Abstract
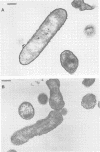
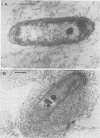
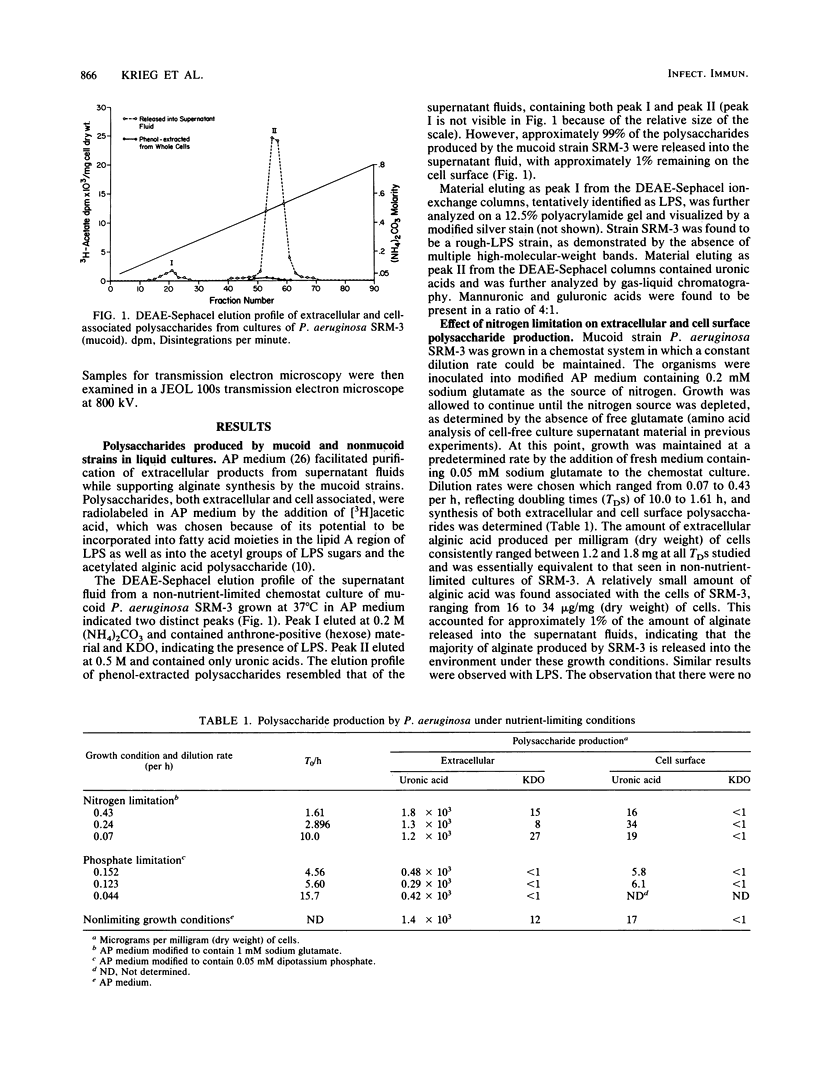
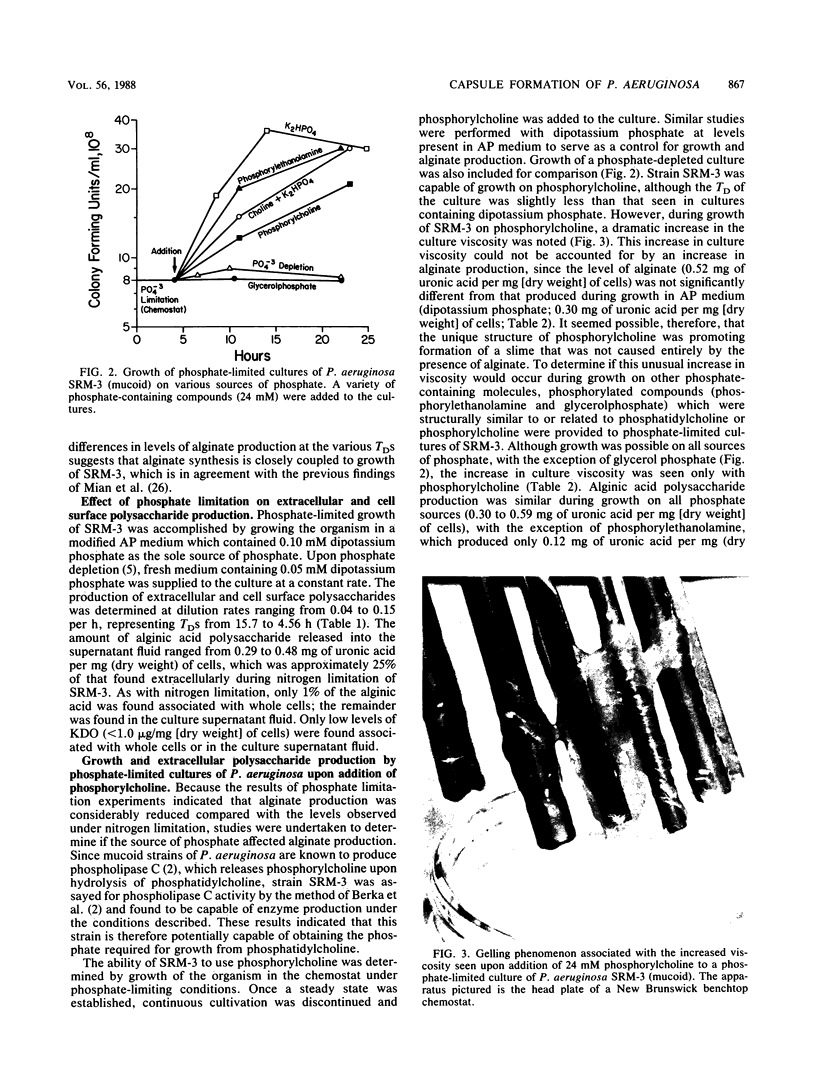
Production of both alginic acid and lipopolysaccharide by a mucoid strain of Pseudomonas aeruginosa, SRM-3, was studied in a chemostat system during growth under nutrient-limiting conditions chosen to reflect the chronic growth conditions in the lungs of cystic fibrosis patients. Since mucoid strains have been shown to elaborate extracellular proteases and phospholipase C, nitrogen and phosphate limitation were selected for analysis. A modified alginate-promoting medium containing either 1 mM glutamate or 0.05 mM K2HPO4 as limiting nutrient and doubling times of 1.6 to 15.7 h were used. Under nitrogen limitation, strain SRM-3 produced 1.4 mg of uronic acid per mg (dry weight) of cells at all doubling times studied. However, phosphate limitation resulted in the synthesis of only 0.4 mg of uronic acid per mg (dry weight) of cells. The role of phosphate in alginic acid polysaccharide production was further investigated by using phosphorylcholine, a product of phospholipase C activity on phosphatidylcholine, the major lung surfactant. No only were mucoid cells capable of utilizing phosphorylcholine for growth, but a highly specific interaction occurred among phosphorylcholine, alginate, and whole cells, resulting in greatly enhanced culture viscosity. Electron micrographs showed the gradual formation of a capsule during growth on phosphorylcholine, indicating that the mucoid strain has the ability to utilize surfactant not only as a nutrient source but also for constructing a capsule with greatly enhanced adhesive properties.
