Abstract
Turkey coronavirus (TCoV) causes diarrhea in young turkey poults, but little is known about its prevalence in the field. To address this, the complete nucleocapsid gene was cloned and expressed in Escherichia coli. Expressed nucleocapsid gene produced two distinct proteins (52 and 43 kDa); their specificity was confirmed by Western blotting using two different monoclonal antibodies. Recombinant N protein was purified and used as an antigen to develop an enzyme-linked immunosorbent assay (ELISA) for the serological detection of TCoV that was then validated using experimentally derived turkey serum. The N-based ELISA showed (97%) sensitivity and (93%) specificity for TCoV, which was significantly higher than an infectious bronchitis coronavirus-based commercial test for TCoV. To assess the utility of this recombinant ELISA, 360 serum samples from turkey farms in Ontario, Canada, and 81 serum samples from farms in Arkansas were tested for TCoV-specific antibodies. A high seroprevalence of TCoV was found in turkeys from the Ontario farms with 73.9% of breeders and 60.0% of meat turkeys testing seropositive using the N-based ELISA. Similarly, a high field prevalence was found in the turkeys from Arkansas, for which 64.2% of the serum samples tested seropositive.
Turkey coronavirus (TCoV) is a group III CoV closely related to infectious bronchitis virus (IBV) of chickens (10). TCoV is one of the most important causative agents of diarrhea in turkey poults. The infection of immunologically naïve poults with TCoV produces diarrhea and, as a consequence, negatively impacts their growth rates, causing significant economic losses. Although TCoV was identified as the causative agent of Bluecomb disease of turkey poults over 50 years ago (28), vaccines are not available to control the disease.
CoVs possess four major structural proteins: spike (S) glycoprotein, which consists of two subunits (S1 and S2); membrane (M) protein; small envelope (E) protein; and nucleocapsid (N) protein. The N protein is highly conserved among group III CoVs, and different strains of IBV and TCoV share 97% or greater identity at the amino acid sequence level (10). During IBV infections in chickens, the IBV N protein is expressed at high levels and produces antibodies that react with a variety of IBV serotypes (25). The B-cell epitopes have been mapped in the carboxy-terminal region of the N protein (3, 26). The N protein is also involved in the cell-mediated immunity and protection of chickens from IBV infection. As with IBV (19), enzyme-linked immunosorbent assay (ELISA) tests have been developed using recombinant N protein for various viruses such as measles virus (16), vesicular stomatitis virus (1), Newcastle disease virus (8) and severe acute respiratory syndrome CoV (24).
Recently, the full-length genomic sequence of TCoV was completed (10) and provided the genetic information necessary to produce recombinant antigen for the development of diagnostic tests for TCoV in turkeys. The objective of this study was to develop and validate an ELISA for TCoV based on N recombinant antigen. This ELISA was compared with a commercial whole IBV-based ELISA for their ability to determine the seroprevalence of TCoV in breeder and meat turkey flocks in Ontario, Canada, and Arkansas.
MATERIALS AND METHODS
Virus.
TCoV was isolated from an Ontario turkey suffering from acute enteritis and diarrhea. The intestines from affected birds were homogenated in phosphate-buffered saline (PBS) and then clarified by centrifugation at 4,000 × g for 15 min. The supernatant was filtered through a 0.22-μm-membrane filter (Millipore, Bedford, MA). This isolate was named TCoV-MG10 (10) and used for this study.
Experimental turkeys.
Day-old turkey poults were obtained from a commercial turkey breeder (Hybrid Turkeys; Kitchener, Ontario, Canada). The birds were housed in the campus animal facility's isolation unit of the University of Guelph where they were fed and watered ad libitum. All animals were housed and handled in accordance with guidelines established by the Canadian Council on Animal Care and the animal care committee of the University of Guelph.
Control positive and negative turkey serum samples.
Forty day-old birds were separated into two groups, 20 birds per group, and birds in the same group were maintained in the same rooms. TCoV-MG10 was inoculated orally into 20 birds, while 20 were kept as a negative control. Serum samples were obtained from both groups of birds at days 7, 14, 20, 35, and 42. All sera were heat-inactivated at 56°C for 30 min before use. Fifteen specific-pathogen-free (SPF) turkey serum samples were kindly provided by Billy Hargis (University of Arkansas, Fayetteville, AR), and they were used as additional negative controls.
Cloning of the TCoV N-protein genes.
