid, Sunnyvale, CA) and with the Xpert MRSA assay using a GeneXpert DX system (version 1.2) real-time PCR platform (Cepheid) according to the manufacturer's instructions as described previously (52). S. aureus ATCC 25923 (MSSA), S. aureus ATCC 29213 (MSSA), S. aureus ATCC 43300 (MRSA) and S. epidermidis ATCC 12228 were used as control strains with both systems.
Nucleotide sequence accession numbers.
The nucleotide sequence of the SCCmec IID remnant was submitted to the GenBank database under accession number AM983545.
RESULTS
The purpose of the present study was to investigate whether MSSA isolates from patients in an environment where MRSA has been endemic for many years harbor segments or remnants of SCCmec. Twenty-five gentamicin-resistant MSSA isolates were investigated by AR typing, SCCmec typing PCR, and genotyping using MLST and spa typing (Table 1). The majority of isolates (21/25; 84%) were multiantibiotic resistant (i.e., resistant to two or more classes of non-beta-lactam antibiotics [Table 1]). All lacked the mecA gene.
SCCmec typing PCR.
Template DNA from all 25 isolates was tested for the presence of mec complex types A, B, and C (PCR scheme 1), ccr genes 1 to 5 (PCR scheme 2), and the various junkyard regions (including mecI) of SCCmec I to IV (PCR scheme 3). All isolates were mecA negative by PCR schemes 1 to 3 and failed to yield any mec complex amplimers when PCR scheme 1 was used, but 7/25 (28%) isolates yielded at least one SCCmec amplimer each when PCR scheme 2 or 3 was used (Table 1). These seven isolates belonged to the following three MLST/spa genotypes: ST5/t088 (n = 2), ST8/t3209 (n = 1), and ST8/t190 (n = 4) (Table 1).
Investigation of SCCmec amplimers generated from seven GrMSSA isolates.
Two isolates, M06/0075 and M06/0179 (recovered from the same patient after an interval of 4 months), each yielded four amplimers: two amplimers of 1,287 bp and 937 bp were obtained using PCR scheme 2, while PCR scheme 3 yielded a 342-bp and a 209-bp amplimer (Table 1). The 1,287-bp amplimer was identical in size to an amplimer usually obtained with PCR scheme 2 from MRSA template DNA carrying the ccrAB4 gene (Table 1). Nucleotide sequence analysis of this 1,287-bp amplimer showed 88% and 91% homology with ccrAB4 from the MRSA SCCmec element SCCmecNI (14) and SCCmec VI (48), respectively, but showed 100% homology with ccrAB4 from the SCC composite island SCC-CI, found in S. epidermidis ATCC 12228 (41). The 937-bp amplimer was identical in size to an amplimer usually obtained with PCR scheme 2 from MRSA template DNA carrying the MRSA ccrAB2 gene and exhibited 100% sequence homology with ccrAB2. The 342-bp amplimer obtained with PCR scheme 3 was identical in size to an amplimer usually obtained with MRSA template DNA using PCR scheme 3 corresponding to the dcs region located downstream of mecA in SCCmec types I, II, and IV in MRSA isolates. Sequence analysis showed that this amplimer exhibited 100% sequence homology with dcs from MRSA. The 209-bp amplimer obtained with PCR scheme 3 was identical in size to an amplimer usually obtained with MRSA template DNA using PCR scheme 3 corresponding to the mec complex gene mecI and exhibited 100% sequence homology with mecI.
Four isolates (M05/0146, M02/0021, M06/0004, and M06/0288) each yielded one 1,287-bp amplimer by PCR scheme 2. This amplimer exhibited 100% sequence homology with ccrAB4 from SCC-CI in S. epidermidis ATCC 12228.
One isolate (M04/0269) yielded one 342-bp amplimer by PCR scheme 3 (Table 3). This amplimer exhibited 100% sequence homology with the dcs region.
TABLE 3.
Summary of findings from the present study and from previous studies reporting SCCmec fragments in methicillin-susceptible S. aureus
| No. of MSSA isolates with SCCmec regions (source)a | Method used to detect SCCmec segments (reference) | SCCmec segments found (nb) | MLST/spa genotype (nb) | Further analysis of SCCmec-associated sequences undertaken | Reference |
|---|---|---|---|---|---|
| 7 (four Irish hospitals) | PCR to detect mec complexes A, B, and C (32) | ccrAB2c (2); | ST8/t190 (4) | Present study | |
| PCR to detect ccrAB1 to-4, ccrC (32) | ccrAB4c (6) | ST8/t3209 (1) | Yese | ||
| Multiplex PCR (47) | mecIc (2); dcsc (3) | ST5/t088 (2) | |||
| 2 (i.v. drug users) | PCR to detect ccrAB1 (28) | ccrAB1c | Not done | No | 6 |
| Multiplex PCR (47) | dcsc | ||||
| 26 (regions worldwide) | Real-time PCR (IDI-MRSA assay) to amplify the right SCCmec/orfX junction | Right SCCmec/orfX junctiond | Not done | No | 25 |
| PCR detection of mecA (39) | |||||
| 6 (French hospitals) | PCR detection of ccrAB1, ccrAB2, and ccrAB3 (36) | IS431-pUB110, dcs | ST8/t008 (1) | No | 13 |
| Multiplex PCR (47) | IS431-pUB110-IS431-dcs | ||||
| PCR detection of HVR-IS431-pUB110-IS431-dcs-orfX | |||||
| 1 (nasal swab, HCW) | Real-time PCR (Genotype MRSA Direct) to amplify right SCCmec/orfX junction | Right SCCmec/orfX junctionc | t498 | No | 57 |
| PCR to confirm the right SCCmec/orfX junction (7) | |||||
| PCR detection of mecA (50) | |||||
| PCR to detect ccrAB1, ccrAB2, and ccrAB3 (46) | |||||
| 169 (60 French hospitals) | Real-time PCR (IDI-MRSA assay) to amplify the right SCCmec/orfX junction | Right SCCmec/orfX junction | ST8/t008 (isolate 1) | No | 11 |
| PCR to investigate HVR-IS431-pUB110-IS431-dcs-orfX (13) (2 isolates only investigated) | dcs only (isolate 1) | ST8/t024 (isolate 2) | |||
| PCR to detect ccrAB1, ccrAB2, and ccrAB3 (28) (14 isolates investigated) | HVR-IS431-pUB110-IS431-dcs (isolate 2) |
HCW, health care worker; i.v., intravenous.
n, no. of isolates.
Identity of amplimer(s) confirmed by DNA sequencing.
IDI-MRSA assay amplimer identity was confirmed by conventional endpoint PCR.
A 26-kb SCCmec remnant was sequenced from one ST8/t190 isolate, and ccrAB4 was found to be located 5,549 bp upstream of SCCmec.
Genotyping.
MLST grouped the 25 isolates into nine clonal complexes, with 13/25 (52%) isolates belonging to either CC8 (n = 9) or CC22 (n = 4) (Table 1). The seven GrMSSA isolates that yielded amplimers by SCCmec typing PCR belonged to either ST8 (n = 5) or ST5 (n = 2) (Table 1). Among the isolates that did not yield SCCmec typing amplimers, the majority (14/18) belonged to a range of STs other than ST8 and ST5 (Table 1). Four isolates that did not yield SCCmec amplimers belonged to ST8 but had different spa types (t2658 and t064) from those of the five ST8 isolates that yielded SCCmec/SCC amplimers. The latter exhibited spa types t190 and t3209. BURP analysis assigned the spa types of these nine ST8 isolates into two clusters, one consisting of t190 and t3209 isolates and the other consisting of t2658 and t064 isolates. The remaining 16 isolates investigated fell into three additional spa clusters and five singleton groups. Two isolates exhibited a novel ST and spa type (Table 1). Two isolates with spa types t164 and t1383, respectively, exhibited two different novel MLST types.