Full text
PDF



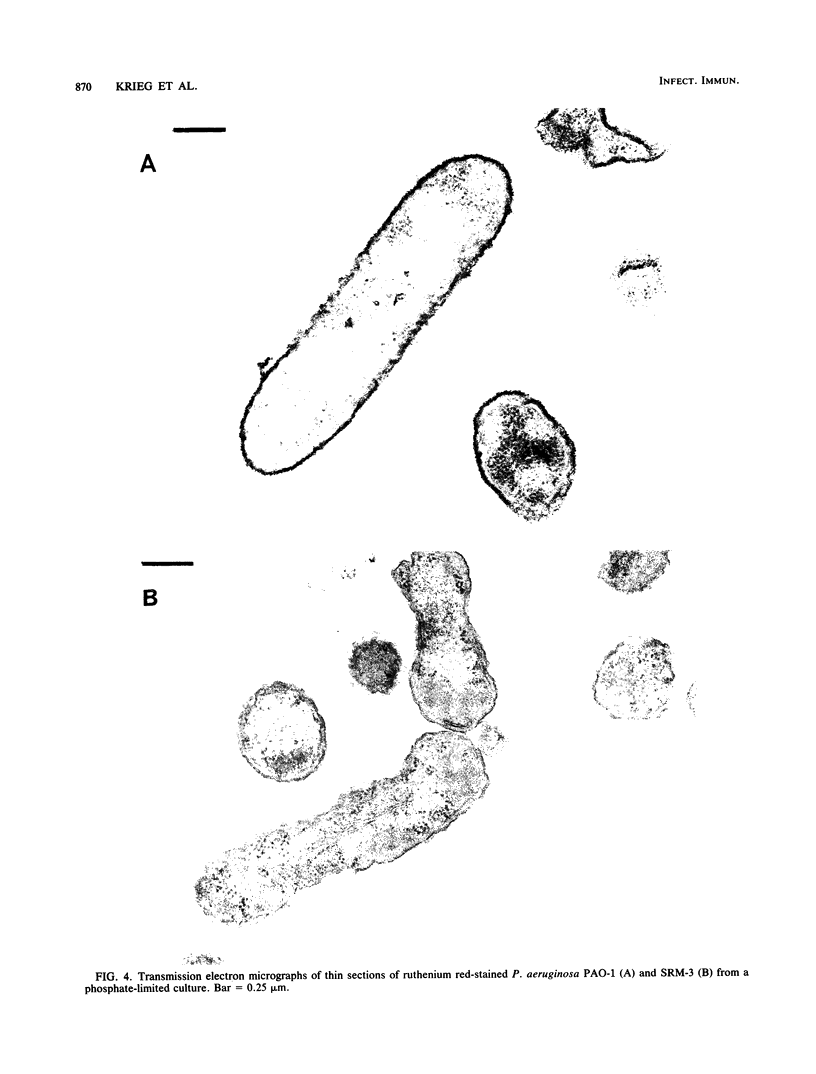
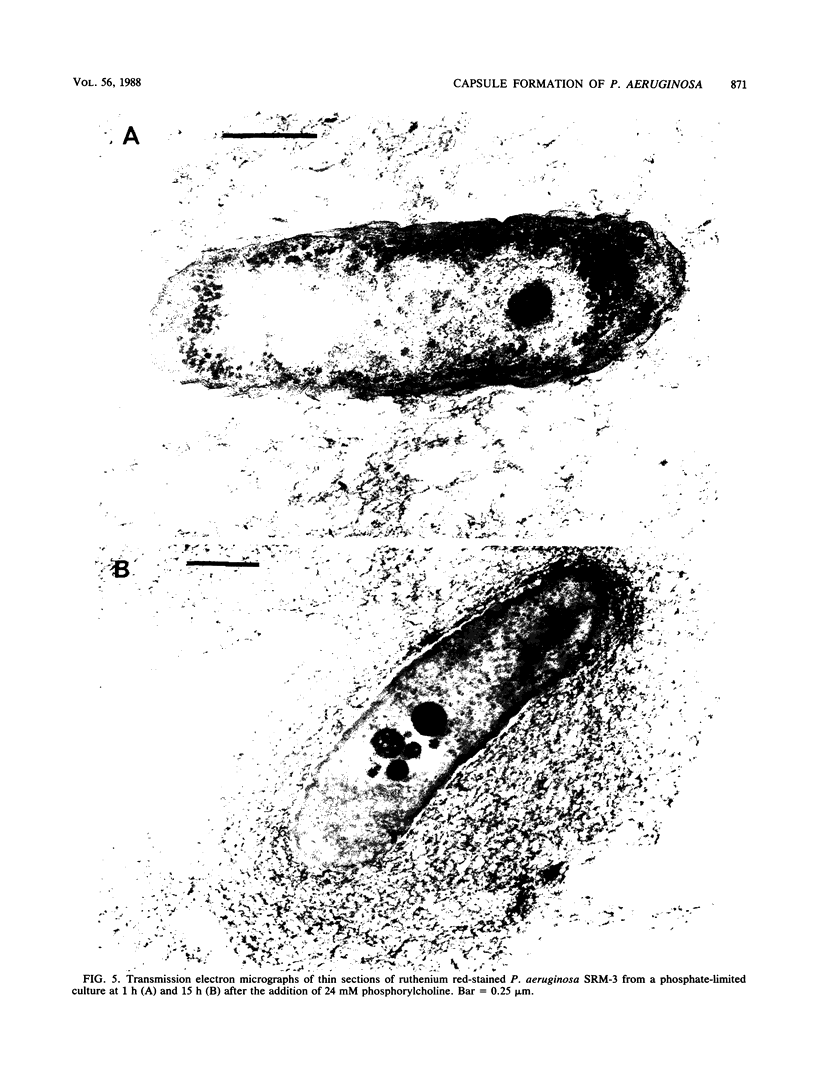


Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- AMINOFF D. Methods for the quantitative estimation of N-acetylneuraminic acid and their application to hydrolysates of sialomucoids. Biochem J. 1961 Nov;81:384–392. doi: 10.1042/bj0810384. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Berka R. M., Gray G. L., Vasil M. L. Studies of phospholipase C (heat-labile hemolysin) in Pseudomonas aeruginosa. Infect Immun. 1981 Dec;34(3):1071–1074. doi: 10.1128/iai.34.3.1071-1074.1981. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Berka R. M., Vasil M. L. Phospholipase C (heat-labile hemolysin) of Pseudomonas aeruginosa: purification and preliminary characterization. J Bacteriol. 1982 Oct;152(1):239–245. doi: 10.1128/jb.152.1.239-245.1982. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dancey G. F., Shapiro B. M. Specific phospholipid requirement for activity of the purified respiratory chain NADH dehydrogenase of Escherichia coli. Biochim Biophys Acta. 1977 May 25;487(2):368–377. doi: 10.1016/0005-2760(77)90013-3. [DOI] [PubMed] [Google Scholar]
- ESSELMANN M. T., LIU P. V. Lecithinase production by gramnegative bacteria. J Bacteriol. 1961 Jun;81:939–945. doi: 10.1128/jb.81.6.939-945.1961. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Esfahani M., Rudkin B. B., Cutler C. J., Waldron P. E. Lipid-protein interactions in membranes: interaction of phospholipids with respiratory enzymes of Escherichia coli membrane. J Biol Chem. 1977 May 25;252(10):3194–3198. [PubMed] [Google Scholar]
- Evans L. R., Linker A. Production and characterization of the slime polysaccharide of Pseudomonas aeruginosa. J Bacteriol. 1973 Nov;116(2):915–924. doi: 10.1128/jb.116.2.915-924.1973. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Farías R. N., Bloj B., Morero R. D., Siñeriz F., Trucco R. E. Regulation of allosteric membrane-bound enzymes through changes in membrane lipid compostition. Biochim Biophys Acta. 1975 Jun 30;415(2):231–251. doi: 10.1016/0304-4157(75)90003-9. [DOI] [PubMed] [Google Scholar]
- Granström M., Ericsson A., Strandvik B., Wretlind B., Pavlovskis O. R., Berka R., Vasil M. L. Relation between antibody response to Pseudomonas aeruginosa exoproteins and colonization/infection in patients with cystic fibrosis. Acta Paediatr Scand. 1984 Nov;73(6):772–777. doi: 10.1111/j.1651-2227.1984.tb17774.x. [DOI] [PubMed] [Google Scholar]