The total RNA was extracted from the intestinal tissue homogenates of infected turkey poults using the Qiagen RNeasy kit (Qiagen, Valencia, CA). The N gene of TCoV-MG10 was amplified using NBEF (5′-AAAGGATCCATGGCAAGCGGTAAGGCAAC-3′) and NBER (5′-AAAGAATTCCTCTACAACTCATTCTCTC-3′) primers containing restriction sites (underlined) for BamHI and EcoRI, respectively. Reverse transcriptase PCR was performed as previously described (10). The PCR product was purified using the Mini Elute purification kit (Qiagen, Valencia, CA) and double-digested with BamHI and EcoRI (New England BioLabs, Ipswich, MA). PGEX-4T3 (Amersham Pharmacia Biotech, Sweden) was digested by both BamHI and EcoRI and dephosphorylated using shrimp alkaline phosphatase (AP; USB Corporation, Cleveland, OH) prior to purification with the Mini Elute purification kit. The ligation of the digested product with linearized pGEX-4T3 was performed using T4 DNA ligase (Invitrogen). Escherichia coli DH5α competent cells were transformed with the ligation mix and plated onto LB agar plates containing ampicillin. Transformants were screened by digestion with BamHI and EcoRI, and clones containing inserts were checked for orientation by sequencing. For protein expression, E. coli strain BL-21 was used.
Expression and purification of N-glutathione S-transferase (GST) fusion protein.
Escherichia coli BL-21 cells containing pGEX-TCoV-N were propagated in LB broth containing ampicillin (0.1 mg/ml) overnight at 37°C with vigorous shaking. One milliliter of overnight culture was inoculated into 100 ml of fresh LB with ampicillin (0.1 mg/ml). When the optical density at 600 nm (OD600) reached 0.6, the culture was induced by adding isopropyl-β-d-thiogalactopyranoside (IPTG) to a final concentration of 1 mM. The culture was further incubated for an additional 4 h at 30°C with shaking. The bacteria were harvested by centrifugation at 6,000 rpm for 15 min at 4°C (J2-MC centrifuge; Beckman, Coulter, Mississauga, Ontario, Canada). The supernatant was decanted, and the pellet was resuspended in 5 ml of PBS.
Purification of the GST fusion protein was conducted by adding 0.5 ml lysozyme solution (12 mg of lysozyme in 50 mM glucose, 10 mM EDTA, 25 mM Tris HCl [pH 8.0]) containing 1 mM phenylmethylsulfonyl fluoride. The suspension was sonicated three times for 2 s on ice. The addition of 250 μl of 20% Triton X-100 preceded incubation for 30 min at 4°C with gentle agitation, followed by centrifugation at 10,000 rpm for 10 min at 4°C. The supernatant was collected, and 100 μl of the 50% slurry glutathione Sepharose was added to the supernatant (GE Health Care, Buckinghamshire, United Kingdom). The suspension was clarified by washing three times with PBS and centrifuged to pellet the Sepharose beads. Cold, reduced glutathione was used to elute the bound protein, and the eluate was collected from the supernatant after centrifugation of the suspension. Purified recombinant proteins were visualized using 12% sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) followed by staining with Coomassie blue G-250 (Bio-Rad, Hercules, CA). The purified protein was separated from the GST fusion partner by thrombin protease using RediPack columns according to the manufacturer's instructions (GE Health Care).
Western blotting of the TCoV N recombinant protein.
The purified TCoV N protein was separated by 12% SDS-PAGE and transferred onto a nitrocellulose membrane (0.45 μm; Bio-Rad) using the Bio-Rad Mini Trans-Blot cell. The membrane was washed three times in Tween-20 Tris-buffered saline (TTBS) and then blocked with 5% bovine serum albumin (Sigma Co., St. Louis, MO) in TTBS overnight at 4°C with gentle agitation. The membrane was then incubated with goat anti-GST antibody (GE Health Care) for 2 h at 4°C, followed by incubation with rabbit anti-goat immunoglobulin G (IgG) conjugated with AP (Jackson Immune Research Laboratories, Inc., West Grove, PA) for 2 h at 4°C. The membrane was washed three times with TTBS, and the color was developed according to the manufacturer's instructions (Bio-Rad). For the purified TCoV N protein, the membrane was incubated with one of two murine monoclonal antibodies (MAbs 1.01 and 4.24) raised against TCoV-NC95, followed by incubation with AP-conjugated goat anti-mouse IgG (H+L) (Cedarlane Laboratories Ltd., Hornsby, Ontario, Canada).
ELISA.
A checkerboard titration was conducted using different coating antigen concentrations (12.5, 25, 50, 100, and 200 ng/μl). To determine the optimal test serum concentrations to use, dilutions of 1:50, 1:100, 1:250, 1:500, and 1:1,000 were tested in combination with a secondary antibody diluted at 1:1,000, 1:2,000, 1:5,000, and 1:10,000. All combinations were repeated in triplicate. The optimum results were determined according to the highest OD difference between the known positive and known negative serum samples with a minimal background.
