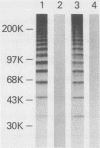
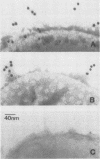
Abstract
Two types of fimbriae have been identified on the pathogenic gram-negative organism Bordetella pertussis. Monoclonal antibodies to these fimbriae were produced to better understand the role of fimbriae as serotype-specific agglutinogens and to investigate the antigenic relationship between these fimbriae. Three monoclonal antibodies were identified that specifically agglutinated B. pertussis cells containing the U.S. Reference Factor 2 agglutinogen, and six monoclonal antibodies were produced that agglutinated only those strains containing the U.S. Reference Factor 6 agglutinogen. Indirect immunofluorescence studies and immunogold electron microscopy demonstrated that these monoclonal antibodies bind to an outer membrane component on serotype-specific strains of B. pertussis. All of the monoclonal antibodies reacted with native or partially assembled type-specific fimbriae but not with monomeric fimbrial subunits as indicated by Western blot (immunoblot) analysis. The fimbrial agglutinogens recognized by the monoclonal antibodies were also uniquely reactive with either U.S. Reference Factor 2 or 6 antiserum (Eldering agglutinogen 2 or 6 polyclonal antiserum) in an indirect ELISA. No cross-reactivity of the monoclonal antibodies with the unrelated fimbriae was observed in any of the comparative immunological studies. Some of the monoclonal antibodies agglutinated certain strains of B. bronchiseptica, suggesting that this closely related species can contain antigenically similar fimbriae. These monoclonal antibodies should prove useful for further structural and functional analysis of Bordetella fimbriae and for studies on the role that these antigens play in prevention of infection and disease.
Full text
PDF


Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Ashworth L. A., Dowsett A. B., Irons L. I., Robinson A. The location of surface antigens of Bordetella pertussis by immuno-electron microscopy. Dev Biol Stand. 1985;61:143–151. [PubMed] [Google Scholar]
- Ashworth L. A., Dowsett A. B., Irons L. I., Robinson A. The location of surface antigens of Bordetella pertussis by immuno-electron microscopy. Dev Biol Stand. 1985;61:143–151. [PubMed] [Google Scholar]
- Ashworth L. A., Irons L. I., Dowsett A. B. Antigenic relationship between serotype-specific agglutinogen and fimbriae of Bordetella pertussis. Infect Immun. 1982 Sep;37(3):1278–1281. doi: 10.1128/iai.37.3.1278-1281.1982. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Beachey E. H. Bacterial adherence: adhesin-receptor interactions mediating the attachment of bacteria to mucosal surface. J Infect Dis. 1981 Mar;143(3):325–345. doi: 10.1093/infdis/143.3.325. [DOI] [PubMed] [Google Scholar]
- Brennan M. J., Cisar J. O., Vatter A. E., Sandberg A. L. Lectin-dependent attachment of Actinomyces naeslundii to receptors on epithelial cells. Infect Immun. 1984 Nov;46(2):459–464. doi: 10.1128/iai.46.2.459-464.1984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brennan M. J., Li Z. M., Cowell J. L., Bisher M. E., Steven A. C., Novotny P., Manclark C. R. Identification of a 69-kilodalton nonfimbrial protein as an agglutinogen of Bordetella pertussis. Infect Immun. 1988 Dec;56(12):3189–3195. doi: 10.1128/iai.56.12.3189-3195.1988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cowell J. L., Zhang J. M., Urisu A., Suzuki A., Steven A. C., Liu T., Liu T. Y., Manclark C. R. Purification and characterization of serotype 6 fimbriae from Bordetella pertussis and comparison of their properties with serotype 2 fimbriae. Infect Immun. 1987 Apr;55(4):916–922. doi: 10.1128/iai.55.4.916-922.1987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- ELDERING G., HORNBECK C., BAKER J. Serological study of Bordetella pertussis and related species. J Bacteriol. 1957 Aug;74(2):133–136. doi: 10.1128/jb.74.2.133-136.1957. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Engvall E. Enzyme immunoassay ELISA and EMIT. Methods Enzymol. 1980;70(A):419–439. doi: 10.1016/s0076-6879(80)70067-8. [DOI] [PubMed] [Google Scholar]