Characterization of a novel SCCmec remnant in GrMSSA M06/0075.
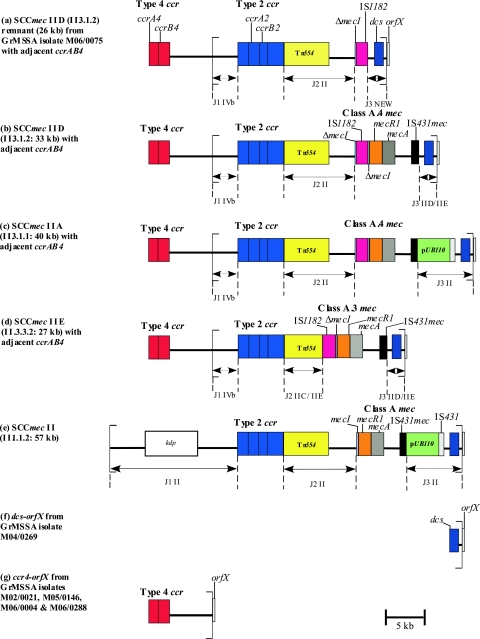
The GrMSSA isolate M06/0075 (genotype ST8/t190) was selected for more-detailed analysis because it was one of the two isolates that yielded the greatest number of amplimers with the SCCmec PCR typing schemes (Table 1). Analysis of the entire nucleotide sequence of the SCC element from the left chromosomal SCC junction to orfX revealed that this GrMSSA isolate harbored a large portion of an SCCmec element consisting of a ca. 26-kb contiguous sequence with a genomic organization similar to that of a variant SCCmec II element, SCCmec IID (II.3.1.2), previously identified among ST8 MRSA isolates from Ireland (59) (Fig. 1).
FIG. 1.
Schematic diagram showing the genetic organization of the novel SCCmec IID (II.3.1.2) remnant from GrMSSA isolate M06/0075 determined in the present study (a) and the corresponding organization of SCCmec elements IID (II.3.1.2) (b), IIA (II.3.1.1) (c) IIE (II.3.3.2), (d), and II (II.1.1.2) (e), determined previously (28, 59). The structure of the SCCmec remnant from M06/0075 was determined by sequencing of the entire element (accession number AM983545) and was found to have closest similarity to SCCmec IID (II.3.1.2). The left and right extremities of the SCCmec elements/remnant are indicated with square brackets. Panels a to d also show the location of ccrAB4 identified in the present study, 5,549 bp upstream of the left chromosomal/SCCmec junction region of the SCCmec IID remnant and SCCmec elements IID (II.3.1.2), IIA (II.3.1.1), and IIE (II.3.3.2), respectively. Panel f shows the location of dcs upstream of orfX, identified in the one GrMSSA isolate (M04/0269) that yielded dcs only by SCCmec typing PCR. Panel g shows the location of ccrAB4 relative to orfX, a structure identified in the four GrMSSA isolates (M02/0021, M05/0146, M06/0004, and M06/0288) that yielded ccrAB4 only by SCCmec typing PCR. The ccrAB4 gene identified in the GrMSSA and MRSA isolates in the present study demonstrated 100% homology with ccrAB4 previously identified in the composite island SCC-CI in S. epidermidis strain ATCC 12228 (41). The DNA sequence between ccrAB4 and the SCCmec IID remnant in M06/0075 and between ccrAB4 and orfX in M02/0021 in each case was 98.5% homologous with the DNA region between ccrAB4 and SCCpbp4 in SCC-CI from S. epidermidis (accession number BK001539, bases 26100 to 24570 and bases 23009 to 18990) except that a ca. 1.5-kb contiguous DNA sequence in this region of SCC-CI (bases 24569 to 23010) was absent in M06/0075 and in M02/0021. This SCC-CI region encodes a spermidine acetyltransferase, a truncated transposase, and the type I restriction proteins HsdS and HsdR, as well as nine other hypothetical proteins (41). All of these open reading frames were also present in the DNA sequences from M06/0075 and M02/0021 referred to above, apart from the partial DNA sequence of one hypothetical protein and the complete sequence of another.
Although SCCmec IID (II.3.1.2) has not been fully sequenced, its genomic organization was determined previously by PCR amplification and comparison to the expected size for amplimers from fully and partially sequenced SCCmec II variant elements SCCmec IIA (II.3.1.1) and IIE (II.3.3.2), respectively (shown in Fig. 1) (59). The variant SCCmec elements IIA to IIE and IVE/IVF were identified and characterized previously by this laboratory (59). SCCmec IID (II.3.1.2) is similar in its genetic organization to SCCmec IIE (II.3.3.2), which has been sequenced in full (59). SCCmec IID (II. 3.1.2) and SCCmec IIE (II.3.3.2) differ in the J2 and mec complex regions, but the J2 and mec complex regions of SCCmec IID (II.3.1.2) are identical to the corresponding regions of SCCmec IIA (II.3.1.1), which have also been sequenced (Fig. 1) (59). Therefore, although the SCCmec remnant of M06/0075 was most similar in genomic organization to SCCmec IID (II.3.1.2), its nucleotide sequence was compared with the nucleotide sequences of the J1, J3, and ccr complex regions of SCCmec IIE (II.3.3.2) and with those of the J2 and mec complex regions of SCCmec IIA (II.3.1.1). The J1 and ccr complex regions of M06/0075 shared 99.5% homology with the corresponding region of SCCmec IIE (II.3.3.2) (Fig. 1). The J2 region shared 99% homology with the J2 region of SCCmec IIA (II.3.1.1). Combining of data from the J1, J2, and ccr complex regions indicated that the SCCmec remnant of M06/0075 most closely resembles a type IID (II.3.1.2) SCCmec element (Fig. 1).
The major difference between SCCmec IID (II.3.1.2) and the SCCmec remnant in M06/0075 was in the mec complex and J3 regions. The mec complex of M06/0075 consisted of IS1182 inserted within the mecI gene near the 3′ end at the exact nucleotide position and resulting in the same 16-bp deletion within mecI as previously identified in the class A.4 mec complex found in SCCmec IIA (II.3.1.1) and SCCmec IID (II.3.1.2) (59). There were five base pair differences between IS1182 from class A.4 mec and M06/0075, including the final base of IS1182, which was absent in M06/0075. In addition, the remainder of the mecI gene (located immediately downstream of IS1182 in class A.4 mec) and the rest of the mec complex, including the mecR1, mecA, and IS431 genes, were absent in M06/0075 (Fig. 1). The region immediately adjacent to IS1182 in M06/0075 shared 99.5% homology with the region extending from ca. 500 bp downstream of IS431 in SCCmec IIE (II.3.3.2) to orfX at the extreme right chromosomal SCCmec junction. This region of both SCCmec IIE (II.3.3.2) and SCCmec IID (II.3.1.2) consists of a noncoding region normally found between IS431 and dcs and the dcs region that has been identified previously in SCCmec I, II, and IV. The orfX gene was identified immediately adjacent to the SCCmec remnant in M06/0075. Furthermore, this SCCmec remnant was integrated at the same nucleotide position within orfX as other SCC/SCCmec elements. The 15-bp direct repeat sequence found at the right extremity of all SCCmec (DR-R) elements identified to date was also identified in M06/0075, as was the 26-bp inverted repeat sequence (IR-R), which had 100% similarity to the IR-R sequences of SCCmec II (II.1.1.2), IIE (II.3.3.2), and IVb (IV.2.1.1). Sequencing of the entire SCCmec remnant from the left chromosomal SCCmec junction to orfX failed to locate the ccrAB4 gene within this element.