The GST-cleaved and purified N protein was diluted in 0.1 M carbonate buffer, and 65 ng were added to each well of the ELISA plate to coat the plates overnight at 4°C (Maxisorp; Nunc). The plates were washed three times with PBS-0.05% Tween-20 (PBS-T) and then blocked with 5% (wt/vol) bovine serum albumin in PBS-T by incubation at room temperature for 1 h. After washing three times with PBS-T, serum samples in 1:250 dilutions were added for a 1-h incubation at room temperature. The plates were washed three times with PBS-T again before the addition of the secondary antibody (goat anti-turkey IgG [H+L] diluted 1:2,000). The plates were washed again three times with PBS-T followed by the addition of substrate (pNPP Microwell substrate system; Kirkegaard and Perry Laboratories, Gaithersburg MD). The plates were incubated for 15 min in the dark, and the reaction was terminated using 2 M H2SO4. The plates were read at 405 nm using a BioTek Powerwave XS microplate ELISA reader (BioTek Instruments, Inc., Winooski, VT).
Sensitivity, specificity, and cutoff values for the recombinant antigen ELISA.
The cutoff values of the TCoV N ELISA were determined using OD405 values obtained from 86 control positive serum samples with 73 control negative turkey sera. Receiving operating characteristics (ROC) curves were generated using an Excel spreadsheet-based analysis tool (11, 12) and used to determine cutoff OD405 values that provided optimal sensitivity and specificity for each ELISA.
Cross-reactivity of turkey sera with IBV assessed using a commercial IBV whole-virus ELISA.
The cross-reactivity of turkey sera against IBV antigens was assessed using commercially available IBV whole-virus-coated ELISA plates (FlockChek infectious bronchitis virus antibody test kit; IDEXX, Westbrook, ME). The ELISA was carried out according to the manufacturer's instructions with the minor modification that the turkey sera were only diluted 1:250 rather than 1:500 as is normally the case when testing chicken sera. A subsample of 168 of the 360 serum samples obtained to assess the seroprevalence of TCoV in Ontario commercial turkey flocks were tested using the IDEXX kit, and the results were compared with the readings obtained from the recombinant N-based ELISA described above. Seropositive or seronegative status was determined using a cutoff OD650 of 0.1 for the whole IBV antigen-based ELISA.
Determination of TCoV seroprevalence in commercially reared turkeys.
Field serum samples were obtained from Ontario turkey farms. Thirty samples were collected from each of six commercial meat turkey and six breeder turkey farms for a total of 360 field serum samples from Ontario. A further 81 field samples were obtained from various turkey farms in Arkansas. All samples were heat inactivated at 56°C for 30 min before being used in the TCoV N-based recombinant ELISA.
Statistical analyses.
Differences in TCoV seroprevalence between breeder and meat turkey flocks were tested with one-tailed t tests assuming unequal variances. In all cases, differences were considered significant at P values of <0.05. Spearman rank order correlation calculated using the Free Statistics software (version 1.1.23-r1; P. Wessa, Office for Research Development and Education [http://www.wessa.net/]) was used to assess the correlation of the OD405 values, and the seropositive or seronegative status was determined by the cutoff OD405 value established for the TCoV N-based and whole IBV antigen-based ELISAs. The OD405 values from 168 paired field serum samples from the Ontario commercial turkeys were included in this correlation analysis.
RESULTS
Cloning and expression of TCoV N protein.
The full-length nucleocapsid protein gene was amplified from TCoV-MG10 (Fig. 1) and directionally cloned into the BamHI and EcoRI restriction sites of the PGEX-4T3 vector. The integrity of the cloned TCoV N gene with an intact reading frame and orientation to produce a GST fusion protein was confirmed by sequencing in both directions (data not shown). TCoV N-GST fusion protein expression in E. coli was confirmed using nonreducing SDS-PAGE and visualized as a 69/78-kDa doublet when stained with Coomassie blue. The identity of the doublet was confirmed by Western blot analysis with MAb 1.01 and MAb 4.24 against TCoV as well as anti-GST antibodies (Fig. 2) prior to the cleavage of the protein with thrombin. After cleavage, the recombinant TCoV N-protein doublet was at 43/52 kDa (data not shown).
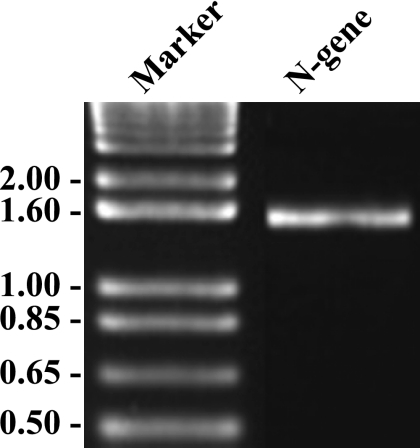
FIG. 1.
Reverse transcriptase PCR amplification of the TCoV N gene (1,227 bp) from TCoV used for the cloning and expression of recombinant protein.
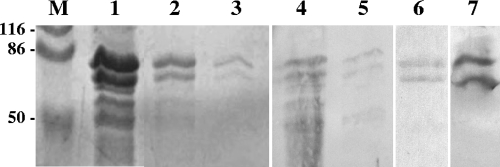
FIG. 2.