- Irons L. I., Ashworth L. A., Robinson A. Release and purification of fimbriae from Bordetella pertussis. Dev Biol Stand. 1985;61:153–163. [PubMed] [Google Scholar]
- Laemmli U. K. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature. 1970 Aug 15;227(5259):680–685. doi: 10.1038/227680a0. [DOI] [PubMed] [Google Scholar]
- Lee S. W., Way A. W., Osen E. G. Purification and subunit heterogeneity of pili of Bordetella bronchiseptica. Infect Immun. 1986 Feb;51(2):586–593. doi: 10.1128/iai.51.2.586-593.1986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li Z. M., Cowell J. L., Brennan M. J., Burns D. L., Manclark C. R. Agglutinating monoclonal antibodies that specifically recognize lipooligosaccharide A of Bordetella pertussis. Infect Immun. 1988 Mar;56(3):699–702. doi: 10.1128/iai.56.3.699-702.1988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mooi F. R., van der Heide H. G., ter Avest A. R., Welinder K. G., Livey I., van der Zeijst B. A., Gaastra W. Characterization of fimbrial subunits from Bordetella species. Microb Pathog. 1987 Jun;2(6):473–484. doi: 10.1016/0882-4010(87)90054-4. [DOI] [PubMed] [Google Scholar]
- Oda M., Cowell J. L., Burstyn D. G., Manclark C. R. Protective activities of the filamentous hemagglutinin and the lymphocytosis-promoting factor of Bordetella pertussis in mice. J Infect Dis. 1984 Dec;150(6):823–833. doi: 10.1093/infdis/150.6.823. [DOI] [PubMed] [Google Scholar]
- Preston N. W., Surapatana N., Carter E. J. A reappraisal of serotype factors 4, 5 and 6 of Bordetella pertussis. J Hyg (Lond) 1982 Feb;88(1):39–46. doi: 10.1017/s0022172400069874. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schifferli D. M., Abraham S. N., Beachey E. H. Use of monoclonal antibodies to probe subunit- and polymer-specific epitopes of 987P fimbriae of Escherichia coli. Infect Immun. 1987 Apr;55(4):923–930. doi: 10.1128/iai.55.4.923-930.1987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Steven A. C., Bisher M. E., Trus B. L., Thomas D., Zhang J. M., Cowell J. L. Helical structure of Bordetella pertussis fimbriae. J Bacteriol. 1986 Sep;167(3):968–974. doi: 10.1128/jb.167.3.968-974.1986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Weiss A. A., Hewlett E. L., Myers G. A., Falkow S. Genetic studies of the molecular basis of whooping cough. Dev Biol Stand. 1985;61:11–19. [PubMed] [Google Scholar]
- Weiss A. A., Hewlett E. L., Myers G. A., Falkow S. Tn5-induced mutations affecting virulence factors of Bordetella pertussis. Infect Immun. 1983 Oct;42(1):33–41. doi: 10.1128/iai.42.1.33-41.1983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wray W., Boulikas T., Wray V. P., Hancock R. Silver staining of proteins in polyacrylamide gels. Anal Biochem. 1981 Nov 15;118(1):197–203. doi: 10.1016/0003-2697(81)90179-2. [DOI] [PubMed] [Google Scholar]
- Zhang J. M., Cowell J. L., Steven A. C., Carter P. H., McGrath P. P., Manclark C. R. Purification and characterization of fimbriae isolated from Bordetella pertussis. Infect Immun. 1985 May;48(2):422–427. doi: 10.1128/iai.48.2.422-427.1985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang J. M., Cowell J. L., Steven A. C., Manclark C. R. Purification of serotype 2 fimbriae of Bordetella pertussis and their identification as a mouse protective antigen. Dev Biol Stand. 1985;61:173–185. [PubMed] [Google Scholar]
- de StGroth S. F., Scheidegger D. Production of monoclonal antibodies: strategy and tactics. J Immunol Methods. 1980;35(1-2):1–21. doi: 10.1016/0022-1759(80)90146-5. [DOI] [PubMed] [Google Scholar]