Investigation of ST8/t190 MRSA isolates for carriage of ccrAB4 outside SCCmec.
Nosocomial MRSA isolates (n = 23) with the same MLST and spa type as and similar SCCmec sequences to those of GrMSSA isolate M06/0075 were investigated to determine if they also harbored ccrAB4 with 100% homology to ccrAB4 of SCC-CI from S. epidermidis. These included isolates representative of each variant SCCmec II subtype previously identified among ST8/t190 MRSA isolates recovered from Irish hospitals between 1989 and 2002 (59). When these isolates were originally SCCmec typed, amplification of ccrAB4 was not part of any published SCCmec typing scheme (59). All 23 isolates yielded amplimers of the expected size (800 bp) using the primer pair α4.3 and β4.3, which are specific for ccrAB4 from SCC-CI from S. epidermidis (Table 2). For each set of PCR experiments, S. aureus HDE288, which harbors SCCmec ccrAB4 (48), failed to yield any amplimers, whereas template DNA from S. epidermidis ATCC 12228, harboring SCC-CI ccrAB4 (41), yielded the expected 800-bp amplimer, indicating that the primers α4.3 and β4.3 are specific for ccrAB4 from SCC-CI in S. epidermidis. The identities of the amplimers from five MRSA isolates representative of each variant SCCmec subtype IIA to IIE (AR13/0132 [IIA, 3.1.1], AR05/0.1345 [IIB, II.3.2.1], AR14/0246 [IIC, II.3.3.1], AR13/3698 [IID, II.3.1.2], and AR13.1/3330.2 [IIE, II.3.3.2])(59) was confirmed by DNA sequencing and in each case showed 100% homology with ccrAB4 in SCC-CI from S. epidermidis. The genomic organization of the SCCmec elements harbored by these MRSA isolates has been fully determined previously and showed that these isolates do not harbor ccrAB4 within SCCmec (59). These results demonstrate that, like the SCCmec IID (II.3.1.2) remnant in the GrMSSA isolate M06/0075, ccrAB4 is present in these ST8/t190 MRSA isolates with variant SCCmec elements IIA to IIE but it is not located within SCCmec.
Determination of location of ccrAB4.
In total, six GrMSSA isolates harbored ccrAB4 with 100% homology with that of the SCC-CI element from S. epidermidis. These included the two ST8/t190 GrMSSA isolates which also harbored the SCCmec IID remnant and the four GrMSSA isolates (genotypes ST8/t190 [n = 2] and ST5/t088 [n = 2]) that yielded only the ccrAB4 amplimer (Table 1). In addition, the 23 ST8-t190 MRSA isolates with SCCmec IIA to IIE investigated were also found to harbor this ccrAB4 gene. PCR amplification of the region between the left extremity of the SCCmec IID remnant and the ccrAB4 gene in the two GrMSSA isolates M06/0075 and M06/0179 and from the left extremity of SCCmec elements IIA to IIE to ccrAB4 in the five MRSA isolates representative of each Irish variant SCCmec II subtype resulted in each isolate yielding a ca. 7-kb amplimer. DNA sequence analysis of this amplimer from one representative GrMSSA isolate, M06/0075, confirmed that ccrAB4 was located 5,549 bp upstream of the left SCCmec IID remnant junction (Fig. 1). The region between ccrAB4 and the SCCmec IID remnant in M06/0075 exhibited 98.5% DNA sequence homology to the corresponding DNA sequence located between ccrAB4 and SCCpbp4 in SCC-CI from S. epidermidis (accession number BK001539, bases 26100 to 24570 and bases 23009 to 18990), except that a ca. 1.5-kb contiguous DNA sequence in this region of SCC-CI (bases 24569 to 23010) was absent in M06/0075. The region located between ccrAB4 and SCCpbp4 in SCC-CI encodes nine hypothetical proteins, as well as a spermidine acetyltransferase, a truncated transposase, and the type I restriction proteins HsdS and HsdR (41). All of these open reading frames were also present in the DNA sequence identified between ccrAB4 and the SCCmec IID remnant apart from the partial DNA sequence of one hypothetical protein and the complete sequence of another. All direct-repeat and inverted repeat (IR) sequences previously described at the left SCCpbp4/SCC-CI junction region in S. epidermidis (41) were also identified at the junction region of the DNA sequence located between ccrAB4 and the SCCmec IID remnant in M06/0075. The DNA sequence of the SCCmec IID remnant J1 region was found immediately after the SCC-CI and SCCpbp4 IR sequence and was found to be 100% homologous to the DNA sequence that normally lies immediately after the left IR sequence of SCCmec IID. The left IR of SCCmec was absent. Figure 1 shows a schematic representation of the location of ccrAB4 in relation to SCCmec in some of the variant subtypes investigated, including SCCmec IIA, IID, and IIE. PCR amplification of the region between ccrAB4 and orfX in the four GrMSSA isolates that yielded ccrAB4 only by SCCmec typing PCR (Table 1) also yielded a 7-kb amplimer. DNA sequence analysis of the 7-kb amplimer from one representative isolate, M02/0021, revealed that the ccrAB4 gene was located 5,549 bp upstream of orfX (Fig. 1) and that the region between ccrAB4 and orfX was identical to that identified between ccrAB4 and the SCCmec IID remnant in GrMSSA isolates M06/0075.
Determination of location of dcs.
Amplification of the region from dcs to orfX in GrMSSA isolate M04/0269, which yielded dcs only, identified that dcs was located ca. 1.7-kb upstream of orfX (Fig. 1).
Investigation of GrMSSA isolates with SCCmec/SCC-associated DNA using real-time PCR rapid MRSA detection assays.
Three of the seven GrMSSA isolates with SCCmec/SCC DNA yielded positive results by both the BD GeneOhm and Xpert MRSA assays. These included both isolates with ccrAB4 and the SCCmec IID remnant (M06/0075 and M06/0179) and the one isolate with the region from dcs to orfX (M04/0269). The remaining four GrMSSA isolates that harbored the ccrAB4 to orfX region tested negative in both assays.
Clinical details of patients with GrMSSA isolates yielding amplimers by SCCmec typing PCR.
Clinical details of the patients from whom the five genotype ST8 isolates yielding SCCmec amplimers were recovered are as follows: M06/0075 and M06/0179 were recovered within 4 months of each other (M06/0075 was the first of the two isolates to be recovered) from a 31-year-old male with a 9-year history of chronic GrMRSA causing osteomyelitis of the left hip; M05/0146 (ccrAB4 only) was cultured from an 82-year-old female with a fractured femur that was slow to heal; this patient had a 9-month history of GrMRSA; M02/0021 was from a patient for whom no clinical details were available, but GrMRSA was also recovered at the same time as the GrMSSA isolate; M04/0269 came from a 7-year-old female with cystic fibrosis from whom GrMRSA was subsequently isolated (Table 1). AR typing of all isolates except M06/0179 suggested AR type AR13 or AR14 (the inferred genotype of AR13 and AR14 MRSA is ST8-MRSA-II) (59). M06/0179 yielded a pattern that could not be assigned an AR type because it was susceptible to ampicillin.