Production and confirmation of antigenicity of TCoV N recombinant fusion protein produced by E. coli BL-21 transfected with pGEX-4T3-TCoV-N using 12% discontinuous SDS-PAGE (lanes 1 to 3) and subsequent Western blotting with anti-GST and anti-TCoV N-protein MAbs 1.01 and 4.23 (lanes 4 to 7). Lane M, molecular mass marker in kilodaltons; lane 1, nonpurified bacterial lysate after induction with IPTG; lane 2, TCoV N fusion protein purified with GST beads from bacterial lysate after induction with IPTG; lane 3, the same as lane 2 but from noninduced bacterial culture; lanes 4 and 5, Western blot using anti-GST antibodies against the TCoV N fusion protein expressed and purified as in lanes 2 and 3, respectively; lanes 6 and 7, Western blot using MAbs 1.01 and 4.23, respectively, against the TCoV N fusion protein expressed and purified as described for lane 2.
Optimization and cutoff value of the TCoV N-based ELISA.
Using the described checkerboard titrations, the optimized ELISA conditions were established as follows: the recombinant antigen concentration for plate coating was 65 ng/μl TCoV N protein, primary antibodies (turkey serum samples) were diluted at 1:250, and the secondary goat anti-turkey IgG (H+L)-AP conjugate was diluted at 1:2,000. Substrate reactions were stopped after 15 min.
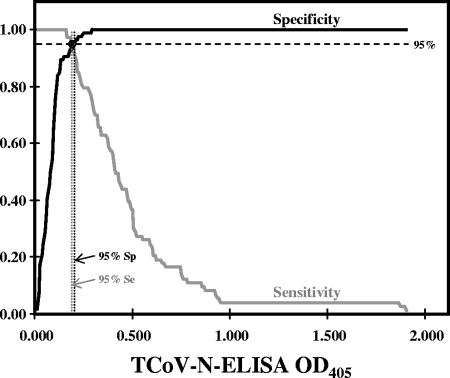
Using a two-graph ROC analysis (11, 12) based on 73 known negative-control serum samples and 86 serum samples from experimentally infected turkeys at 14, 21, 28, 35, and 42 days postinfection as positive-control sera, we established the cutoff value for the TCoV N-based ELISA to be an OD405 of 0.180; serum samples testing above this OD value were considered positive, while values at or below this value were considered negative. This cutoff value resulted in 97% sensitivity and 93% specificity for the TCoV N-based ELISA (Fig. 3).
FIG. 3.
ROC analysis of TCoV N-based ELISA. The cutoff value derived from the curves was 0.180 (indicated by ⊗) that resulted in 97% sensitivity and 93% specificity for the TCoV N-based ELISA.
Seroprevalence of TCoV in Ontario, Canada.
The TCoV N-based ELISA was conducted on 360 random serum samples collected from 12 turkey farms (30 samples per farm). For the six breeder turkey farms, a mean prevalence of 73.9 ± 0.76% (133/180 had OD405 values of >0.180) was observed (Table 1). In serum samples obtained from six meat turkey farms, a mean seroprevalence of 60.0 ± 1.18% (108/180 had OD405 values of >0.180) was observed (Table 2). There was a significantly lower (P = 0.042) overall positive seroprevalence for meat turkey farm serum samples compared with samples obtained from breeder turkey farms.
TABLE 1.
Seroprevalence of TCoV at Ontario breeder turkey farms using a recombinant TCoV N-based ELISA
| Ontario breeder turkey farm | Mean turkey age (wk) | Total no. of samples | No. of positive samples | No. of negative samples | Prevalence (%) |
|---|---|---|---|---|---|
| 1 | 30 | 30 | 23 | 7 | 76.67 |
| 2 | 32 | 30 | 23 | 7 | 76.67 |
| 3 | 55 | 30 | 25 | 5 | 83.33 |
| 4 | 55 | 30 | 24 | 6 | 80.00 |
| 5 | 58 | 30 | 17 | 13 | 56.67 |
| 6 | 59 | 30 | 21 | 9 | 70.00 |
| Total | 180 | 133 | 47 | 73.89 |
TABLE 2.
Seroprevalence of TCoV at Ontario meat turkey farms using a recombinant TCoV N-based ELISA
| Ontario meat turkey farm | Mean turkey age (wk) | Total no. of samples | No. of positive samples | No. of negative samples | Prevalence (%) |
|---|---|---|---|---|---|
| 1 | 10 | 30 | 21 | 9 | 70.00 |
| 2 | 11 | 30 | 14 | 17 | 46.67 |
| 3 | 12 | 30 | 22 | 10 | 73.33 |
| 4 | 13 | 30 | 20 | 10 | 66.67 |
| 5 | 15 | 30 | 11 | 19 | 36.67 |
| 6 | 16 | 30 | 20 | 10 | 66.67 |
| Total | 180 | 108 | 72 | 60.00 |
Seroprevalence of TCoV in turkeys from Arkansas.