- Groniowski J. A. Fine structural basis of pulmonary surfactant. Int Rev Exp Pathol. 1983;25:183–238. [PubMed] [Google Scholar]
- Hancock R. E., Mutharia L. M., Chan L., Darveau R. P., Speert D. P., Pier G. B. Pseudomonas aeruginosa isolates from patients with cystic fibrosis: a class of serum-sensitive, nontypable strains deficient in lipopolysaccharide O side chains. Infect Immun. 1983 Oct;42(1):170–177. doi: 10.1128/iai.42.1.170-177.1983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Higashi Y., Strominger J. L. Biosynthesis of the peptidoglycan of bacterial cell walls. XX. Identification of phosphatidylglycerol and cardiolipin as cofactors for isoprenoid alcohol phosphokinase. J Biol Chem. 1970 Jul 25;245(14):3691–3696. [PubMed] [Google Scholar]
- Hirsch S. R., Zastrow J. E., Kory R. C. Sputum liquefying agents: a comparative in vitro evaluation. J Lab Clin Med. 1969 Aug;74(2):346–353. [PubMed] [Google Scholar]
- Høiby N., Rosendal K. Epidemiology of Pseudomonas aeruginosa infection in patients treated at a cystic fibrosis centre. Acta Pathol Microbiol Scand B. 1980 Jun;88(3):125–131. doi: 10.1111/j.1699-0463.1980.tb02617.x. [DOI] [PubMed] [Google Scholar]
- Krieg D. P., Bass J. A., Mattingly S. J. Aeration selects for mucoid phenotype of Pseudomonas aeruginosa. J Clin Microbiol. 1986 Dec;24(6):986–990. doi: 10.1128/jcm.24.6.986-990.1986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kurioka S., Liu P. V. Effect of the hemolysin of Pseudomonas aeruginosa on phosphatides and on phospholipase c activity. J Bacteriol. 1967 Feb;93(2):670–674. doi: 10.1128/jb.93.2.670-674.1967. [DOI] [PMC free article] [PubMed] [Google Scholar]
- LOWRY O. H., ROSEBROUGH N. J., FARR A. L., RANDALL R. J. Protein measurement with the Folin phenol reagent. J Biol Chem. 1951 Nov;193(1):265–275. [PubMed] [Google Scholar]
- Laemmli U. K. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature. 1970 Aug 15;227(5259):680–685. doi: 10.1038/227680a0. [DOI] [PubMed] [Google Scholar]
- Luft J. H. Ruthenium red and violet. I. Chemistry, purification, methods of use for electron microscopy and mechanism of action. Anat Rec. 1971 Nov;171(3):347–368. doi: 10.1002/ar.1091710302. [DOI] [PubMed] [Google Scholar]