The two patients yielding ST5 isolates were resident in long-stay care institutions. One patient (M06/0288) had a history of chronic leg ulcers but no known history of MRSA (Table 1). No clinical details are available on the other patient (M06/0004). AR types could not be reliably inferred from the AR pattern obtained with these isolates. None of the remaining GrMSSA isolates from patients with a history of MRSA yielded SCCmec typing amplimers, but in each case, MRSA isolates recovered from these patients were all susceptible to gentamicin (Table 1).
DISCUSSION
A small number of previous studies have identified MSSA isolates harboring segments of SCCmec-associated DNA, but characterization of these segments was limited to DNA sequencing of amplimers obtained by SCCmec typing PCR (e.g., ccrAB1, dcs, and IS431-pUB110) or to amplification of a region usually found downstream of mecA in MRSA (e.g., the SCCmec right-junction region to orfX and/or the hypervariable region usually found immediately downstream of mecA to orfX). Details of previous studies are summarized in Table 3. In the present study, in addition to DNA sequence analysis of the four SCCmec typing PCR amplimers found in 7 of the 25 GrMSSA isolates investigated (28%), the entire SCCmec remnant in 1 GrMSSA isolate was sequenced. This 26-kb SCCmec remnant exhibited very high similarity to the type IID SCCmec element of MRSA, except that regions of the mec complex structure, including part of mecI and the complete mecR1, mecA, and IS431 genes, were absent (Fig. 1). The present study is the first report of such a large SCCmec remnant in MSSA, and although only part of the class A.4 mec complex was identified within the remnant (including truncated mecI and IS1182 genes), this is also the first definitive report of mec complex genes in MSSA.
The GrMSSA isolates with the SCCmec IID remnant (i.e., M06/0075 and M06/0179) appear to be closely related to ST8 MRSA isolates that exhibited variant SCCmec II elements (IIA [II.3.1.1], IIB [II.3.2.1], IIC [II.3.3.1], IID [II.3.1.2], and IIE [II.3.3.2]) that were predominant in the Irish nosocomial MRSA population during the 1990s and continue to constitute part of the MRSA population in Ireland (59). They share the same MLST and spa type (ST8/t190), and similar antibiograms. Comparative sequence analysis of SCCmec IID and the SCCmec IID remnant identified in the present study revealed that they are almost identical except that part of the mec complex, including mecA, and a section of the J3 region are not found in the SCCmec IID remnant (Fig. 1). The patient from whom isolates M06/0075 and M06/0179 were recovered had a 9-year history of MRSA prior to culture of these GrMSSA isolates, suggesting that they may have derived from MRSA by partial loss of SCCmec. Unfortunately, none of the patient's MRSA isolates were available for investigation. Prolonged antibiotic therapy during the patient's protracted osteomyelitis infection, sublethal concentrations of antimicrobials at the site of infection due to poor penetration of antimicrobial agents into bone, and/or continued survival of the isolate in bone may have provided suboptimal growth conditions that favored the partial loss of SCCmec. Other researchers have suggested that MSSA can arise from MRSA both in vivo and in vitro by a spontaneous loss of mecA (8, 9, 11-13, 17, 23, 33, 43). Loss of mecA has been reported to occur under stressful conditions in vitro, such as long-term storage in antibiotic-free medium, nutrient starvation, elevated temperatures, or UV irradiation (17, 27, 49). In addition, Noto et al. have shown that exposure of MRSA isolates to vancomycin in vitro can result in a loss of mecA or portions of SCCmec (43). Daskalaki et al. reported a series of clonally related isolates recovered from a patient with bacteremia and found evidence that a GrMSSA isolate and a gentamicin-susceptible (GsMRSA) isolate were derived from a GrMRSA isolate by deletion of mecA and the gentamicin resistance genes aacA-aphD, respectively (8).
Although genotyping showed that 9 CCs were represented among the 25 GrMSSA isolates investigated in the present study, the 7 isolates that yielded amplimers by SCCmec typing PCR belonged to only 2 CCs, CC8 (n = 5) and CC5 (n = 2). spa typing divided the nine CC8 isolates included in the study into two clusters; all five ST8 isolates yielding SCCmec typing PCR amplimers formed one cluster. All patients from whom these five ST8 isolates were recovered had a history of GrMRSA, and the AR pattern obtained with four of these five isolates suggested AR13 (ST8). None of the remaining 20 patients from whom the remaining 20 GrMSSA isolates were recovered had a history of GrMRSA. The other two GrMSSA isolates that yielded SCCmec amplimers belonged to CC5 (ST5), and both exhibited the same spa type (t088). Both isolates came from patients in long-stay care in geographically remote institutions. Hence, it is unlikely, although possible, that these isolates are epidemiologically related. However, it is interesting to note that the spa type t088 is relatively uncommon in the Ridom SpaServer database (frequency, 0.08%). MRSA with the genotype ST5-MRSA-II have been present in Ireland since 1989 but never predominated (59). Neither of these patients had a history of GrMRSA, but one did have GsMRSA.
In the present study, although three widely used multiplex SCCmec typing PCR schemes indicated that seven GrMSSA isolates harbored SCCmec-associated DNA, subsequent amplimer sequencing showed that only three isolates carried SCCmec-specific DNA (two with the SCCmec IID [II.3.1.2] remnant and one with the SCCmec-specific dcs region amplimer). All three isolates belonged to ST8, and BURP analysis of spa typing results placed all three isolates in the same spa cluster (shown in Table 1). Donnio et al. also reported an ST8 MSSA isolate with the dcs region only, but their isolate had a different spa type (t008 rather than t3209 as in the present study) (Table 1) (11). PCR amplification of the dcs to orfx region of M04/0269 revealed that dcs was located ca. 1.7 kb upstream of orfX. This is the same location as the dcs to orfX region in previously described SCCmec elements (59). Further investigations of isolate M04/0269 are required in order to identify the extent of SCCmec DNA upstream of dcs in this GrMSSA isolate. The remaining four GrMSSA isolates yielded a single amplimer by SCCmec typing PCR, corresponding to ccrAB4. This ccrAB4 allele was also found in the two isolates carrying the SCCmec IID (II.3.1.2) remnant and in the 23 MRSA isolates harboring SCCmec II variant elements. In total, ccrAB4 was identified in two distinct genetic backgrounds, ST8/t190 (MSSA and MRSA) and ST5/t088 (MSSA only). ccrAB4 shared 100% DNA sequence homology with the ccrAB4 gene identified in SCC-CI of S. epidermidis ATCC 12228 but was not located within a SCCmec remnant/element. ccrAB4 homologues have been reported previously in MRSA in SCCmec VI and SCCmecN1, but those ccrAB4 genes share only 91% and 88% similarity, respectively, to ccrAB4 of SCC-CI in S. epidermidis (14, 48).