Eighty-one turkey serum samples were obtained from Arkansas, and 15 serum samples from SPF turkeys were tested using the TCoV N-based ELISA developed herein. A relatively high seroprevalence of 64.2% (51/81 had OD405 values of >0.180) for TCoV was detected in the Arkansas field samples. No seroconversion (0/15 had OD405 values of >0.180) was detected in the sera from the SPF turkeys.
Comparison of the recombinant TCoV N-based ELISA to a commercial IBV whole-virus ELISA for detecting seroconversion against TCoV.
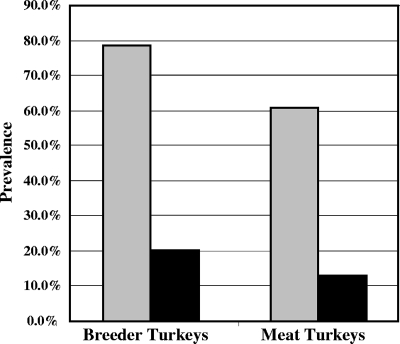
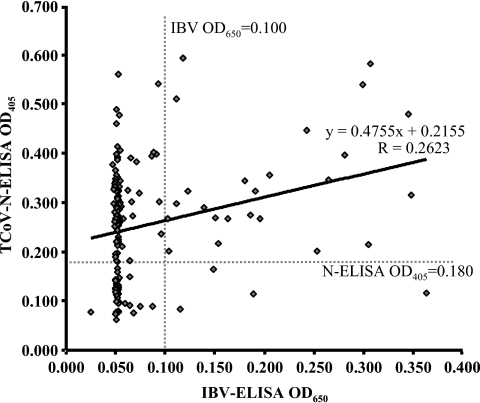
The ability of a commercial IBV whole virus-coated ELISA to detect antibodies recognizing TCoV was compared to the developed TCoV N-based ELISA. From the 168 turkey serum samples tested, the ELISAs produced TCoV seroprevalence estimates that differed dramatically (Table 3). The IBV whole-virus-coated ELISA revealed only 20.24% (17/84) positive samples from breeder turkey farms and 13.10% (11/84) positive samples from commercial turkey farms. In contrast, the recombinant antigen TCoV ELISA reported much higher TCoV seroprevalence in both breeder turkey (78.57% [66/84]) and meat turkey (60.71% [51/84]) flocks (Fig. 4). The TCoV N-based ELISA reported the overall seroprevalence of TCoV as 70.24%; this was approximately fourfold higher than the 16.67% overall seroprevalence reported by the commercial IBV whole-virus ELISA. There was only a modest correlation between the OD readings obtained using the TCoV N-based and IBV whole-virus-based ELISAs (R = 0.2623) (Fig. 5).
TABLE 3.
Comparison of a commercial whole-virus IBV ELISA with a recombinant-antigen TCoV N-based ELISA for detecting TCoV infections in turkeys using paired serum samples
| ELISA | Primary serum dilution | Cutoff OD value | No. of positive samples | Total no. of samples | Prevalence (% infected) |
|---|---|---|---|---|---|
| TCoV N | 1:250 | 0.180 | 118 | 168 | 70.24 |
| IDEXX IBV | 1:250 | 0.100 | 17 | 168 | 16.67 |
FIG. 4.
Comparison of the detection of TCoV in turkey serum samples obtained from breeder turkey farms (84 samples) and meat turkey farms (84 samples) in Ontario, Canada, examined in duplicate using TCoV N-based (gray bars) and IBV whole-virus-based (black bars) ELISAs. The recombinant TCoV N-based ELISA detected an overall seroprevalence of TCoV of 70.24% that was about fourfold higher than the 16.67% overall seroprevalence reported by the commercial IBV whole-virus-based ELISA.
FIG. 5.
Scatter plot of the OD405 values obtained from 168 paired turkey serum samples from Ontario turkey farms using TCoV N- and a whole-virus IBV-based commercial ELISA. Only a modest correlation (R = 0.2623) was observed between the recombinant antigen TCoV N-based ELISA and the commercial IBV whole-virus-based ELISA. The usual OD405 cutoff value for the N-based ELISA is 0.180 (horizontal dotted line), and the usual OD650 cutoff value for the whole-virus IBV-based ELISA is 0.100 (vertical dotted line).
DISCUSSION
TCoV is one of the most important etiological agents of diarrhea in young turkey poults (5) and is believed to be involved in poult enteritis and mortality syndrome. First efforts to determine the seroprevalence of TCoV were based on virus neutralization (23) and fluorescent antibody tests (22). Subsequently, ELISA-based detection methods were attempted; however, determining the seroprevalence of TCoV in commercial turkey populations remained difficult because TCoV cannot be propagated readily in cell culture (2, 4, 13; unpublished data) although at least some isolates can be propagated in turkey embryos (23). Thus, a variety of antigens other than whole TCoV have been used to determine the seroprevalence of TCoV in turkey flocks. These include the use of IBV whole-virus antigen in an antibody-capture ELISA (this study and reference 17). Recombinant TCoV N protein derived from a prokaryotic expression system was suggested as a potential antigen for an antibody-capture ELISA specific for TCoV (18), and N protein expressed in a baculovirus system (4) has been used to develop a competitive ELISA test in concert with MAbs raised against TCoV N protein (14).