- Luzar M. A., Montie T. C. Avirulence and altered physiological properties of cystic fibrosis strains of Pseudomonas aeruginosa. Infect Immun. 1985 Nov;50(2):572–576. doi: 10.1128/iai.50.2.572-576.1985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Magoon M. W., Wright J. R., Baritussio A., Williams M. C., Goerke J., Benson B. J., Hamilton R. L., Clements J. A. Subfractionation of lung surfactant. Implications for metabolism and surface activity. Biochim Biophys Acta. 1983 Jan 7;750(1):18–31. doi: 10.1016/0005-2760(83)90200-x. [DOI] [PubMed] [Google Scholar]
- Mian F. A., Jarman T. R., Righelato R. C. Biosynthesis of exopolysaccharide by Pseudomonas aeruginosa. J Bacteriol. 1978 May;134(2):418–422. doi: 10.1128/jb.134.2.418-422.1978. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ohman D. E., Chakrabarty A. M. Utilization of human respiratory secretions by mucoid Pseudomonas aeruginosa of cystic fibrosis origin. Infect Immun. 1982 Aug;37(2):662–669. doi: 10.1128/iai.37.2.662-669.1982. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ojeniyi B., Baek L., Høiby N. Polyagglutinability due to loss of O-antigenic determinants in Pseudomonas aeruginosa strains isolated from cystic fibrosis patients. Acta Pathol Microbiol Immunol Scand B. 1985 Feb;93(1):7–13. doi: 10.1111/j.1699-0463.1985.tb02844.x. [DOI] [PubMed] [Google Scholar]
- Ombaka E. A., Cozens R. M., Brown M. R. Influence of nutrient limitation of growth on stability and production of virulence factors of mucoid and nonmucoid strains of Pseudomonas aeruginosa. Rev Infect Dis. 1983 Nov-Dec;5 (Suppl 5):S880–S888. doi: 10.1093/clinids/5.supplement_5.s880. [DOI] [PubMed] [Google Scholar]
- Pate J. L., Ordal E. J. The fine structure of Chondrococcus columnaris. 3. The surface layers of Chondrococcus columnaris. J Cell Biol. 1967 Oct;35(1):37–51. doi: 10.1083/jcb.35.1.37. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Penketh A., Pitt T., Roberts D., Hodson M. E., Batten J. C. The relationship of phenotype changes in Pseudomonas aeruginosa to the clinical condition of patients with cystic fibrosis. Am Rev Respir Dis. 1983 May;127(5):605–608. doi: 10.1164/arrd.1983.127.5.605. [DOI] [PubMed] [Google Scholar]
- Peter H. W., Ahlers J. Phospholipid requirements of ATPase of Escherichia coli. Arch Biochem Biophys. 1975 Sep;170(1):169–178. doi: 10.1016/0003-9861(75)90108-3. [DOI] [PubMed] [Google Scholar]