SCC-CI is a 57-kb non-mecA-containing SCC element inserted into orfX in the genome of S. epidermidis ATCC 12228 (41). Nested within SCC-CI is a 19-kb SCCpbp4 element, which harbors ccrAB2 and genes encoding penicillin binding protein 4 (pbp4) and teichoic acid synthesis (tagF) (41). The rest of the SCC-CI element, which harbors ccrAB4, lies upstream of the left junction of SCCpbp4. In the present study, ccrAB4 was located 5,549 bp upstream of the left extremity of the SCCmec IID remnant/SCCmec element junction in the two GrMSSA isolates with the SCCmec IID remnant and in the same position in the MRSA isolates and 5,549 bp upstream of orfX in the four GrMSSA isolates with ccrAB4 only. This ca. 5.5-kb DNA sequence exhibited 98.5% sequence identity to the region located between ccrAB4 and SCCpbp4 in SCC-CI of S. epidermidis except for the absence of a ca. 1.5-kb region encoding hypothetical proteins. Further investigations are under way to completely characterize the element(s) harboring ccrAB4 in these S. aureus isolates, but data from the present study indicate that ccrAB4 is located on an SCC-CI-like element that lies immediately adjacent to either a SCCmec element/remnant (in MRSA and MSSA) or orfX (in MSSA). Investigations of the DNA region upstream of ccrAB4 in these S. aureus isolates will be of particular interest since this region in SCC-CI harbors genes encoding resistance to cadmium and mercury and also harbors three IS431 elements. Insertion sequence elements have been reported to promote genomic rearrangements (37), and any additional insertion sequence elements that may be present in these S. aureus isolates may have played a role in the deletion and rearrangement of regions of SCC in these isolates.
The identification of DNA in S. aureus with extensive homology to the S. epidermidis SCC-CI element provides further evidence for horizontal transfer of SCCmec/SCC DNA between S. aureus and coagulase-negative staphylococci (CoNS) (19, 41, 65). It has been suggested that the direction of transfer is from CoNS to S. aureus by an as yet unknown mechanism and that CoNS act as a reservoir for SCCmec/SCC DNA (2, 18, 20, 28, 60, 66). However, it is interesting to note that in the present study, the ccrAB4 gene with 100% homology to that found in SCC-CI from S. epidermidis was identified in 29 S. aureus isolates (23 MRSA isolates and 6 MSSA isolates) belonging to two distinct genetic lineages (ST8/t190 [n = 27] and ST5/t088 [n = 2]), but previous studies have identified this ccrAB4 gene in only one S. epidermidis strain (41). While this evidence may suggest that the ccrAB4 gene is more common among or may even have originated in S. aureus, it may merely reflect the fact that the majority of previous studies of SCCmec in S. aureus and CoNS have not involved investigation of ccrAB4 (19, 20). With the incorporation of detection of ccrAB4 into an SCCmec typing scheme (32) and with the description of primers specific for ccrAB4 from SCC-CI, it will be interesting to see the extent to which this ccrAB4 allotype will be detected in various staphylococcal species in future studies. Further work involving S. epidermidis isolates and S. aureus isolates representing the different S. aureus genetic backgrounds from Ireland and other countries is required to determine the prevalence, significance, and origin of ccrAB4 among S. aureus and S. epidermidis isolates.
Donnio et al. (11) investigated 247 multiresistant MSSA isolates from 60 French hospitals using the IDI-MRSA real-time PCR assay to target the right junction region between SCCmec and orfX and found that 169/247 isolates (68%) were positive, suggesting carriage of portions of SCCmec. These isolates belonged to more than 30 spa types, suggesting that partial loss of SCCmec was not restricted to specific S. aureus lineages, although isolates with the genotypes ST8 and ST5 predominated. In the present study, SCCmec/SCC-associated DNA was detected in only 28% of isolates but these isolates also exhibited either the ST8 or ST5 genotypes (Table 1). It is important to highlight the fact that although Donnio et al. (11) used PCR to detect the ccr genes (ccrAB1, ccrAB2, ccrAB3, and ccrC), only 2 of the 169 IDI-MRSA assay-positive isolates were investigated further by PCR amplification of the region downstream of mecA to confirm the presence of SCCmec DNA sequences. It has been reported that the IDI-MRSA assay is not specific for SCCmec but also detects SCC sequences (10). Interestingly, a study of a genetically diverse collection of 42 MSSA isolates mainly from patients with bacteremia or skin infections or from healthy carriers reported that no isolate was found to harbor the ccrAB, ccrC, or mecA fragments when investigated by PCR and Southern hybridization analysis (44).
The presence of SCC- and SCCmec-associated DNA in MSSA has practical implications for the rapid detection of MRSA from clinical specimens and for tracking and genotyping of MRSA by SCCmec typing. There are now a number of commercially available assays that use real-time PCR to detect SCC-associated sequences directly from clinical specimens for the rapid detection of MRSA. They include the BD GeneOhm MRSA assay (10, 25, 51) and Cepheid's Xpert MRSA assay (52). During development of the IDI-MRSA assay, the forerunner of the BD GeneOhm MRSA assay, which was the first FDA-approved commercial system, Huletsky et al. found that 4.6% (26/569) of MSSA isolates investigated were positive, probably due to the presence of either SCCmec or SCC sequences (25). Another study using the IDI-MRSA assay to detect MRSA from broth enrichment culture found that 26% (77/298) of positive specimens carried MSSA (10). In the present study, the three GrMSSA isolates with SCCmec-specific DNA tested positive with both the BD GeneOhm and Xpert MRSA assays. Interestingly, Donnio et al. found that one of the MSSA isolates detected by the IDI-MRSA assay also harbored the SCCmec dcs region only (11). In contrast, the four GrMSSA isolates with the SCC-CI-associated ccrAB4 gene tested negative with both assays. These findings suggest that kit-positive MRSA culture-negative specimens should not be dismissed lightly as false-positive kit results without taking the patient's prior MRSA history into account. The clinical significance of such positive results in patients with a history of MRSA is difficult to interpret.
SCCmec typing together with MLST is recommended for comparison of MRSA strains and is commonly used for routine epidemiological typing of MRSA (5). The presence of a second ccr gene (ccrAB4) outside of SCCmec in MRSA isolates could result in the misinterpretation of typing results for isolates carrying ccrAB4 in addition to another ccr allotype within SCCmec, as was found in the present study. When this occurs, it is important to determine which ccr gene type is part of the SCCmec element and which is located outside of SCCmec.
In conclusion, this study has found that 28% of GrMSSA isolates investigated carry remnants of SCC or SCCmec and that some of these GrMSSA isolates may have been derived from a previously predominant MRSA clone in Irish hospitals. The study provides the first detailed description of a 26-kb SCCmec remnant with ccr and partial mec complex genes in an MSSA isolate. It is also the first report of S. epidermidis SCC-CI DNA, including SCC-CI ccrAB4 genes, in MRSA and MSSA isolates, with a location outside of, but immediately adjacent to, SCCmec or orfX. The presence of SCCmec-associated DNA in MSSA should be considered when real-time PCR assays are used for the rapid detection of MRSA from clinical specimens. In addition, MRSA isolates with additional ccr genes located outside of SCCmec may pose problems for the interpretation of SCCmec typing results.
Acknowledgments
S. aureus control isolates were kindly provided by Teruyo Ito, Juntendo University, Japan, Herminia de Lencastre, Rockefeller University, New York, and Robert Daum, University of Chicago,. We thank the staff of the National MRSA Reference Laboratory, St. James's Hospital, Dublin, Ireland.