In the present study, a recombinant antigen ELISA was developed based on the recently completed genomic sequence of a recent field isolate of TCoV (isolate MG10) (10). The utility of this recombinant ELISA was then assessed using field turkey serum samples from Ontario, Canada, and Arkansas, and the efficacy of this recombinant ELISA was compared to the use of a commercially available whole-virus IBV ELISA for detecting TCoV infections.
Development of a TCoV N-based ELISA.
The nucleocapsid protein of most CoVs is produced at a high level as early as 12-h postinfection, making it a good candidate for the early detection of different CoVs (15, 20). The N protein of IBV is immunogenic and contains domains conserved among most IBV strains (29). The conserved nature of the N protein has permitted its use as a recombinant antigen in various ELISA systems to test serum samples for antibodies against IBV (7, 9, 21) as well as antibodies against TCoV (17). The TCoV N protein has been used in a competitive ELISA to detect antibodies against TCoV (14); not surprisingly, this ELISA was able to detect antibodies against IBV as well.
Our clone of TCoV N protein was expressed in two forms (52 and 43 kDa after thrombin cleavage). A similar pair of expressed forms (52 and 43 kDa) was observed when the complete TCoV N protein was expressed using a baculovirus expression system (4). Finding a doublet protein product is not unusual for some CoVs such as IBV and MHV that each produce two forms of their nucleocapsid proteins (6, 19, 27). It has been shown that a whole-length IBV N-protein clone in E. coli expressed two protein bands (50 and 46 kDa) detectable using an MAb specific for the IBV N protein (29). Similar phenomena of having different forms of the nucleocapsid protein have been reported for Newcastle disease virus, where this pattern was explained as the proteolytic cleavage of the translation product to yield different forms of the same protein (20).
The recombinant TCoV antigen ELISA developed herein had high specificity and sensitivity as determined using a two-graph ROC analysis. Assuming a turkey flock seroprevalence of TCoV of about 65% in line with our observations and others (14), the TCoV N-based ELISA would typically have a positive predictive value of 96.26% and a negative predictive value of 94.35%. These strong predictive values would be more than adequate to justify the use of this recombinant antigen ELISA to accurately and economically determine the presence of antibodies against TCoV in turkey sera.
Comparison between the TCoV ELISA and IBV ELISA for TCoV antibodies.
Using experimentally derived positive and negative turkey sera and a commercial IBV whole-virus antigen plate, TCoV-positive sera showed reactivity with the IBV antigens but at much lower OD readings. Thus, relatively few samples had OD readings above the cutoff values, and therefore, the inferred seroprevalence was much lower for this ELISA. For example, using a subset of 168 field serum samples, only 28 had OD650 readings above a cutoff value of 0.100 (established using a two-graph ROC analysis and experimental turkey sera) using the IDEXX FlockChek IBV whole-virus ELISA performed according to the manufacturer's instructions and with the kit reagents (conjugated secondary antibody and enzyme substrate). In contrast, the TCoV-specific ELISA based on recombinant N protein found that 70.24% of these serum samples were positive for antibodies recognizing TCoV. There was a positive, albeit weak, correlation of this commercial IBV-based ELISA with the recombinant TCoV ELISA (Fig. 5), presumably because the N protein is relatively conserved between IBV and TCoV (4, 14, 18).
Seroprevalence of TCoV in Ontario, Canada, and Arkansas.
Despite its cosmopolitan presence in turkey flocks and potential importance in disease pathogenesis, few epidemiological surveys have been conducted to determine TCoV seroprevalence. Using an indirect fluorescent-antibody assay or a commercially available IBV ELISA (IDEXX), it was determined that in Indiana, 175/325 (53.84%) or 163/325 (50.15%) of the field samples had antibodies for TCoV, respectively (17). In a second study using a competitive ELISA and a recombinant TCoV N protein, 63% of the sampled turkey poults were found to possess antibodies to TCoV (14). Our recombinant TCoV N-based ELISA detected 73.9% and 60.0% positive sera from breeder turkey farms and meat turkey farms in Ontario, Canada, respectively. Field serum samples obtained in Arkansas had similarly high seroprevalence. The higher observed prevalence of antibodies recognizing TCoV using our recombinant ELISA compared to results using the commercially prepared IBV whole-virus ELISA plate (17) and our results using the IDEXX IBV antigen plates suggest that TCoV-specific antibodies cross-react with IBV antigens but to a lower level than the homologous TCoV antigen. The prevalence of TCoV antibodies was high in both breeder turkey and meat turkey flocks as demonstrated by our data, but there was a significantly higher prevalence in the turkey breeder flocks, perhaps because the birds in the latter group are older and more likely to have had time to be exposed to the virus and mount a detectable serum antibody response.