- Pier G. B., Desjardins D., Aguilar T., Barnard M., Speert D. P. Polysaccharide surface antigens expressed by nonmucoid isolates of Pseudomonas aeruginosa from cystic fibrosis patients. J Clin Microbiol. 1986 Aug;24(2):189–196. doi: 10.1128/jcm.24.2.189-196.1986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pier G. B., Matthews W. J., Jr, Eardley D. D. Immunochemical characterization of the mucoid exopolysaccharide of Pseudomonas aeruginosa. J Infect Dis. 1983 Mar;147(3):494–503. doi: 10.1093/infdis/147.3.494. [DOI] [PubMed] [Google Scholar]
- Raetz C. R. Enzymology, genetics, and regulation of membrane phospholipid synthesis in Escherichia coli. Microbiol Rev. 1978 Sep;42(3):614–659. doi: 10.1128/mr.42.3.614-659.1978. [DOI] [PMC free article] [PubMed] [Google Scholar]
- SHEFFNER A. L., MEDLER E. M., JACOBS L. W., SARETT H. P. THE IN VITRO REDUCTION IN VISCOSITY OF HUMAN TRACHEOBRONCHIAL SECRETIONS BY ACETYLCYSTEINE. Am Rev Respir Dis. 1964 Nov;90:721–729. doi: 10.1164/arrd.1964.90.5.721. [DOI] [PubMed] [Google Scholar]
- Sandermann H. Interfacial regulation: influence of lipid polar group conformation on lipid activation of C55-isoprenoid alcohol phosphokinase apoprotein. FEBS Lett. 1976 Mar 15;63(1):59–61. doi: 10.1016/0014-5793(76)80194-9. [DOI] [PubMed] [Google Scholar]
- Tsai C. M., Frasch C. E. A sensitive silver stain for detecting lipopolysaccharides in polyacrylamide gels. Anal Biochem. 1982 Jan 1;119(1):115–119. doi: 10.1016/0003-2697(82)90673-x. [DOI] [PubMed] [Google Scholar]
- Vadas L., Prihar H. S., Pugashetti B. K., Feingold D. S. A gas chromatographic method for the quantitative determination of hexuronic acids in alginic acid. Anal Biochem. 1981 Jul 1;114(2):294–298. doi: 10.1016/0003-2697(81)90484-x. [DOI] [PubMed] [Google Scholar]
- Wardi A. H., Allen W. S., Varma R. A simple method for the detection and quantitative determination of hexuronic acids and pentoses. Anal Biochem. 1974 Jan;57(1):268–273. doi: 10.1016/0003-2697(74)90072-4. [DOI] [PubMed] [Google Scholar]
- Woo D. D., Holt S. C., Leadbetter E. R. Ultrastructure of Bacteroides species: Bacteroides asaccharolyticus, Bacteroides fragilis, Bacteroides melaninogenicus subspecies melaninogenicus, and B. melaninogenicus subspecies intermedius. J Infect Dis. 1979 May;139(5):534–546. doi: 10.1093/infdis/139.5.534. [DOI] [PubMed] [Google Scholar]
- Woods D. E., Schaffer M. S., Rabin H. R., Campbell G. D., Sokol P. A. Phenotypic comparison of Pseudomonas aeruginosa strains isolated from a variety of clinical sites. J Clin Microbiol. 1986 Aug;24(2):260–264. doi: 10.1128/jcm.24.2.260-264.1986. [DOI] [PMC free article] [PubMed] [Google Scholar]