This work was supported by the Microbiology Research Unit, Dublin Dental School & Hospital, and by Irish Health Research Board grant TRA/2006/4.
Footnotes
Published ahead of print on 13 October 2008.
REFERENCES
- 1.Abbott, Y., N. Leonard, B. Markey, A. Rossney, and J. Dunne. 2007. Persistence of MRSA infection. Vet. Rec. 160:851-852. [DOI] [PubMed] [Google Scholar]
- 2.Archer, G. L., J. A. Thanassi, D. M. Niemeyer, and M. J. Pucci. 1996. Characterization of IS1272, an insertion sequence-like element from Staphylococcus haemolyticus. Antimicrob. Agents Chemother. 40:924-929. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Carroll, D., M. A. Kehoe, D. Cavanagh, and D. C. Coleman. 1995. Novel organization of the site-specific integration and excision recombination functions of the Staphylococcus aureus serotype F virulence-converting phages phi 13 and phi 42. Mol. Microbiol. 16:877-893. [DOI] [PubMed] [Google Scholar]
- 4.Collery, M. M., D. S. Smyth, J. M. Twohig, A. C. Shore, D. C. Coleman, and C. J. Smyth. 2008. Molecular typing of nasal carriage isolates of Staphylococcus aureus from an Irish university student population based on toxin gene PCR, agr locus types and multiple locus, variable number tandem repeat analysis. J. Med. Microbiol. 57:348-358. [DOI] [PubMed] [Google Scholar]
- 5.Cookson, B. D., D. A. Robinson, A. B. Monk, S. Murchan, A. Deplano, R. de Ryck, M. J. Struelens, C. Scheel, V. Fussing, S. Salmenlinna, J. Vuopio-Varkila, C. Cuny, W. Witte, P. T. Tassios, N. J. Legakis, W. van Leeuwen, A. van Belkum, A. Vindel, J. Garaizar, S. Haeggman, B. Olsson-Liljequist, U. Ransjo, M. Muller-Premru, W. Hryniewicz, A. Rossney, B. O'Connell, B. D. Short, J. Thomas, S. O'Hanlon, and M. C. Enright. 2007. Evaluation of molecular typing methods in characterizing a European collection of epidemic methicillin-resistant Staphylococcus aureus strains: the HARMONY collection. J. Clin. Microbiol. 45:1830-1837. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Corkill, J. E., J. J. Anson, P. Griffiths, and C. A. Hart. 2004. Detection of elements of the staphylococcal cassette chromosome (SCC) in a methicillin-susceptible (mecA gene negative) homologue of a fucidin-resistant MRSA. J. Antimicrob. Chemother. 54:229-231. [DOI] [PubMed] [Google Scholar]
- 7.Cuny, C., and W. Witte. 2005. PCR for the identification of methicillin-resistant Staphylococcus aureus (MRSA) strains using a single primer pair specific for SCCmec elements and the neighbouring chromosome-borne orfX. Clin. Microbiol. Infect. 11:834-837. [DOI] [PubMed] [Google Scholar]
- 8.Daskalaki, M., J. R. Otero, F. Sanz, and F. Chaves. 2007. Bacteremia due to clonally derived methicillin-resistant, gentamicin-susceptible isolates and methicillin-susceptible, gentamicin-resistant isolates of Staphylococcus aureus. J. Clin. Microbiol. 45:3446-3448. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Deplano, A., P. T. Tassios, Y. Glupczynski, E. Godfroid, and M. J. Struelens. 2000. In vivo deletion of the methicillin resistance mec region from the chromosome of Staphylococcus aureus strains. J. Antimicrob. Chemother. 46:617-620. [DOI] [PubMed] [Google Scholar]
- 10.Desjardins, M., C. Guibord, B. Lalonde, B. Toye, and K. Ramotar. 2006. Evaluation of the IDI-MRSA assay for detection of methicillin-resistant Staphylococcus aureus from nasal and rectal specimens pooled in a selective broth. J. Clin. Microbiol. 44:1219-1223. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Donnio, P. Y., F. Fevrier, P. Bifani, M. Dehem, C. Kervegant, N. Wilhelm, A. L. Gautier-Lerestif, N. Lafforgue, M. Cormier, and A. Le Coustumier. 2007. Molecular and epidemiological evidence for spread of multiresistant methicillin-susceptible Staphylococcus aureus strains in hospitals. Antimicrob. Agents Chemother. 51:4342-4350. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Donnio, P. Y., L. Louvet, L. Preney, D. Nicolas, J. L. Avril, and L. Desbordes. 2002. Nine-year surveillance of methicillin-resistant Staphylococcus aureus in a hospital suggests instability of mecA DNA region in an epidemic strain. J. Clin. Microbiol. 40:1048-1052. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Donnio, P. Y., D. C. Oliveira, N. A. Faria, N. Wilhelm, A. Le Coustumier, and H. de Lencastre. 2005. Partial excision of the chromosomal cassette containing the methicillin resistance determinant results in methicillin-susceptible Staphylococcus aureus. J. Clin. Microbiol. 43:4191-4193. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Ender, M., B. Berger-Bachi, and N. McCallum. 2007. Variability in SCCmecN1 spreading among injection drug users in Zurich, Switzerland. BMC Microbiol. 7:62. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Enright, M. C., N. P. Day, C. E. Davies, S. J. Peacock, and B. G. Spratt. 2000. Multilocus sequence typing for characterization of methicillin-resistant and methicillin-susceptible clones of Staphylococcus aureus. J. Clin. Microbiol. 38:1008-1015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Feil, E. J., B. C. Li, D. M. Aanensen, W. P. Hanage, and B. G. Spratt. 2004. eBURST: inferring patterns of evolutionary descent among clusters of related bacterial genotypes from multilocus sequence typing data. J. Bacteriol. 186:1518-1530. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Grubb, W. B., and D. I. Annear. 1972. Spontaneous loss of methicillin resistance in Staphylococcus aureus at room-temperature. Lancet ii:1257. [DOI] [PubMed] [Google Scholar]
- 18.Hanssen, A. M., and J. U. Ericson Sollid. 2006. SCCmec in staphylococci: genes on the move. FEMS Immunol. Med. Microbiol. 46:8-20. [DOI] [PubMed] [Google Scholar]
- 19.Hanssen, A. M., G. Kjeldsen, and J. U. Sollid. 2004. Local variants of staphylococcal cassette chromosome mec in sporadic methicillin-resistant Staphylococcus aureus and methicillin-resistant coagulase-negative staphylococci: evidence of horizontal gene transfer? Antimicrob. Agents Chemother. 48:285-296. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Hanssen, A. M., and J. U. Sollid. 2007. Multiple staphylococcal cassette chromosomes and allelic variants of cassette chromosome recombinases in Staphylococcus aureus and coagulase-negative staphylococci from Norway. Antimicrob. Agents Chemother. 51:1671-1677. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Heusser, R., M. Ender, B. Berger-Bachi, and N. McCallum. 2007. Mosaic staphylococcal cassette chromosome mec containing two recombinase loci and a new mec complex, B2. Antimicrob. Agents Chemother. 51:390-393. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Hiramatsu, K., L. Cui, M. Kuroda, and T. Ito. 2001. The emergence and evolution of methicillin-resistant Staphylococcus aureus. Trends Microbiol. 9:486-493. [DOI] [PubMed] [Google Scholar]