In summary, a recombinant TCoV antigen-based ELISA was developed based on the complete nucleocapsid protein of TCoV. This ELISA performed significantly better at detecting antibodies against TCoV in turkey sera than a commercially available IBV ELISA kit. The recombinant antigen ELISA was instrumental in detecting the high seroprevalence of TCoV in breeder turkey farms in Ontario and meat turkey farms in Ontario and Arkansas.
Acknowledgments
We thank Tony Cengija and Barb Mitchell of the Campus Animal Facility's Isolation Unit of the University of Guelph for the excellent care of our experimental animals and Stephanie Reabel and Inas Elawadli for assistance with animal handling and sample collection. We also thank Gaye Smith, Hongwen Fu, and Keith Harron of the University of Guelph's Animal Health Laboratory for technical assistance and Jeff Zhang from Cold Spring Farms for the field turkey sera. Hybrid Turkeys (Kitchener, Ontario, Canada) generously donated the day of age poults used in the study as well as provided SPF and field turkey serum samples from Ontario. Billy Hargis, University of Arkansas, Fayetteville, AR, kindly provided SPF and field turkey serum samples from the state of Arkansas. Two MAbs were kindly provided by James Guy, North Carolina State University, Raleigh, NC.
This work is supported by grants to J.R.B. from the Natural Sciences and Engineering Research Council of Canada (NSERC) and the Ontario Ministry of Agriculture and Food and Rural Affairs (OMAFRA). M.H.G. is a scholarship recipient from the Egyptian Cultural and Educational Bureau of the Arab Republic of Egypt in Canada.
Footnotes
Published ahead of print on 29 October 2008.
REFERENCES
- 1.Ahmed, S., M. Bassiri, A. K. Banerjee, and T. Yilma. 1993. Immunological characterization of VSV nucleocapsid protein expressed by baculovirus in Spodoptera exigua larva: use in differential diagnosis between vaccinated and infected animals. Virology 192:207-216. [DOI] [PubMed] [Google Scholar]
- 2.Ali, A., and D. L. Reynolds. 1998. The in vitro propagation of stunting syndrome agent. Avian Dis. 42:657-666. [PubMed] [Google Scholar]
- 3.Boots, A. M. H., J. G. Kuster, J. M. van Noort, K. A. Zwaagstra, E. Rijke, B. A. van der Zeijst, and E. J. Hensen. 1991. Localization of a T-cell epitope within the nucleocapsid protein of avian coronavirus. Immunology 74:8-13. [PMC free article] [PubMed] [Google Scholar]
- 4.Breslin, J. J., L. G. Smith, and J. S. Guy. 2001. Baculovirus expression of turkey coronavirus nucleocapsid protein. Avian Dis. 45:136-143. [PubMed] [Google Scholar]
- 5.Cavanagh, D. 2001. The role of coronaviruses in enteric diseases of turkeys, p. 31. Proceedings of the 24th Technical Turkey Conference. http://www.poultry-health.com/fora/turkhelth/turtec24/cavanagh.htm.
- 6.Cheley, S., and R. Anderson. 1981. Cellular synthesis and modification of murine hepatitis virus polypeptides. J. Gen. Virol. 54:301-311. [DOI] [PubMed] [Google Scholar]
- 7.Chen, H., B. Coote, S. Attree, and J. A. Hiscox. 2003. Evaluation of a nucleocapsid-based enzyme-linked immunosorbent assay for the detection of antibodies against infectious bronchitis virus. Avian Pathol. 32:519-526. [DOI] [PubMed] [Google Scholar]
- 8.Erington, W., M. Steward, and P. M. Emerson. 1995. A diagnostic immunoassay for Newcastle disease virus based on nucleocapsid protein expressed by a recombinant baculovirus. J. Virol. Methods 55:357-365. [DOI] [PubMed] [Google Scholar]
- 9.Gibertoni, A. M., M. F. S. Montassier, J. A. D. Sena, P. E. N. Givisiez, C. R. A. G. Furuyama, and H. J. Montassier. 2005. Development and application of Saccharomyces cerevisiae-expressed nucleocapsid protein-based enzyme-linked immunosorbent assay for detection of antibodies against infectious bronchitis virus. J. Clin. Microbiol. 43:1982-1984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Gomaa, M. H., J. R. Barta, D. Ojkic, and D. Yoo. 2008. Complete genome sequence of turkey coronavirus. Virus Res. 135:237-246. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Griener, M. 1995. Two-graph receiver operating characteristics (TG-ROC): a Microsoft Excel template for the selection of cutoff values in diagnostic tests. J. Immunol. Methods 185:145-146. [DOI] [PubMed] [Google Scholar]
- 12.Griener, M., D. Sohr, and P. Gobel. 1995. A modified ROC analysis for the selection cut-off values and the definition of intermediate results of serodiagnostic tests. J. Immunol. Methods 185:123-132. [DOI] [PubMed] [Google Scholar]