- 23.Hiramatsu, K., E. Suzuki, H. Takayama, Y. Katayama, and T. Yokota. 1990. Role of penicillinase plasmids in the stability of the mecA gene in methicillin-resistant Staphylococcus aureus. Antimicrob. Agents Chemother. 34:600-604. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Holtfreter, S., D. Grumann, M. Schmudde, H. T. Nguyen, P. Eichler, B. Strommenger, K. Kopron, J. Kolata, S. Giedrys-Kalemba, I. Steinmetz, W. Witte, and B. M. Broker. 2007. Clonal distribution of superantigen genes in clinical Staphylococcus aureus isolates. J. Clin. Microbiol. 45:2669-2680. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Huletsky, A., R. Giroux, V. Rossbach, M. Gagnon, M. Vaillancourt, M. Bernier, F. Gagnon, K. Truchon, M. Bastien, F. J. Picard, A. van Belkum, M. Ouellette, P. H. Roy, and M. G. Bergeron. 2004. New real-time PCR assay for rapid detection of methicillin-resistant Staphylococcus aureus directly from specimens containing a mixture of staphylococci. J. Clin. Microbiol. 42:1875-1884. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Humphreys, H., C. T. Keane, R. Hone, H. Pomeroy, R. J. Russell, J. P. Arbuthnott, and D. C. Coleman. 1989. Enterotoxin production by Staphylococcus aureus isolates from cases of septicaemia and from healthy carriers. J. Med. Microbiol. 28:163-172. [DOI] [PubMed] [Google Scholar]
- 27.Inglis, B., P. R. Matthews, and P. R. Stewart. 1990. Induced deletions within a cluster of resistance genes in the mec region of the chromosome of Staphylococcus aureus. J. Gen. Microbiol. 136:2231-2239. [DOI] [PubMed] [Google Scholar]
- 28.Ito, T., Y. Katayama, K. Asada, N. Mori, K. Tsutsumimoto, C. Tiensasitorn, and K. Hiramatsu. 2001. Structural comparison of three types of staphylococcal cassette chromosome mec integrated in the chromosome in methicillin-resistant Staphylococcus aureus. Antimicrob. Agents Chemother. 45:1323-1336. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Ito, T., X. X. Ma, F. Takeuchi, K. Okuma, H. Yuzawa, and K. Hiramatsu. 2004. Novel type V staphylococcal cassette chromosome mec driven by a novel cassette chromosome recombinase, ccrC. Antimicrob. Agents Chemother. 48:2637-2651. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Ito, T., K. Okuma, X. X. Ma, H. Yuzawa, and K. Hiramatsu. 2003. Insights on antibiotic resistance of Staphylococcus aureus from its whole genome: genomic island SCC. Drug Resist. Updat. 6:41-52. [DOI] [PubMed] [Google Scholar]
- 31.Katayama, Y., F. Takeuchi, T. Ito, X. X. Ma, Y. Ui-Mizutani, I. Kobayashi, and K. Hiramatsu. 2003. Identification in methicillin-susceptible Staphylococcus hominis of an active primordial mobile genetic element for the staphylococcal cassette chromosome mec of methicillin-resistant Staphylococcus aureus. J. Bacteriol. 185:2711-2722. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Kondo, Y., T. Ito, X. X. Ma, S. Watanabe, B. N. Kreiswirth, J. Etienne, and K. Hiramatsu. 2007. Combination of multiplex PCRs for staphylococcal cassette chromosome mec type assignment: rapid identification system for mec, ccr, and major differences in junkyard regions. Antimicrob. Agents Chemother. 51:264-274. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Lawrence, C., M. Cosseron, P. Durand, Y. Costa, and R. Leclercq. 1996. Consecutive isolation of homologous strains of methicillin-resistant and methicillin-susceptible Staphylococcus aureus from a hospitalized child. J. Hosp. Infect. 33:49-53. [DOI] [PubMed] [Google Scholar]
- 34.Lina, G., G. Durand, C. Berchich, B. Short, H. Meugnier, F. Vandenesch, J. Etienne, and M. C. Enright. 2006. Staphylococcal chromosome cassette evolution in Staphylococcus aureus inferred from ccr gene complex sequence typing analysis. Clin. Microbiol. Infect. 12:1175-1184. [DOI] [PubMed] [Google Scholar]
- 35.Luong, T. T., S. Ouyang, K. Bush, and C. Y. Lee. 2002. Type 1 capsule genes of Staphylococcus aureus are carried in a staphylococcal cassette chromosome genetic element. J. Bacteriol. 184:3623-3629. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Ma, X. X., T. Ito, C. Tiensasitorn, M. Jamklang, P. Chongtrakool, S. Boyle-Vavra, R. S. Daum, and K. Hiramatsu. 2002. Novel type of staphylococcal cassette chromosome mec identified in community-acquired methicillin-resistant Staphylococcus aureus strains. Antimicrob. Agents Chemother. 46:1147-1152. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Mahillon, J., C. Leonard, and M. Chandler. 1999. IS elements as constituents of bacterial genomes. Res. Microbiol. 150:675-687. [DOI] [PubMed] [Google Scholar]
- 38.Maiques, E., C. Ubeda, M. A. Tormo, M. D. Ferrer, I. Lasa, R. P. Novick, and J. R. Penades. 2007. Role of staphylococcal phage and SaPI integrase in intra- and interspecies SaPI transfer. J. Bacteriol. 189:5608-5616. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Martineau, F., F. J. Picard, N. Lansac, C. Menard, P. H. Roy, M. Ouellette, and M. G. Bergeron. 2000. Correlation between the resistance genotype determined by multiplex PCR assays and the antibiotic susceptibility patterns of Staphylococcus aureus and Staphylococcus epidermidis. Antimicrob. Agents Chemother. 44:231-238. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Mellmann, A., T. Weniger, C. Berssenbrugge, J. Rothganger, M. Sammeth, J. Stoye, and D. Harmsen. 2007. Based Upon Repeat Pattern (BURP): an algorithm to characterize the long-term evolution of Staphylococcus aureus populations based on spa polymorphisms. BMC Microbiol. 7:98. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Mongkolrattanothai, K., S. Boyle, T. V. Murphy, and R. S. Daum. 2004. Novel non-mecA-containing staphylococcal chromosomal cassette composite island containing pbp4 and tagF genes in a commensal staphylococcal species: a possible reservoir for antibiotic resistance islands in Staphylococcus aureus. Antimicrob. Agents Chemother. 48:1823-1836. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Murphy, O. M., S. Murchan, D. Whyte, H. Humphreys, A. Rossney, P. Clarke, R. Cunney, C. Keane, L. E. Fenelon, and D. O'Flanagan. 2005. Impact of the European Antimicrobial Resistance Surveillance System on the development of a national programme to monitor resistance in Staphylococcus aureus and Streptococcus pneumoniae in Ireland, 1999-2003. Eur. J. Clin. Microbiol. Infect. Dis. 24:480-483. [DOI] [PubMed] [Google Scholar]
- 43.Noto, M. J., P. M. Fox, and G. L. Archer. 2008. Spontaneous deletion of the methicillin resistance determinant, mecA, partially compensates for the fitness cost associated with high-level vancomycin resistance in Staphylococcus aureus. Antimicrob. Agents Chemother. 52:1221-1229. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Noto, M. J., B. N. Kreiswirth, A. B. Monk, and G. L. Archer. 2008. Gene acquisition at the insertion site for SCCmec, the genomic island conferring methicillin resistance in Staphylococcus aureus. J. Bacteriol. 190:1276-1283. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Novick, R. P., and A. Subedi. 2007. The SaPIs: mobile pathogenicity islands of Staphylococcus. Chem. Immunol. Allergy 93:42-57. [DOI] [PubMed] [Google Scholar]
- 46.Okuma, K., K. Iwakawa, J. D. Turnidge, W. B. Grubb, J. M. Bell, F. G. O'Brien, G. W. Coombs, J. W. Pearman, F. C. Tenover, M. Kapi, C. Tiensasitorn, T. Ito, and K. Hiramatsu. 2002. Dissemination of new methicillin-resistant Staphylococcus aureus clones in the community. J. Clin. Microbiol. 40:4289-4294. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Oliveira, D. C., and H. de Lencastre. 2002. Multiplex PCR strategy for rapid identification of structural types and variants of the mec element in methicillin-resistant Staphylococcus aureus. Antimicrob. Agents Chemother. 46:2155-2161. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Oliveira, D. C., C. Milheirico, and H. de Lencastre. 2006. Redefining a structural variant of staphylococcal cassette chromosome mec, SCCmec type VI. Antimicrob. Agents Chemother. 50:3457-3459. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Poston, S. M., and F. L. Li Saw Hee. 1991. Genetic characterisation of resistance to metal ions in methicillin-resistant Staphylococcus aureus: elimination of resistance to cadmium, mercury and tetracycline with loss of methicillin resistance. J. Med. Microbiol. 34:193-201. [DOI] [PubMed] [Google Scholar]
- 50.Reischl, U., H. J. Linde, M. Metz, B. Leppmeier, and N. Lehn. 2000. Rapid identification of methicillin-resistant Staphylococcus aureus and simultaneous species confirmation using real-time fluorescence PCR. J. Clin. Microbiol. 38:2429-2433. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Rossney, A. S., C. M. Herra, M. M. Fitzgibbon, P. M. Morgan, M. J. Lawrence, and B. O'Connell. 2007. Evaluation of the IDI-MRSA assay on the SmartCycler real-time PCR platform for rapid detection of MRSA from screening specimens. Eur. J. Clin. Microbiol. Infect. Dis. 26:459-466. [DOI] [PubMed] [Google Scholar]
- 52.Rossney, A. S., C. M. H. Herra, G. I. Brennan, P. M. Morgan, and B. O'Connell. 2008. Evaluation of the Xpert methicillin-resistant Staphylococcus aureus (MRSA) assay using the GeneXpert real-time PCR platform for rapid detection of MRSA from screening specimens. J. Clin. Microbiol. 46:3285-3290. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Rossney, A. S., and C. T. Keane. 2002. Strain variation in the MRSA population over a 10-year period in one Dublin hospital. Eur. J. Clin. Microbiol. Infect. Dis. 21:123-126. [DOI] [PubMed] [Google Scholar]
- 54.Rossney, A. S., M. J. Lawrence, P. M. Morgan, M. M. Fitzgibbon, A. Shore, D. C. Coleman, C. T. Keane, and B. O'Connell. 2006. Epidemiological typing of MRSA isolates from blood cultures taken in Irish hospitals participating in the European Antimicrobial Resistance Surveillance System (1999-2003). Eur. J. Clin. Microbiol. Infect. Dis. 25:79-89. [DOI] [PubMed] [Google Scholar]
- 55.Rossney, A. S., P. McDonald, H. Humphreys, G. M. Glynn, and C. T. Keane. 2003. Antimicrobial resistance and epidemiological typing of methicillin-resistant Staphylococcus aureus in Ireland (North and South), 1999. Eur. J. Clin. Microbiol. Infect. Dis. 22:379-381. [DOI] [PubMed] [Google Scholar]
- 56.Rossney, A. S., A. C. Shore, P. M. Morgan, M. M. Fitzgibbon, B. O'Connell, and D. C. Coleman. 2007. The emergence and importation of diverse genotypes of methicillin-resistant Staphylococcus aureus (MRSA) harboring the Panton-Valentine leukocidin gene (pvl) reveal that pvl is a poor marker for community-acquired MRSA strains in Ireland. J. Clin. Microbiol. 45:2554-2563. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Rupp, J., I. Fenner, W. Solbach, and J. Gieffers. 2006. Be aware of the possibility of false-positive results in single-locus PCR assays for methicillin-resistant Staphylococcus aureus. J. Clin. Microbiol. 44:2317. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Sheftel, T. G., J. T. Mader, J. J. Pennick, and G. Cierny III. 1985. Methicillin-resistant Staphylococcus aureus osteomyelitis. Clin. Orthop. Relat. Res. 1985:231-239. [PubMed] [Google Scholar]
- 59.Shore, A., A. S. Rossney, C. T. Keane, M. C. Enright, and D. C. Coleman. 2005. Seven novel variants of the staphylococcal chromosomal cassette mec in methicillin-resistant Staphylococcus aureus isolates from Ireland. Antimicrob. Agents Chemother. 49:2070-2083. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Suzuki, E., K. Kuwahara-Arai, J. F. Richardson, and K. Hiramatsu. 1993. Distribution of mec regulator genes in methicillin-resistant Staphylococcus clinical strains. Antimicrob. Agents Chemother. 37:1219-1226. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Takano, T., W. Higuchi, T. Otsuka, T. Baranovich, S. Enany, K. Saito, H. Isobe, S. Dohmae, K. Ozaki, M. Takano, Y. Iwao, M. Shibuya, T. Okubo, S. Yabe, D. Shi, I. Reva, L. J. Teng, and T. Yamamoto. 2008. Novel characteristics of community-acquired methicillin-resistant Staphylococcus aureus strains belonging to multilocus sequence type 59 in Taiwan. Antimicrob. Agents Chemother. 52:837-845. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Takeuchi, F., S. Watanabe, T. Baba, H. Yuzawa, T. Ito, Y. Morimoto, M. Kuroda, L. Cui, M. Takahashi, A. Ankai, S. Baba, S. Fukui, J. C. Lee, and K. Hiramatsu. 2005. Whole-genome sequencing of Staphylococcus haemolyticus uncovers the extreme plasticity of its genome and the evolution of human-colonizing staphylococcal species. J. Bacteriol. 187:7292-7308. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Tallent, S. M., T. B. Langston, R. G. Moran, and G. E. Christie. 2007. Transducing particles of Staphylococcus aureus pathogenicity island SaPI1 are comprised of helper phage-encoded proteins. J. Bacteriol. 189:7520-7524. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Ubeda, C., P. Barry, J. R. Penades, and R. P. Novick. 2007. A pathogenicity island replicon in Staphylococcus aureus replicates as an unstable plasmid. Proc. Natl. Acad. Sci. USA 104:14182-14188. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Wisplinghoff, H., A. E. Rosato, M. C. Enright, M. Noto, W. Craig, and G. L. Archer. 2003. Related clones containing SCCmec type IV predominate among clinically significant Staphylococcus epidermidis isolates. Antimicrob. Agents Chemother. 47:3574-3579. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Wu, S., H. de Lencastre, and A. Tomasz. 1998. Genetic organization of the mecA region in methicillin-susceptible and methicillin-resistant strains of Staphylococcus sciuri. J. Bacteriol. 180:236-242. [DOI] [PMC free article] [PubMed] [Google Scholar]