- 13.Guy, J. S., H. J. Barnes, L. G. Smith, and J. J. Breslin. 1997. Antigenic characterization of a turkey coronavirus identified in poult enteritis- and mortality syndrome-affected turkeys. Avian Dis. 41:583-590. [PubMed] [Google Scholar]
- 14.Guy, J. S., L. G. Smith, J. J. Breslin, and S. Pakpinyo. 2002. Development of a competitive enzyme-linked immunosorbent assay for detection of turkey coronavirus antibodies. Avian Dis. 46:334-341. [DOI] [PubMed] [Google Scholar]
- 15.Hiscox, J. A., D. Cavanaugh, and P. Britton. 1995. Quantification of individual sub-genomic mRNA species during replication of the coronavirus transmissible gastroenteritis virus. Virus Res. 36:119-130. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Hummel, K. B., D. D. Erdamn, J. Heath, and W. J. Bellini. 1992. Baculovirus expression of the nucleocapsid gene of measles virus and utility of the recombinant protein in diagnostic enzyme immunoassay. J. Clin. Microbiol. 30:2874-2880. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Loa, C. C., T. L. Lin, C. C. Wu, T. A. Bryan, H. L. Thacker, T. Hooper, and D. Schrader. 2000. Detection of antibody to turkey coronavirus by antibody-capture enzyme-linked immunosorbent assay utilizing infectious bronchitis virus antigen. Avian Dis. 44:498-506. [PubMed] [Google Scholar]
- 18.Loa, C. C., T. L. Lin, C. C. Wu, T. A. Bryan, T. Hooper, and D. Schrader. 2004. Expression and purification of turkey coronavirus nucleocapsid protein in Escherichia coli. J. Virol. Methods 116:161-167. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Lugovskaya, N. N., A. V. Scherbakov, A. S. Yakovleva, M. A. Tsyvanyuk, N. S. Mudrak, V. V. Drygin, and A. V. Borisov. 2006. Detection of antibodies to avian infectious bronchitis virus by a recombinant nucleocapsid protein-based enzyme-linked immunosorbent assay. J. Virol. Methods 135:292-296. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Mountcastle, W. E., R. W. Compans, L. A. Caliguiri, and P. W. Choppin. 1970. Nucleocapsid protein subunits of simian virus 5, Newcastle disease virus, and Sendai virus 1. J. Virol. 6:677-684. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Ndifuna, A., A. K. Waters, M. Zhou, and E. W. Collisson. 1998. Recombinant nucleocapsid protein is potentially an inexpensive, effective serodiagnostic reagent for infectious bronchitis virus. J. Virol. Methods 70:37-44. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Patel, B. L., B. S. Pomeroy, E. Gonder, and C. E. Cronkite. 1976. Indirect fluorescent antibody test for the diagnosis of coronaviral enteritis of turkeys (bluecomb). Am. J. Vet. Res. 37:1111-1112. [PubMed] [Google Scholar]
- 23.Pomeroy, B. S., C. T. Larsen, D. R. Deshmukh, and B. L. Patel. 1975. Immunity to transmissible (coronaviral) enteritis of turkeys (bluecomb). Am. J. Vet. Res. 36:553-555. [PubMed] [Google Scholar]
- 24.Qiu, M., J. Wang, H. Wang, Z. Chen, E. Dai, Z. Guo, X. Wang, X. Pang, B. Fan, J. Wen, J. Wang, and R. Yang. 2005. Use of the COOH portion of the nucleocapsid protein in an antigen-capturing enzyme-linked immunosorbent assay for specific and sensitive detection of severe acute respiratory coronavirus. Clin. Diagn. Lab. Immunol. 12:474-476. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Seah, J. N., L. Yu, and J. Kwang. 2000. Localization of linear B-cell epitopes on infectious bronchitis virus nucleocapsid protein. Vet. Microbiol. 75:11-16. [DOI] [PubMed] [Google Scholar]
- 26.Seo, S. H., L. Wang, R. Smith, and E. W. Collisson. 1997. The carboxyl-terminal 120-residue polypeptide of infectious bronchitis virus nucleocapsid induces cytotoxic T lymphocytes and protects chickens from acute infection. J. Virol. 71:7889-7894. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Stern, D. F., L. Burgess, and B. M. Sefton. 1982. Structural analysis of virion proteins of the avian coronavirus infectious bronchitis virus. J. Virol. 42:208-219. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Tumlin, J. T., B. S. Pomeroy, and R. K. Lindorfer. 1957. Bluecomb disease of turkey. IV. Demonstration of a filterable agent. J. Am. Vet. Med. Assoc. 130:360-365. [PubMed] [Google Scholar]
- 29.Zhang, D. Y., J. Y. Zhou, J. Fang, J. Q. Hu, J. X. Wu, and A. X. Mu. 2005. An ELISA for antibodies detection to infectious bronchitis virus based nucleocapsid protein produced in Escherichia coli. Vet. Med. Czech. 50:336-344. [Google Scholar]